Abstract
Background
Numerous studies suggest an increased vascular risk in patients with migraine, in particular in those with aura. A possible link between both conditions might be a dysfunction of the vascular endothelium. This observational study analyzed the endothelial markers angiopoietin-1, angiopoietin-2, Tie-2, sFlt-1 and NT-proBNP for the first time in migraineurs, patients with other primary headache disorders and healthy controls.
Methods
Patients with episodic migraine with and without aura, episodic cluster headache, tension-type headache and healthy controls were included. Blood samples were obtained during migraine attacks and headache-free periods in migraineurs, in and out of bout in cluster headache and during headache-free periods in tension-type headache and healthy individuals to analyze markers of endothelial function.
Results
No significant difference in endothelial markers between migraine, other headache disorders and healthy controls was detected. There was no significant difference between migraine attacks and headache-free intervals. Additionally, no distinction could be found between migraine with and without aura.
Discussion
The endothelial markers analyzed do not display a characteristic pattern in different headache disorders especially migraine compared to healthy controls. The novel findings of our study indicate that factors other than endothelial dysfunction seem to be responsible for the at least statistical association of migraine with vascular disease.
Introduction
An association between migraine, in particular migraine with aura, and cerebrovascular or cardiovascular disease has been discussed controversially throughout the past decades. There is ample evidence, mainly from large epidemiological studies, for a higher prevalence of cerebrovascular and cardiovascular diseases, arterial hypertension, preeclampsia, ischemic white matter lesions and various other disorders associated with vascular abnormalities in patients with migraine, especially with aura (1–9). Most reports, however, have focused on migraine and ischemic stroke (10–12).
Dysfunction of the vascular endothelium has been suggested to be a prominent underlying condition linking these disorders and migraine (13,14). This hypothesis is supported by preliminary findings of altered endothelial function in migraineurs. Increased endothelial progenitor cells, an indicator of endothelial damage, have been reported in patients with migraine (15,16). A recent study revealed an elevation of circulating endothelial microparticles in female migraineurs with aura (17). Tietjen and colleagues described elevated markers of endothelial activation in premenopausal women independently from other cardiovascular risk factors and suggested that vasculopathy is present in migraineurs even without atherosclerotic disease (18). On the other hand recent studies investigating flow-mediated dilation or endothelial nitric oxide release showed that systemic endothelial function is not impaired in migraine patients without comorbidities, arguing against a pivotal role of endothelial dysfunction in migraine (19). However, exact pathophysiological mechanisms explaining the statistical association of migraine, in particular with aura, and increased cerebrovascular disease still remain unclear.
This study prospectively analyzed well-established serum markers reflecting endothelial and cardiac function in patients with migraine in comparison with other primary headache disorders and healthy controls. We hypothesized that serum levels of angiopoietin-1 (Ang-1), angiopoietin-2 (Ang-2), soluble fms-like tyrosine kinase-1 (sFlt-1), soluble angiopoietin receptor sTie-2 and brain natriuretic peptide (NT-proBNP) would be elevated in migraine patients during pain-free periods as well as during migraine attacks compared with other primary headache disorders or healthy controls. To our knowledge these parameters have not been investigated prospectively in migraine patients so far.
Material and methods
Study sites and design
Three groups of headache patients were enrolled in this prospective study: patients with episodic migraine with and without aura, episodic cluster headache, and frequent episodic tension-type headache. All headache diagnoses were made by a headache specialist according to the criteria of the second edition of the International Classification of Headache Disorders (ICHD-II) (20). All patients were recruited consecutively in two headache outpatient clinics: the headache outpatient clinic at the Department of Neurology at Innsbruck Medical University, Innsbruck, Austria, and the West German Headache Center at the University of Duisburg-Essen, Essen, Germany. In migraineurs and patients with cluster headache collection of blood samples was performed twice: during migraine attacks and during the interictal phase, inside and outside of cluster bouts, respectively. In patients with tension-type headache (during pain-free periods only) and healthy controls, one single blood sample was obtained. All individuals participating in this study were enrolled following the identical inclusion and exclusion criteria of the study protocol. Coworkers of our institution, who did not have a history of any primary headache disorder, served as healthy controls.
Migraine patients were contacted three days after venipuncture during headache-free periods to rule out blood sampling during the premonitory phase of an upcoming migraine attack. Hence, samples of patients who experienced a migraine attack within 72 hours after blood sampling were discarded and not used for analysis of endothelial markers.
Standard protocol approvals, registrations and patient consents
The study protocol was approved by the institutional review boards at the Medical University of Innsbruck and the University of Duisburg-Essen. All patients and controls gave written informed consent to study participation prior to study-related procedures.
Inclusion and exclusion criteria
Patients (males and females) had to be between 18 and 50 years. In migraineurs no more than eight migraine attacks per month were allowed. Forty-eight hours prior to blood sample collection patients were not allowed to take any pain medication including nonsteroidal anti-inflammatory drugs, triptans or opioids. A following phone contact should be possible three days after blood sampling. Patients taking any preventive medication for prophylaxis of migraine or tension-type headache were excluded. Further exclusion criteria were diagnosis of medication overuse headache (according to ICHD II revised (r)), and patients with tension-type headache and more than 10 days per month with reported intake of medication for acute treatment of headache. Patients were not included if they had a history of cardio- or cerebrovascular disease, arterial hypertension, diabetes, cancer/malignancy, dyslipidemia, obesity (body mass index ≥30), or severe renal or hepatic disease. Though being an important risk factor for vascular disease, smoking status was not defined as an exclusion criterion in our study, mainly because the prevalence of smokers among patients with cluster headache has been reported to be up to 65% (21). Patients with more than one diagnosis of a primary headache, i.e. coexistent migraine and tension-type headache, were excluded from the study. Pregnancy and breastfeeding were additional exclusion criteria.
Pathophysiological rationale of testing Ang-1, Ang-2, sTie-2, sFlt-1 and NT-proBNP
Angiopoietin-1 and -2 have been identified as important regulators of vascular function and act as agonist (Ang-1) and antagonist (Ang-2) on their common receptor Tie-2 (22,23). They exert antagonistic functions on endothelial cells with Ang-1 having an anti-inflammatory, anti-apoptotic effect and Ang-2 playing an opposite role, i.e. pro-apoptotic and pro-inflammatory on endothelial cells (24). A change of the Ang-1/Ang-2 ratio in favor of Ang-2 has been shown in various conditions associated with endothelial dysfunction such as sepsis, arterial hypertension or renal disease (25–29).
The soluble receptor of vascular endothelial growth factor (VEGF) sFlt-1 is well known to be elevated in women with preeclampsia (30,31). Endothelial dysfunction is regarded as the underlying pathomechanism of this disease; elevated levels of sFlt-1 are indicators of endothelial damage and have been shown to predict, in conjunction with the placental growth factor, the onset of preeclampsia (32). The N-terminal part of the pro-hormone of brain-derived natriuretic factor (NT-proBNP) is widely used in clinical practice as a surrogate marker of cardiac disease, i.e. heart failure (33). In a recent study BNP has been shown to be an independent predictor of endothelial function in healthy individuals and patients with cardiovascular risk factors (34).
Blood samples and analysis of Ang-1, Ang-2, sTie-2, sFlt-1 and NT-proBNP
All blood samples were processed strictly following an identical predefined protocol in both centers. Venous cubital blood samples were centrifuged after a clotting time of 30–60 minutes at 1500 rcf for 15 minutes to obtain serum. Immediately after centrifugation, serum samples were divided into aliquots of 300 µl each and stored at −20℃ until use. Measurement of Ang-1, Ang-2 and sTie-2 was performed using sandwich enzyme-linked immunosorbent assay according to the manufacturer’s instructions (R&D Systems, Minneapolis, MN, USA). The lower limits of detection are 3.45 pg/ml (Ang-1), 8.29 pg/ml (Ang-2) and 14 pg/ml (sTie-2). Intra- and interassay coefficients of variation were determined to be 2.0% to 3.4% and 4.5% to 6.8% (Ang-1), 4.2% to 6.9% and 7.4% to 10.4% (Ang-2), 4.3% to 5.3% and 5.2% to 8.5% (sTie-2), respectively. sFLT-1 serum concentrations were measured using an electrochemoluminiscence immunoassay (“sFLT-1” ECLIA, Roche, Mannheim, Germany) on a Cobas e601 immunoanalyzer (Roche Diagnostics). The lower limit of detection is 10 pg/ml. Intra- and interassay coefficients of variation were determined to be 0.8% to 1.6% and 2.3% to 4.3%, respectively. NT-proBNP serum levels were determined by the “proBNPII” ECLIA (Roche, Mannheim, Germany) on a Cobas e601 immunoanalyzer (Roche Diagnostics). The lower limit of detection is 5 pg/ml. Intra- and interassay coefficients of variation are 1.2% to 1.9% and 1.7% to 3.1%, respectively. No specific interferences have been reported. Patient and control samples were analyzed during the same assay run with identical standard curves.
Statistical analysis
Our main hypothesis was that Ang-2 would be elevated in migraine patients compared to non-migraineurs. Hence, Ang-2 was used for power calculation. At the time of study design no data were available for Ang levels in migraineurs. Therefore, the power calculation for this study was based on patients with arterial hypertension as a risk factor for cardio- and cerebrovascular disease, who are supposed to have moderately higher Ang-2 levels compared with healthy individuals (35). According to the power calculation (with an effect size of 0.80), a sample size of 14 patients with a power of 0.80 would have been required for each group to show significant differences at a 5% level using a two-sided test. Assuming that blood sampling during the migraine attack without prior pain medication would not be feasible in all patients, we defined a sample size of at least 20 patients for each group.
The Kolmogorov-Smirnov test was used to test for normal distribution of continuous variables. The Spearman’s rank correlation was used to test the association between headache days per month/attack frequency and serum levels of Ang-1, Ang-2, Tie-2, sFlt-1 and NT-proBNP. Data are presented as median (interquartile range (IQR)) unless otherwise stated. A two-sided p value of <0.05 was considered as statistically significant. P values are reported without adjustment for multiplicity. Univariate analyses were used to describe the impact of group allocation (migraine, cluster headache, tension-type headache, healthy) on levels of endothelial markers. In a second step, adjustment for important covariates (gender, age and history of smoking) was performed using a general linear model (GLM). All statistical analyses were performed using the IBM SPSS statistics 20 software (IBM Statistics) and the GraphPad Prism 6 software (GraphPad Prism Software Inc, San Diego, CA, USA).
Results
Demographic and clinical characteristics
Serum levels of angiopoietin-1 (Ang-1), angiopoietin-2 (Ang-2), Ang-2/Ang-1 ratio, soluble Tie-2 receptor, soluble receptor of the vascular endothelial growth factor (sFlt-1) and brain natriuretic peptide (NT-proBNP) in primary headache patients and healthy controls.

Data are presented as median (interquartile range (IQR)). f: female. P values refer to comparison between all groups (migraine with and without aura, cluster headache, tension-type headache, healthy controls) during headache-free periods using the Kruskal-Wallis test.
A comparison of serum levels of Ang-1, Ang-2, Tie-2, sFlt-1 and NT-proBNP between male and female patients revealed a statistical significance for NT-proBNP with BNP levels being higher in females (p < 0.001) as published previously (36). For the remaining markers there was no difference with respect to gender.
Serum levels of Ang-1, Ang-2, sTie-2, sFlt-1 and NT-proBNP
Ang-1, Ang-2, Ang-2/Ang-1 ratio, Tie-2, sFlt-1 and BNP did not differ significantly between groups (migraine with and without aura, cluster headache, tension-type headache, healthy participants) during pain-free periods (see Table 1). Adjusted analysis (for covariates age, gender and history of smoking) still did not reveal a significant difference of the respective serum levels between the main diagnoses. The difference of NT-proBNP levels between males and females was confirmed in the model. Median serum levels of all markers are displayed in Figure 1 (a)–(f).
(a)–(f) Median serum levels of angiopoietin-1 (Ang-1), angiopoietin-2 (Ang-2), Ang-2/Ang-1 ratio, soluble Tie-2 (sTie-2), soluble fms-like tyrosine kinase (sFlt-1) and brain natriuretic peptide (NT-proBNP) in patients with migraine (M) during pain-free periods and migraine attacks, cluster headache (CH) in and out of cluster bouts, tension-type headache (TTH) and healthy individuals (HC). Serum levels were compared between migraine patients during headache-free periods and migraine attacks, headache-free periods in migraineurs and healthy controls and between migraine attacks and healthy controls. The Mann-Whitney U test was used for all comparisons.
Median serum levels of angiopoietin-1 (Ang-1), angiopoietin-2 (Ang-2), Ang-2/Ang-1 ratio, soluble Tie-2 receptor, soluble receptor of the vascular endothelial growth factor (sFlt-1) and brain natriuretic peptide (NT-proBNP) in paired samples of patients with migraine (M) with and without aura and cluster headache (CH).
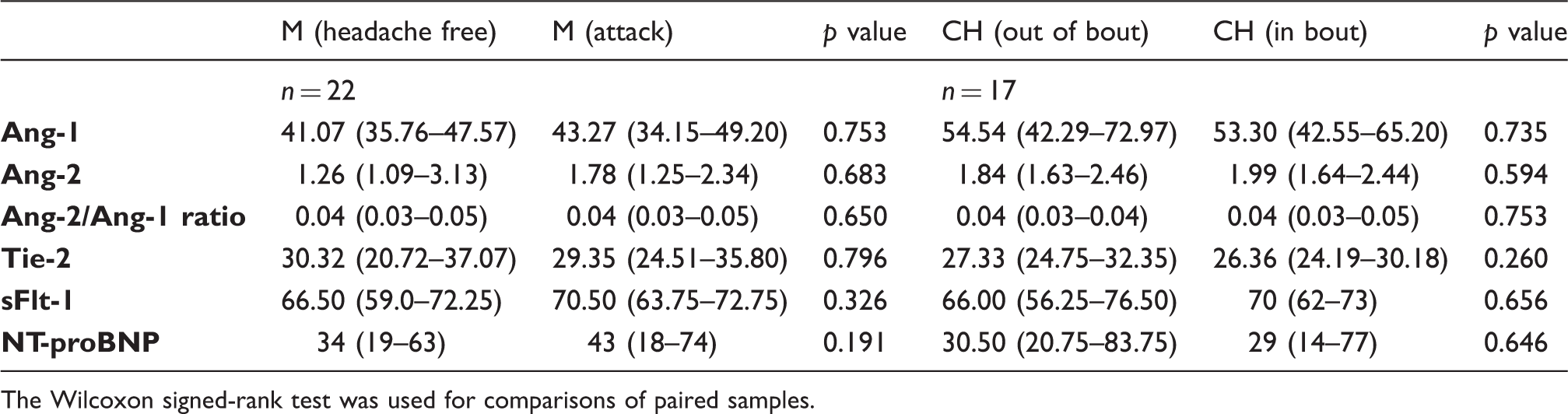
The Wilcoxon signed-rank test was used for comparisons of paired samples.
A subgroup analysis of migraine patients with aura comparing headache-free periods and attacks did not reveal a significant difference in any of the markers analyzed (Figure 2). In migraine patients there was no significant correlation with any of the proteins analyzed and the number of headache days per month (Ang-1: r = −0.26, Ang-2: r = −0.10, ratio: r = −0.02, sTie-2: r = 0.08, sFlt-1: r = –0.05, NT-proBNP: r = 0.12).
(a)–(f) Comparison of median serum levels of angiopoietin-1 (Ang-1), angiopoietin-2 (Ang-2), Ang-2/Ang-1 ratio, soluble Tie-2 (sTie-2), soluble fms-like tyrosine kinase (sFlt-1) and brain natriuretic peptide (NT-proBNP) between migraine patients with (MwA) and without (MoA) aura during migraine attacks and headache-free periods. The Mann-Whitney U test was used for all comparisons.
When comparing patients with cluster headache and healthy controls, there was no statistical significance (Ang-1: p = 0.06, Ang-2: p = 0.33, ratio: p = 0.42, sTie-2: p = 0.36, sFlt-1: p = 1.0, NT-proBNP: p = 0.74). In cluster headache patients the Ang-2/Ang-1 ratio was imbalanced in bout in favor of Ang-2 (p = 0.031). The remaining biomarker levels did not differ significantly in and out of bout. When analyzing paired samples of 17 patients we did not find a difference in any of the serum markers in and out of bout (Ang-1: p = 0.735, Ang-2: p = 0.594, Tie-2: p = 0.260, sFlt-1: p = 0.656, BNP: p = 0.646).
Discussion
Numerous studies describe an association of migraine in particular with aura and cerebro- and cardiovascular diseases. The endothelium is one prominent suspect that has been discussed to be the driver behind this association. However, analyzing those parameters (Ang-1, -2, sTie-2, sFlt-1 and NT-proBNP) in different headache diagnostic groups as well as in healthy controls did not reveal any statistical difference. Also no difference was found comparing pain-free state with blood samples taken during headache attacks.
Assuming that increased vascular risk in migraineurs is attributable to a dysfunction of the vascular endothelium, it is surprising that none of these well-established endothelial markers analyzed in this study was increased in migraine patients.
Most studies focusing on increased risk of cardio- and cerebrovascular diseases in migraine patients found a higher risk in migraine with aura. Also, signs of endothelial dysfunction such as increased levels of endothelial progenitor cells, circulating endothelial microparticles or increased intima-media thickness have been reported to occur more frequently in migraine with aura (17,37–40). Thus, even without a difference in endothelial activation markers between migraineurs and other study groups, it is surprising that we could not even detect a significant difference comparing migraine with and without aura for the respective parameters.
The lacking difference in levels of the proteins analyzed might be due to the fact that endothelial dysfunction is not the main cause for increased vascular risk in migraineurs. Other factors or coexisting conditions, such as hormonal contraception, smoking status, higher prevalence of atherosclerosis in migraine patients, cortical spreading depression presumably leading to structural brain alterations, treatment interactions, genetic predisposition or associated comorbidities such as obesity might be even more causative when it comes to cardio- and cerebrovascular events in migraineurs than the endothelium alone (14,41). Alterations in vascular function might also be not (only) attributable to endothelial function. A case-control study by Napoli and colleagues showed impaired vascular reactivity caused by vascular smooth muscle cell dysfunction in migraine patients (42).
In line with our results, there are previous studies that could not show an association of migraine and vascular pathology. Perko and colleagues did not find significant changes in flow-mediated dilation, an indicator for vascular reactivity and function, and intima-media thickness in otherwise healthy migraineurs (43). Furthermore, a cross-sectional study investigating endothelium-dependent vasodilation and basal endothelial nitric oxide production in migraine patients and healthy participants did not reveal any alterations in endothelial function (19).
Numerous methods are available to assess endothelial function including measurement of flow-mediated dilation, intima-media thickness, endothelial progenitor cells, endothelial-derived substances, endothelial microparticles, digital pulse amplitude tonometry or pulse wave velocity (44). The variety in methodological approaches may explain the conflicting results observed in clinical studies focusing on endothelial function in migraineurs.
It is important to note that systemic levels of endothelial markers may not reflect the corresponding condition in the cerebral circulation (45). Local dysfunction of the cerebral vasculature may play a stronger causative role in increased vascular risk, in particular with regard to the increased risk of cerebrovascular diseases. Cerebrovascular reactivity has been found to be impaired in migraineurs, which may also explain the increased risk of cerebrovascular disease (46).
There are several limitations to our study: The average age of the control group was relatively low. Thus, we might have included individuals who may possibly develop a primary headache disorder later in their life. Patients with chronic migraine and patients receiving prophylactic treatment, who experience more migraine attacks, were not included in our study. We followed a very strict exclusion protocol in order to study unmodified effects of migraine per se on endothelial function. In order to control for confounding by comorbidities such as arterial hypertension, a history of cardio- or cerebrovascular disease, diabetes, dyslipidemia, atherosclerosis or a body mass index ≥ 30, we did not include patients with concomitant disorders possibly affecting vascular integrity and thus, causing an alteration of endothelial markers per se. However, it is important to note that an association of these comorbidities and migraine has been reported (47).
In conclusion the results of our prospective study analyzing several well-established markers of endothelial function and integrity did not confirm the hypothesis that the at least statistical association between migraine and vascular disease described in epidemiological studies can be explained by endothelial dysfunction only.
We speculate that our results may indicate that it is not migraine per se that causes endothelial dysfunction but rather a mixture of various concomitant factors possibly interacting with migraine including among others medication, hormones, genetic predisposition and vascular aging.
Key findings
We did not find a difference in Angiopoietin-1, Angiopoietin-2, their common soluble receptor Tie-2, the soluble vascular endothelial growth factor (VEGF) receptor sFlt-1 and brain natriuretic peptide between migraineurs and patients with other primary headaches or healthy individuals. Markers of endothelial activation are not elevated during migraine attacks compared with headache-free periods or control groups. The association between migraine and vascular disease might be attributable to factors other than endothelial dysfunction.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
