Abstract
Objective
The objective of this study was to determine whether white matter hyperintensities (WMHs) in subjects with migraine are related to alterations in resting cerebral blood flow (CBF).
Methods
Migraine with aura (MWA), migraine without aura (MwoA), and control subjects were enrolled in a 1:1:1 ratio. WMH load was scored based on fluid-attenuated inversion recovery/T2-weighted magnetic resonance imaging (MRI) using a previously established semi-quantitative scale. Global and regional CBFs were quantified using arterial spin labelled perfusion MRI. Integrity of the circle of Willis was assessed with magnetic resonance angiography (MRA).
Results
A total of 170 subjects were enrolled (54 controls, 56 MWA, and 60 MwoA). There was no significant difference in subjects with ≥1 WMH across groups (22% controls, 29% MWA, 35% MwoA; p = NS). Similarly, high WMH load was not significantly different across groups (16.7% controls, 21.4% MWA, 25.0% MwoA; p = NS). High WMH load was strongly associated with increasing age (odds ratio: 1.08 per year, 95% confidence interval: 1.02–1.13, p = 0.01). Resting CBF was similar across groups, but was significantly higher in women. In MWA subjects with high WMH load, CBF was substantially lower (p = 0.03). No association between WMH load and CBF was seen in control or MwoA subjects.
Conclusions
WHMs in MWA may be related to alterations in resting CBF.
Introduction
A number of studies have demonstrated an increased prevalence of white matter hyperintensities (WMHs) in migraine (1–4); an increased prevalence of subclinical brain infarctions has also been seen in some studies (3,5). Two meta-analyses of studies assessing WMH prevalence in migraine have been performed, suggesting that subjects with migraine are two- to four-times more likely to have WMHs than control subjects (6,7).
The mechanisms underlying the increased prevalence of WMH in migraine are not well understood. It has been suggested that WMH in migraine may be a consequence of ischaemic microvascular disturbances with prolonged and repeated oligaemia during migraine attacks affecting vulnerable small, deep-penetrating arteries and leading to hypoperfusion in deep brain regions (8). Experimental data also suggest that activated matrix metalloproteinases during cortical spreading depression can lead to transient breaches of the blood–brain barrier and ischaemic brain injury (9). Additional proposed mechanisms include microembolism, coagulation activation, and endothelial dysfunction (10).
Studies of resting cerebral blood flow (CBF) in migraine have reported widely variable results, with some showing hypoperfusion and others hyperperfusion (11,12). The largest study performed to date used 2D phase-contrast magnetic resonance imaging (MRI) to assess CBF in over 2500 community dwellers in Rotterdam (13). This study demonstrated a statistically significant but very small increase in CBF in migraine subjects compared to control subjects. While it seems plausible that alterations in resting CBF might contribute to vulnerability to WMH, to date, few studies have directly examined the relationship between these two factors in migraine subjects. The purpose of the present study was to evaluate the burden of WMH in migraine subjects compared to controls and to examine the interaction between WMH and resting CBF as measured using arterial spin-labelled (ASL) perfusion MRI, an established technique for the non-invasive quantification of CBF (14,15).
Methods
Study design and subjects
We performed a secondary analysis of data collected from the Anatomy and Cerebral Hemodynamic Evaluation of Migraine (ACHE-M) study, the full details of which have been published previously (16). ACHE-M is a prospective case–control study using MRI to compare the vascular structure and function between headache-free controls and migraine subjects. Participants were recruited from the neurology clinic at the University of Pennsylvania and by advertisements in the wider community. Eligible participants were 25–50 years old and either had a diagnosis of migraine with aura (MWA) or migraine without aura (MwoA) using International Classification of Headache Disorders criteria or were headache-free controls (17). Individuals with a history of cerebrovascular or cardiovascular disease or other neurologic illness were excluded. The study aimed to enrol MWA, MwoA, and control individuals in a 1:1:1 ratio, and recruitment of control subjects was targeted to match the migraine cohort by age and sex. Participants were screened and examined by a single study neurologist to ensure that they met the inclusion/exclusion criteria, and they were enrolled between March 2008 and June 2012. The study was approved by the University of Pennsylvania Institutional Review Board; all participants provided written informed consent.
MRI protocol
MRI scans were acquired on a 3T whole-body Siemens TIM Trio scanner (Erlangen, Germany) with an eight-channel receive-only head coil and body coil transmission. High-resolution whole-brain anatomic images were collected using 3D magnetisation-prepared rapid gradient echo (MPRAGE) with the following parameters: TR/TE/TI = 1620/3.1/950 ms, 160 axial slices, and 1-mm isotropic resolution.T2-weighted images (TR/TE = 4000/79 ms, FOV = 22 × 22 cm2, acquisition matrix = 256 × 256, slice thickness = 3 mm, slice gap = 1 mm, number of slices = 40), fluid-attenuated inversion recovery (FLAIR) images (TR/TE/TI = 9190/104/2200 ms, FOV = 18 × 24 cm2, acquisition matrix = 320 × 144, slice thickness = 3 mm, slice gap = 1 mm, number of slices = 40), and 3D time-of-flight magnetic resonance angiography (MRA) (TR/TE = 26/3.4 ms, slice = 1 mm, overlap = 0.5 mm, field-of-view (FOV) = 25 cm, matrix = 832 × 571) were acquired. Resting CBF measurements were acquired using pseudo-continuous ASL perfusion imaging with gradient echo echo-planar imaging (TR/TE = 4000/18 ms, slice thickness = 6 mm, slice gap = 1.2 mm, voxel resolution = 3.5 × 3.5 × 6 mm3, labelling duration = 1125 ms, post-labelling delay time = 1200 ms) (18). The labelling plane was positioned 90 mm below the centre of the imaging slab composed of 16 axial slices. Thirty pairs of interleaved control and tag images were acquired.
Image analysis
Structural MRI images were interpreted by a neuroradiologist who was blinded to diagnosis and clinical data. The scans of the subjects with WMH were subsequently reviewed and confirmed by two neurologists, also blinded to clinical data, with disagreements resolved through discussion. In order to facilitate comparison with prior studies, we replicated the methods for assessing WMH used in the Cerebral Abnormalities in Migraine Epidemiologic Risk Analysis (CAMERA) study (3). Briefly, WMHs were visible as a hyperintense lesions on T2-weighted and FLAIR images and isointense or slightly hypointense on T1-weighted images. WMHs were rated by both number and size, and a semi-quantitative measure of white matter lesion load was determined by multiplying the number of individual lesions by a size-dependent constant (0.0042 for ≤3-mm lesions, 0.114 for 4–10-mm lesions, and 0.90 for >10-mm lesions) (3). High WMH load was defined as the upper quintile of WMH load in the overall cohort. Periventricular WMHs (PVWMHs) were assessed in three regions and rated on a 0–3 scale in each region for a total score of 0–9 as was done in CAMERA (3). Cerebral infarctions were defined as parenchymal defects that were isointense to cerebrospinal fluid signal on all sequences and surrounded by a hyperintense rim on FLAIR images when supratentorial.
Interpretation of MRA data for circle of Willis (CoW) variants has been described in detail previously (16). A CoW variant burden score was computed by assigning points to each vessel in the circle (0 = present, 1 = hypoplastic, 2 = absent) and summing the total number of points; high CoW variant burden was defined as a score ≥2. ASL data pre-processing and CBF calculation were performed by ASLtbx (19). Raw ASL images were first co-registered to the MPRAGE images and label and control images were separately motion-corrected using a six-parameter rigid body spatial transformation. Pairwise subtraction images were then generated after smoothing by a 6-mm full-width half-maximum (FWHM) kernel. Averaged difference images were converted to mL/100 g/minute CBF maps using a single-compartment model (20). Finally, the CBF maps were normalised into the Montreal Neurological Institute (MNI) template.
To extract grey matter (GM) and white matter (WM), CBF and GM and WM regions of interest (ROIs) were established by a previously reported method (15): the T1-weighted image from the MNI template was segmented to probabilistic GM and WM maps using an optimised segmentation routine in SPM8, and then a GM ROI was created by binarising the probabilistic GM map with a threshold of > 0.8. In order to minimise contamination of WM CBF measurements with GM, the probabilistic WM map was first smoothed with a 4-mm FWHM kernel, then binarised with a threshold of > 0.8, and finally eroded with a threshold of 3. The final WM ROIs mainly consisted of central WM. Global CBF was assessed by combining the GM and WM after binarising the probabilistic maps with a threshold of > 0.15 in order to exclude cerebrospinal fluid (CSF). The relative CBF values for GM and WM were calculated by dividing the GM and WM CBF values by the global CBF values. Although additional spatial smoothing is often used to reduce noise in ASL MRI CBF maps, for this study, the global, GM, and WM masks were applied to unsmoothed CBF maps in order to further minimise the contamination between GM and WM segments.
Statistical analysis
Baseline clinical characteristics were analysed using the chi-squared or Fisher’s exact tests for dichotomous or categorical variables and the t-test or Wilcoxon ranked-sum test for continuous variables as appropriate. Means and standard deviations or medians and interquartile ranges are presented as appropriate. Associations between continuous variables were tested using multivariable linear regression, adjusting for factors that were associated in the univariate analysis at a significance level of p < 0.10. For all reported linear regression analyses, analysis of residuals confirmed the assumption of linearity. Similarly, multivariable logistic regression was used to test associations between categorical variables. An association was considered significant if p < 0.05. All tests were two sided. As recommended by biostatistical experts, corrections for multiple comparisons were not performed (21). Statistical analyses were performed using JMP (Version 10, SAS Institute, Inc., Cary, NC).
Results
Clinical characteristics of groups.
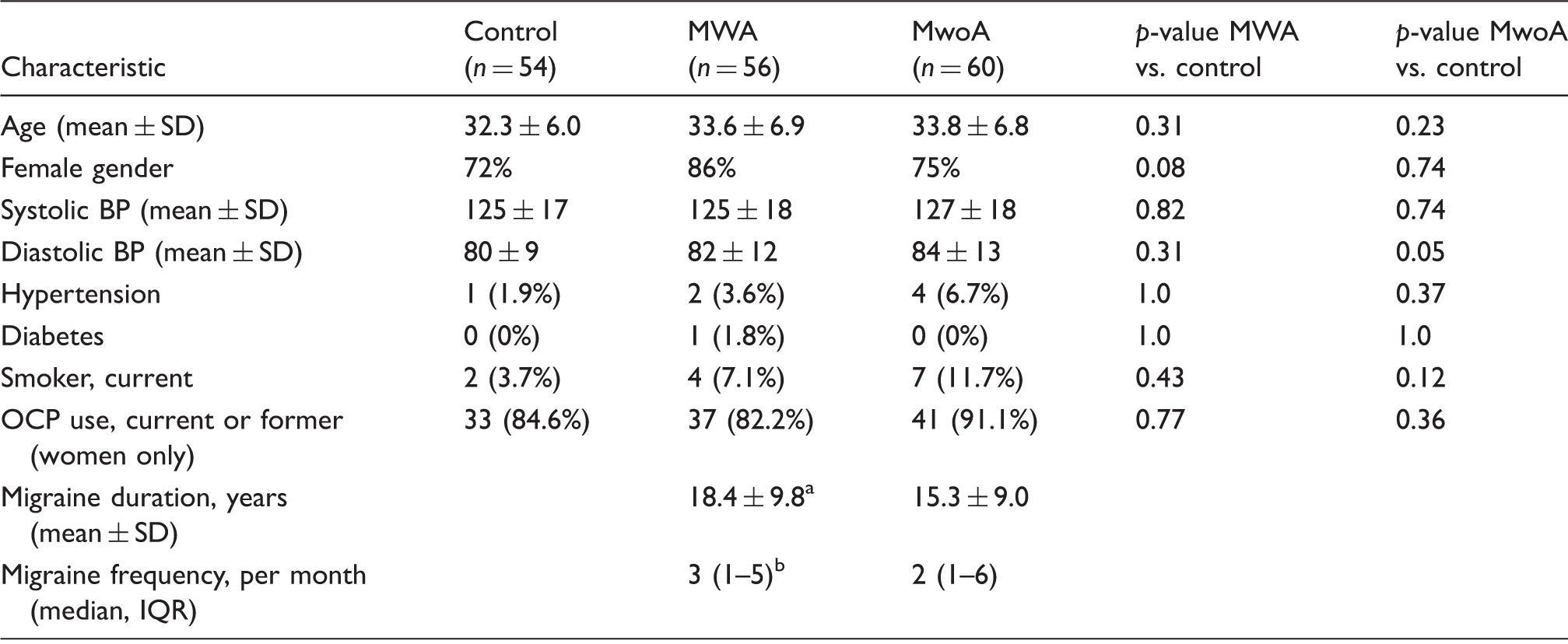
p = 0.08 vs. MwoA.
p = 0.52 vs. MwoA.
BP: blood pressure; IQR: interquartile range; MWA: migraine with aura; MwoA: migraine without aura; OCP: oral contraceptive pill.
White matter hyperintensities
White matter hyperintensities across groups.
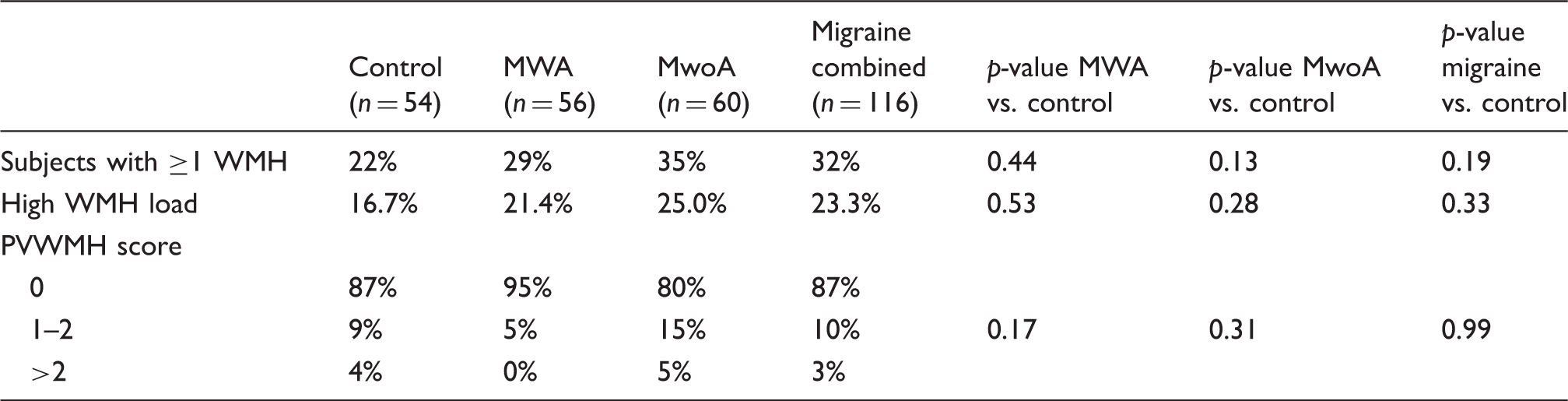
p-values for PVWMH; score represent comparisons of percentages of subjects with scores of 0 to ≥1.
MWA: migraine with aura; MwoA: migraine without aura; PVWMH: perivascular white matter hyperintensity; WMH: white matter hyperintensity.
There was no relationship between WMH load and migraine status, sex, migraine frequency or duration, systolic or diastolic blood pressure, history of hypertension, incomplete CoW, or, in women, oral contraceptive pill (OCP) use. Both age (odds ratio (OR): 1.09 per year, 95% confidence interval (CI): 1.03–1.15, p = 0.002) and current smoking (OR: 3.6 compared to never/former smokers, 95% CI: 1.1–11.7, p = 0.04) were associated with high WMH load, and there was a trend towards an association between high WMH load and high CoW variant burden (OR: 1.9, 95% CI: 0.9–4.1, p = 0.09). In multivariate analysis adjusting for these factors, only age remained significantly associated with high WMH load (OR: 1.08 per year, 95% CI: 1.02–1.13, p = 0.01). Similar results were seen with a multivariate model incorporating age, sex, history of hypertension, migraine status, and current smoking. Using a model incorporating age, sex, and migraine status (as has been done in prior reports) again showed no significant relationship between high WMH load and MWA (OR: 1.3, 95% CI: 0.5–3.6) or MwoA (OR: 1.5, 95% CI: 0.6–3.9) (3). For subjects with MWA, the association was similar for men (OR: 1.3, 95% CI: 0.1–10.2) and women (OR: 1.3, 95% CI: 0.4–4.2). For subjects with MwoA, the association was stronger for women (OR: 1.8, 95% CI: 0.6–5.7) compared to men (OR: 0.9, 95% CI: 0.1–5.8).
Analysis comparing those with one or more WMH compared to those with no WMHs yielded similar overall results to the analysis of high WMH load.
Cerebral infarctions
There was only a single MRI lesion that met the criteria for a cerebral infarction. This lesion was present in a control subject and was located in the medial cerebellum.
Cerebral blood flow
Comparison of mean cerebral blood flow (mL/100 g/minute) across different patient groups (n = 156).

CBF: cerebral blood flow; MWA: migraine with aura; MwoA: migraine without aura; WMH: white matter hyperintensity.
Interaction between CBF and WMH
In the overall cohort, there was no significant relationship between high WMH load and global CBF (p = 0.20), grey matter CBF (p = 0.24), or white matter CBF (p = 0.20). However, in the MWA group, global CBF was significantly lower in patients with high WML load after adjustment for age, sex, SBP, DBP, history of hypertension, and smoking status (45.5 ± 9.2 vs. 52.9 ± 11.2, p = 0.03; unadjusted p = 0.049) (Figure 1). Similar findings were seen for grey (49.4 ± 10.2 vs. 57.3 ± 12.6, p = 0.06) and white matter CBF (29.8 ± 5.9 vs. 34.6 ± 6.8, p = 0.04). There were no significant differences in CBF between subjects with high versus low WMH loads in the MwoA (p = 0.98 adjusted, p = 0.79 unadjusted) or control groups (p = 0.74 adjusted, p = 0.50 unadjusted).
Interaction between cerebral blood flow and white matter hyperintensity load across groups.
Discussion
In the present study, we did not find a significantly increased prevalence of WMHs in migraine subjects compared to controls. In the CAMERA study, the risk of high WMH load was significantly increased in women with both MWA (OR: 2.0) and MwoA (OR: 2.1), but there was no significantly increased risk in men (MWA, OR: 0.6; MwoA, OR: 0.7) (3). Using a very comparable methodology, we found quite similar results for MwoA (OR: 1.8 for women; OR: 0.9 for men), although our results were not statistically significant. We found a lower risk without significant sex difference for MWA subjects (OR: 1.3 for both women and men); however, our CIs are wide and clearly encompass the previously reported results. The overall prevalence of WMHs in both migraine and control groups was substantially lower in our study compared to the CAMERA study, and we found no subclinical cerebral infarctions in either migraine group, compared to the 8% rate observed in CAMERA (3). Compared to that population, our study subjects were substantially younger (average age ∼33 vs. ∼48 years) and had a much lower prevalence of hypertension (∼2–7% vs. ∼35–40%), which may account for these differences. We used a semi-quantitative scale to assess WMH (as was used in the CAMERA study), so we are not able to compute a precise WMH volume; however, the total burden of WMHs in those subjects in which lesions were present was very low, on the order of < 0.1% of the total white matter volume. It is likely that the low prevalence and burden of WMHs in our cohort limited our power to identify a difference across groups.
We found that increasing age was significantly associated with WMH load, a finding that is consistent with prior research (22). We also found a significant association in the univariate analysis with current smoking, which has also been identified in prior studies as an independent risk factor for the progression of WMHs (23). While we did not find a relationship between hypertension and WMHs, this likely reflects the very low prevalence of hypertension in our cohort, such that there was very limited power to identify this well-established association (24). One prior study has examined the association between integrity of the CoW and WMH burden and, like our study, failed to identify a significant association (25).
The main focus of our study was to examine the potential role of alterations in CBF in migraine. We used ASL perfusion MRI, a well-validated technique for quantifying global and regional CBF, and specifically determined segmented grey and white matter blood flow (15,26). While measurement of white matter blood flow with ASL is more challenging than measurement of grey matter blood flow, the methods we utilised in this study, when applied to large white matter regions of interest as was done here, have been shown to provide reliable estimates of white matter perfusion (27,28). Prior studies of interictal CBF in migraine largely used nuclear medicine approaches, such as single-photon emission computed tomography (SPECT), and showed widely variable results, but were generally limited by small sample sizes and imperfect matching for age and sex with control subjects (11). Recently, the population-based Rotterdam study reported CBF measurement using 2D phase-contrast MRI in 2642 community dwellers, 456 of whom had migraine (13). Phase-contrast MRI can be used to generate CBF measurements by determining flow volume in the individual large cerebral vessels and then norming these measurements to total brain volume. In the Rotterdam study, CBF was 1.07 mL/100 g/minute higher (95% CI: 0.08–2.05) in migraine compared to control subjects, a finding that remained significant after adjustment for age and sex, as well as a number of other covariates. There was no significant difference between those with and without aura. Interestingly, however, the finding of slightly increased CBF was only significant in those without a migraine attack within the past year. In those with more recent migraine, there was no significant difference in CBF compared to controls (0.63 mL/100 g/minute; 95% CI: –0.63 to 1.88). Given its large-sample, population-based cohort and rigorous ability to adjust for covariates, the Rotterdam study provides the strongest data to date on interictal global CBF in migraine. Considering that all of the subjects in our study had experienced active MWA attacks within the prior year, our results are entirely concordant with the Rotterdam results, demonstrating a small but insignificant increase in CBF in migraine compared to control subjects.
Our results show that MWA subjects with high WMH load have reduced interictal CBF; this was only seen in the MWA group, and not in the MwoA or control groups, suggesting that the underlying mechanisms leading to the development of WMHs may vary between groups.
There are a number of potential explanations for the finding of an interaction between interictal CBF and WMHs in MWA subjects. First, this might support the hypothesis that large-amplitude fluctuations in CBF related to hyper- and hypo-perfusion and modulated by cortical spreading depression from repeated aura attacks might affect microvascular haemodynamics, even in the interictal phase, leading to ischaemic injury in a manner that is analogous to the association observed between blood pressure variability and increased risk of clinical stroke (29). However, we did not observe an association between frequency of attacks and either presence of WMHs or CBF. Alternatively, MWA subjects might be particularly vulnerable to mild hypoperfusion with a lower threshold for ischaemic injury, as has been suggested in animal models of familial hemiplegic migraine (30). Vascular endothelial dysfunction, altered vasoreactivity, and structural variations in collateral flow pathways might also mechanistically link WMHs and reduced interictal CBF, as might systemic vascular risk factors. Indeed, population-based studies of brain perfusion have shown reduced CBF in subjects with worse cardiometabolic risk factor profiles (31). Additionally, it is possible that lower CBF is the result, rather than the cause, of white matter injury. Finally, it is possible that our results could be due to chance; our analysis of the relationship between CBF and WMHs should necessarily be considered exploratory, given its post-hoc nature.
In summary, our results do not show a significant increase in WMH burden in migraine compared to control subjects, although given the observed point estimates and wide CIs, our results do not contradict prior reports of this association either. Resting CBF was similar between migraine and control subjects, and was consistent with the results reported in the large Rotterdam study for subjects having experienced migraine attacks within the past year, confirming that any changes in interictal CBF in migraine are likely to be quite small. We found significantly reduced CBF in MWA subjects with high WMH load, suggesting that, in this group, hypoperfusion may be mechanistically linked with the formation of white matter lesions. However, given the exploratory nature of our analysis, replication in additional larger datasets is necessary before this finding could be considered conclusive.
Key findings
White matter hyperintensity load increases strongly with older age. Cerebral blood flow is significantly lower in migraine with aura subjects with high white matter hyperintensity load. There is no association between cerebral blood flow and white matter hyperintensity load in control or migraine without aura subjects.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: grants from the National Institute of Neurological Disorders and Stroke (NS061572 to BC; NS058386, NS045839, and RR002305 to JD).
