Abstract
Aim
We aimed to identify clinical characteristics and risk factors associated with onset of high-altitude headache (HAH) after acute exposure at 3700 m.
Method
In two hours, 163 individuals ascended by plane to 3700 m. Demographic information, physiological and psychological measurements, cognitive function, physical work capacity tests and profile of mood states within one week prior to the departure and within 24 hours after arrival were examined.
Results
HAH patients featured significantly higher vertebral artery diastolic velocity (Vd), heart rate (HR) and pulmonary artery diameter. HAH was also associated with a more negative mood state, including scores for tension anxiety, depression, hostility, fatigue and confusion, as well as lower vigor (all p values <0.05). Furthermore, negative emotions were positively related to HAH severity. HAH slightly decreased cognitive functioning. HR, Vd, lack of vigor, confusion and self-reported anxiety (all p values <0.05) were independent risk factors for HAH. We have identified three independent baseline predictors for HAH including internal diameter of the left ventricle (LVD), Athens Insomnia Scale (AIS) and confusion score.
Conclusions
Higher HR, Vd, confusion and self-reported anxiety and insufficient vigor were independent risk factors for HAH. Furthermore, higher baseline LVD, AIS and confusion score are independent predictors of HAH.
Introduction
Headache has been considered the most persistent and predominant complaint in mountaineers (1–3), and it is a primary symptom of acute mountain sickness in the diagnostic criteria of the Lake Louise Scoring System (4–7). High-altitude headache (HAH) has been described as a headache that occurs within 24 hours after ascending to 2500 m or higher and that subsides within eight hours after descending from such an elevation (8,9).
Previous studies have investigated the epidemiological characteristics, clinical manifestations, risk factors, prevention and treatment of HAH and identified some clinical characteristics of HAH (3,10,11). HAH usually presents as a sudden attack and shares some common clinical features with a primary headache (12,13). Several independent risk factors of HAH, including migraine history, higher heart rate (HR), lower pulse oxygen saturation (SpO2), insomnia and anxiety (8,14,15), have been identified. The underlying mechanisms, clinical characteristics and risk factors of HAH are not fully understood. Furthermore, most previous studies have not investigated HAH after acute exposure; thus, HAH may not be typical (1,16,17). In particular, traditional, simple, and useful noninvasive physiological parameters (such as echocardiogram) have not been fully investigated, and emotions and psychological patterns have not received sufficient attention. In addition, the effect of HAH on individuals’ ability to perform mental (cognitive functioning) and physical work is an important issue that should be evaluated.
Given the crucial need to investigate the clinical characteristics of HAH, we postulated that HAH may be closely associated with physiological and psychological measurements. Therefore, we performed the current study to identify heterogeneity in the clinical presentation of HAH to facilitate our understanding of the disease process, to provide insight into the specific phenotypes associated with the course of the disease and to identify the factors associated with onset of HAH.
Methods
Participants and procedures
Participants
In total, 163 participants were recruited according to the inclusion and exclusion criteria (detailed protocols are shown in Supplementary Appendix 1) in June and July 2012 in Chengdu, Sichuan province (sea level, average 500 m) and Lhasa (3700 m above sea level, section 1.1 in Supplementary Appendix 1).
The study was reviewed and approved by the Ethics Committee of Xinqiao Hospital, Third Military Medical University. The study was thoroughly explained to all individuals who agreed to participate, and all participants signed informed consent forms before their examinations.
Procedures and clinical data collection
The field trials were performed within one week prior to the flight (a two-hour plane ride transported the participants to high altitude June 29 and July 1, 2012, respectively) and within 18 to 24 hours after arrival at 3700 m. The HAH assessments, physiological and psychological measurements, were performed in the morning on the second day after high-altitude exposure (section 1.2 in Supplementary Appendix 1).
Demographic data (i.e. age, body mass index (BMI), smoking history and alcohol consumption) and HAH (0 = no headache; 1 = mild headache; 2 = moderate headache; 3 = severe headache) were recorded by using structured case-report form questionnaires (Supplementary Appendix 2). The systolic blood pressure (SBP), diastolic blood pressure (DBP), HR, SpO2, echocardiogram (end-diastolic internal diameters of the left atrium (LA), left ventricle (LVD), right atrium (RA), right ventricle (RV) and pulmonary artery (PA) and stroke volume (SV), ejection fraction (EF)) and Transcranial Doppler sonography (systolic and diastolic velocities (Vs and Vd) in the vertebral artery) examinations were also performed.
Self-Rating Anxiety Scale (18) (SAS, Supplementary Appendix 3), Epworth Sleepiness Scale (19) (ESS, Supplementary Appendix 4), Athens Insomnia Scale (20) (AIS, Supplementary Appendix 5), Fatigue Self-Assessment Scale (21) (FSAS, Supplementary Appendix 6) and profile of mood states (22) (POMS, Supplementary Appendix 7) were also tested. The mental effort required by the participants to perform certain tasks or their cognitive functioning was evaluated using five items (Supplementary Appendix 8). Finally, physical work capacity at an HR of 170 beats per minute (PWC170) was also performed (Supplementary Appendix 9). The questionnaire forms were translated into Chinese before the measurements.
The HAH has been diagnosed based on International Classification of Headache Disorders, second edition (ICHD-2) (9, fulfills at least two out of the five criteria in section 1.3 in Supplementary Appendix 1). However, HAH in this study was not diagnosed strictly according to ICHD-2 because of difficulties of field study and the clinical research.
Statistical analysis
Normally distributed variables, including age, BMI, HR, SBP, DBP, SpO2, LA, LVD, RA, RV, PA, SV, EF and PWC170, were expressed as the mean ± standard deviation (SD). Non-normally distributed variables were expressed as medians (quartile range). Other statistical methods and protocols are summarized in section 1.4 in Supplementary Appendix 1 and Figure 1.
Flowchart of this study.
Results
Demographic data
The mean age and BMI of the participants were 22.2 ± 3.3 years and 21.6 ± 2.1 kg/m2, respectively. Altogether, 24.7% and 64.0% of the participants smoked and drank alcohol, respectively. The incidence of HAH was 71.3%.
Clinical characteristics
Thirty-eight individuals reported frontal headache, 31 reported bilateral headaches, 25 of these were frontal-temporal headache, and 13 reported other types of headache locations. Furthermore, 65 participants experienced dull headache, 32 of these were pressing headache, and 10 participants reported other quality headaches. Additionally, 103 patients reported mild or moderate headache, and 95 individuals thought their headache was slightly aggravated after the PWC170 test. However, the intensity was not altered significantly after exertion.
Differences in the baseline measurements between the HAH+ and HAH– groups
Age differed significantly between the HAH+ and HAH– groups (p = 0.001).
Differences of each variable between HAH+ and HAH– groups.

AIS: Athens Insomnia Scale; BMI: body mass index; DBP: diastolic blood pressure; EF: ejection fraction; ESS: Epworth Sleepiness Scale; FSAS: Fatigue Self-Assessment Scale; HAH: high-altitude headache; HAH+: with HAH or HAH positive; HAH–: without HAH or HAH negative; HR: heart rate; LA: internal diameter of the left atrium; LVD: internal diameter of the left ventricle; PA: internal diameter of the pulmonary artery; POMS: profile of mood status; PWC170: physical working capacity at an HR of 170 beats/min; RA: internal diameter of the right atrium; RV: internal diameter of the right ventricle; SAS: Self-Rating Anxiety Scale; SBP: systolic blood pressure; SpO2: pulse oxygen saturation; SV: stroke volume; Vd: diastolic velocities; Vs: systolic.
Age: year; BMI: kg/m2; smoking and drinking: %; SBP and DBP mmHg; SpO2: %; HR: beats per minute; LA, LVD, RA, RV and PA: mm; SV: ml/min; EF: %; Vs, Vd and Vm: cm/s; PWC170: k.m/min.
p value is 0.05 or less; bp value is 0.01 or less.
Differences in variables between the HAH+ and HAH– groups at 3700 m
DBP was significantly higher in the HAH+ individuals than in the HAH– individuals (p = 0.039), whereas SBP was not. The HAH+ participants were also characterized by a faster HR (p = 0.021). However, as to echocardiographic parameters, LA, LVD, RA, RV, PA, EF and SV were not significantly different between the HAH+ and HAH– participants (all p > 0.05, Table 1). A novel finding was that Vd was faster in the HAH+ patients than in the HAH– patients (p = 0.001), whereas Vs was similar between the two groups. AIS scores were also much higher in the HAH+ group than in the HAH– group, although ESS scores were similar between the groups. Moreover, scores for tension anxiety, depression, hostility, fatigue and confusion (all p < 0.05) were significantly higher in the HAH+ group, whereas scores for vigor were much lower. There were no significant differences in the cognitive function parameters between HAH+ and HAH– groups. Finally, the physical work capacity variables were similar between the two groups (Table 1).
Relationship between HAH and the baseline parameters
Relationships between HAH score and all of the parameters.

AIS: Athens Insomnia Scale; BMI: body mass index; DBP: diastolic blood pressure; EF: ejection fraction; ESS: Epworth Sleepiness Scale; FSAS: Fatigue Self-Assessment Scale; HAH: high-altitude headache; HAH+: with HAH or HAH positive; HAH–: without HAH or HAH negative; HR: heart rate; LA: internal diameter of the left atrium; LVD: internal diameter of the left ventricle; PA: internal diameter of the pulmonary artery; POMS: profile of mood status; PWC170: physical working capacity at an HR of 170 beats/min; RA: internal diameter of the right atrium; RV: internal diameter of the right ventricle; SAS: Self-Rating Anxiety Scale; SBP: systolic blood pressure; SpO2: pulse oxygen saturation; SV: stroke volume; Vd: diastolic velocities; Vs: systolic.
p value is 0.05 or less; bp value is 0.01 or less.
Associations between HAH and the parameters after high-altitude exposure at 3700 m
Regarding the basic physiological measurements, DBP, HR, PA and Vd (all p values <0.05) were closely related to HAH severity. Scores on the AIS and SAS were also associated with HAH. Further, all the POMS factors were closely correlated with HAH: tension anxiety, depression, hostility, fatigue, confusion and vigor (all p values <0.05). Regarding cognitive functioning, only pursuit aiming capacity (r = −0.161, p = 0.047) was negatively related to HAH (r = −0.187, p = 0.022). Scores on the FSAS were also closely associated with HAH (r = 0.176, p = 0.031) (Table 2).
Only alterations of HR (21.0 (14.0) vs. 17.0 (15.0) beats/min, p = 0.017), SAS (2.0 (5.0) vs. 1.0 (2.0), p = 0.015) and LVD (0.0 (3.0) vs. 1.0 (5.0) mm, p = 0.035) from sea level to 3700 were different between HAH+ and HAH– groups.
Logistic analyses for HAH
Univariate logistic regression for each variable at baseline.

AIS: Athens Insomnia Scale; BMI: body mass index; CI: confidence interval; DBP: diastolic blood pressure; EF: ejection fraction; ESS: Epworth Sleepiness Scale; FSAS: Fatigue Self-Assessment Scale; HAH: high-altitude headache; HAH+: with HAH or HAH positive; HAH–: without HAH or HAH negative; HR: heart rate; LA: internal diameter of the left atrium; LVD: internal diameter of the left ventricle; PA: internal diameter of the pulmonary artery; POMS: profile of mood status; PWC170: physical working capacity at an HR of 170 beats/min; RA: internal diameter of the right atrium; RV: internal diameter of the right ventricle; SAS: Self-Rating Anxiety Scale; SBP: systolic blood pressure; SpO2: pulse oxygen saturation; SV: stroke volume; Vd: diastolic velocities; Vs: systolic.
p value is 0.05 or less; bp value is 0.01 or less.
Univariate logistic regression for each variable at 3700 m.
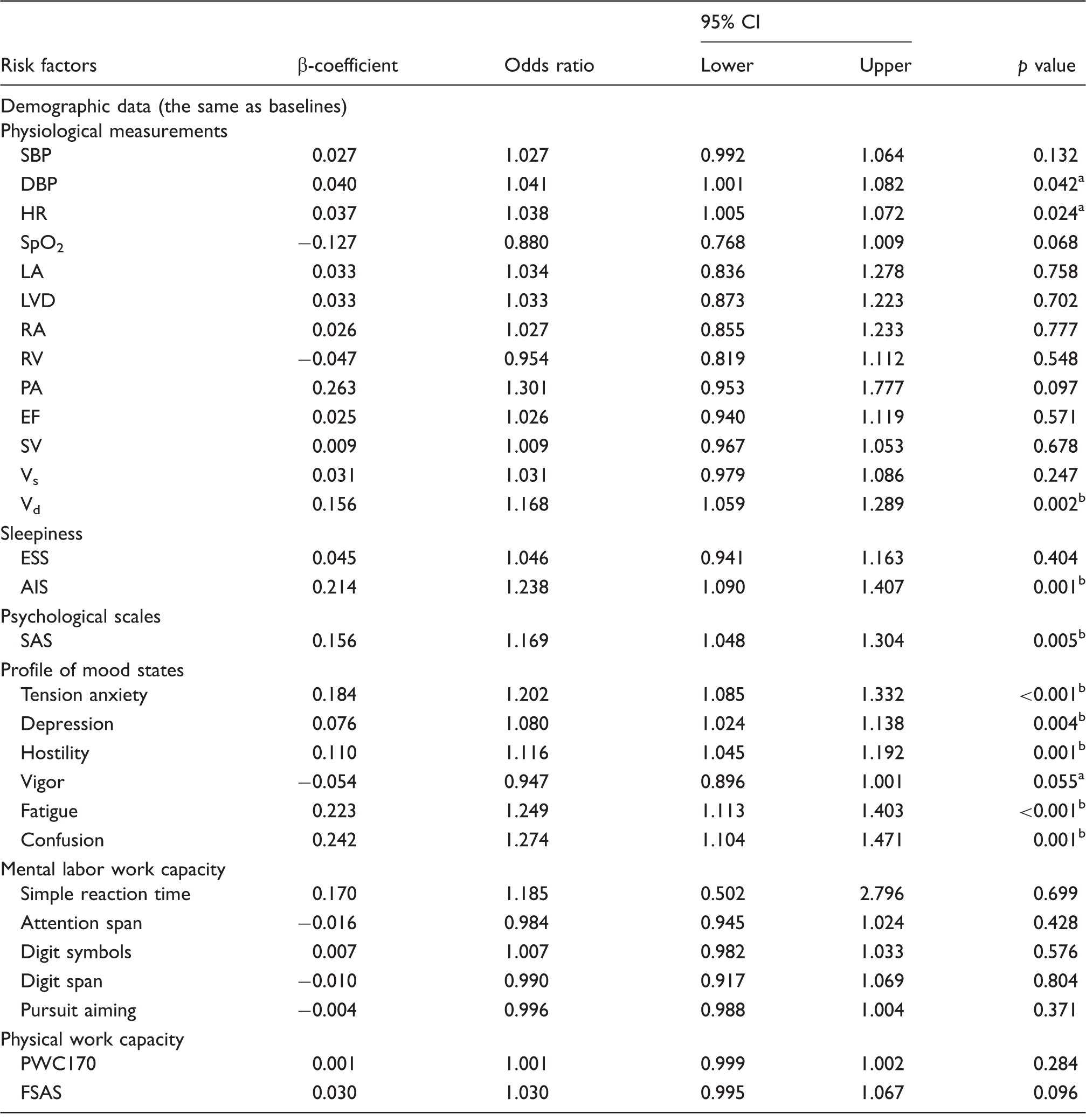
AIS: Athens Insomnia Scale; BMI: body mass index; CI: confidence interval; DBP: diastolic blood pressure; EF: ejection fraction; ESS: Epworth Sleepiness Scale; FSAS: Fatigue Self-Assessment Scale; HAH: high-altitude headache; HAH+: with HAH or HAH positive; HAH–: without HAH or HAH negative; HR: heart rate; LA: internal diameter of the left atrium; LVD: internal diameter of the left ventricle; PA: internal diameter of the pulmonary artery; POMS: profile of mood status; PWC170: physical working capacity at an HR of 170 beats/min; RA: internal diameter of the right atrium; RV: internal diameter of the right ventricle; SAS: Self-Rating Anxiety Scale; SBP: systolic blood pressure; SpO2: pulse oxygen saturation; SV: stroke volume; Vd: diastolic velocities; Vs: systolic.
p value is 0.05 or less; bp value is 0.01 or less.
Adjusted logistic regression for HAH at baseline and 3700 m.

AIS: Athens Insomnia Scale; CI: confidence interval; HAH: high-altitude headache; HR: heart rate; LVD: internal diameter of the left ventricle; SAS: Self-Rating Anxiety Scale; Vd: diastolic velocities; Vs: systolic.
p value is 0.05 or less; bp value is 0.01 or less.
In adjusted logistic analysis, increased HR, higher Vd, a lack of vigor, increased confusion and a higher SAS score at 3700 m (all p < 0.05) were independent risk factors for HAH (Table 5).
Discussion
Several risk factors associated with HAH, including demographic factors, basic physiological measurements, psychological factors, POMS, and mental and physical work capacity, have been indicated in the present study.
HAH is associated with basic physiological parameters
HAH was associated with baseline parameters of the echocardiogram, including LVD, RA, RV and SV. The results suggest that hypoxic stress may activate the compensatory function of the cardiovascular system. These baseline echocardiogram variables may be used to predict HAH. The baseline LVD was also demonstrated to be a predictor for HAH, indicating that the diastolic function of the left ventricle at sea level may influence HAH via systemic and cerebral circulation.
Increased HR has been identified as an independent risk factor for HAH (14,15). HAH is associated with higher HR, and higher HR is an independent risk factor for HAH. However, we found that DBP is closely associated with HAH, which is a novel result; DBP may lead to HAH through the vasodilatation dysfunction of systemic arteries. This finding contrasts with other reported results (15), and this inconsistency may be due to differences in sample size. SpO2 was found to be significantly lower in HAH+ individuals, which accounted for the hypobaric hypoxia-induced headache (1).
Furthermore, PA was positively correlated with HAH severity demonstrating the role of vasodilatation in HAH. However, it is inconsistent with normal conceptions of the relationship between DBP and HAH (15). This divergent result may be attributed to the relationship between systemic and pulmonary circulation.
In addition, cerebral hemodynamics, indicated by the Vd, was found to be strongly associated with HAH. The HAH+ participants had a significantly higher Vd, which may be attributed to the cerebral artery vasodilatation dysfunction that caused hypoxia-induced expression of vasoconstrictors and vasodilators and activation of the sympathetic nervous system (5,23). The above results are consistent with our previous study on the cerebral hemodynamic characteristics of acute mountain sickness (24,25). The evaluated velocity of cerebral arteries may lead to alterations in intracranial pressure and cerebral perfusion pressure, both of which can cause headaches (26–29).
Lastly, the changes in HR and LVD were also different between HAH+ and HAH– groups. These results may indicate that the variant of heart function may be protective factors for high altitude.
The role of psychological factors in patients experiencing HAH
Anxiety often occurs after high-altitude exposure because of hypobaric hypoxia and cold stresses (29). Both baseline and followed-up psychological factors were associated with HAH. The SAS scores were significantly higher in HAH+ patients than in HAH– individuals (15,29,30). The anxiety may affect the self-reported symptom of headache. The alteration of SAS was more various in HAH+ groups indicting the importance of the stability of SAS in development of HAH. We included other psychological factors, such as tension anxiety and depression, which were measured by the POMS.
The incidence of HAH is closely associated with emotional status
HAH is a subjective, self-reported symptom. Thus, emotional status may play a major role in HAH. Few studies have devoted proper attention to the relationship between HAH and emotional status. We assessed emotional status using the POMS. The emotional status of some of the participants can be attributed to psychological factors. Negative emotions, including tension anxiety, depression, hostility, confusion and fatigue, were significantly associated with HAH; all of these emotions were significantly higher in the HAH+ group than in the HAH– group, both at baseline and at 3700 m.
Tension anxiety, or anxiety, was closely related to HAH. Travelling to a high altitude exposes individuals to a high stress condition in which anxiety cannot be avoided. This result is consistent with previous studies showing that anxiety is correlated with headache and pain at high altitude (31). Depression has also been demonstrated to be a risk factor for or to contribute strongly to headaches at sea level (32). Furthermore, high-altitude exposure has been shown to lead to new-onset depression (29,33). However, the relationship between HAH and depression has not been widely studied (34–36). Our results indicate that depression is closely correlated with HAH but that it is not an independent risk factor for HAH.
Hostility and confusion are common negative emotional feelings after stress from high-altitude exposure (30). Both of these emotions play critical roles in the development of self-reported HAH. In particular, confusion was found to be an independent risk factor for HAH. Anxiety, depression and confusion also contributed to HAH development, perhaps due to the disequilibrium of expression and the secretion of serotonin, dopamine, acetylcholine and γ-aminobutyric acid (37,38). Additionally, a negative mood state usually results in somatization symptoms such as headache. Thus, the emotional states as a subjective scale may be used to predict HAH and other altitude-related diseases.
People who experienced HAH were found to have high fatigue and low vigor. Such feelings may be ascribed to hypobaric hypoxia, lower SpO2 and other alterations that led the individual to feel powerless, weak and fatigued. As reported, physical work capacity decreases as individuals who have not been exposed to high altitude ascend in altitude, potentially because of a decrease in oxygen and mitochondria stresses (39) that lead to a reduction in energy metabolism.
Physical work and mental exertion contribute to the development of HAH
At sea level and 3700 m, PWC170 was not correlated with HAH. However, another measure of subjective-reported fatigue, FSAS, was closely associated with HAH severity at 3700 m. This result is in line with the POMS result, indicating that HAH has no objective effect on work capacity but that it affects individuals’ subjective feelings and experiences.
Regarding mental exertion, only the pursuit aiming score was negatively related to HAH at 3700 m. This finding indicates that HAH symptoms may cause partial impairment in cognitive functioning, particularly in precise tracking. Other types of cognitive functioning were not associated with HAH, indicating that HAH is a subjective symptom that may not objectively affect physical and mental work capacity. HAH may thus be less severe and fatal than high-altitude cerebral edema (40).
Relationship between sleep and HAH
The association between headache and sleep has previously been found both at sea level and high altitude (15,41). We observed that scores on the AIS were higher in the HAH+ patients and strongly positively correlated with HAH severity both at sea level and 3700 m. However, scores on the ESS and AIS were not independent risk factors for HAH. Severe insomnia may cause subjective complaints of discomfort, including headache. These results are consistent with our previous studies showing that insomnia is an independent risk factor for HAH (15). Likewise, the studies of Dong et al. showed that sleep status is correlated with anxiety and headache at high altitude (31). In addition, the baseline AIS was a predictor of HAH, which may indicate that sleep quality influences the incidence of HAH.
Novel risk factors and predictors for HAH
We have identified three novel risk factors for HAH, including higher Vd and confusion scores and insufficient vigor. Increased posterior circulation was also found to be a risk factor for HAH, which may be explained by clinical presentations of HAH, including pulsating and location and the supply of blood and oxygen. Insufficient vigor may be an independent risk factor for HAH because of the discomfort induced by HAH. Furthermore, confusion, as an important psychological or mood state, affects many diseases, particularly self-reported symptoms.
High-altitude exposure places great stress on people. Thus, the alterations both in physiology and psychology may cover the predictive values of baseline parameters. However, the baseline LVD, AIS and confusion scores were independent predictors of HAH. They were in accordance with the risk of HAH after acute high-altitude exposure. Nevertheless, the confusion score both at baseline and 3700 m was associated with HAH independently, which may be caused by the stress of pre-exposure and after exposure.
Limitations
The sample size was only 163 because the trials to record basic physiological and psychological measurements, mental and physical work capacity, and mood states required substantial time and effort to perform. The participants in our study were all young Chinese men, which could perhaps have generated bias due to age or gender; this aspect should be improved in our future studies. The HAH in this study was not diagnosed strictly based on diagnostic criteria of the ICHD-2. The underlying mechanisms of HAH have not been determined using the POMS; thus, further studies on the basic mechanisms are warranted. Lastly, there was another limitation that the ESS, AIS, Wechsler Adult Intelligence Scale (WAIS) and Wechsler Memory Scale (WMS) questionnaires were valid in English but may not be correctly validated in Chinese, and this should be improved in the future studies.
Conclusions
This observational study showed that HAH is closely associated with emotional status, cerebral artery velocity and physiological measurements. Independent risk factors for HAH include a higher HR, Vd, confusion and SAS score as well as a lack of vigor. Furthermore, higher baseline LVD, AIS and confusion score are independent predictors of HAH.
Article highlights
This article identified physiological and psychological clinical characteristics of high-altitude headache (HAH). HAH was closely associated with emotional states. Baseline diameter of the left ventricle (LVD), Athens Insomnia Scale (AIS) and confusion score were independent predictors for HAH.
Footnotes
Acknowledgments
We would like to thank all the individuals who participated in this study for their support. We are grateful to Dr Ming Li, Mr Can Chen, Mr Guo-Zhu Chen, Mr Rong-Sheng Rao and Mr Cai-Fang Tang for their technical support and the clinical data collection. We also thank Mr Ru-Fu Xu for his suggestions regarding statistics.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Ministry of Health of P.R. China (grant number 201002012) and People’s Liberation Army P.R. China (grant number BWS14J040).
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
