Abstract
Headache is the most prevalent symptom of acute mountain sickness. We conducted a pilot clinical trial at an altitude of 3500 m to evaluate the efficacy of gabapentin in treatment of high-altitude headache (HAH). Twenty-four adult HAH patients (10 female, 14 male; age 18–50 years) were randomly assigned to receive either 300 mg of gabapentin capsule or identical placebo. After 1 h the presence of HAH and need to receive supplementary analgesic were assessed. The duration of the HAH-free phase after taking additional analgesic was also registered. Four patients in the gabapentin group asked for additional analgesics, whereas nine placebo recipients did not find primary medication satisfactory after the first hour of treatment (P = 0.04). The mean HAH-free period was 17.10 h in the gabapentin group, which was significantly higher than in the placebo group with a mean of 10.08 h (P = 0.02). This preliminary observation indicates that gabapentin is effective in treatment and alleviation of HAH.
Introduction
Every year thousands of people who live at low altitude climb high-altitude areas; many of them experience acute mountain sickness (AMS). The most common symptoms are headache, loss of appetite, nausea, fatigue, dizziness, and insomnia that usually appear within the first days of arrival at high altitude (1). AMS is not only uncomfortable; it is also partly responsible for an increased mortality at high altitude (2). Headache is the most prevalent symptom of AMS (1, 2).
Gabapentin was originally developed as a γ-aminobutyric acid (GABA) mimetic compound with potent anticonvulsive effects. Gabapentin has been recently employed in the treatment and prophylaxis of migraine and other types of headache (3). We conducted a randomized, placebo-controlled, double-blind, pilot clinical trial in Tochal mountain hotel (Tehran, Iran) clinic to evaluate the efficacy of low-dose gabapentin in treatment of high-altitude headache (HAH).
Methods
Tochal hotel is located at an altitude of 3500 m above sea level and designed to host ordinary guests and skiers. Individuals were transferred within 45–60 min from a height of 1600 m to 3500 m using cable cars. Inclusion criteria were age ≥18 years and suffering from HAH before 24 h of ascent. Exclusion criteria were severe cardiac, pulmonary or liver disease; severely impaired kidney function; history of migraine; current history of alcohol or drug abuse; known allergy to gabapentin; and treatment with anticonvulsants or tricyclic antidepressants. The study was conducted in accordance with the Declaration of Helsinki (4). The study was evaluated and approved by Iranian Centre of Neurological Research (Iman Hospital) Institutional Review Board, Tehran University of Medical Sciences.
Participants and intervention
Twenty-four adult HAH patients (10 female, 14 male; average age 29.1 ± 1.7 years; range 18–50 years) came to Tochal hotel clinic from 1 to 7 January and 10 to 20 February 2007; none of them met exclusion criteria. All participants gave written informed consent prior to study entrance. Patients were randomly assigned (1 : 1 ratio) to receive either 300 mg of gabapentin capsule or identical monohydrate lactose capsule as placebo (Darou Darman Pars Pharmaceuticals, Tehran, Iran) in a non-stratified randomization method. Medications were in identical opaque boxes labelled with randomization codes that were not disclosed to investigators or assessor. Only the pharmacist who provided the drugs knew the details of the computer-generated randomization codes. All patients received 400 mg ibuprofen after 1 h of gabapentin/placebo intake and were followed up for recurrence of HAH and other AMS symptoms. All assessments were made by one assessor (S.J.). Participants took single-dose medication in Tochal hotel clinic in the presence of the investigators (F.G. & S.S.). A written instruction sheet was given to guests, asking them not to take analgesics, alcohol, sedatives or other kinds of medicine without the permission of the investigators.
Assessment
Pretreatment variables included age, gender, heart rate, blood pressure, smoking habits, severity of HAH based on visual analogue scale (VAS) pain score, and AMS score based on the Lake Louise acute mountain sickness scoring system (5). The assessor visited all patients after 1 h of treatment and asked them about the presence and severity (VAS pain score) of HAH. Whether a HAH patient needed supplementary analgesics after 1 h of gabapentin/placebo commencement and the duration of the HAH-free phase after ibuprofen initiation were considered as primary end-points. A maximum follow-up duration of 24 h was determined; however, no patient stayed at the altitude of 3500 m for more than 18 h after ibuprofen intake (average duration 12.2 ± 5.7 h; range 3–18 h). AMS incidence (Lake Louise acute mountain sickness score of ≥3 with headache and at least one other symptom) and AMS severity (score of ≥5) were recorded as secondary end-points. An interview was held to ask about HAH-free phase length and AMS symptoms after ibuprofen intake at the end of each participant's stay in the hotel in order to investigate if gabapentin (GB) could stop or alleviate the development of AMS.
Statistics
Continuous variables are expressed as mean ± SD. Means were compared by non-parametric Mann–Whitney U-test. χ2 test (Fisher's exact if appropriate) was used to compare percentages. Mean survival time (duration of HAH-free period) was calculated by Kaplan–Meier method and reported with 95% confidence interval (CI). Log rank test was used to compare mean HAH-free period between gabapentin and placebo. All P-values were two-tailed and P < 0.05 was considered to be significant. No individuals were lost to follow-up.
Results
No difference in baseline characteristics was found between the two groups of gabapentin and placebo recipients (Table 1). Four participants in the gabapentin group needed further pain-relieving agents after the first hour of treatment, whereas nine placebo recipients did not find medication satisfactory after 1 h and asked for supplementary ibuprofen (P = 0.041). The mean HAH VAS score was 2.92 ± 2.91 for gabapentin and 4.75 ± 3.11 for placebo recipients after 1 h of treatment (P = 0.12). The mean HAH-free period was 17.10 h (95% CI 15.98, 18.22) in the gabapentin group, which was significantly higher than in the placebo group with a mean HAH-free period of 10.08 h (95% CI 6.22, 13.94; P = 0.02) (Fig. 1).

High-altitude headache-free phase after the first hour of ibuprofen initiation.
Baseline characteristics
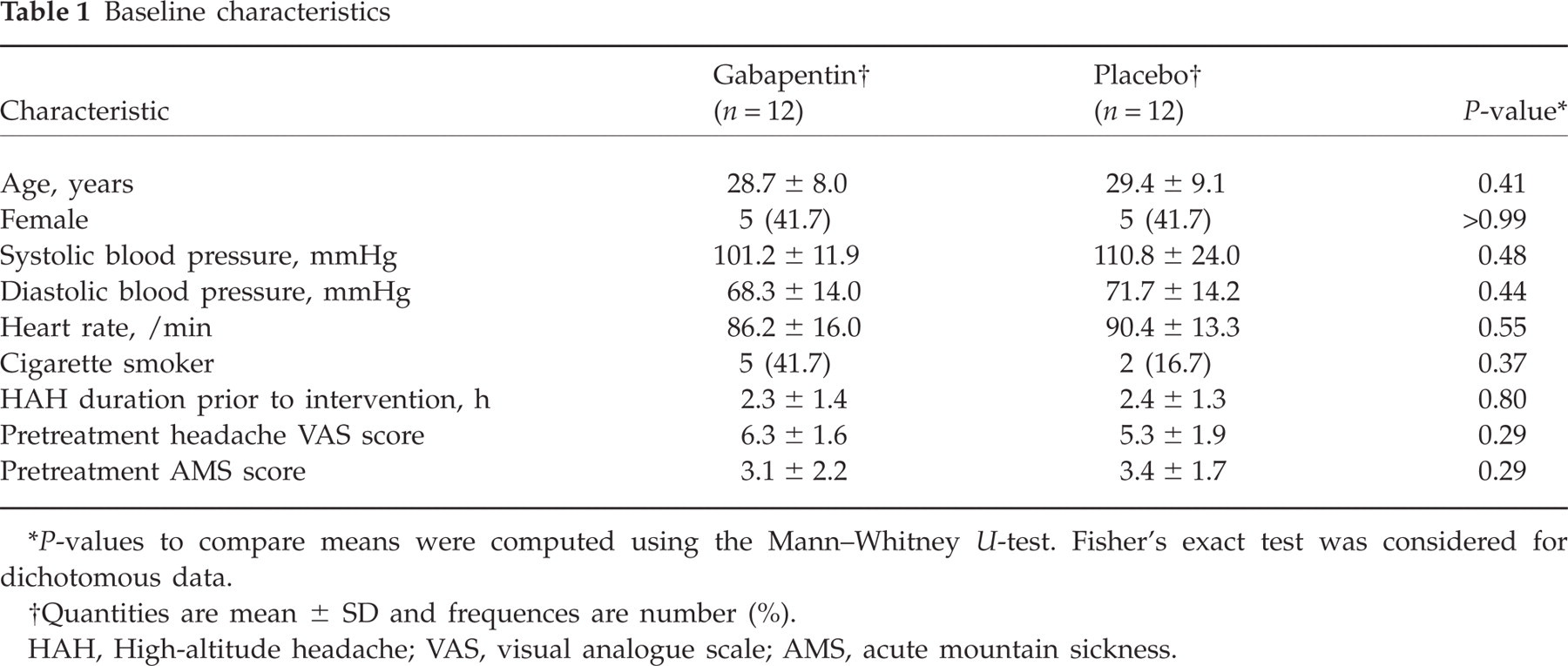
P-values to compare means were computed using the Mann–Whitney U-test. Fisher's exact test was considered for dichotomous data.
Quantities are mean ± SD and frequences are number (%).
HAH, High-altitude headache; VAS, visual analogue scale; AMS, acute mountain sickness.
AMS developed in eight (33.3%) participants between the headache recurrence point and the end of the participant's stay at the altitude of 3500 m. Three (12.5% of total participants) patients had severe AMS. There was only one (8.3%) participant in the gabapentin group who had suffered from AMS after ibuprofen initiation, which was significantly lower than in the placebo group, in which seven (58.3%) reported experiencing AMS (P = 0.03). Severe AMS was reported by three (25.0%) placebo recipients, whereas none of the gabapentin recipients met the criteria for severe AMS (P = 0.21). No noteworthy adverse effect was reported by participants in either treatment group.
Discussion
This preliminary observation, which requires confirmation in further large-scale controlled trials, indicates that gabapentin is effective in treatment and alleviation of HAH and AMS. Why HAH develops is not fully understood, but hypoxia-induced cerebral vasodilation or its effectors, such as nitric oxide, probably produce the headache, perhaps through the activation of the trigeminovascular system (1).
Gabapentin is a structural analogue of GABA, which was initially approved for use in epileptic syndromes. The modality of action in the treatment of neuropathic pain is still a matter of debate (6). It has been used for migraine prophylaxis (7), trigeminal neuralgia (8), cluster headache (9), hemicrania continua (10) and idiopathic stabbing headache (11), with promising results. The precise mechanisms of action of gabapentin are still not completely defined but are probably related to both peripheral and central pathways of pain suppression (12). The anti-allodynic effect of the drug, related to antagonism of glutamate N-methyl-D-aspartic acid (NMDA) receptors and blockade of calcium channels in the central nervous system, particularly in the hippocampal area, has been shown to be the main mechanism of action of gabapentin (12). Other potential mechanisms include enhancement of GABA-ergic pathways adjusting cortical reaction to painful stimuli (13). Gabapentin has few side-effects and minor interactions with other drugs when used for the treatment of pain (14).
Similar to previous studies, our findings confirm that calcium channels in the central nervous system may play a causative role in the pathophysiology of HAH (15). Furthermore, these results suggest that glutamate NMDA receptors and GABA-ergic pathways might be responsible, at least in part, for HAH and AMS pathophysiology.
If this hypothesis is correct, one could propose that gabapentin might be an appropriate alternative to non-steroidal anti-inflammatory drugs and glucocorticoids, the most accepted medications in treatment of HAH (16), while gastrointestinal problems, asthma and hyper-allergic conditions, and sinusitis headaches that may resemble HAH, are of concern after intake of such drugs. Further studies are needed to confirm these results. Moreover, higher dosages of gabapentin in treatment of HAH and its usage in prevention of AMS and HAH should be subject to further research. Finally, the data suggest that the combination of ibuprofen and gabapentin is better than either drug alone. This combination might be worth testing in the future.
Acknowledgements
We acknowledge Dr Alireza Madjd, managing director of Darou Darman Pars Pharmaceuticals, for providing gabapentin and placebo.
