Abstract
Purpose
The purpose of this article is to investigate possible differences in effect between three types of masks in the acute treatment of cluster headache (CH).
Patients and methods
Fifty-seven CH patients according to ICHD-II-criteria participated in a single-blinded, semi-randomized, placebo-controlled, crossover inpatient study, and 102 CH attacks were treated with 100% oxygen delivered by demand valve oxygen (DVO), O2ptimask or simple mask (15 liters/min) or placebo delivered by DVO for 15 minutes. Primary endpoint: Two-point decrease of pain on a five-point rating scale within 15 minutes.
Results
Only 10 CH patients had multiple attacks and reached the point of placebo. There were no significant differences between masks in the primary endpoints (p = 0.412). After 15 minutes 48% had a two-point decrease using the DVO compared to 45% with placebo (p = 0.867). After 30 minutes 68% were pain free or had pain relief using DVO and 45% by placebo (p = 0.061). The DVO was preferred by 62% compared to 5% and 33% for simple mask (p < 0.0001) and O2ptimask (p = 0.061). In the first attack the DVO was significantly better at achieving pain relief at 15 minutes (p = 0.018). Treatment with DVO or O2ptimask reduced the need for rescue medication compared to the simple mask (23%, 19%, 50%, respectively). No treatment-related adverse events were observed.
Conclusion
The primary endpoint with pain relief at 15 minutes was non-significant; however, a post hoc analysis of the first attack significantly favored DVO. Further, therapy by O2ptimask and DVO resulted in a decreased need for rescue medication. We recommend that CH patients be offered DVO or O2ptimask before oxygen therapy is abandoned.
Introduction
Cluster headache (CH) is a severe primary headache belonging to the trigeminal autonomic cephalalgias. Treatment of CH consists of both preventive and acute medication. Acute treatment aims to relieve the individual attack or reduce pain. First-line treatment is subcutaneous triptan (1–3) and 100% oxygen inhalation at a flow rate of 6–15 l/minute (l/min) (4–8). First-line preventive treatment is verapamil, whereas steroids or greater occipital nerve-block are used for the transitions between acute and preventive therapy (9). All three types of treatment can be combined to relieve the patient of the CH burden and increase his or her quality of life.
CH is reportedly the only trigeminal autonomic cephalalgia that responds to oxygen treatment (10). Oxygen as a therapeutic agent of headache has been known since 1940 (11); however, it was widely accepted for CH only after Kudrow’s study in 1981 (4,11). Later studies have found higher flow rates (12–15 l/min) to be equal to or even more efficient than 7 l/min (6,7). In a recent study demand valve oxygen (DVO) was introduced (8). The study included four patients and indicated that the DVO may be more efficacious than continuous high-flow oxygen treatment (15 l/min) (8). However, no larger comparative studies of any mask types are yet available. Based on our clinical experience, the authors hypothesized that DVO is more effective for pain relief than other mask-types.
The objective of this trial was to investigate possible differences in effect between three types of masks in the treatment of acute CH attacks (Figure 1).
The three different mask types. Simple open mask: simple plastic mask. Standard equipment. Continuous oxygen delivered at a flow rate of 15 l/min. O2ptimask: Specialized non-rebreather mask with a 3 L reservoir, where dilution of oxygen is minimized. Continuous oxygen delivered at a flow rate of 15 L/min. Ultraflow™ Oxygen Demand Valve: Delivers oxygen according to need (tidal volume and respiration rate), thus is not limited to a specific flowrate. The DVO ensures a FiO2 of 100%. It supports hyperventilation and it is speculated this might improve the treatment response (8). Pictures with permission of AGA, Linde Healthcare A/S.
Methods
Design and endpoints
The study was designed as a single-blinded, semi-randomized, placebo-controlled, crossover study (Figure 2). This crossover design is suitable for this trial as oxygen is assumed to have a very short wash-out period. To eliminate a possible effect of the previous treatment and the risk of treating a rebound headache, the treatments were separated by a minimum of three hours. A physician or a trained medical student (MB, NL or AP) included, randomized and instructed the patients carefully in a standardized manner. The treatment sequence was block-randomized (excluding DVO with placebo). The sequences consisted of a random sequence of the simple facemask (SM), the O2ptimask™ (OM) and the DVO always succeeded by DVO with placebo (Figure 2). Placebo was placed last in the sequence so that in the case of patients suffering fewer than four attacks, they would have tried as many of the active masks as possible. Author MB generated the random sequence with the Microsoft Excel SLUMP function. The list was kept in the trial master file during the trial. Blinding of the mask type currently used was deemed impossible but patients were blinded to the contents of the gas cylinders. The gas cylinders were exactly the same size and covered with black plastic wrapping during the trial. A sticker with the numbers 1–4 distinguished the cylinders. The primary endpoint was a two-point reduction on a five-point rating scale within 15 minutes (min). The secondary endpoint was patient mask preference. Other endpoints were: 1) pain freedom after 15 min, 2) pain freedom after 30 min, 3) rebound within three hours, 4) time from pain start to meaningful pain relief, 5) rescue medication at 15 min and 6) re-occurrence of CH attacks within 24 hours.
Study design. Overview of the study design and the patient flow through the trial. After inclusion the participants, were randomized to one of six possible sequences (1: Simple face mask, 2: O2ptimask. 3: Demand Valve Oxygen, 4: Demand Valve with placebo). Attacks 1 to 3 was treated with 100% oxygen. Attack 4 was always treated with placebo. After two weeks the participants were contacted for follow-up by telephone.
Setting
The study took place as an inpatient study at the Department of Neurology at Rigshospitalet. When the CH patient felt a CH attack, he or she called the staff and immediately began treatment. The patients were instructed to initiate treatment as soon as they felt the attack, i.e. not to wait until staff arrived. The staff generally arrived within 0.5–2 min. After 15 min, the patient was offered rescue medication or could choose to continue the trial therapy. The intensity of the CH attack was rated on a five-point rating scale (0-none, 1-mild, 2-moderate, 3-severe, 4-very severe). Trained staff assisted and recorded the precise start and end of the CH attack; however, it was the CH patients who defined start and finish. The staff also observed the patients at 0, 5, 15 and 30 min as a minimum.
Patients and inclusion
The inclusion criteria were age between 18 and 65 years; CH diagnosis according to International Classification of Headache Disorders, second edition (ICHD-II) criteria (12); regular attack and cluster frequency for at least two years; average attack frequency of two to eight attacks per day prior to inclusion; cluster duration of more than two weeks; ability to differentiate between CH and other primary headaches and acceptance of the trial conditions after oral and written information. Exclusion criteria were changes in prophylactic treatment one week prior to participation; pregnancy or nursing; serious somatic or psychiatric disorders; moderate, severe or very severe chronic obstructive pulmonary disease; chronic primary or secondary headache (more than 14 days/month) (other than CH); alcohol overuse and non-acceptance of the trial conditions.
Eligibility criteria changed during the trial to include both chronic cluster headache (CCH) and episodic cluster headache (ECH) patients to increase recruitment, whereas the first edition of criteria included only ECH patients. We included 57 CH patients from the Danish Headache Center, a tertiary headache center (13), between June 2012 and December 2014. Hereof, 31 CH patients also participated in an inpatient sleep study (Project ID: H-2-2012-016). Twenty-six CH patients participated only in the mask comparison trial (Figure 3). A neurologist and headache specialist had verified the diagnosis. Patient history was taken at time of inclusion and details regarding prior use of treatment were supplied in an extensive questionnaire (H2-2012-016). The participants were followed up by a structured telephone interview two weeks after administration of treatments. Patient inclusion ended when we reached 100 CH attacks.
Flowchart of inclusion. In total, 107 Cluster Headache (CH) patients were invited to the study, hereof only 57 CH-patients signed the informed consent form. 42 CH-patients treated at least one CH-attack. The discrepancy is due to two exclusions and 13 CH-patients remaining headache free during the study period. The cross-over was incomplete and only 10 CH-patients completed the cross over and treated 4 CH-attacks.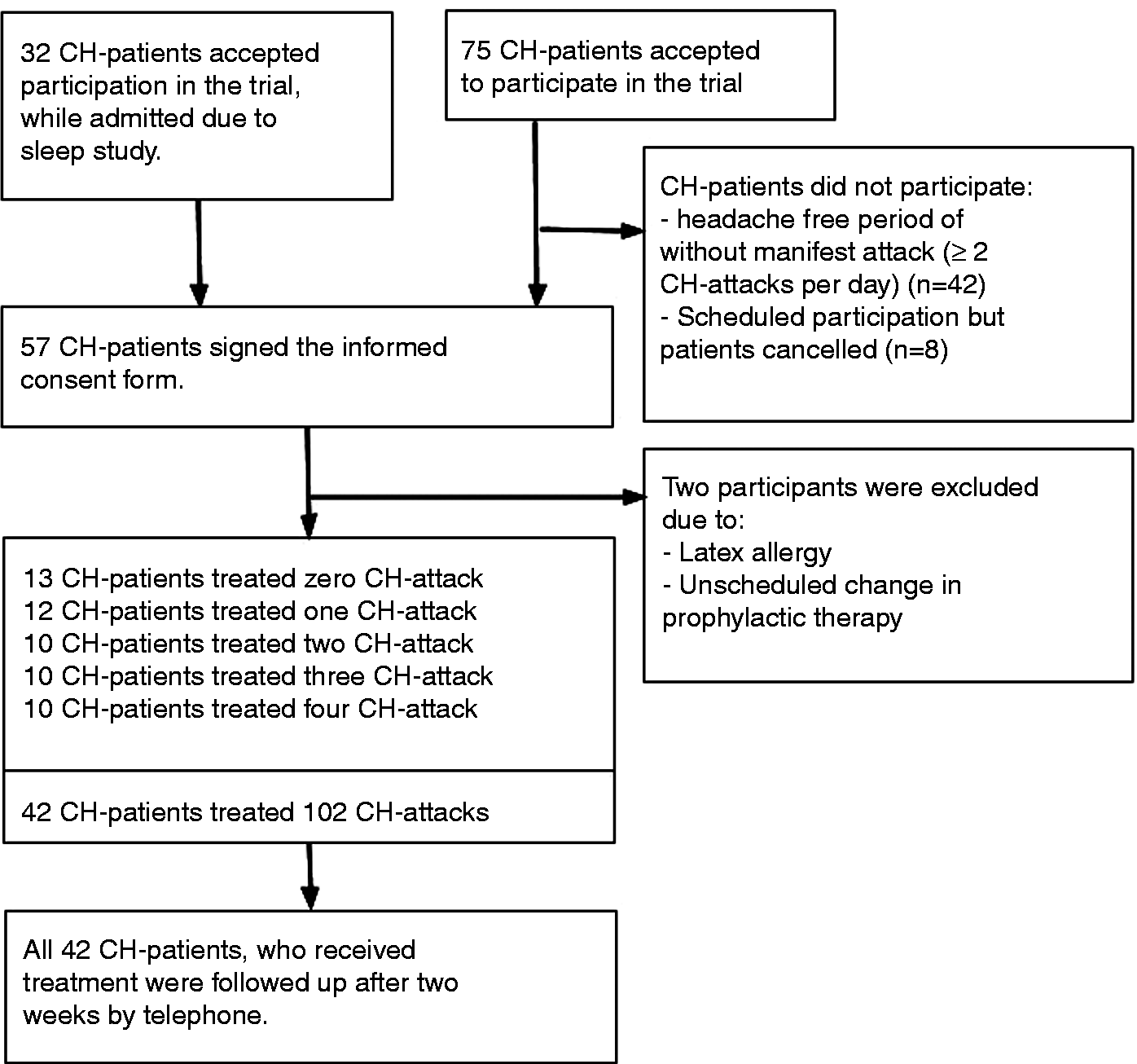
Intervention and equipment
Patients inhaled 100% oxygen delivered by three facemasks for 15 min at the beginning of a CH attack and placebo for one attack. The facemasks were: a simple mask (SM) (Linde Healthcare, AGA A/S), an O2ptimask™ (OM) (Linde Healthcare, AGA A/S), or an Ultraflow™ Oxygen Demand Valve (DVO) (BPR Medical Gas Control) (Figure 1). The SM and the OM delivered 100% oxygen at a flow-rate of 15 l/min. The SM is standard equipment when a doctor prescribes oxygen treatment for CH in Denmark. The OM is a non-rebreather mask with a 3 l reservoir. The DVO delivers oxygen according to respiration rate and tidal volume (flow-rate: 0–200 l/min), thus the DVO is not limited to flow but delivers oxygen according to demand. In the trial, the DVO mask delivered 100% oxygen and placebo (21% oxygen and 79% nitrogen). No specific instructions were given regarding hyperventilation or posture.
The Committee on Health Research Ethics of the Capital Region in Denmark and the Danish Health and Medicine Authorities approved the study (H-2-2011-163). Patients signed written consent forms in accordance with the Declaration of Helsinki.
Statistics
A power calculation stated that a population of 29 would give a power of at least 80% when the expected outcome was 80% of patients would achieve at least a two-point pain reduction at 15 min in the DVO group, compared with just 50% in the SM group. A population of seven was needed for a power of 80% when the expected outcome was 20% for placebo and 80% for active therapy. An intention-to-treat analysis was not compatible with the endpoints of the study and therefore only treated attacks were included in the analysis. A per-protocol analysis was not possible as only 10 CH patients completed the crossover design as described in the protocol.
We tested for informative drop-out by means of a logistic regression model for the risk of more than one attack with respect to the mask used during the first attack. In addition, we tested the association between the mask used during the first attack and completion of the crossover study by logistic regression.
To eliminate the possible carry-over effect, we conducted a post hoc comparative cross-sectional analysis, where only the first treated attack was included. Since this was the first attack, the randomization was intact and the risk of bias was minimized.
The performances of the three masks were compared by 1) a logistic regression model for the performance of the three masks on the first attack and 2) a generalized linear mixed-effects model with study participants as random effects in order to account for correlation between attacks on the same individual.
Patient characteristics and clinical details (mean values and range are indicated).

Wilcoxon two-sample test (numerical data) and chi-square test (categorical data). bPercentage of population that received acute or prophylactic therapy. cGabapentin, indomethacin, propranolol (for aura without headache).
BMI: body mass index; CH: cluster headache; DVO: demand valve oxygen; SPG: sphenopalatine ganglion.
SAS 9.4 and R 3.0.0 were used for the statistical analysis and p values below 5% were considered significant. The p values reported in the paper are not adjusted for multiple testing.
Results
Patients and attacks
Fifty-seven CH patients were included in the trial (42 males, 15 females). The population consisted of 27 CCH patients and 30 ECH patients (Table 1) who had been treated at the Danish Headache Center for an average of 3.2 years. The mean age was 45 years. The mean CH duration was 13.2 years, but ranged between 1 and 39 years. Only 42 CH patients received the trial therapy. The discrepancy was due to two exclusions because of latex allergy and one screening failure due to an unplanned change in prophylactic medication. The remaining 13 were attack free during the study. There were no statistical differences in clinical characteristics or demographics between those who received treatment and those who did not (Table 1). During admission (range: 1–4 days), the average attack frequency was 1.1 per day, a marked reduction compared to the self-reported pre-admission frequency (3.9 per day). This meant that fewer attacks than expected were observed. Before the trial, oxygen treatment was used by 76% of the patients who received trial therapy. Only 20% of the patients reported absolute pain reduction. Fifty-one percent reported some effect and 29% described only a slight response to oxygen.
Patient characteristics and clinical details of first attack analysis (mean values and range are indicated).

One-way ANOVA. bChi-square. cKruskal-Wallis test. ANOVA (p = 0.267) not valid because of difference between the variances in the population (Levene’s test p = 0.022). dPercentage of population that received acute or prophylactic therapy.
BMI: body mass index; CH: cluster headache; ANOVA: analysis of variance.
Primary and secondary endpoints
After 15 min of therapy, the percentage of patients that were either pain free or had a two-point decrease on the five-point rating scale was 40%, 44% and 52% on the SM, OM and DVO, respectively (Figure 4). We completed a logistic analysis of all attacks for the primary outcome regarding pain relief within 15 min of oxygen therapy, but could not show any significant differences (p = 0.411). The analysis found an odds ratio (OR) of 0.4 (95% confidence interval (CI): 0.1–1.2) comparing SM to DVO and an OR of 0.7 (95% CI: 0.2-2.1) when comparing OM to DVO. The majority of the CH patients preferred DVO (62%) and only 5% preferred the SM (p < 0.001). The remaining 33% favored the OM (p = 0.061, compared to DVO) (Table 3). Only patients who tried all three mask types were included in this analysis (n = 21).
Effect of oxygen treatment. Primary, secondary and post hoc endpoints (frequency, percentage and p values are indicated). p values: compared therapy with 100% oxygen delivered by DVO (chi-square or fisher exact test or Wilcoxon-Mann-Whitney test). All compared to the data of the DVO. p values are not corrected for multiple testing. Excluded: if the patient has used rescue medication and time to meaningful effect exceeds 15 minutes. bExcluded: rescue medication at 15 minutes or missing data. Not excluded: continued with trial therapy. cAll patients who tried all three masks. DVO: demand valve oxygen; SM: Simple mask; OM: O2ptimask.

Post hoc outcomes
The analysis of the first attack contains 42 CH attacks (Figure 3). The DVO was significantly better than the OM at treating the CH attacks on the five-point rating scale at 15 min with an OR of 5.5 (p = 0.042). The DVO was borderline better than the SM with an OR of 5.0 (p = 0.056). There was no difference between SM and OM (p = 0.918).
Pooling the data and comparing the DVO to both SM and OM, the DVO is significantly better (p = 0.018). The complexity reduction does not make the model significantly worse (p = 0.918), meaning that statistically it is valid to compare the DVO population and the pooled population of OM and SM. No statistical difference was found between the groups (Table 2).
After 15 min of therapy placebo was effective in 45% of the patients (compared to DVO, p = 0.867). The cumulated frequencies of responders were higher after 30 min compared to 15 min of therapy. After 30 min, the response rates were 50%, 66% and 68% for SM, OM and DVO, respectively. The difference was non-significant. Placebo response was 45%, which was borderline significantly different compared to DVO (p = 0.061).
The mean attack duration for DVO-treated attacks was 20 min, which was lower than SM (28 min, p = 0.215), OM (28 min, p = 0.113) and placebo (45 min, p = 0.099). The mean self-declared duration of attack was 86 min without treatment (range 20–180 min, Table 1). The DVO mask reduced the mean duration with 66 min compared to the declared mean duration of attacks, which is a decrease of 69%.
The patients could choose rescue therapy after 15 minutes. Half the patients using SM and 45% of the patients receiving placebo were in need of rescue medication. Treatment with OM and DVO reduced the use of rescue medication to 19% and 23%, respectively, There was a statistically significant higher use of rescue medication using SM compared to DVO (p = 0.028).
Only 10 CH patients completed the crossover (see Supplementary Table 4). The risk of suffering more than one attack was slightly decreased with an OR of 0.4 when the patient used DVO to treat the first attack compared to SM or OM (p = 0.238). Furthermore, CH patients who treated the first attack with the DVO had a lower risk of completing the crossover, i.e. experiencing four attacks, compared to SM or OM (p = 0.036), indicating informative censoring.
Of the patients who had an effect from the oxygen in the trial, there was a high frequency of smokers, up to 76%. This is higher than the average of 57% current smokers in the population that received the trial therapy. The patients who had an effect from the DVO had a significantly higher prevalence of current smokers compared to the non-effect group (p = 0.006). No significant differences were discovered regarding the SM (p = 0.060) or the OM (p = 0.169). There was a positive association between use of prophylactic medication and effect of OM (p = 0.019). This was not seen for the SM or DVO (p = 0.256 and p = 0.242). No other differences between responders and non-responders were discovered regarding phenotype, age, gender or nightly attack.
Rebound headache occurred in 24 out of 126 attacks (19%). No statistical difference was found in distribution of rebound headaches between the three mask types (see Table 3). There were no differences in re-occurrence of CH attacks within 24 hours.
Safety of oxygen
Four adverse events happened after the trial, including one serious, but all were assessed to be unrelated to oxygen therapy. One patient developed throbbing chest pain one week after oxygen inhalation but the cardiac follow-up was negative. A patient reported an increase of CH attacks in the weeks following participation. Another reported influenza in the days following the trial, and finally, one reported nosebleed one week after experimental treatment.
Discussion
Efficacy
Despite the widespread use of oxygen therapy, only a few studies of oxygen treatment in CH are available. Our trial shows a tendency toward a more efficient oxygen therapy, reduced need for rescue medication and higher satisfaction with the DVO, although the primary endpoint with pain relief at 15 min was non-significant. The per-protocol defined effect ranged from 29% to 48% in contrast to a previous study in which a much higher effect up to 82% was found (14). Our cumulated frequencies of responders are higher after 30 min of oxygen therapy, up to 68%. This could indicate that oxygen therapy might not be as fast acting (in a hospital setting) as previously thought.
In a previous study it was suggested that old age and a diagnosis of CCH decreased the probability of a positive response (4). In our study population, the mean age was 45 years and 74% were older than 40 years. The high proportion of CCH patients (44%) in this study might have affected the results. Likewise, our patients were included from a tertiary headache center, their mean duration of CH was 13 years, and they were probably more difficult to treat than newly diagnosed patients from a primary care setting.
Attack frequency
The frequency was markedly decreased during admittance to our study. This is a well-known clinical phenomenon but not well documented in the literature. The phenomenon could be due to exaggerated pre-admission reporting, a decrease in frequency due to a lowered or increased stress level. Since all the patients had been followed for a mean duration of 3.2 years in the Danish Headache Center, the frequency reduction cannot be attributed to the effect of a highly specialized center.
Methodological considerations
Guidelines for oxygen studies of CH do not exist and designs and execution of such studies are complicated. Different methodologies have been employed before. Previous studies were conducted as outpatient studies. We found it of great importance to conduct our study as an inpatient study to ensure optimal therapy compliance, instruction and attack observation. The hospital provides optimal settings for observation; our trial took place in a single patient room guaranteeing quiet surroundings. In a home setting, the patients’ reports of attack start and end times may be imprecise. In addition, there is a risk that the pain associated with the attack compromises the ability to fill out attack observations correctly. The files may have been completed subsequently leading to recall bias. In addition, the patients had a markedly lower attack frequency during the study compared to the self-reported attack frequency, which implied that the number of patients completing the trial was lower than designed.
In the Cohen study patients were oxygen naïve (6). Our patients were not oxygen naïve, which may have influenced the results; however, they probably reflect a clinical CH population more accurately. This could also suggest that the effect of oxygen might change as the headache disorder progresses. Oxygen treatment was used in 76% of our study population and hereof 29% reported having only a slight effect oxygen. This might affect the results due to the patients’ expectations.
In previous studies patients treated four to 10 attacks (4–6); however, in our study each patient treated only one attack with each mask type. This could affect the treatment response since patients describe both attack and treatment variability. There is to the authors’ knowledge no current literature that explores the field of attack and treatment variability.
The placebo response was higher than previous reports (5,6,15). Only 11 tried placebo, which might affect the results via selection bias. Furthermore, our data suggest an unequal distribution of attacks depending on the first tried mask type. The fact that 45% patients found the placebo treatment efficacious may also be because they would expect the more complex DVO mask to be better. Since placebo consisted of 21% oxygen, it might be effective in itself when administered by a DVO mask. Atmospheric gas from a cylinder is colder than ordinary air and it is speculated that cool gas may be equally efficient as oxygen therapy for abolishing CH attacks (16).
Pathophysiology
The mechanism by which oxygen alleviates headache still needs clarification. Oxygen inhibits cranial parasympathetic outflow (17) and it has also been suggested that oxygen inhibits dural inflammation (18).
It seems as if a higher fraction of inspired oxygen (FiO2) or higher flow rates might provide better treatment (7). In atmospheric air, the FiO2 is 0.21. In theory, the ODV and the OM are closed respiratory systems (FiO2 = 1). However, the OM’s reservoir can collapse when consumption exceeds flow. It is a clinical observation that some patients might need higher flow rates than 15 l/min to prevent reservoir collapse. An empty reservoir obstructs breathing because the system is closed. A simple non-rebreather mask (reservoir: 1 l) has an FiO2 between 0.6 and 0.8 when applied with 10–15 l/min and the FiO2 of the SM is only 0.3–0.6 (19). However, a dose-response curve of the effect of oxygen seems unlikely since the oxygen saturation is almost constant because of the hemoglobin-oxygen-dissociation curve (20). Free oxygen concentration (PaO2) is not as stable as oxygen saturation, but most oxygen molecules are bound to the hemoglobin.
Safety and financial aspects
A possible side effect of high-flow oxygen is mucosa damage. A recent study concluded that oxygen treatment of CH may lead to bilateral thinning of the temporal retinal nerve fiber layer, but importantly not to visual deficits (21). CH patients have a high prevalence of smoking (22,23), which may cause safety concerns as oxygen increases the flammability of fuels. Thus, the DVO is preferable because of the non-continuous flow in the safety aspect regarding retinal thinning, mucosa damage and smoking. Cost is also an important aspect of every treatment. Oxygen therapy may be unaffordable for some (24). The DVO is expensive, around $300, but the running costs are assumed to be lower due to lower oxygen-consumption by the non-continuous flow. It is a clinical observation that some patients fall asleep immediately after a CH attack and forget to turn off the oxygen. Further, compared to injectable triptans, and the marked indirect costs and loss of quality of life by CH (25), the use of oxygen is highly cost-effective irrespective of the applied mask.
Future research
Future studies should investigate the role of hyperventilation and smoking in relation to the efficacy of oxygen in CH. Ongoing animal studies may provide insight into the mechanism of effect.
Conclusion
Oxygen therapy is a safe, well-established and effective acute treatment for CH attacks. Although the primary endpoint was non-significant, our post hoc outcome of the first attack is significant and favors oxygen treatment with DVO. Subjectively, CH patients significantly tended to prefer DVO therapy to the SM and OM. We demonstrated that oxygen therapy by DVO resulted in a significant decreased need for rescue medication compared to SM. We recommend that CH patients be offered DVO or OM before oxygen therapy is abandoned.
Article highlights
The primary endpoint (pain relief at 15 minutes) was non-significant, but a post hoc analysis of the first attack significantly identifies demand valve oxygen (DVO) as superior to a simple mask and O2ptimask. Patients treated with DVO had significantly lower need of rescue medication compared to the simple mask. Patients preferred the DVO therapy to O2ptimask and the simple mask. There is a marked attack frequency reduction during hospital admission. A high placebo rate with the DVO mask was noted.
Footnotes
Acknowledgments
The authors would like to thank the staff of N38, Rigshospitalet-Glostrup and the Danish Headache Center for their assistance with the patient recruitment and treatment. Furthermore, we thank Senior Statistician Christian Dehlendorff, PhD, for valuable support with the statistical analysis.
Author contributions: Study design and concept: Mads Barløse and Rigmor Jensen; acquisitioning of data: Mads Barløse, Anja Sofie Petersen and Nunu Lund; analysis and interpretation of data: Anja Sofie Petersen, Mads Barløse and Rigmor Jensen; statistical analysis: Anja Sofie Petersen, Mads Barløse and Christian Dehlendorff; drafting the manuscript: Anja Sofie Petersen, Mads Barløse, Nunu Lund and Rigmor Jensen; study supervision and critical revision of the manuscript: Rigmor Jensen.
Role of sponsor: The sponsor was not part of designing the trial or analyzing the data. The sponsor supplied the mask and the gas cylinders for the trial.
Full protocol is available upon request by email.
Trial registration: 2011-006182-18. ClinicalTrials.gov identifier: NCT01589588.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Anja Sofie Petersen and Nunu Lund declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The remaining authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mads Barløse has received honorarium from ATI Inc and an unrestricted research grant from AGA, Linde Gas. Rigmor Jensen has given lectures for Pfizer, Berlin-Chemie, Norspan, Merck and Autonomic Technologies; and is a member of the advisory boards of Autonomic Technologies, Medotech and ElectroCore.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: an unrestricted research grant from Linde Healthcare, AGA A/S.
