Abstract
Background
Neurally mediated syncope and migraine have a complex relationship.
Aim
The aim of this study was to investigate whether patients developing syncope in the laboratory would experience migraine.
Methods
Thirty-one consecutive patients were evaluated for precipitation of headache during head-up tilt (HUT)-induced syncope (reduction of systolic blood pressure [SBP] >20 mmHg and prodromal symptoms with or without loss of consciousness). Autonomic functions were assessed using heart rate response to deep breathing (HRDB), Valsalva maneuver and HUT. Blood pressure and heart rate (via electrocardiography) were continuously monitored. Headache diagnosis was based on ICHD-3 criteria.
Results
Eighteen patients (58%) experienced syncope without headache and 13 (42%) had syncope and headache (SH). No difference was observed in time of syncope onset, reduction in SBP, Valsalva ratio, HRDB or tachycardia during initial 10 minutes of HUT. Of the 13 SH patients, 11 (85%) had a past history of migraine. Two reported headache just before tilt, eight developed headache during tilt and three developed headache only after tilt. Headache resolved within 1–15 minutes in 10 out of 13 patients. No patient experienced migraine.
Conclusions
Syncope did not precipitate migraine. Headache during syncope may be due to cerebral hypoperfusion, and cerebral hyperperfusion may cause post-syncopal headache.
Introduction
Orthostatic intolerance refers to a group of clinical disorders associated with diminished ability to tolerate erect posture as a result of presumed symptoms of cerebral hypoperfusion (lightheadedness, pre-syncope and syncope) that are ameliorated by recumbence. Orthostatic hypotension (OH), postural tachycardia syndrome (PoTS) and neurally mediated syncope (NMS) constitute three well-delineated types of orthostatic intolerance. OH is defined as a sustained reduction of systolic blood pressure (SBP) >20 mmHg or a reduction of diastolic blood pressure (DBP) >10 mmHg within 3 minutes of standing or head-up tilt (HUT). PoTS causes a sustained heart rate (HR) increment of ≥30 beats per minute within 10 minutes of standing or HUT, without OH. NMS is a transient, self-limited loss of consciousness and postural tone resulting from global cerebral hypoperfusion with spontaneous, prompt and complete recovery (1).
Headache frequently accompanies orthostatic intolerance. Coat-hanger ache of variable severity and duration is a hallmark of OH, occurring more frequently during daily activities than during HUT (2). Orthostatic headache during daily activities or during HUT may be reported by two-thirds of patients with PoTS; younger age (<30 years) and increasing duration of upright posture predicts increased incidence of orthostatic headache (3). Reports based on clinical observations, epidemiology and questionnaires suggest a complex relationship between NMS and headache. Syncope and migraine may coexist in individuals and families (4–6). Both syncope and migraine are highly prevalent in the general population and co-occur more often than chance would predict. Vallejo and colleagues estimated the frequency of migraine at 52% in a sample of 50 syncope patients (7). In the population-based CAMERA study, Thijs and colleagues demonstrated a significantly higher lifetime prevalence of syncope in 323 patients with migraines compared with 153 control subjects (46% versus 31%, respectively) (8). In a retrospective questionnaire-based study, Curfman and colleagues reported syncopal migraine or migrainous headache preceding or following syncope in nearly a third of 248 recurrent syncope patients and proposed migraine management in order to improve syncope (9).
We sought to investigate the characteristics and temporal presentations of headaches in 31 consecutive patients with documented syncope in the laboratory. We hypothesized that patients developing syncope or pre-syncope in the laboratory would experience migraine. Tests of autonomic function were performed in all patients to evaluate the relationship between the occurrence of headache and the type and severity of autonomic dysfunction.
Methods
The institutional review board approved the retrospective analysis of prospectively collected data in 31 consecutive patients with hemodynamically documented syncope. These patients had histories of three or more episodes of syncope within the last 12 months or frequent pre-syncope with prodromal symptoms including nausea and diaphoresis (10). All patients were referred to the senior author for the diagnosis and management of previously unexplained syncope. The patients were subjected to autonomic assessment if no other cause was established after a comprehensive history and physical examination, full neurological assessment, routine laboratory tests, electrocardiography, 24-hour ambulatory Holter monitor study and echocardiography. A detailed autonomic questionnaire and supine and orthostatic hemodynamic measurements showed no evidence of other forms of orthostatic intolerance.
Informed consent was obtained before testing patients in order to ascertain the involvement of cardiovagal and adrenergic functions and to document the diagnosis of NMS. These tests included HR response to deep breathing (HRDB), Valsalva maneuver (VM) and prolonged HUT (11). HRDB measures changes in HR in response to deep breathing, an indicator of cardiovagal activity. VM was analyzed for Valsalva ratio (VR), an indicator of cardiovagal activity, and SBP overshoot during phase IV, a measure of adrenergic function. Prolonged HUT, currently a clinical tool of choice for demonstrating propensity to NMS (12), was performed with continuous monitoring of HR and blood pressure. Pre-syncopal features included pallor, diaphoresis, nausea, abdominal discomfort, yawning and hyperventilation (10). Patients with pre-syncope (presence of prodromal symptoms and reduction of SBP >20 mmHg) were monitored after the table was lowered for continued drop in SBP and possible transient loss of consciousness to confirm that pre-syncope indicated imminent syncope. HR increase during the initial 10 minutes of HUT was measured for correlation with headache because NMS patients may have transient tachycardia preceding syncope, mimicking PoTS.
Patients were off all medications for 5 days (13). They were studied in a headache-free phase. They were instructed to avoid alcohol, caffeine and nicotine overnight and to have only a light snack no less than 3 hours before the test. These tests were conducted between 9 a.m. and noon in a quiet room at 22–24℃. Patients were monitored continuously and noninvasively for HR by a beat-to-beat HR monitor and for blood pressure by a plethysmographic device (Finapres, Ohmeda 2300) applied around the left middle finger. The left hand was supported in a sling in order to keep the cuff at heart level. No intravascular instrumentation or pharmacologic provocation agents were employed. An electrically operated table with foot support for weight bearing was used to produce postural stress.
HRDB
Patients breathed at a fixed rate of six breaths per minute, 5 seconds in and 5 seconds out, with the aid of a computerized program. The difference between the maximum and minimum HR during each respiratory cycle was calculated. The mean of the differences during six consecutive respiratory cycles provided the HRDB (11).
VM
In a semi-recumbent position, the patient expired forcefully from the normal inspiratory position to maintain an expiratory pressure of 40 mmHg for 10 seconds. VM was performed three times at 2-minute intervals. The maneuver with best hemodynamic response was selected for analysis of VR and SBP overshoot (14,15).
HUT test
The patient was strapped loosely to the tilt table across the chest and thighs. Baseline blood pressure and HR were recorded for 5 minutes. The patient was asked about any symptoms including headache before the table was tilted to 90° and maintained in that position for a maximum of 45 minutes, if tolerated (16). The patient enumerated symptoms as they occurred during and after the test. The investigator made observations regarding the condition of the patient, including onset of pre-syncopal features such as yawning, vasomotor changes, diaphoresis and occasionally myoclonic jerks. The test was promptly terminated when the patient became pre-syncopal with prodromal symptoms and a SBP decrease >20 mmHg compared with baseline or lost consciousness. Changes in SBP and HR from baseline at the time of pre-syncope or syncope were recorded. After the table was returned to the horizontal position, patients were monitored until they felt normal. Patients were then asked if the HUT reproduced their spontaneous symptoms. If the patient developed headache just before, during or after the test, details of the headache were recorded, including aura, location, quality of pain, severity, duration, photophobia, phonophobia, nausea and vomiting. The pain was graded on a visual analog scale of 1–10, with 10 being most severe. The patient was asked to report any additional observations regarding the HUT-precipitated headache at the next visit. HR and blood pressure signals during autonomic tests were transferred from the monitors to a computer for analysis. Headache type was assessed based on ICHD-3 diagnostic criteria (17). Two-sample t-tests were used to compare the means of the continuous variables between syncope patients with and without headache.
Results
Laboratory observations and autonomic data.
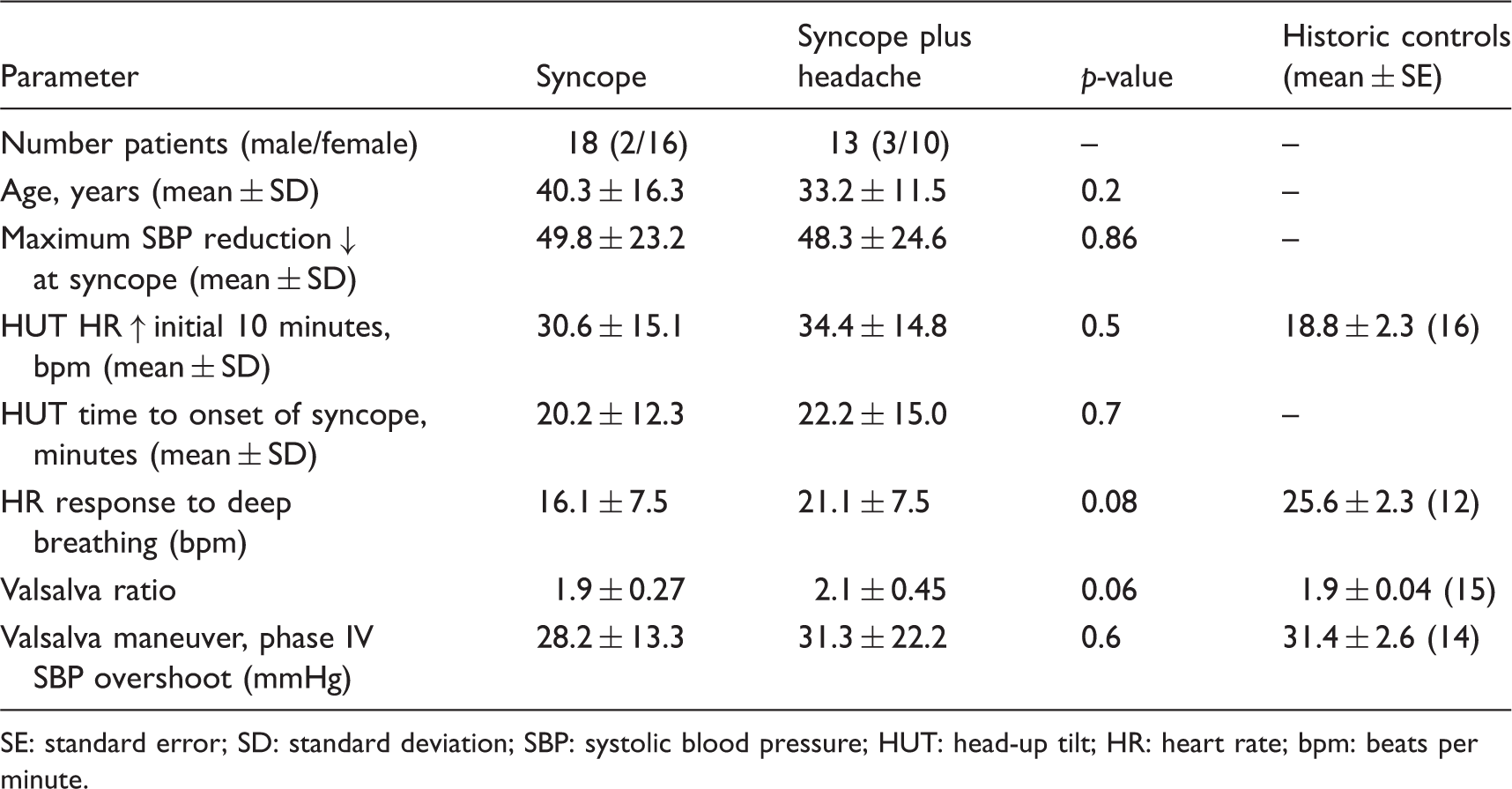
SE: standard error; SD: standard deviation; SBP: systolic blood pressure; HUT: head-up tilt; HR: heart rate; bpm: beats per minute.
Of the 18 patients in the S group, 11 (61%) had past history of headache: migraine without aura (six patients), chronic migraine (three patients) and migrainous headache (short duration and/or lacking associated migraine symptoms; two patients). Age of syncope onset was 6–47 years. Of the 13 patients in the SH group, 11 (85%) had past occurrence of headache: migraine without aura (four patients), migraine with aura (one patient), chronic migraine (two patients) and migrainous headache (four patients). Age of syncope onset was 6–43 years.
Head-up tilt-induced syncope and headache.

Headache began just before HUT.
HUT: head-up tilt; NR, not recorded.
Discussion
Patients in this study, predominantly women (84%), demonstrated a high prevalence of migraine history compared with the general population, where prevalence is 16.2–22.7% (18). In a cross-sectional study of 173,434 women hospitalized for syncope, Ulas and colleagues observed an odds ratio of 1.65 for migraine (19). Vallejo et al. also found a higher prevalence of migraine (76%) in female subjects with vasovagal syncope (7). Prevalence of migraine was substantially higher in the SH (85%) group than in the S group (61%). The majority of patients had histories of migraine without aura. During HUT-induced syncope, 13 out of 31 patients developed headaches. Of the 13 patients who developed headaches, no patient, including one patient with a past occurrence of visual aura, developed aura symptoms, and no headache met the accepted criteria for migraine. Head pain displayed characteristics of migraine, but was of short duration and without associated migraine symptoms, except for nausea in approximately half of the SH patients. However, nausea is a common accompaniment of NMS. The current data suggest that syncope does not precipitate migraine. A higher frequency of past occurrence of migraines and transient precipitation of headache during or after HUT suggest an underlying deficient habituation with superimposed temporary sensitization of the central neural structures involved in the pain matrix (20).
In syncope, the loss of consciousness is due to transient cerebral hypoperfusion with a critical reduction of cerebral blood flow to the reticular activating system. A 50–60% reduction of cerebral blood flow causes prodromic symptoms, and an interruption of cerebral blood flow for 8–10 seconds is sufficient to cause loss of consciousness. Furthermore, the reduction of cerebral blood flow may precede the occurrence of systemic hypotension and bradycardia (21). In our patients, although HUT was terminated as soon as the clinical and hemodynamic parameters supported the diagnosis of NMS, a mean decline in SBP of over 45 mmHg was noted, indicating continued further decline in SBP after termination of the tilt. Headache occurred in close temporal association with clinical and hemodynamic signs of syncope followed by quick resolution, implying reversibility. Ocon and colleagues observed post-syncopal headaches in 16 adolescent subjects that resolved within 5 minutes (22). Occurrence of headache simultaneously with an onset of focal deficit and its resolution with the resolution of deficit is well known in patients with transient ischemic attacks (23). Edmeads observed headaches in approximately 25% of 400 patients with transient ischemic attack and stroke (24). The current data suggest that global cerebral hypoperfusion, similarly to focal ischemia, may constitute a pathophysiological mechanism for headache.
Headache during syncope may be a multifactorial event. The relative contribution of various factors may differ between individuals based on the severity and duration of hypoperfusion. Cerebral hypoperfusion, although global, may be more pronounced in the right posterior parietal and parietotemporal regions, as demonstrated by 99mTc-ECD single-photon emission computed tomography imaging (25). Even transient cerebral hypoperfusion can impair vascular permeability, increase endothelial activation, alter neurohumoral responses with rising epinephrine levels and cause the release of vasoactive neuropeptides from perivascular sensory nerves via axon reflex-like mechanisms (26,27). Ischemia from hypoperfusion may cause the accumulation of lactate, NMDA receptor-mediated calcium influx into cells and the release of excitatory amino acids such as nitric oxide, which enhances headache tendency (28). Various anatomical and physiological studies support the notion that a significant decline in right peri-insular cortical perfusion may adversely affect descending nociceptive processing. Employing the Nauta–Gygax silver impregnation technique in patients with ischemic or hemorrhagic lesions of the hemisphere, Kuypers demonstrated degenerating fibers predominantly in the contralateral spinal trigeminal complex (29). Noseda and Burstein reported that direct descending projections from the contralateral primary somatosensory and insular cortices to the spinal trigeminal complex in rats and humans can influence meningeal nociception (30). It is also known that electrical stimulation of restricted areas of the human insular cortex can elicit cephalic pain (31). It is likely that dysfunction of the descending corticotrigeminal circuitry may facilitate headache by either the disinhibition or facilitation of nociceptive pathways.
Immediate post-HUT headache may be caused by different mechanisms, including residual vascular and neurohumoral effects and increased cerebral blood flow. A sudden decrease in cerebral blood flow at syncope is followed by rapid hyperemic pulsatile cerebral blood flow upon return to a supine position (22). The trigeminovascular system is believed to play a role in mediating post-ischemic cerebral hyperperfusion, its sensory component transmitting vascular pain and its motor component neurgenically mediating cerebral hyperemia (32). Post-syncopal headaches may be evoked by transient exposure of the distal cerebral vasculature to increased pressure. Moreover, a highly pulsatile flow during hyperemia may act as a potent stimulus for the release of nitric oxide. Nitric oxide may directly or via the release of calcitonin gene-related peptide stimulate meningeal and vascular nociceptors (33).
Limitations of the present study include its cross-sectional nature. These observations were collected in a controlled laboratory setting, and recorded events may not correspond to spontaneous syncopal episodes. All patients, however, confirmed that their symptoms during HUT mimicked their spontaneous episodes. In this study, HUT was terminated when syncope was objectively imminent based on known symptoms of pre-syncope/syncope and systemic hypotension. However, in a study in which the maintenance of an upright posture until fainting was a prerequisite, Ocon and colleagues reported similar observations (22). All subjects who fainted developed post-syncopal headache that subsided within 5 minutes. Finally, the conclusions of the present study apply only to syncope-induced headache and not vice versa. A review of the literature reveals that if altered consciousness (drowsiness, confusion, etc.) is differentiated from loss of consciousness, then syncope in adult migraineurs is rare. Experienced clinicians such as Oleson (34) and Blau (35) state that the occurrence of syncope in adult migraineurs is likely secondary to epilepsy, postural hypotension or vomiting-induced cardiac arrhythmia. Further studies should help to elucidate the role of migraine in NMS.
Clinical implications
The study highlights the fact that syncope does not precipitate migraine. Migraine management for the treatment of syncope may not be productive.
Footnotes
Acknowledgments
We thank Lyn Camire of our department for editorial assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
