Abstract
Objective
To perform a meta-analysis of migraine biomarkers in cerebrospinal fluid (CSF) and of corresponding blood concentrations.
Methods
We conducted a systematic search for studies that measured biochemical compounds in CSF of chronic or episodic migraineurs and non-headache controls. Subsequent searches retrieved studies with blood measurements of selected CSF biomarkers. If a compound was assessed in three or more studies, results were pooled in a meta-analysis with standardised mean differences (SMD) as effect measures.
Results
Sixty-two compounds were measured in 40 CSF studies. Most important results include: increased glutamate (five studies, SMD 2.22, 95% CI: 1.30, 3.13), calcitonin gene-related peptide (CGRP) (three studies, SMD: 3.80, 95% CI: 3.19, 4.41) and nerve growth factor (NGF) (three studies, SMD: 6.47, 95% CI: 5.55, 7.39) in chronic migraine patients and decreased β-endorphin (β-EP) in both chronic (four studies, SMD: –1.37, 95% CI: –1.80, –0.94) and interictal episodic migraine patients (three studies, SMD: –1.12, 95% CI: –1.65, –0.58). In blood, glutamate (interictal) and CGRP (chronic, interictal and ictal) were increased and β-EP (chronic, interictal and ictal) was decreased.
Conclusions
Glutamate, β-EP, CGRP and NGF concentrations are altered in CSF and, except for NGF, also in blood of migraineurs. Future research should focus on the pathophysiological roles of these compounds in migraine.
Introduction
Migraine is a prevalent episodic brain disorder (1). The World Health Organisation (WHO) rates migraine as one of the most disabling chronic disorders (2). Despite extensive research over the last decades, migraine pathophysiology is not completely understood (3). Although several compounds (e.g. calcitonin gene-related peptide (CGRP), glutamate and serotonin) have been implicated in migraine pathophysiology, our understanding of the biochemistry of migraine is still limited (4,5). Identification and validation of biochemical biomarkers might help us in uncovering pathophysiological processes involved in migraine, which in turn might lead to diagnostic tests or new therapeutic strategies (6,7).
The field of biochemical biomarker research is expanding rapidly. Promising biomarkers have been discovered for brain disorders such as Alzheimer’s disease, narcolepsy, and Parkinson’s disease (8–10). Cerebrospinal fluid (CSF) is believed to reflect biochemical changes in the brain and therefore is the body fluid of primary interest for brain disorders (11). Although many small studies have analysed biochemical changes in CSF from migraine patients, results were often inconsistent and have not led to pathophysiological and diagnostic biomarkers. However, the literature has never been systematically reviewed with quantitative synthesis of the evidence. With this first meta-analysis we aimed to identify biochemical migraine biomarkers which show consistent changes in CSF and to assess whether these changes are also present in blood.
Methods
Search strategy, study selection and eligibility criteria
We conducted and reported the review process in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (12). We performed an electronic search for published studies up to 16 August 2014 in MEDLINE, EMBASE and Web of Science on biochemical findings in CSF of migraine patients. Medical Subject Heading (MeSH) terms and free text terms were collated with the assistance of research librarians at the Leiden University Medical Centre. The full search string can be found in Supplement 1.
Two investigators (R.M.D. and R.Z.) independently assessed titles and abstracts to determine potential eligibility. Disagreement was resolved by discussion. The same investigators independently assessed the full-text articles of potentially relevant studies to verify if eligibility criteria were met, and to evaluate whether the results were adequately reported.
We included case-control studies and case-crossover studies (same patients studied in between and during migraine attacks), in which one or more endogenous compounds (metabolites, peptides, proteins) were quantified in CSF samples from migraine patients. Case reports were not considered eligible. Publications on pharmacological trials were excluded if no endogenous compounds were measured at baseline. Studies not written in English, conference abstracts, editorials and letters were also not eligible. Reference lists of articles eligible for full-text review and relevant reviews were additionally searched for potentially relevant studies.
Subsequent search for studies on blood concentrations of selected biomarkers
To assess whether CSF biomarkers show similar results in blood, we performed in a second stage a literature search for published data on measurements in plasma and serum. We specifically searched for studies reporting blood concentrations of compounds which had shown consistent and significant differences in meta-analysis of CSF data. These additional blood studies were identified and selected by performing the same search and selection process as described for CSF studies. The full search string for blood is reported in Supplement 1. After study selection, data were extracted and subsequently included in study assessment and meta-analysis following the same methodology as for CSF.
Data extraction
Data extraction was performed by one investigator (R.M.D) using a standardised extraction form. A second investigator (R.Z.) was consulted if discussion was necessary. Information was extracted on: (1) study design; (2) study population characteristics (sample size, age, gender, medication, comorbidity and other potential confounders) and study groups definition (diagnostic criteria, presence of migraine attack during sampling, presence of chronic migraine component); (3) sampling methods (fasting, timing and storage temperature), measurement methods and data analysis; and (4) concentrations of endogenous compounds (metabolites, peptides, proteins) in study (sub)groups, including statistical parameters. To obtain relevant missing information of studies included for meta-analysis, we attempted to contact corresponding authors twice via email.
Risk of bias assessment
To assess risk of bias, we adapted the Newcastle-Ottowa Scale (13) (Supplement 1). We considered definition of cases and controls to be adequate when published criteria were used for diagnosis of migraine patients. Selection of cases was adequate when patients were representative for the defined migraine type (no severe comorbidity or clinical reasons to sample body fluids). Selection of controls was adequate when controls were sampled from the same population as the cases. Comparability between cases and controls was assessed based on gender- and age-matching of study groups (either by design or analysis). Studies adequately describing sampling and measurement methods and performing measurements according to validated analytical methods were considered to have low risk of measurement bias. One investigator (R.M.D.) assessed selection and comparability, two investigators (R.M.D. and M.N.) assessed the description and validity of the measurements, and a third investigator (R.Z.) was contacted if discussion was necessary.
Group definition
We divided case-control comparisons into episodic migraine versus controls and chronic migraine versus controls. If there was no evidence that migraine patients had a chronic component, we classified them as episodic migraine. Findings in episodic migraine were further classified based on migraine state: interictal and ictal. Migraine with aura patients and migraine without aura patients were grouped, because results were often not reported separately for these groups. When applicable, ictal versus interictal findings, from case-control and from case-crossover studies, were compared.
Meta-analysis: Pooling of results and statistical procedures
We used standardised mean differences (SMDs) with their 95% confidence interval (CI) as the main effect measure. Compound concentrations were analysed in meta-analysis if data were available from three or more studies for one of the defined group comparisons. The way we have dealt with missing data, irregularities in the data, and pooling of data was in accordance with approaches described by the Cochrane Collaboration (14) (Supplement 1).
For quantitative synthesis, we used the inverse variance method. We applied a random-effects model by default given the expected clinical heterogeneity between studies. However, as the between-study variation cannot be estimated reliable in case of <5 studies, we applied a fixed-effects model in these instances. Homogeneity of effect sizes was assessed using the I2 statistic and by visual inspection of forest plots. To examine the effect of inclusion of clear heterogeneous studies, we applied a sensitivity analysis to assess their specific effect on the overall effect size. For statistical analysis we used RevMan 5.2 (Cochrane IMS, Baltimore, MD, USA).
Results
Study selection and study characteristics
The selection of CSF studies is depicted in the flowchart (Figure 1). A total of 1197 unique articles were identified, of which 40 were considered eligible (38 case-control studies and two case-crossover studies). Investigator agreement on title and abstract screening, before consensus, was κ = 0.72. Episodic migraine patients were sampled for 22 case-control studies and chronic migraine patients for 16 case-control studies. The number of cases ranged from 4 to 60 (average: 24) and the number of controls from 5 to 108 (average: 24). Description of individual study characteristics can be found in the electronic supplementary table. Twelve CSF studies were finally included in meta-analyses on compounds that were measured in multiple studies. The subsequent search for blood studies on selected CSF biomarkers is illustrated in Supplementary Figure S1.
Flowchart of CSF study selection process.
Risk of bias assessment
Summary of study characteristics and risk of bias assessment of CSF studies.
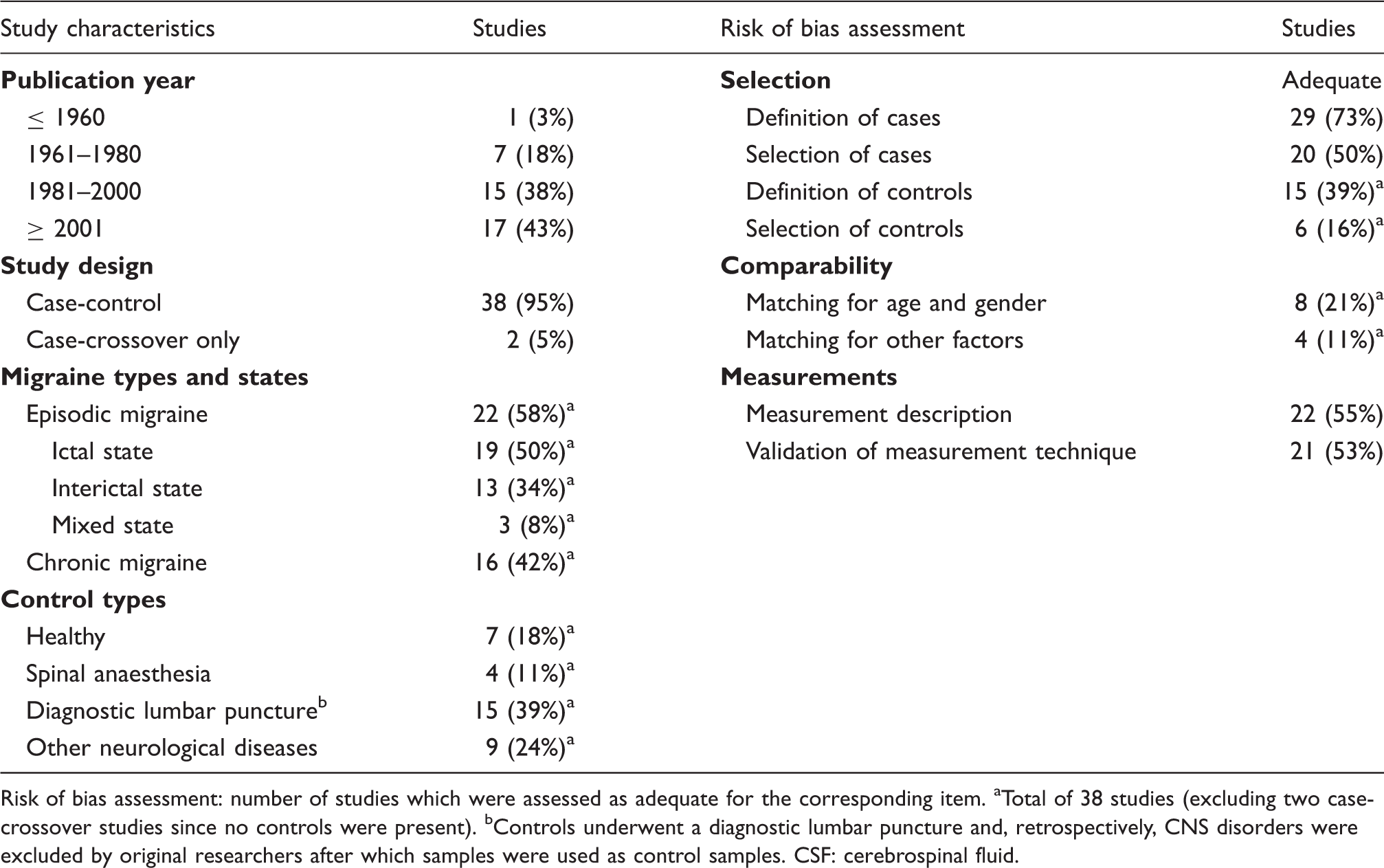
Risk of bias assessment: number of studies which were assessed as adequate for the corresponding item. aTotal of 38 studies (excluding two case-crossover studies since no controls were present). bControls underwent a diagnostic lumbar puncture and, retrospectively, CNS disorders were excluded by original researchers after which samples were used as control samples. CSF: cerebrospinal fluid.
Migraine cases were not always deemed representative for the diagnosed migraine type because lumbar punctures were performed to exclude other neurologic diseases (five studies), migraine patients were admitted to the hospital for unstated reasons (four studies) or because recruitment of cases was not clearly reported (11 studies) (Table 1). Controls often had lumbar punctures for other purposes than migraine patients; either for other diagnostic purposes (13 studies) or before spinal anaesthesia (four studies). Based on available cohort descriptions, only six studies recruited cases and controls from the same population, of which four studies were sampled from the general population. For 15 studies it was explicitly stated that controls had no personal history of migraine (Table 1). Furthermore, a minority of studies (eight studies) adjusted for age and gender.
Sampling and measurement methods were adequately described in 22 studies (55%; Table 1). The older publications especially lacked full and clear descriptions of methods. Measurement techniques were considered to be (partially) validated in 21 studies. Quantitation characteristics (precision, accuracy and limit of detection) were often not reported.
Biochemical findings
In total, 62 unique compounds have been measured in CSF from migraine patients (Table 2) (18–55). Frequently measured compounds (in three or more studies) are glutamate, β-endorphin (β-EP), 5-hydroxyindoleacetic acid (5-HIAA), homovanillic acid (HVA), CGRP and nerve growth factor (NGF). Forty-four compounds were measured only once. Meta-analysis showed that glutamate (Figure 2), β-EP (Figure 3), CGRP (Figure 4) and NGF (Figure 5) concentrations were consistently altered in CSF from migraine patients compared to controls; results will be discussed below, together with results on blood concentrations. For HVA there was insufficient quantitative data available for meta-analysis (CSF concentrations not reported separately for ictal and interictal patients) and for 5-HIAA CSF studies showed inconsistent effects (Supplementary Figure S2).
Forest plot of glutamate concentrations in migraine patients and controls. Forest plot of β-EP concentrations in migraine patients and controls. Forest plot of CGRP concentrations in migraine patients and controls. Forest plot of NGF concentrations in migraine patients and controls. Overview of published biochemical measurements in CSF from migraine patients. Published biochemical findings in migraine patients compared with controls. ↑= significantly elevated concentrations, ↓ = significantly decreased concentrations, “=“ = similar concentrations, between () = no statistical analysis reported, u.d. = undetectable concentrations reported for studied migraine group. aSignificant when interictal and ictal groups were pooled. bSignificant difference between ictal and interictal migraine patients reported. cMigraine state not reported. dNitric oxide (NO) was measured; not reported if NO2– and NO3– were also quantified. Excluding glucose from the routine CSF measurements (nine studies reported normal glucose concentrations in migraine patients). CSF: cerebrospinal fluid.




Glutamate (Figure 2)
Glutamate concentrations were increased in CSF from chronic migraine patients (SMD: 2.22, 95% CI: 1.30, 3.13) (18–22). In blood from interictal episodic migraine patients the pooled difference was not statistically significant (SMD: 1.08, 95% CI: –0.07, 2.22) (56–62). After exclusion of paediatric migraine patients (58), glutamate concentrations were increased in the remaining adult migraineurs (SMD: 1.61, 95% CI: 0.73, 2.49). Glutamate concentrations were increased in CSF from ictal patients (SMD: 2.01, 95% CI: 1.27, 2.75) (24). In blood, two studies showed clearly opposing results on ictal measurements and therefore we did not perform a meta-analysis (24,56). There are no studies on glutamate concentrations in blood from chronic migraine patients and in CSF from interictal migraineurs.
β-Endorphin (Figure 3)
β-EP concentrations were decreased in CSF (SMD: –1.37, 95% CI: –1.80, –0.94) (30,37–39) and blood (SMD: –0.76, 95% CI: –1.17, –0.36) (38,63,64) from chronic migraine patients. Concentrations were also decreased in CSF (SMD: –1.12, 95% CI: –1.65, –0.58) (37,38,40) and blood (SMD: –0.40, 95% CI: –0.64, –0.16) (38,40,65–73) from interictal patients. Pooled estimates remained similar in sensitivity analysis. One blood study was excluded from meta-analysis because the assay that was used had a very high cross-reactivity with β-lipotropin (28). In ictal migraineurs β-EP concentrations were decreased in CSF (SMD: −1.39, 95% CI: –2.40, –0.39) (40) and blood (SMD: –1.44, 95% CI: –2.11, –0.77) (40,66).
CGRP (Figure 4)
CGRP concentrations were increased in CSF (SMD: 3.80, 95% CI: 3.19, 4.41) (18,42,43) and blood (SMD: 0.70, 95% CI: 0.45, 0.95) (74–77) from chronic migraine patients, and in blood from interictal (SMD: 0.47, 95% CI: 0.20, 0.75) (74,76–83) and ictal (SMD: 0.79, 95% CI: 0.49, 1.09) (78,82–84) episodic migraineurs. Sensitivity analysis had small effects on pooled estimates. There are no studies on CGRP concentrations in CSF from episodic migraine patients.
NGF (Figure 5)
Concentrations of NGF were increased in CSF (SMD: 6.47, 95% CI: 5.55, 7.39) (20,21,42) and blood (SMD: 1.08, 95% CI: 0.58, 1.59) (75) from chronic migraine patients. Blood concentrations were not significantly different in interictal patients (SMD: 0.06, 95% CI: –0.31, 0.42) (85). There are no studies published on ictal concentrations (CSF and blood) and interictal concentrations in CSF.
Discussion
We conducted a systematic review and meta-analysis of biochemical measurements in CSF from chronic and episodic migraineurs. Meta-analysis showed increased concentrations of glutamate and CGRP and decreased concentrations of β-EP in CSF. These changes are also present in blood – a more accessible body fluid. Concentrations of NGF were increased in CSF from chronic migraine patients but blood data were limited.
Increases in glutamate and CGRP are in agreement with theories on pathophysiological mechanisms for migraine (4,5). Glutamate is the principal excitatory neurotransmitter within the central nervous system and has been linked to neuronal hyperexcitability in migraine (86). Glutamate has been implicated in the onset and generation of cortical spreading depression (CSD), which is believed to be the underlying cause of migraine aura. Increased synaptic glutamate concentrations lower the threshold for CSD (5). CGRP has been implicated as a mediator which activates and sensitises peripheral meningeal nociceptors causing migraine headache (4). Trigeminal fibres surrounding meningeal vessels release CGRP and other neuropeptides, and there is increasing evidence that CSD can initiate this release in animal experiments (5). Another rat model showed that CGRP released by primary trigeminal afferents impacts both CSF and blood concentrations and that the contribution of non-trigeminal structures to CSF concentrations is only minor (87).
In CSF from chronic migraine patients, NGF is increased with glutamate and CGRP. NGF is not only a well-known growth factor, but, following tissue injury, also an inducer of hyperalgesia via different peripheral mechanisms including mast cell degranulation (88). After local injury or inflammation both peripheral (reactivated Schwann cells, non-neural cells) and central (neurons, astrocytes, microglia) sources upregulate NGF expression (89). In contrast to CGRP, their relative contributions to CSF composition are still unknown. Additionally, NGF can upregulate CGRP expression in sensory and motor neurons (90,91). In one included CSF study NGF and CGRP concentrations were indeed positively correlated (42). Furthermore, by upregulating synthesis of brain-derived neurotrophic factor (BDNF), NGF can enhance synaptic transmission via N-methyl-D-aspartate receptors (92,93). Primarily the latter is hypothesised to contribute to chronic sensitisation of central neurons (i.e. in the nucleus trigeminus) (19–22). It is believed that this process also occurs in other chronic pain disorders such as fibromyalgia, where increased CSF concentrations of NGF, BDNF and glutamate have been found (21). This indicates that the observed changes are possibly not specific for migraine and instead reflect exposure to chronic pain.
β-EP concentrations are decreased in CSF and blood both from interictal patients and chronic migraine patients. Low β-EP concentrations have been hypothesised to reflect low analgesic activity in individuals. However, recent evidence suggests chronic pain patients with low β-EP concentrations have stronger analgesic activity when in pain through rapid upregulation of β-EP (94). In analgesic research with migraine patients β-EP could be a useful marker to study in more detail.
Study strengths and limitations
The main strength of this study is the systematic approach to the identification, quality assessment and analysis of published data. However, our findings should be interpreted in the light of the limitations of the included evidence.
We found considerable clinical and methodological heterogeneity across studies. Statistical heterogeneity was also observed in meta-analysis of glutamate, β-EP and CGRP, but importantly all studies showed an effect in the same direction. Nonetheless, diversity in migraine patients was present due to differences in migraine frequency, timing of measurements and diagnostic criteria. Less diversity was present in CSF studies on CGRP and NGF, since the studies were performed at the same headache centre (new participants with approximately similar clinical characteristics (age, gender, disease history, headache frequency and medication overuse) were recruited for each study; confirmed with original investigators). Furthermore, migraine patients were not always representative for the diagnosed migraine type because samples were taken for diagnostic purposes (i.e. other neurological disorders were suspected). Diversity in controls seemed primarily related to the availability of samples as well. Control cohorts often consisted of non-healthy controls in whom samples were collected for other diagnostic purposes than the migraine patients; this is especially the case for CSF studies where collection in healthy individuals is often not possible.
Additionally we found that the quality of reporting was inconsistent. Studies particularly failed to specify the validation, sensitivity and monitoring of applied measurement technique. Additionally, group comparisons were not always clearly reported and applied statistical analysis was frequently not well explained. Therefore, despite our attempts to contact corresponding authors, we were not able to retrieve all required data and had to apply published methods to calculate or estimate these data (14).
Publication bias is probably a major issue in the reporting of biomarker studies, because negative findings are less likely to get published (95). However, we did not generate funnel plots to assess any publication bias because the power of this strategy is low with the relative small number of studies per compound.
Recommendations
Future research should further clarify the pathophysiological relevance of the altered glutamate, β-EP, CGRP and NGF concentrations in migraine. For better understanding of involved biochemical processes, and for potential application as diagnostic biomarkers, it is also important to know whether concentrations are altered in all migraine types and whether similar changes are present in other headache disorders and chronic pain disorders.
Article highlights
This is the first meta-analysis of biochemical measurements in cerebrospinal fluid (CSF) and blood from chronic and episodic migraine patients. A total of 62 unique compounds have been measured in CSF from migraine patients. Glutamate, calcitonin gene-related peptide and nerve growth factor (NGF) concentrations are increased and β-endorphin concentrations are decreased in CSF from migraine patients. These changes are also present in blood, with the exception of NGF. The presented data identify clear biomarker targets for future pathophysiological or diagnostic studies on migraine.
Footnotes
Acknowledgements
We would like to thank the collaborating corresponding authors who were so kind to answer our questions and the research librarians who aided with formulating the search string.
Author contributions are as follows: R.M. van Dongen and R. Zielman were responsible for study design, data acquisition, data analysis, data interpretation and drafting/revising manuscript content. M. Noga was responsible for study assessment (biochemical measurements). O.M. Dekkers was responsible for data analysis (meta-analysis), data interpretation and revising manuscript content. T. Hankemeier, G.M. Terwindt, A.M.J.M. van den Maagdenberg and M.D. Ferrari were responsible for data interpretation and revising manuscript content.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R.M. van Dongen, M. Noga, O.M. Dekkers, T. Hankemeier and A.M.J.M. van de Maagdenberg declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. R. Zielman disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: support for conference visits from Menarini and Allergan. G.M. Terwindt disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: independent support from NWO, ZonMW, the National Institutes of Health (NIH), the European Community, and the Dutch Heart Foundation. M.D. Ferrari disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: grants and consultancy or industry support from Medtronic, and independent support from NWO, ZonMW, NIH, the European Community, and the Dutch Heart Foundation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Netherlands Organisation for Scientific Research (VICI grant 918.56.601 to M.D.F. and VIDI grant 917.11.319 to G.M.T.) and the European Union’s Seventh Framework programme (EUROHEADPAIN, grant agreement no. 602633).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
