Abstract
Keywords
Introduction
Involvement of the adrenaline, serotonin, dopamine, estrogen, glutamate and cannabinoid receptors is integral to migraine pathophysiology and its treatment (1–3). These receptors initiate signal transduction using lipid second messengers that are derived from phospholipids by the actions of several lipases (4–7). Phosphatidylcholine (PC), a major source of lipid signaling molecules, is the most abundant phospholipid in the brain by weight (8) and is hydrolyzed by the substrate-specific phospholipase C (PC-PLC). This process generates the major lipid second messengers diacylglycerol and phosphorylcholine.
We hypothesized that there would be a difference in PC-PLC activity in migraineurs compared to controls because the receptor pathways known to be associated with migraine influence PC-PLC-derived signaling molecules (5–7). To test this hypothesis, we measured PC-PLC activity in cerebrospinal fluid (CSF) and plasma; we then compared activity levels in samples from controls (C) and from migraineurs in their interictal (M−) and/or ictal state (M+). To investigate whether enzyme activity correlated with migraine severity, we tested if PC-PLC levels correlate with annual frequency or lifetime duration of migraine.
Methods
Study population
The Huntington Hospital Institutional Review Board for Human Research approved the study protocol and informed consent forms. Participants were included in this study after they gave informed consent. We recruited consecutive controls and migraineurs from 18 to 75 years of age from our research clinic and from the Pasadena area through advertising.
To minimize heterogeneity, migraineurs were included only if the primary headache disorder was migraine without aura, as defined by the criteria of the International Classification for Headache Disorders, second edition (ICHD-II code 1.1) (9). Diagnosis was determined by consensus after clinical assessment by one of two migraine diagnosticians (RPC, MGH) and through a validated, structured interview and computerized classification (10). Additional inclusion criteria were: at least one migraine per month and less than 15 headache days per month; availability of retrospective recall estimates for lifetime duration of migraine and attack frequency for the preceding year; all medications recorded for 90 days before study; no change in prophylactic medications during the 90 days; and no rescue medication taken within 48 hours of sample collection.
Controls were healthy individuals with no primary headache disorder determined from the same assessments used for migraineurs, had no family history of migraine in first-degree relatives, and had no changes in baseline medications for 90 days before and throughout the study.
Additional requirements for all participants included completion of an extensive structured interview for medical and neurological disorders and completion of the Structured Clinical Interview for the
Cerebrospinal fluid/blood collection protocol
All CSF/plasma samples were collected between 1:00 and 6:00
Migraine states at the time of sample collection were defined as follows: the M- state had no headache (0 on 0–10 scale) for >24 hours, while the M+ state had migraine that was typical for each participant, with duration of two to six hours and severity >5 (0–10 scale) at the time of CSF collection. CSF and blood collection was completed before rescue medication was taken. For a subgroup of migraineurs, CSF and plasma were collected in their M+ state and subsequently in their M− state, approximately four weeks later.
Laboratory methods
Cells were counted in a hemocytometer after trypan blue staining. Total protein concentrations were determined with the fluorescent Quant-iT™ protein assay kit (Invitrogen/Molecular Probes, Eugene, OR, USA) with bovine serum albumin (0–500 ng/ml) as a standard for quantification. Fluorescence (excitation at 470 nm and emission at 570 nm) was measured using a Gemini XPS Dual-Scanning Microplate Spectrofluorometer, and protein concentration was calculated using SoftMax® Pro software (Molecular Devices, Sunnyvale, CA, USA).
PC-PLC activity in cerebrospinal fluid and plasma
The Amplex® Red PC-PLC Assay Kit (A12218, Invitrogen/Molecular Probes) was used to measure enzyme activity. Since PC-PLC activity levels in CSF have not been reported previously, we describe the procedure in detail in the Supplemental Methods section. Enzyme activity was calculated as relative fluorescence unit (RFU) per microgram protein per minute (RFU/µg/min). To confirm specific activity, samples were pre-incubated with the PC-PLC inhibitor tricyclodecan-9-yl-xanthogenate (D609) from Sigma Aldrich (St. Louis, MO, USA) at increasing concentrations (0–200 mM) for 60 minutes at 30°C before adding the Amplex reagent working solution.
PC-PLC specific activity in plasma samples was determined using a similar procedure. Excess Ca2+ buffer (10 mM) was added to dilute plasma samples to overcome any chelation effects of K3-EDTA.
Statistics
Analyses were performed using Statview (Berkeley, CA, USA), Prism (GraphPad Software, La Jolla, CA, USA), or SAS v9.2 (SAS Institute, Inc., Cary, NC, USA). Overall group comparisons were done using Kruskal-Wallis tests. Wilcoxon rank sum (unpaired) or signed rank tests (paired) were used for comparisons between diagnostic groups. Spearman’s correlation coefficients were calculated to evaluate the relationships between migraine frequency and duration and PC-PLC activity levels. To assess the specificity of the PC-PLC assay, the arc sine transformation was used on percentage inhibition data; analysis of variance with inhibitor concentration as a continuous variable was used to test for trend, and Student’s t-test for location was used to test whether or not each inhibitor concentration differed from 0. Significance was set at 0.05 and was adjusted in pair-wise comparisons by the Bonferroni correction. All tests were two-sided.
Results
Clinical
Clinical characteristics, CSF cell counts and total protein by diagnostic group

CSF, cerebrospinal fluid; M−, interictal migraine; M+, ictal migraine; SD, standard deviation; SCID 1 and 2, Structured Clinical Interview for DSM-IV Axis I and 2 Disorders, Research Version/Non-patient Edition.
PC-PLC in cerebrospinal fluid
The hydrolysis of PC by PLC forms a fluorescent product and our initial studies indicate that PC-PLC activity increased with CSF protein (Figure 1A). The initial rate of PC hydrolysis was linear (dotted line). The specific inhibitor, D609, reduced PC-PLC activity (Figure 1B). The D609 added to duplicates of four different CSF samples showed significant inhibition with increasing concentration of D609 ( PC-PLC activity is present in human CSF and is suppressed by a PC-PLC-specific inhibitor. (A) PC-PLC activity [mean (SE), 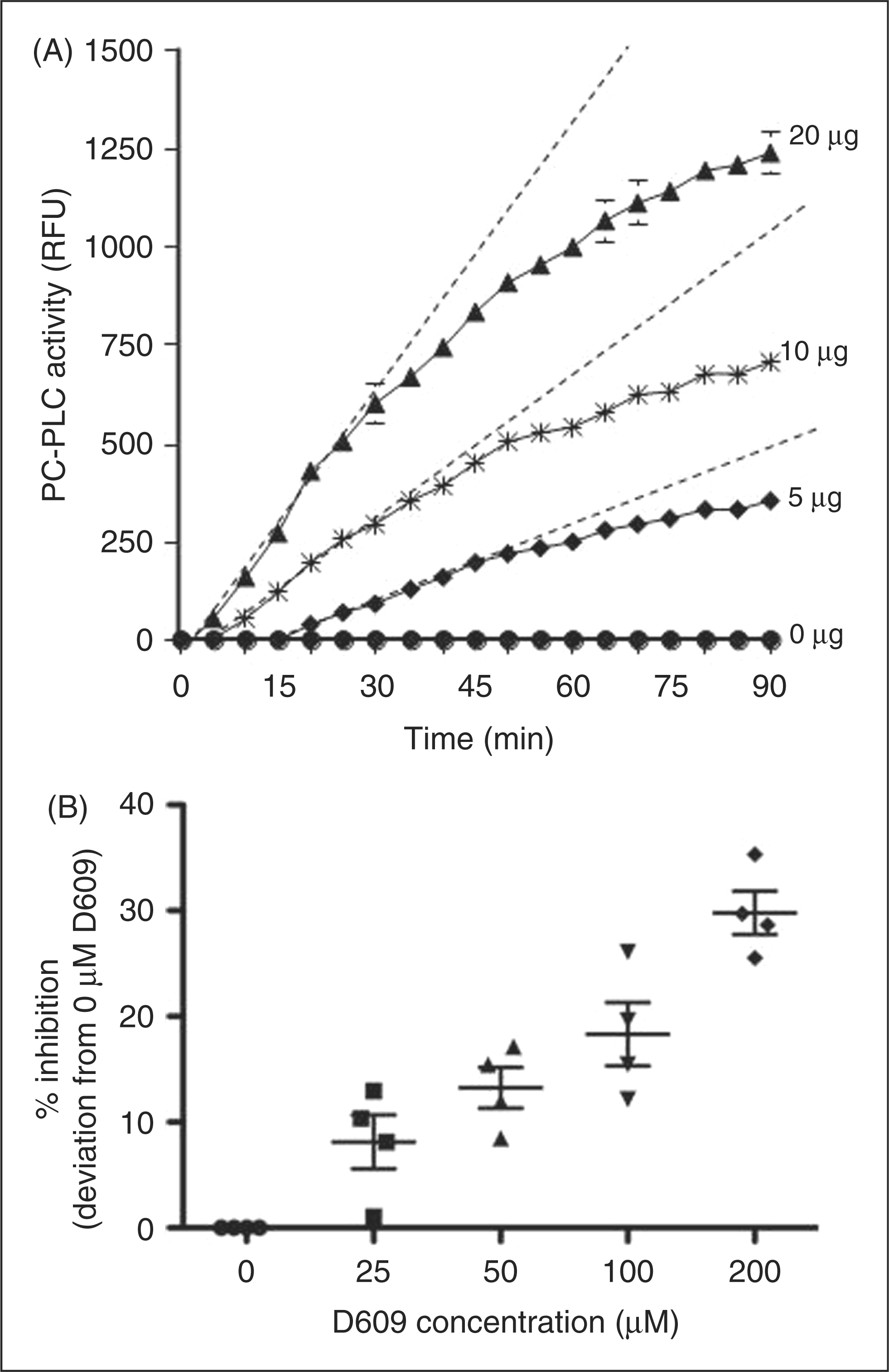
PC-PLC activity significantly differed by diagnostic group ( PC-PLC activity in CSF from controls and interictal and ictal migraineurs. (A) CSF PC-PLC activity was measured in controls (C, 
PC-PLC activity in plasma
PC-PLC activity was also present in blood plasma but it was 250–350 times lower than in CSF (data not shown). Moreover, the activity was not significantly different between either paired [mean (SD) M− = 0.04 (0.004) RFU/µg/min; M+ = 0.03 (0.008) RFU/µg/min,
PC-PLC, recent annual frequency and lifetime duration of migraine
PC-PLC activity in CSF from the M+ state was negatively correlated with migraine frequency [mean (SD) = 50.3 (43.8) days] over the previous year (r = −0.82,
Discussions
The data support the original hypothesis
Our objective was to test the hypothesis that PC-PLC—an enzyme important in receptor-mediated signal transduction—is expressed in CSF and that its activity is altered in migraine.
The data in Figure 1 provide the first evidence, to our knowledge, of PC-specific PLC activity in CSF. The enzyme is also present in blood plasma. We propose that the substantially smaller activity in plasma reflects the abundant supply of PC-PLC in blood cells (11). By contrast, the paucity of cells in CSF necessitates that any enzyme will be in the fluid, although the functions and regulation of the abundant CSF PC-PLC remain unknown.
Our observation that PC-PLC activity in CSF was significantly higher in the M+ state compared to healthy controls (Figure 2A) supports the second part of our hypothesis, that PC-PLC activity is altered in migraine. The paired analysis of seven participants who provided CSF in both M+ and M− states emphasizes this finding by showing that PC-PLC activity was significantly increased in the M+ compared to the M− state (Figure 2B).
Our observation that blood plasma PC-PLC did not change in migraine suggests that the CSF changes in migraine arise within the nervous system and not from systemic circulation.
PC-PLC activity and migraine frequency
An unexpected result from our study was the inverse correlation between migraine frequency over the preceding year and PC-PLC activity in CSF in the M+ state. In the M− state, the PC-PLC activity was also negatively correlated with lifetime migraine duration, though this was not significant (
Clinical heterogeneity
For these experiments, the prospective and invasive collection of CSF necessitates that we limit the number of participants. This highlights the need for optimal clinical diagnosis and inter-group matching. We are confident of the diagnosis and matching of the clinical populations of M+ and M−; however, the controls were older and took less medication. The medication difference was predictable, since established migraine sufferers take medications that healthy volunteers do not require. Importantly, age, gender and medication use were not significantly correlated with PC-PLC activity levels in migraineurs. To further address the age and medication differences between migraineurs and controls, we performed a paired analysis using a subgroup of seven migraineurs who were sampled first during migraine and later when they were completely recovered. This method controlled for any possible influence of age and medication effects on PC-PLC because, while the migraineur transitioned from M+ to M− state over a four-week period, their daily medications were unchanged. Furthermore, each of these seven participants took completely different drug components, none of which are known to affect PC-PLC. Thus, the results of the paired analysis provide supporting evidence that PC-PLC changes between M+ and C are migraine state–dependent and not due to the influence of age or medication.
Study limitations
Small sample size, possible recruitment bias, and the recall method of measuring headache frequency and intensity are major limitations of this study that render these findings preliminary evidence for altered CSF PC-PLC activity in migraine.
Pathophysiological interpretation
Because CSF functions are not well understood, interpreting roles for any CSF component is still largely limited to the theory that they reflect the composition of the local nervous system (12). However, accumulating evidence suggests that CSF has an active role in biological functions of the brain (13–18), including exocytosis, release of transmitters and bulk flow of active neuropeptides. While the source of PC-PLC in CSF is not known, we recently showed that CSF contains membrane-bound nanostructures with signaling capabilities (19). Thus, it seems reasonable that the aforementioned bulk-flow and nanostructure-regulated components will include PC-PLC-mediated signaling pathways. A physiologic function of CSF may include production and transport of signaling molecules and this will benefit from the presence of the lipid-metabolizing enzyme, PC-PLC. Alternatively, composition will also reflect deviations, as we report in this migraine shift of PC-PLC; such changes will likely contribute to pathophysiology. The PC-PLC changes in CSF that we report in migraine are limited to the comparison with migraineurs when well and healthy controls. Studies in other disorders and pain conditions are required to evaluate the specificity of the PC-PLC activity in migraine.
Any function for the PC-PLC activity in CSF also remains to be established. However, the abundance of brain phospholipids (8) indicates that important physiological roles require PC-PLC. Moreover, increased activity during migraine (Figure 2) suggests a pathophysiological role. Migraine is a multifaceted disease that involves complex signaling pathways mediated by several G-protein coupled receptors. Since the signaling pathways for most of these ligands involve PC-PLC activation (20), the increased activity of PC-PLC in migraine is likely due to activation by a combination of several receptors. Receptor modulators such as adrenergic or dopaminergic antagonists and serotonin agonists are useful in migraine treatment (21); since signaling from these receptors utilizes PLC-derived mediators, it is reasonable to predict that migraine variations and these treatments will modulate brain and CSF PC-PLC. We conclude that the PC-PLC activation in CSF reported in this study reflects a common event in migraine pathophysiology.
Footnotes
Funding
This work was supported by the National Institutes of Health, R01 NS043295; the Norris, Glide, Hezlep, Lucas Brothers, and Posthuma Foundations; and Thermo Fisher Scientific.
Acknowledgements
We thank Drs Xianghong Yang and Mercedes Gacad for their critical review and comments during the preparation of this manuscript. We thank our study participants for their time and for donating CSF and plasma samples. We acknowledge administrative help by Elena Oborina and thank Drs Chequer and Cadogan for CSF collection.
Supplemental methods and medication description
PC-PLC assay: PC-PLC from Bacillus cereus was used as a positive control to estimate the PLC protein content of CSF. Reaction buffer alone without CSF was used as a negative control. Briefly, CSF was diluted in sample buffer [50 mM Tris-HCl (pH 7.4), 0.14 M NaCl, 10 mM dimethylglutarate, 2 mM CaCl2] to ∼0.1 mg/ml. All samples and controls were placed in 96 well plates in triplicates. A10-mM stock solution of Amplex Red reagent in dimethyl sulfaxide (DMSO) was made by adding 0.13 mg of Amplex Red to 50 mL DMSO. An Amplex Red working solution was prepared by adding 50 ml Amplex Red reagent, 50 mL horseradish peroxidase (HRP), 25 mL lecithin, 50 mL choline oxidase, and 4.8 mL 1X Reaction Buffer. PLC activity was started by adding 100 ml Amplex Red working solution to 100 mL of diluted CSF samples. The plate was incubated at 37°C with monitoring of fluorescence using excitation at 545 nm and emission at 590 nm every five minutes for 90 minutes. The increase in fluorescence was recorded and enzyme activity calculated as RFU/µg/min.
PLC activity (in RFU/µg/min) was determined from the initial slope of the reaction when it was linear and before substrate became limiting. Specificity for PC was determined by measuring the percent inhibition in the presence of increasing amounts of the D609 inhibitor. Statistical analysis confirmed that each of the increasing quantities of D609 significantly inhibited the fluorescence formation and that inhibition increased with increasing concentrations of D609.
