Abstract
Objectives
The objectives of this article are to evaluate the association between migraine and trigeminal neuralgia and to investigate the effects of age, sex, migraine subtype, and comorbid risk factors on trigeminal neuralgia development.
Methods
This population-based cohort study was conducted using data from Taiwan’s National Health Insurance Research Database. Individuals aged ≥ 20 years with neurologist-diagnosed migraine between 2005 and 2009 were included. A non-headache age-, sex-, and propensity score-matched control cohort was selected for comparison. All participants were followed until the end of 2010, death, or the occurrence of trigeminal neuralgia. Cox proportional hazards regression was used to calculate hazard ratios (HRs) and 95% confidence intervals (CIs) for comparison of the risk of trigeminal neuralgia between groups.
Results
Both cohorts (n = 137,529 each) were followed for a mean of 3.1 years. During the follow-up period, 575 patients (421,581 person-years) in the migraine cohort and 88 matched controls (438,712 person-years) were newly diagnosed with trigeminal neuralgia (incidence rates, 136.39 and 20.06/100,000 person-years, respectively). The HR for trigeminal neuralgia was 6.72 (95% CI, 5.37–8.41; p < 0.001). The association between migraine and trigeminal neuralgia remained significant in sensitivity analyses. Among migraine subtypes, patients with migraine with aura were at greater risk of trigeminal neuralgia development. No other significant interaction was identified in subgroup analyses.
Conclusions
Migraine is a previously unidentified risk factor for trigeminal neuralgia. The association between these conditions suggests a linked underlying mechanism, which is worthy of further exploration.
Introduction
Trigeminal neuralgia (TN) is a disease characterized by sudden and recurrent, usually unilateral, severe, brief episodes of stabbing pain over one or more branches of the trigeminal nerve (1). The pain is located more often on the right than on the left side, and is most commonly limited to the second or/and third division of the trigeminal nerve (involvement of the first division is rare) (2). Remission of TN can last for months to years, but the pain-free intervals may gradually shorten between episodes (1,3). TN onset usually occurs in middle or old age, and the disease is more prevalent in women (2,4). Patients with TN often have trigger factors, such as blowing wind, eating, shaving, drinking, or applying makeup, which they try to avoid. The lifetime prevalence of TN is estimated to be 0.3%, and the annual incidence rate is about 4.3–27 per 100,000 population (1,4,5).
The diagnosis of TN is based largely on clinical history. The International Headache Society now differentiates between classical TN, in which no identifiable cause other than microvascular compression at the trigeminal root entry zone of the brainstem can be found, and painful trigeminal neuropathy, caused by herpes zoster infection, tumor, multiple sclerosis, or trauma (6). Hypertension has been suggested to be a risk factor for the development of TN, possibly due to the acceleration of arterial tortuosity (7), but this association requires further investigation. TN is presumed to be a type of neuropathic pain, which may be precipitated by injury to the trigeminal axons in the nerve root or ganglion, causing demyelination and remyelination of the trigeminal nerve (8,9). The injury renders the trigeminal axons and axotomized somata hyperexcitable, contributing to pain paroxysms as a result of ectopic generation of spontaneous nerve impulses. These impulses create the characteristic “electrical explosion” of pain via electrical crossover between demyelinated neurons (ephaptic crosstalk) (9).
The association between migraine and TN has not been well studied. One study reported that the frequency of migraine in patients with TN was 9% (2). In another study, 2% of TN patients were found to have a familial predisposition to migraine (10). Traditionally, central sensitization has been deemed the origin of pain in patients with migraine (11,12). However, increasing evidence indicates that activation of the peripheral trigeminovascular system plays an important role in the pathophysiology of migraine (13–15). Microscopically, disrupted myelin sheaths and discontinuous neurofilaments of the zygomaticotemporal branch of the trigeminal nerve were noted in surgical patients with refractory migraine (16). These pathological findings may resemble those in patients with TN who have undergone microvascular decompression surgery (8,17). Thus, this study was conducted to determine whether TN is associated with migraine using the nationwide population-based Taiwanese National Health Insurance Research Database (NHIRD).
Methods
Data source
Taiwan’s National Health Insurance (NHI) program, launched in 1995, currently provides comprehensive medical coverage to 99% of the population of 23 million residents. The NHI offers comprehensive medical coverage, including outpatient, inpatient, emergency, dental, and traditional Chinese medicine services, as well as prescription drugs. Data for this study, including enrollment files, claims data, and drug prescriptions, were extracted from the NHIRD. This database provides comprehensive utilization and enrollment information for all patients in the NHI program. Diseases were coded according to the 2001 International Classification of Diseases, ninth revision, Clinical Modification (ICD-9-CM) (18). Several studies have validated the accuracy of numerous diagnoses recorded in the NHIRD (19–21). All information in this database that would potentially reveal individual patients’ identities has been anonymized, and the confidentiality of data is ensured according to the regulations of the NHI Bureau and the National Health Research Institute. The institutional review board (IRB) of Taipei Veterans General Hospital approved this study (IRB ID number: 2013-03-039BC).
Study design and cohorts
This nationwide, population-based, observational study involved two cohorts: the migraine cohort, comprising all patients in Taiwan with a diagnosis of migraine (ICD-9-CM code 346.x) who visited neurologists between January 2005 and December 2009, and the non-headache control cohort, comprising individuals with no diagnosis of migraine (code 346.x), tension-type headache (code 307.81), or headache (code 784.0), extracted from a dataset of 1 million beneficiaries sampled randomly from the original NHIRD (Figure 1). Three subgroups of migraine were defined: migraine with aura (ICD-9-CM codes 346.00–346.01), migraine without aura (ICD-9-CM codes 346.10–346.11) and unspecified migraine (ICD-9-CM codes 346.90–346.91). Each person with migraine entered the cohort on the day of the first clinical visit associated with a neurologist’s registry of the diagnosis of migraine in the dataset. The exclusion criteria for the recruitment of individuals into both cohorts were: 1) age <20 years, 2) antecedent TN (ICD-9-CM code 350.1) before the index visit, and 3) diagnosis of multiple sclerosis (ICD-9-CM code 340) or benign or malignant neoplasm of the nervous system (ICD-9-CM codes 191, 192, 225) before or within two years after the index visit. These criteria were applied to reduce the likelihood of painful trigeminal neuropathy development. Individuals in whom migraine and TN were both diagnosed within a 30-day period were also excluded to minimize the possibility of alternative diagnoses that might contribute to atypical facial pain and migraine-like headache. People in both cohorts who were followed for <30 days were excluded.
Propensity score matching
We used a multivariable logistic regression model to calculate the propensity scores for the predicted probabilities of migraine status. The covariates used in generating the propensity scores are listed in supplementary Table e-1 on the Cephalalgia website. For each patient with migraine, we matchd one control subject according to propensity scores, which are conditional estimations of treatment assignment based on observed baseline covariates (22). For each patient in the migraine cohort, we identified one control patient with similar demographic characteristics, matched in terms of age (±5 years), and propensity score (±0.1) for the likelihood of migraine diagnosis, calculated using multivariate logistic regression analysis and baseline covariates. Participants in the migraine cohort who could not be matched with controls were excluded from the study.
Outcome
The primary outcome was the occurrence of TN, defined as at least two ambulatory medical visits with the principal diagnosis of TN (ICD-9-CM code 350.1) during the follow-up period. Patients with concomitant diagnoses of TN and herpes zoster infection (ICD-9-CM code 053.xx) within a 30-day period were excluded to minimize the chance of misclassification of Ramsay-Hunt syndrome or trigeminal post-herpetic neuralgia as TN. Both cohorts were followed until December 31, 2010, death, or the occurrence of TN.
Baseline characteristics
Baseline demographic data, including age, sex, medical care utilization (ambulatory visits in the past year), income, and urbanization level, were collected. The Charlson Comorbidity Index (CCI) was used to determine participants’ overall systemic health (23); higher CCI scores are associated inversely with the 10-year survival rate, i.e. a score of 0 is associated with a 10-year survival rate of 99% and a score of 5 is associated with a rate of 34% (23). Data on other systemic diseases not included in the CCI, such as atrial fibrillation, valvular heart disease, hypertension, asthma, dyslipidemia, and drug abuse, were also recorded.
Statistical analysis
Descriptive statistics were used to examine the baseline characteristics of the study cohorts. The characteristics of the two groups were compared using the Pearson chi-squared test for categorical variables and the independent t-test for parametric continuous variables. The incidence rates of TN in both groups were calculated using Poisson distributions. We calculated adjusted absolute incidence rates of TN to evaluate the contribution of each risk factor to the occurrence of TN using the macro developed by Zhao (24). The SAS GENMOD function of this macro was used with the Poisson distribution option to calculate predicted adjusted rates at mean levels of the independent variables. The adjusted absolute rates are presented with upper and lower 95% confidence intervals (CIs) per 100,000 person-years. The relative risk of TN was compared between groups using hazard ratios (HRs) derived from Cox regression models constructed with a conditional approach using stratification. We further examined the interactions between migraine and the following variables: age, sex, CCI score, diabetes mellitus, hypertension, and migraine subtypes by likelihood ratio tests. Sensitivity analyses were conducted to validate our results in the following patients: (1) those with the initial diagnosis of TN made by a neurologist, and (2) those receiving carbamazepine or oxcarbazepine prescriptions following the diagnosis of TN.
The Microsoft SQL Server (2012; Microsoft Corporation, Redmond, WA, USA) was used for data linkage, processing, and sampling. Propensity scores and adjusted absolute incidence rates were calculated with SAS software (version 9.2; SAS Institute Inc, Cary, NC, USA). All other statistical analyses were conducted using STATA statistical software (version 12.0; StataCorp, TX, USA). The p values were two tailed with a level of significance of 5%.
Results
Characteristics of the study population
Demographic and clinical characteristics of migraine patients and controls.

Unless otherwise indicated, data are presented as n (%).
Imbalance defined as absolute value >0.007.
Including aspirin, clopidogrel, ticlopidine, and cilostazol.
SD: standard deviation; NT$: new Taiwan dollars; ACE: angiotensin-converting enzyme; ARB: angiotensin II receptor blocker.
Incidence rates of TN
Incidence rates and risks of trigeminal neuralgia in matched migraine and control cohorts.

Per 105 person-years.
CI: confidence interval.
Clinical characteristics of migraine patients and controls who developed trigeminal neuralgia.
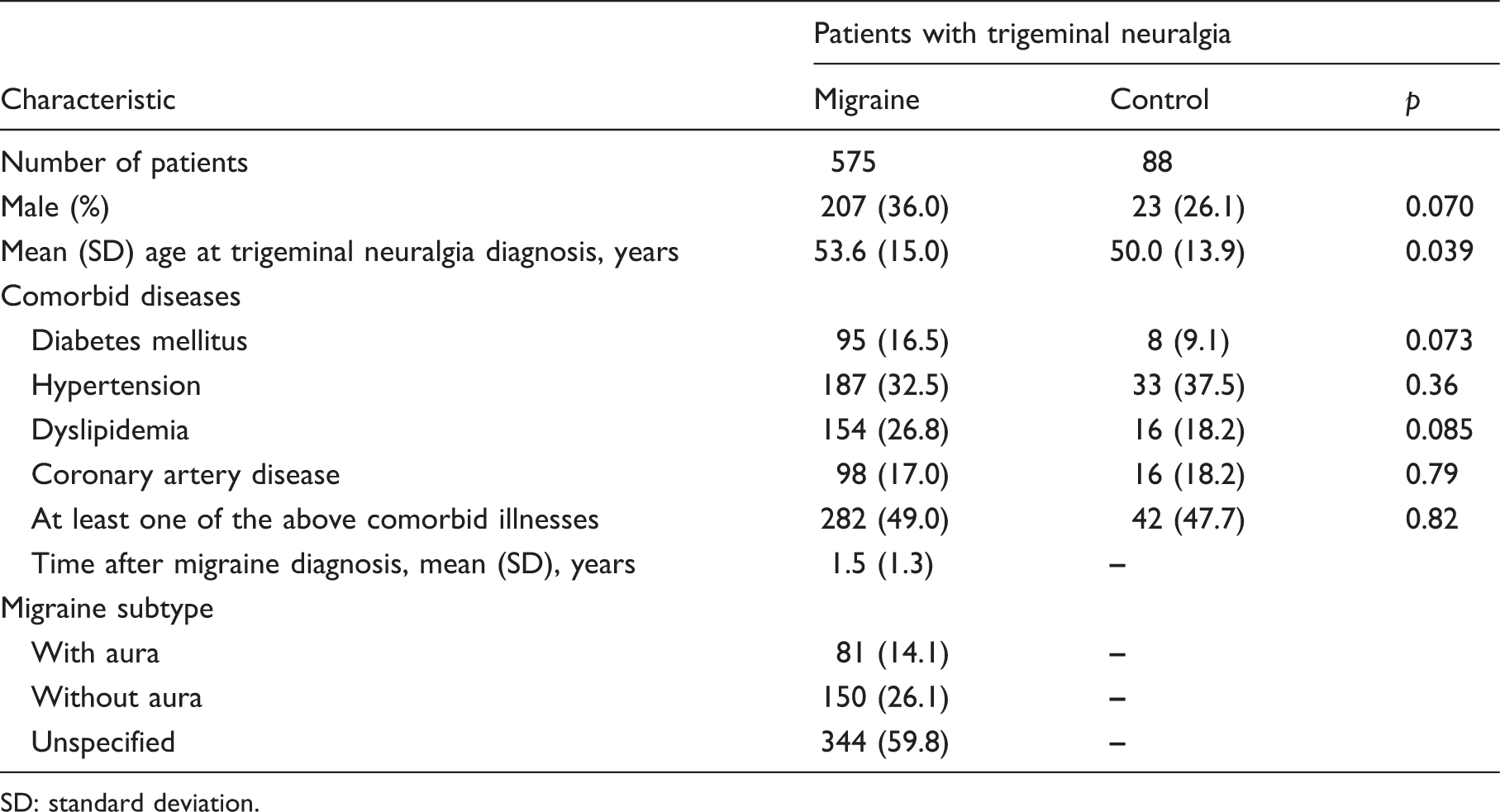
SD: standard deviation.
Sensitivity analysis of the risk of TN
Sensitivity analyses of associations between the risk of trigeminal neuralgia and migraine.

Per 104 person-years.
Adjusted for propensity score.
CI: confidence interval.
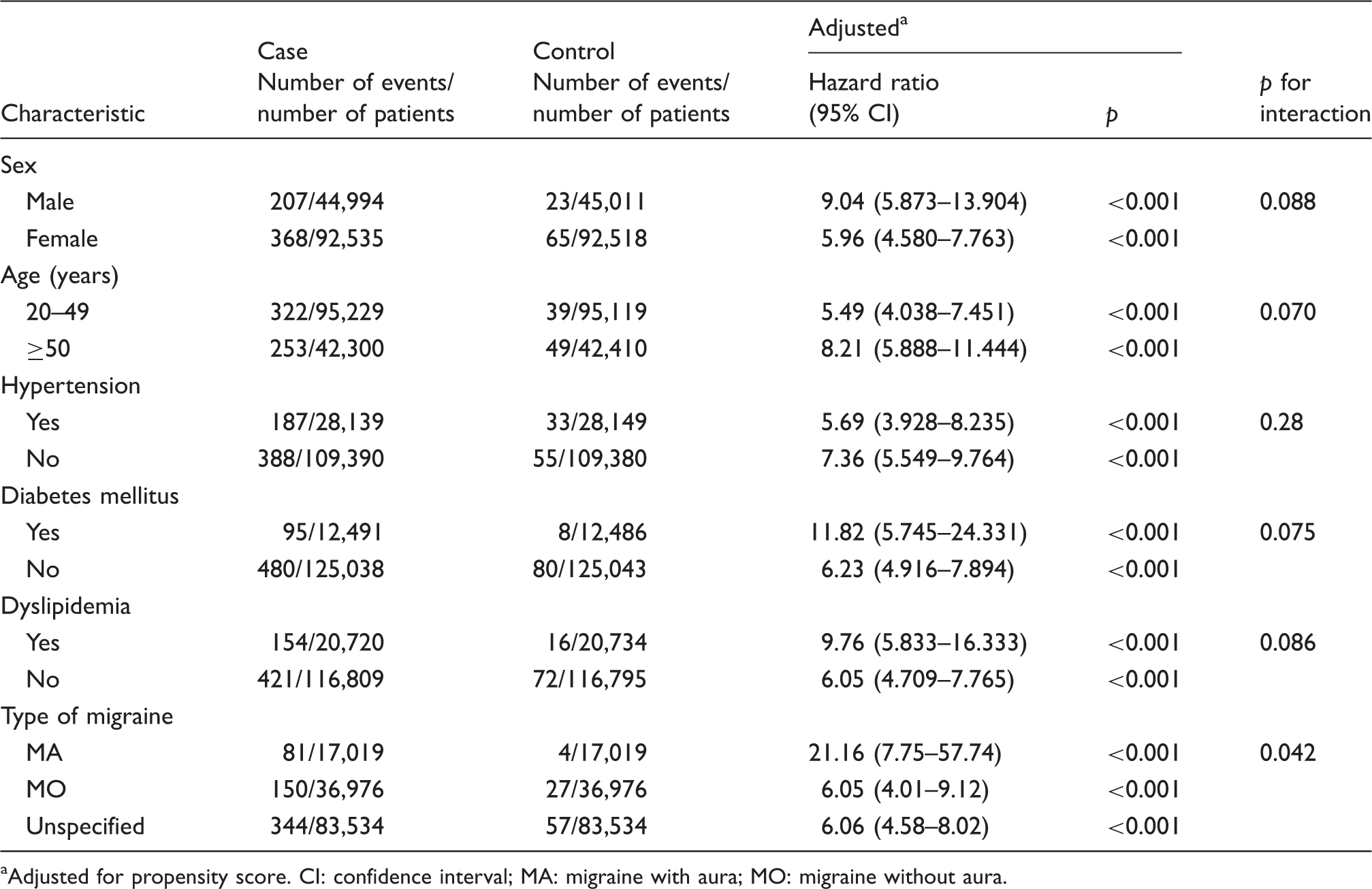
Subgroup analyses of the risk of TN
Migraine subtype interacted significantly with the risk of TN. In analyses stratified by migraine subtype, the association between migraine and the risk of TN was stronger in patients with migraine with aura. Other subgroup analyses revealed no significant effect of gender, age, hypertension, diabetes, or dyslipidemia on TN risk (Table 5).
Flowchart of patient enrollment. Subgroup analysis of risk of trigeminal neuralgia in migraine and control cohorts. aAdjusted for propensity score. CI: confidence interval; MA: migraine with aura; MO: migraine without aura.
Discussion
The results of our study indicate that the risk of TN increased up to six times in patients with migraine compared with age-, sex-, and propensity score-matched control individuals after a mean follow-up period of 3.1 years. This association was not affected by sex, age, diabetes, dyslipidemia, or hypertension. To our knowledge, this population-based insurance cohort study is the first to demonstrate such an association.
The cumulative annual incidence rate of TN in the control cohort is similar to previous study results (4.3–27/100,000 person-years) (1,4,5,25). TN is the most common facial pain in patients 50 years or order, and its etiology is believed to be related primarily to microvascular compression of the trigeminal nerve root by an aberrant loop of an artery or vein (8,17,26–28). This compression leads to demyelination and remyelination at the root entry zone of the trigeminal nerve, generating spontaneous ectopic nerve impulses and electrical crossover between demyelinated neurons (9). Other rare etiologies include primary demyelinating disorder, such as multiple sclerosis (8). Some patients with peripheral nerve demyelination due to Charcot-Marie-Tooth disease also develop TN (29). Pathological examination has revealed a zone of demyelination in the proximal portion of the trigeminal root (part of the central nervous system), near the junction with the peripheral nervous system (8).
The mechanisms underlying the association between migraine and TN remain unknown. Migraine attacks can excite the trigeminal vascular system, and trigger the release of inflammatory neuropeptides, thereby causing inflammation of nearby cranial nerves (15,30–32). We hypothesize that this cascade may predispose the trigeminal nerve to undergo further demyelination after microvascular compression. Evidence of deregulation of myelination and axonal abnormalities of the trigeminal nerve has been found in patients undergoing surgical decompression for severe migraine (16,33). In the current study, the relative HRs of the risk of TN failed to reach statistical significance in all of the subgroup analyses performed, except for migraine subtypes (i.e. the presence or absence of aura) (Table 5). Therefore, we hypothesize that migraine, rather than its cardiovascular comorbidities, may be associated with the subsequent development of TN.
The major strengths of our study include the use of a large nationwide population-based database and enrollment only of patients diagnosed by neurologists or treated with carbamazepine or oxcarbazepine. Neurologists’ diagnoses of migraine in Taiwan based on the International Classification of Headache Disorder, second edition, criteria have shown 91.6% accuracy (34). We also used an age-, sex-, and propensity score-matched control cohort to reduce selection bias. Third, stricter inclusion criteria were used to reinforce our study results. The HRs of TN remained consistently higher in the migraine cohort than in the control cohort in all analyses.
Several limitations of our study should be addressed. First, the diagnosis of TN depends largely on clinical information and, to some degree, medication response (6,25). Some atypical facial pain is difficult to differentiate from TN (35), potentially leading to misdiagnosis. Hence, overestimation of the occurrence of TN was possible in both cohorts. Second, the NHIRD contains limited information, and we could not determine whether the course of TN differed between cohorts. We also could not examine the impact of migraine-preventive medication on the occurrence rate of TN. Third, we used a neurologist-diagnosed migraine cohort, which may have led to Berkson’s bias, i.e. patients who sought neurological consultation were more likely to be diagnosed with TN. Fourth, our study did not reconfirm the diagnoses of TN by neurologists or headache specialists. Instead, we used sensitivity analyses to consolidate the diagnosis of TN because the risk of development of TN remained regardless of different limitations to the criteria of TN diagnoses.
In summary, our study provides new clinical data with several implications. Patients with TN should be asked about their migraine history. However, there is insufficient evidence to indicate that migraine prevention reduces the occurrence of TN. Based on the interaction tests performed, our study illustrates that hypertension or other cardiovascular comorbidities do not appear to play a role in the development of TN among migraine patients. Nevertheless, further studies are required to clarify the association between migraine and TN, and to explore the common pathophysiological mechanisms bridging these two conditions.
Clinical implications
Migraine increases the relative risk of trigeminal neuralgia six-fold. In approaching patients with trigeminal neuralgia, the query of past history of migraine may be crucial. Whether adequate migraine treatment can prevent subsequent TN needs further study.
Footnotes
Acknowledgments
Author contributions: Kuan-Hsiang Lin: study concept and design, data analysis and interpretation, drafting and revising of manuscript; Yung-Tai Chen: statistical analysis, study concept and design, data analysis and interpretation, drafting and revising of manuscript; Jong-Ling Fuh: study concept and design, data analysis and interpretation, critical revision of the manuscript for important intellectual content; Shuu-Jiun Wang: study concept and design, critical revision of the manuscript for important intellectual content, study supervision.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jong-Ling Fuh is a member of a scientific advisory board of Elli Lilly and Novartis, and has received research support from the Taiwan National Science Council and Taipei-Veterans General Hospital; Shuu-Jiun Wang has served on the advisory boards of Allergan and Elli Lilly Taiwan. He has received speaking honoraria from local companies (Taiwan branches) of Elli Lilly and GSK. He has received research grants from the Taiwan National Science Council, Taipei-Veterans General Hospital and Taiwan Headache Society. The other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from Ministry of Science and Technology of Taiwan (104-2314-B-010-015-MY2 and 103-2321-B-010-017-), Taipei-Veterans General Hospital (VGHUST104-G7-1-1, V104C-082, V104E9-001), Ministry of Science and Technology support for the Center for Dynamical Biomarkers and Translational Medicine, National Central University, Taiwan (MOST 103-2911-I-008-001), Academia Sinica (Grant No. IBMS-CRC103-P04), Brain Research Center, National Yang-Ming University, Ministry of Health and Welfare (MOHW104-TDU-B-211-113-003), and a grant from Ministry of Education, Aim for the Top University Plan.
