Abstract
Trigeminal neuralgia is considered as a paroxysmal single nerve phenomenon. Abnormal sensory perception has been previously described in 15-25% of patients with clinical examination. Quantitative sensory testing (QST) was used to evaluate sensory perception in patients with idiopathic trigeminal neuralgia (ITN). Nine patients and 10 normal control subjects were evaluated in all six trigeminal branches. QST abnormalities were found in the symptomatic division and in the other two branches on the same side. Minor contralateral changes were also found. Differences consisted of cold and warm hypoaesthesia and higher cold and heat pain thresholds in patients. All differences proved statistically significant. Our findings suggest that trigeminal neuralgia is not only a paroxysmal single nerve disorder, but also that other higher structures may be involved.
Introduction
Although idiopathic trigeminal neuralgia (ITN) is commonly considered as a positive neuralgic phenomenon, sensory loss was first described by clinical examination in 1938 in up to 25% of patients (1). While touch, pinprick and cotton wool examination may show no impairment, evaluation with graduated hairs improves the accuracy of sensory loss detection. Using Von Frey's filaments, abnormal thresholds were found in 15–25% of patients despite the lack of subjective sensory impairment (2–5). Unlike electromyography and nerve conduction studies, quantitative sensory testing (QST) allows the evaluation of unmyelinated (C) and small calibre myelinated (A-δ) fibres (6, 7).
Using thermal as well as pinprick and two-point discrimination stimuli to evaluate sensory thresholds in patients with trigeminal neuralgia, Nurmikko in 1991 (8) disclosed several abnormalities. Raised warm and tactile thresholds were observed in affected divisions. In adjacent branches, a significant elevation of warm, hot, pain and two-point discrimination thresholds was found compared with the contralateral side. However, no comparison with data obtained from normal subjects was made.
QST provides a specific procedure to test a wide spectrum of cutaneous modality thresholds in ITN. Cold sensation, warm, heat, cold pain and also vibration stimuli may thus be evaluated in an objective manner.
We studied the three sensitive divisions of the fifth nerve on both sides of the face in trigeminal neuralgia patients, and analysed the results by comparing data with those obtained from age-matched controls.
Patients and methods
Patients
Nine trigeminal neuralgia patients with 1–16 years’ symptom duration were studied, all of whom met International Headache Society (IHS) criteria (9). Five females and four males, mean age 57.6 years (range 37–72), were evaluated. Treatment included carbamazepine in five patients and gabapentin in one, while three patients were not under treatment. All nine cases were experiencing a painful period; however, pain was neither present nor triggered during the study. Only one patient reported a sensation of numbness in the symptomatic trigeminal branch. None of the patients had undergone trigeminovascular decompression surgery and all had magnetic resonance imaging studies (T1, T2 weighted and FLAIR sequences). There was no evidence of demyelinated lesions or macroscopic vascular compression at trigeminal emerging level at the pons.
Normal values were obtained from 10 normal subjects, two males and eight females with a mean age of 57 years (range 28–75).
Methods
A Medoc 2001 Thermal Sensory Analyser was used. A 30 × 30-mm Peltier thermode was applied on the skin territory of the ophthalmic (V1), maxillary (V2) and mandibular (V3) branches of both trigeminal nerves in every patient and control subject in randomized order. In order not to cross accidentally the boundaries of adjoining dermatomes, stimuli were applied on the emergence point of each branch. Results from measurements were pooled to form groups in the following way: affected divisions characterized by the presence of trigger zones (Affected), contralateral-mirror image divisions (Mirror), unaffected trigger free divisions ipsilateral and adjacent to affected branch (Neighbour) and contralateral to affected division adjacent to mirror (Contralateral). Warm and cool thresholds as well as hot and cold pain thresholds were determined for each trigeminal division. Starting with a temperature adaptation of 32 °C, ascending and descending ramps of 1 °C/s were delivered.
The subject was instructed to stop stimulus delivery in order to determine the four modalities under study. Examiners and subjects were not blinded to the diagnosis but had no access to the data screen until data were completely collected. Patients and controls underwent an instruction period, followed by three repeated tests whose scores were averaged.
Stimuli were automatically stopped at 0 and 50 °C to prevent skin damage (6–8).
Results were analysed by comparing the affected branch with the other trigeminal divisions in each patient (mirror, neighbour, and contralateral branches). These data were also compared with those obtained from control subjects.
Statistical analysis
Due to a low number of samples and their abnormal distribution, a Kruskal–Wallis one-way method was used to assess the significance of findings between each trigeminal branch. Threshold significance level was scaled down to P < 0.05. Statistix 7.0 Analytical Software was employed. All data are displayed on box and whisker plot figures showing median and interquartile intervals. The asterisk indicates extreme values for possible outliers and circles indicate probable outliers.
Results
Our results are resumed in Table 1, in which are displayed mean temperature thresholds with their standard deviations and significance between each branch and controls.
Mean temperature (°C) thresholds on the four trigeminal branches evaluated in patients and in control subjects
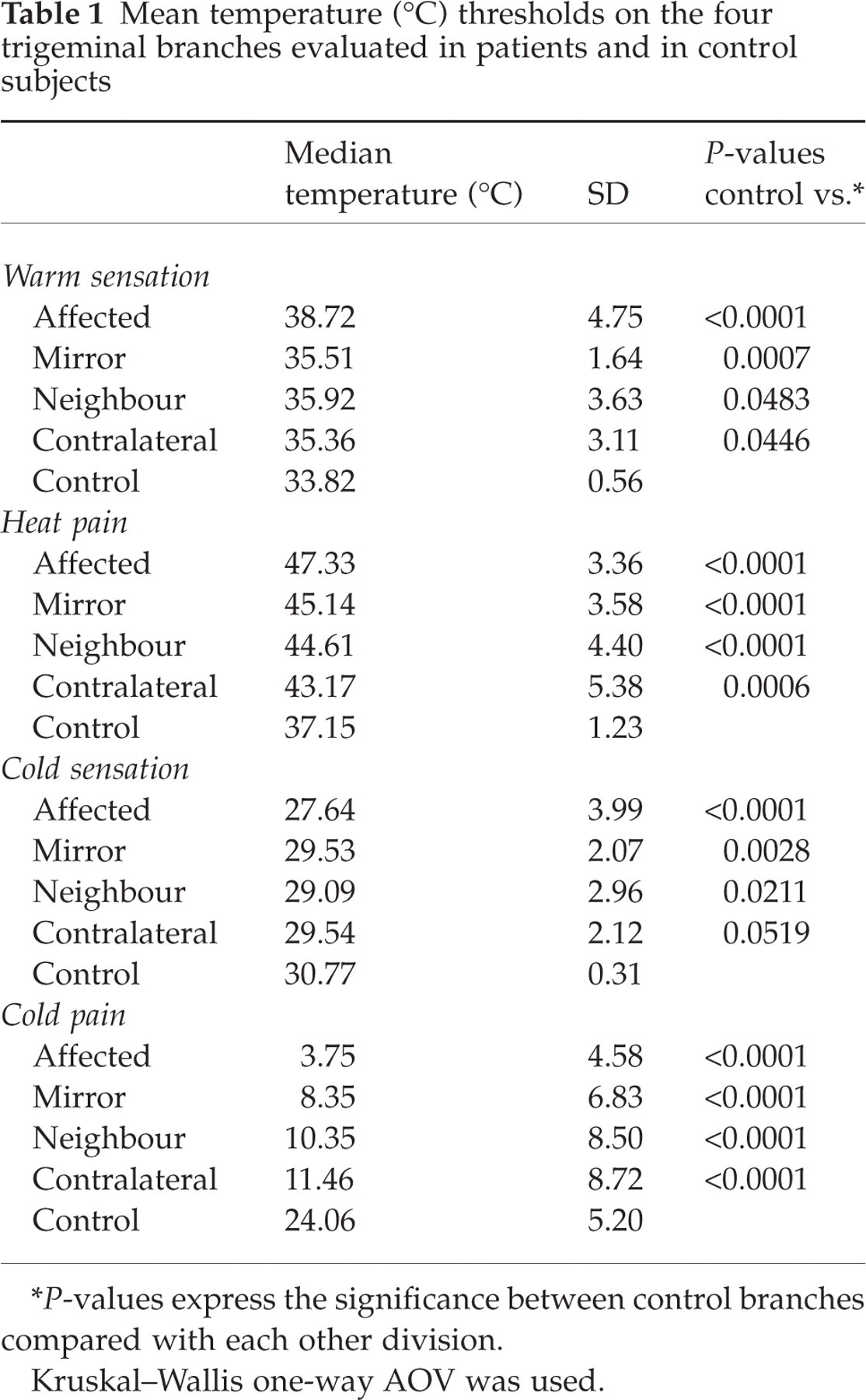
∗P-values express the significance between control branches compared with each other division.
Kruskal–Wallis one-way AOV was used.
Figure 1 compares warm sensation thresholds from the affected branch with neighbour and contralateral branches, as well as with the control group. Warm sensation thresholds showed a decrease in affected branches passing through unaffected ones down to control values. Warm threshold was increased to 38.7 °C in the affected root, whereas in healthy sections values ranged from 35.3 to 35.9 °C depending on which branch was evaluated. Control subjects stopped the stimulus at 33.8 °C. However, these differences achieved statistical significance only when comparing control trigeminal branches with those of patients .

Warm sensation thresholds-median and interquartile intervals. P-value expresses the significance between control branches compared with each other division. ∗Extreme values for possible outliers; circles: extreme values for probable outliers.
All heat pain thresholds measured in ITN patients showed significant hypoalgesia compared with control subjects regardless of the side and vicinity of the painful branch (P < 0.001), as shown in Fig. 2.
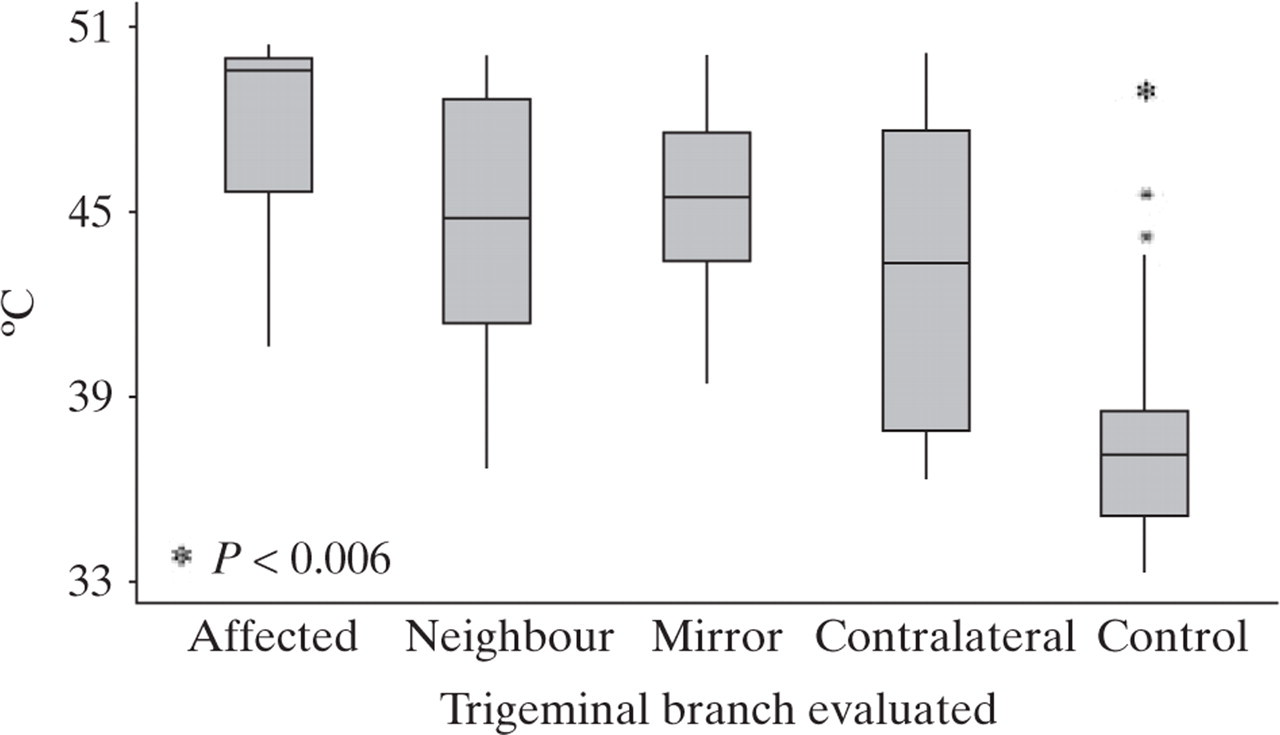
Heat pain thresholds. Median and interquartile intervals. P-value expresses the significance between control branches compared with each other division. ∗Extreme values for possible outliers; circles: extreme values for probable outliers.
Figure 3 shows data from cold sensation thresholds. When analysing cold sensation, the same hypoaesthesia pattern was highly significant (P < 0.05) for all trigeminal branches in our patients.
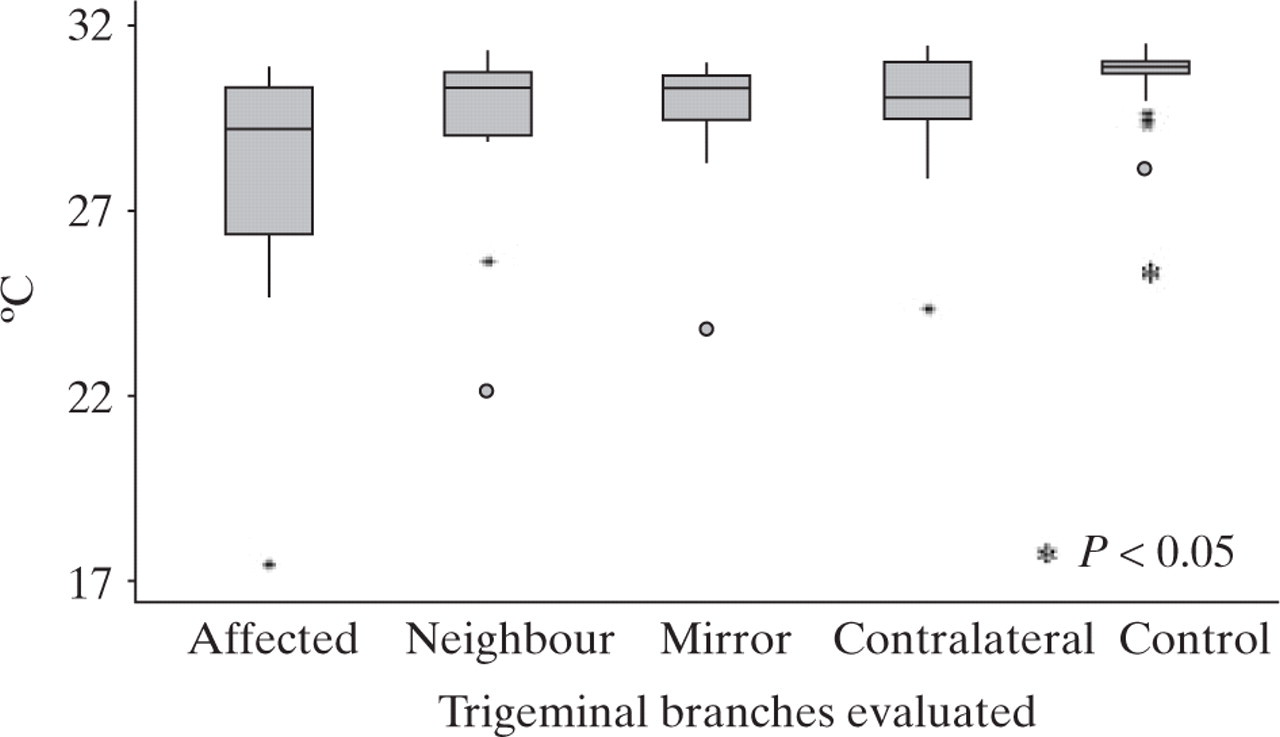
Cold sensation thresholds. Median and interquartile intervals. P-value expresses the significance between control branches compared with each other division. ∗Extreme values for possible outliers; circles: extreme values for probable outliers.
Regarding cold pain, significant hypoalgesia was found in patients’ branches when compared with the trigeminal divisions of healthy subjects (P < 0.001) (Fig. 4).

Cold pain thresholds. Median and interquartile intervals. P-value expresses the significance between control branches compared with each other division. ∗Extreme values for possible outliers; circles: extreme values for probable outliers.
Interestingly, the four figures (Figs. 1–4) disclose an obvious trend showing a higher degree of involvement at the level of trigeminal neuralgia branches.
Discussion
In 1988, the IHS established the classification and diagnostic criteria for headache disorders, cranial neuralgia and facial pain (9).
Two major diagnostic criteria for idiopathic trigeminal neuralgia are the absence of interictal (i) neurological symptoms or (i) sensory impairment. However, our QST findings support permanent sensory dysfunction in this group of patients.
Chronic drug intake could be suspected to induce sensory threshold elevation in these patients, although Wang et al. studied the effect of analgesic drugs on QST but failed to find significant differences compared with placebo (10). While anti-epileptic drugs may cause mild neuropathy, it seems unlikely that a systemic drug can specifically affect a nerve, as our symptomatic patients showed more hypoaesthesia in involved branches, Furthermore, drug-induced polyneuropathy is usually distal and, particularly when axial nerves are affected, may prove severe. Indeed, this suspicion may turn untreated controls into an unreliable comparison group.
It has been proposed that chronic pain may lead to habituation to external stimuli. Using sensory quantification in chronic pain, Leffler et al. (11) found threshold changes only in the affected area at the time of acute pain, but not after pain relief or between paroxysms.
The study was not blinded to the diagnosis, although this is true in relation to the design. It is very important to consider the fact that the determination of the threshold is absolutely independent of the observer's intervention since it is the patient that halts the stimulus when he actually feels the sensation. The method, then, although subjective for the patient, is objective for the observer, who has no possibility of influencing the result. This precludes any investigator bias.
Our findings suggest that in patients with trigeminal neuralgia there is generalized sensory dysfunction involving not only the symptomatic branch but also, though to a lesser degree, all facial trigeminal divisions.
Future evaluations in this field, with a greater number of subjects under study, may allow patient subgroups to be discerned based on disease duration, another factor which may add further information on the natural course of the disease and also when comparing untreated cases.
Trigeminal neuralgia should not be considered merely as a peripheral nerve dysfunction. It is possible that hemicranial or hemifacial pain may induce changes in cutaneous sensation distant from the painful area, as shown by Burstein et al. in their migraine series (12), whereas our trigeminal neuralgia patients display a strictly unilateral pain pattern. It should therefore be of interest to explore thermal and cutaneous sensation on cervical 2 and 3 areas, which share the same spinal trigeminal nuclei.
Concomitant changes in nervous system structures, occurring soon after onset of disease or over the years, could be responsible for such neuropathic findings. A longitudinal study, starting with the first painful episodes, would thus be necessary in order to determine whether idiopathic trigeminal neuralgia is a progressive disorder.
