Abstract
Background: Previous studies have reported an increased frequency of restless legs syndrome (RLS) in adult migraine patients. Until now, the frequency of RLS in pediatric patients has not been investigated. We set out to assess the frequency of RLS in children and adolescents with migraine compared to headache-free controls.
Methods: We investigated 111 consecutive patients with a sole diagnosis of migraine with or without aura presenting to the Headache Unit at the Department of Child and Adolescent Psychiatry and 73 headache-free controls for the presence of RLS using a semistructured interview. In addition, we assessed the level of daytime sleepiness by means of the Epworth sleepiness scale (ESS). A second group of headache-free controls was screened for the presence of RLS using an online questionnaire.
Results: The frequency of RLS in migraine patients was significantly higher than in controls (22% vs. 5% (p < 0.001) and 8% (p < 0.001)).
Discussion: This is the first study suggesting an association between RLS and migraine in the pediatric population. Future studies are needed to determine the extent of sleep disruption in children and adolescents with migraine and comorbid RLS.
Introduction
Migraine may be complicated by comorbidities which augment the perceived severity of migrainous symptoms. Most recently, evidence for a possible association between migraine and restless legs syndrome (RLS) has been summarized (1). Several clinic-based studies in adult populations found a significantly increased frequency of RLS in migraine patients (2,3), while a single observational study did not confirm an association between migraine with aura and RLS (4). A large population-based study showed an association between migraine and RLS, which was similar for migraine with and without aura (5).
RLS is a common sleep-related movement disorder characterized by the following diagnostic criteria: an urge to move that is usually associated with unpleasant sensations, and symptoms that are worse at rest, relieved by movement and most severe at night (6). The prevalence of RLS in children and adolescents was found to range between 2% (7) and 4% (8,9).
To date, possible pathophysiological links between migraine and RLS remain speculative. The latter is thought to be caused by a dysfunction within the dopaminergic system (10). This notion is supported by the rapid improvement of RLS symptoms after treatment with dopaminergic agents (11). Hypersensitivity of peripheral and central dopamine receptors has been reported in migraine patients (12). Migraineurs with comorbid RLS have reported a higher number of dopaminergic accompanying symptoms (13). Most recently, elevated serum phosphorus levels have been reported in migraineurs with comorbid RLS (14), which may reflect dopaminergic dysfunction (15). Both disorders may be linked by abnormalities of the iron metabolism. In RLS patients symptoms can be aggravated by iron deficiency (16). Especially in chronic migraineurs, increased iron storage in the periaqueductal grey matter has been reported, thus pointing to an abnormal iron metabolism in the brain (17).
To our knowledge, there are no studies investigating a possible association between pediatric migraine and RLS. Based on the published data on adult migraine patients and our personal clinical experience that children and adolescents with migraine frequently complained of “painful or fidgety legs”, we decided to perform a controlled study on children and adolescents aged between 5 and 18 years focusing on the question: Do children and adolescents with migraine (with and without aura) suffer from RLS more frequently than headache-free controls?
Methods
Participants
Between January 2009 and June 2010 we included all consecutive patients seen at the Headache Unit at the Department of Child and Adolescent Psychiatry of the University Hospital of Vienna. The patients underwent a thorough medical, neurological and psychiatric examination. To record the headache history and symptomatology cross-sectionally, we used a semistructured interview published elsewhere in detail (18).18 Headache diagnosis was based on the diagnostic criteria of the International Headache Society (ICHD-II [19]). Inclusion criteria for patients were (1) a sole diagnosis of migraine (with or without aura) according to ICHD-II and (2) age between 5 and 18 years. Exclusion criteria for patients were (1) any additional headache type and (2) any other significant medical, neurological or psychiatric condition.
We included two groups of controls: The first group was recruited from the outpatient clinic of the Department of Pediatrics and Adolescent Medicine by the authors A.B. and W.Sch. (group 1). These children and adolescents were exclusively screened at follow-up after recovery from a minor illness and did not suffer from a significant medical, neurological or psychiatric condition at time they were included in this study. The subjects in control group 1 were seen between October 2011 and January 2012 at the outpatient clinic of the Department for Pediatrics and Adolescent Medicine. During that time a total of 180 patients were seen in the outpatient clinic, and 73 patients were free of an acute disease and had no chronic disease specified as exclusion criterion. The most common diagnoses in this group were remitted infections of the lower respiratory tract (i.e. bronchitis and pneumonia) (50%), infections of the ear, throat and sinuses (40%) and gastrointestinal problems (e.g. vomiting or diarrhea) (7%).
The second control group was recruited from Viennese primary and grammar schools (group 2). One of the authors (G.G.) personally went to these primary and grammar schools and explained the aim of the study and asked the teachers to support the younger children to fill out the questionnaires or even read the questions to them if their reading skills had not been developed that far.
Inclusion criteria for controls (group 1 and 2) were (1) age between 5 and 18 years and (2) absence of any headache during the last year. Exclusion criteria for controls were any significant medical, neurological or psychiatric condition. The study protocol was approved by the ethics committee of the Medical University of Vienna and informed consent was obtained from all study participants and/or their parents.
Questionnaires
RLS questionnaire administered to patients and controls.

The questions written in bold represent the four core diagnostic criteria for RLS.
RLS: restless legs syndrome.
Modified Epworth sleepiness scale.20
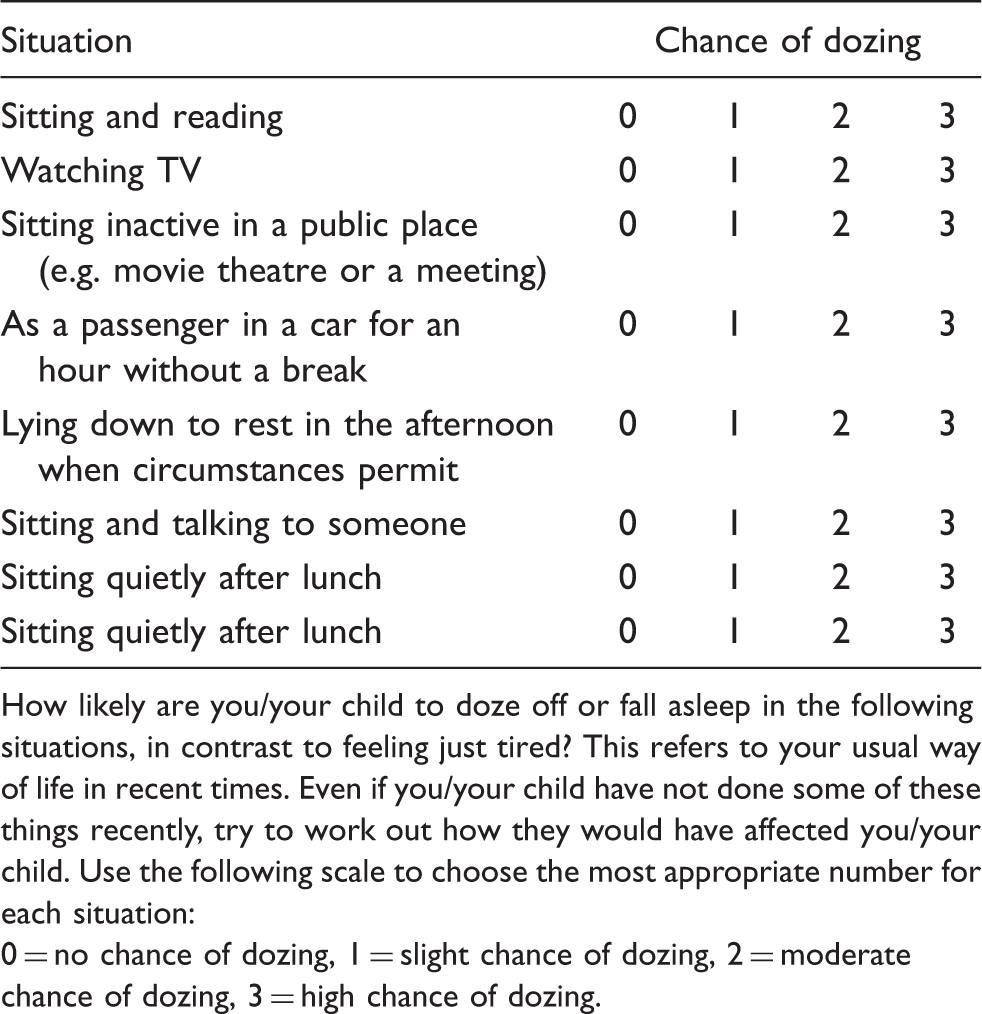
How likely are you/your child to doze off or fall asleep in the following situations, in contrast to feeling just tired? This refers to your usual way of life in recent times. Even if you/your child have not done some of these things recently, try to work out how they would have affected you/your child. Use the following scale to choose the most appropriate number for each situation:
0 = no chance of dozing, 1 = slight chance of dozing, 2 = moderate chance of dozing, 3 = high chance of dozing.
Diagnosis of restless legs syndrome
All patients and controls in control group 1 were accompanied by their mothers and/or fathers. They filled out the RLS questionnaire (Table 1) as well as the (ESS) (20) together with their parents immediately before their appointment with a physician experienced in the field of pediatric headaches/sleep disorders (Y.W.-B., S.S., S.N. or A.B.). The physician verified each single answer in the RLS questionnaire with the patient/control and parents, if necessary, in child-appropriate language, and asked the patient/control specifically for the following RLS mimics: leg cramps, positional discomfort and pronounced or frequent unconscious foot or leg movements (hypnic jerks, habitual foot tapping, leg shaking or general nervous movements). Thus, we tried to ensure that all questions were understood correctly and to eliminate false-positive and false-negative answers in the questionnaire. In none of the patients or controls was the diagnosis of RLS based entirely on parents’ reports.
RLS diagnosis in parents of patients and control group 1 was established by asking about the presence of RLS features as published by Allen et al. (6) during the visit with their child.
Statistical analysis
Statistical analysis was conducted by using the Statistical Package for the Social Sciences software version 17.0 (SPSS Inc., Chicago, IL, USA). Differences between groups were tested by using chi-square and Fisher’s exact test as well as two-sided student’s t-test for normal distributed data and Mann-Whitney U-test for not-normal distributed data. We also calculated the odds ratio (OR) and 95% confidence interval (CI) for the association between migraine and RLS. Due to the different RLS screening method, we did not use the percentage of RLS cases in control group 2 for the statistical analysis regarding the association between migraine and RLS. Logistic regression models were used to determine if the difference in the frequencies of RLS between migraine patients and control group 1 remained significant after adjustment for age, gender, RLS family history and ESS total score. Values represent percentages or means ± SD. p values ≤ 0.05 were considered statistically significant.
Results
Demographic characteristics
Between January 2009 June 210 and November 2011 and January 2012, we screened 141 patients, 75 controls (group 1) and 136 controls (group 2). Thirty patients (21%) and two (3%) controls in group 1 were excluded because of comorbid tension-type headache, and 28 (21%) controls in group 2 were excluded because they did not complete the questionnaire (N = 7) or reported a current illness (allergy, N = 7; asthma, N = 7; neurodermitis, N = 2; scoliosis, N = 2; hypothyreosis, N = 1; tumor, N = 1; and psoriasis, N = 1).
Demographic and clinical details of migraine patients and control groups 1 and 2.
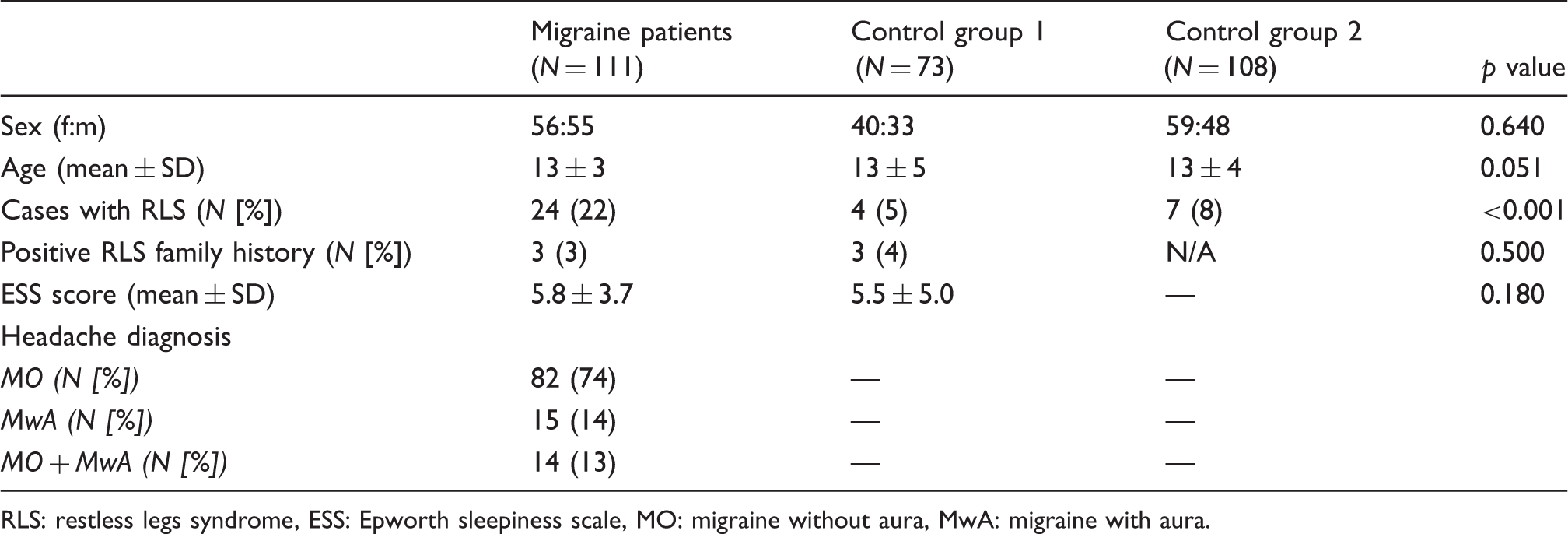
RLS: restless legs syndrome, ESS: Epworth sleepiness scale, MO: migraine without aura, MwA: migraine with aura.
Migraine
Headache diagnoses comprised migraine with aura (MwA) in 15 (14%), migraine without aura (MO) in 82 (74%) and MwA + MO in 14 (13%) patients (Table 3). Patients suffered from migraine for a mean 3.2 ± 2.8 years. Frequency of migraine attacks was less than once a week in 43 (39%) patients and more than once a week in 68 (61%) patients. Headache was aggravated by physical activity in 108 (97%) patients. Regarding accompanying symptoms, nausea was reported by 93 (84%), vomiting by 69 (62%), photophobia by 106 (95%) and phonophobia by 108 (97%) patients. None of the migraine patients received a prophylactic medication at the time of the study and none used metoclopramide to treat nausea during migraine attacks.
Restless legs syndrome and daytime sleepiness
Twenty-four migraine patients (21.6%), 4 (5%) controls in group 1 and 9 (8.3%) controls in group 2 met the diagnostic criteria for definite RLS in children and adolescents. The difference between the migraine patients and control group 1 was statistically highly significant (OR 3.9, 95% CI 1.4–10.9, p < 0.001) (Table 3). RLS prevalence did not differ statistically significantly between the two control groups. After controlling for age, gender, RLS family history and ESS total score by logistic regression analysis, migraine patients still showed a significantly higher frequency of RLS than control group 1 (OR 5.3, 95% CI 1.5–18.6, p < 0.001).
Four migraine patients with RLS were aged between 2 and 12 years. The number of verbal descriptors for their leg discomfort ranged between two and six. Neither patients nor controls with RLS had been treated because of RLS before.
Family history of RLS was positive in three (3%) patients and three (4%) controls in group 1. Intriguingly, only one (5%) patient with definite RLS and none of the controls with definite RLS in group 1 had a positive family history.
Mean ESS total scores of migraine patients and controls in group 1 did not differ significantly (5.8 ± 3 .7 vs. 5.6 ± 5.1, p = 0.4) (Table 3).
Discussion
This is the first study addressing an association between RLS and migraine in children and adolescents. Our main finding is an approximately fourfold-higher RLS frequency in migraine patients compared to headache-free controls.
In contrast to the adult population (2,3), knowledge about the relationship between migraine and RLS in children and adolescents is practically non-existent. Tomás-Vila et al. (21) found an association between headache and rhythmic movement disorders, waking up more than twice during the night and excessive daytime sleepiness in school-aged children. Migraine has been found in 9% of a sample of children and adolescents with periodic limb movements (22). Our data are in line with results from studies in adult migraine patients (2,3) and show an approximately fourfold-higher frequency of RLS in pediatric migraine patients compared to headache-free controls.
The RLS frequency found in control group 1 roughly matches the data of previous studies (7,8,23,24). Interestingly, RLS frequency was 8% in control group 2. The methodological difference regarding the screening for RLS (face-to-face interview vs. online questionnaire) may have led to the inclusion of RLS mimics like growing pains and muscle cramps known to bias data on RLS prevalence. As shown previously, the expert interview is able to reduce the number of false-positive cases of RLS (25).
RLS is highly familial in early-onset cases, defined as those in which restless legs symptoms first occur before 35–40 years of age (26). In a population-based study of pediatric RLS, a parental history indicative of RLS was found in 71% of children 8–11 years of age and 80% of adolescents 12–17 years of age (7). In contrast, and quite surprisingly, we found only a single RLS case (5%) in our patient group to be familial.
Our study is clearly strengthened by the fact that it is the first to identify an increased frequency of RLS among children and adolescents with migraine and the inclusion of two separate control groups. We used the state-of-the-art diagnostic criteria for migraine according to ICHD-II (19) and pediatric RLS as administered by Picchietti et al. (7).
Our study also has some limitations: its cross-sectional design and the administration of strict inclusion/exclusion criteria does not allow us to generalize our findings to the whole population. Since our headache outpatient center for children and adolescents is the only one in Vienna, we see most of the pediatric headache patients. We acknowledge that our migraine patients may suffer from severe migraine attacks more frequently, which may have influenced the patients’ quality of sleep and hence the level of excessive daytime sleepiness. RLS also causes sleep disturbances which may lead to more and severe migraine attacks. We did not perform full-night polysomnography/actigraphy to diagnose RLS/periodic limb movement disorder in patients and controls. We may also have included cases of secondary RLS among our study population. We asked about all active medical conditions and current medication, but we did not perform laboratory tests. Secondary RLS may be caused by low serum ferritin, renal failure, pregnancy, polyneuropathy and antidopaminergic agents (27). Due to the low occurrence of these factors in a pediatric population we conclude that the majority of our patients were suffering from primary RLS (28). The absence of a validated tool for the diagnosis of RLS in this young population poses a challenge to all studies dealing with this subject. We think that the administration of a questionnaire with diagnostic questions in the presence of the parents and verification of the answers during a face-to-face interview by an experienced physician ensured the validity of the RLS diagnoses.
In conclusion, this study shows an increased frequency of RLS in a pediatric migraine population. Future studies should address the quality of sleep and possible comorbidities (e.g. depression and anxiety) in pediatric migraine patients with RLS.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
Ch. Wöber has received honoraria and travel funding from AstraZeneca (Austria), Linde Gas (Austria), A. Menarini Pharma GmbH (Austria) and Pfizer (Austria). The other authors do not report any conflict of interest.
