Abstract
Aims
Our studies investigated the location of oxytocin receptors in the peripheral trigeminal sensory system and determined their role in trigeminal pain.
Methods
Oxytocin receptor expression and co-localization with calcitonin gene-related peptide was investigated in rat trigeminal ganglion using immunohistochemistry. Enzyme-linked immunosorbent assay was used to determine the effects of facial electrocutaneous stimulation and adjuvant-induced inflammation of the temporomandibular joint on oxytocin receptor expression in the trigeminal ganglion. Finally, the effects of oxytocin on capsaicin-induced calcitonin gene-related peptide release from dural nociceptors were investigated using isolated rat dura mater.
Results
Oxytocin receptor immunoreactivity was present in rat trigeminal neurons. The vast majority of oxytocin receptor immunoreactive neurons co-expressed calcitonin gene-related peptide. Both electrocutaneous stimulation and adjuvant-induced inflammation led to a rapid upregulation of oxytocin receptor protein expression in trigeminal ganglion neurons. Oxytocin significantly and dose-dependently decreased capsaicin-induced calcitonin gene-related peptide release from dural nociceptors.
Conclusion
Oxytocin receptor expression in calcitonin gene-related peptide containing trigeminal ganglion neurons, and the blockade of calcitonin gene-related peptide release from trigeminal dural afferents suggests that activation of these receptors may provide therapeutic benefit in patients with migraine and other primary headache disorders.
Introduction
The nine amino acid peptide oxytocin, first isolated in 1927 (1), is primarily recognized as a hormone involved in the induction of uterine contractions (2), lactation (3), and, more recently, for its role in social behavior (4). In fact, oxytocin infusion has been used for inducing labor since the 1950s (5–8). Additionally, oxytocin was also approved in the USA for nasal application to stimulate milk ejection and for prevention of mastitis from 1960–1997 (9).
In addition to its involvement in uterine contraction, lactation and psychosocial processes, oxytocin has also been implicated as an endogenous modulator of pain. Endogenous oxytocin has been measured in human dorsal root and trigeminal ganglion (11), and terminals of hypothalamic neurons containing specific oxytocin neurophysin carrier proteins have been immunohistochemically localized to the dorsal horn of human spinal trigeminal nucleus (12). Similarly, Swanson and McKellar (13) identified oxytocin receptors within regions of the spinal cord involved in nociceptive transmission in monkeys. Several other groups (14–17) found similar spinal cord distribution of oxytocin receptors in the rat. There is evidence from animal studies for the analgesic effects of oxytocin (18–22) mostly after intrathecal or intracerebroventricular injection. In addition, accumulating evidence suggests that breastfeeding, with the accompanying elevated concentrations of oxytocin, may have a protective effect on migraine recurrence in the postpartum period (23–25). Although the above studies suggest that trigeminal oxytocin receptors may play a role in trigeminal pain perception, there are only limited data on oxytocin receptor expression and their role in the trigeminal ganglion and nucleus, since most studies focused on expression in reproductive tissues, such as mammary gland or endometrium. Clearly, however, expression of oxytocin receptors in these tissues is labile and rapid expression changes have been observed, which are driven, at least in part, by the presence of inflammatory cytokines (26,27).
The focus of the current studies is to investigate whether: (1) oxytocin receptors are expressed on trigeminal primary afferent neurons; (2) oxytocin receptors are co-expressed on calcitonin gene-related peptide (CGRP) positive neurons, presumptively involved in nociception; (3) oxytocin receptor expression on these neurons is enhanced under painful inflammation induced either by injection of complete Freund’s adjuvant (CFA) into the temporomandibular joint (TMJ) or through prolonged electrocutaneous stimulation of the cheek; (4) oxytocin can prevent capsaicin-induced CGRP release from trigeminal afferent neurons that innervate the dura mater.
Methods
Animals
Male Sprague–Dawley rats (225–275 g, Harlan Laboratories, CA, USA) were used for the experiments. Rats were maintained two per cage in a controlled environment (temperature: 21.5 ± 4.5℃; relative humidity: 35–55%) under a standard 12 h light/12 h dark lighting cycle. Cage changes occurred twice per week, using standard bedding. All procedures were approved by the Stanford University IACUC.
Oxytocin receptor expression/co-expression with CGRP
Naïve rats (N = 6) were sacrificed using CO2 inhalation and perfused with phosphate-buffered saline followed by 4% paraformaldehyde. Trigeminal ganglia were removed and stored at 4℃ overnight in 4% paraformaldehyde containing 20% sucrose. Ganglia were then embedded in paraffin and mounted on a chuck for cryosectioning. Sagittal sections (5 µm thick) were mounted on glass slides, followed by paraffin removal using sequential washes in xylene and ethanol, and then rehydrated in deionized water. Standard procedures for antigen retrieval employed heating the rehydrated tissue for 30 min at 95℃ in citrate buffer. Sections were rinsed in phosphate-buffered saline supplemented with 0.05% Tween-20 for two washes of 2 min. Sections were blocked in TBST buffer (TRIS-balanced salts containing 0.05% Tween-20) supplemented with 5% goat serum. Primary antibodies: oxytocin receptor, goat polyclonal anti-oxytocin receptor (N-19; Santa Cruz Biotechnology, catalog # sc-810, antibody dilution 1:50; blocking peptide sc-8013p), CGRP, sheep polyclonal anti-CGRP (Abcam, catalog #ab22560, dilution 1:2000; rat CGRP peptide, Abcam, catalog #ab47101). Secondary antibodies: rhodamine (TRITC)-conjugated rabbit anti-goat IgG (Abcam catalog # ab6738, dilution 1:200); fluorescin thioisocyanate (FITC)-conjugated rabbit anti-sheep IgG (Abcam, catalog # ab6743, dilution 1:500). Separate images of the two fluorophores marking each antigen were obtained using a Nikon PCM-2000 laser scanning confocal microscope at standardized gain settings to minimize overlap of the fluorescence intensity. CGRP-positive cells were selected for measurement only if the image for oxytocin showed a sharp and clear nuclear profile, indicating a mid-diameter cross-section. Such cells were compared with neighboring unstained (i.e. CGRP-negative) cells of similar diameter.
Induction of inflammation/chronic pain
In addition to naive controls (N = 3), which were anesthetized but not treated, trigeminal ganglia were also harvested from rats 24 h after either: (1) CFA injection (N = 3) into the TMJ; or (2) electrocutaneous shock to the face (N = 15, three per time point) as described below.
CFA injection into the TMJ
In order to produce a robust inflammation of trigeminally innervated tissue, the TMJ of some rats were injected with CFA. Rats were placed in an anesthesia chamber and anesthetized with 2.5% isoflurane. Fifty µL of CFA (DIFCO; Sigma Aldrich, St. Louis, MO, USA) was injected (1 mL syringe with a 26 G × 5/8-inch needle) into the left TMJ in order to produce robust and prolonged orofacial inflammation. To do this, prior to injection, the rat’s mouth was propped open to palpate the target area of the TMJ. In this position, an oval-shaped groove located in the center of the cheek and above the mandible can be distinctly felt. With the syringe positioned at a 30° angle from the rat’s cheek, the tip of the needle was inserted just under the articular disc (approximately 1.5 mm in diameter and 1.0 mm deep). After CFA injection, rats were returned to their home cages.
Electrocutaneous stimulation
In order to produce a time-controlled inflammation of trigeminally innervated tissue, some rats were administered electrocutaneous stimulation of the cheek. Rats were lightly anesthetized with 2.5% isoflurane prior to electrocutaneous stimulation. While anesthetized, the left cheek was depilated (NAIR® hair removal cream; Church & Dwight Co., Ewing, NJ, USA). Once hair was removed, the cheek was thoroughly cleaned with 70% ethanol and the rat was returned to the nose cone supplying isoflurane (1.5%). Rats were placed on a heated pad to maintain body temperature during noxious electrical stimulation. Once positioned, the exposed area of cheek was covered with an EEG conductive paste (NuPrep™; Weaver and Company, Aurora, CO, USA) and needle electrodes were inserted into the skin overlaying the left masseter muscle approximately 1 cm apart. Electrical pulses (10 ms duration, 1 Hz, 0.4 mA) were delivered continuously for 2 h using an isostimulator (Model A320; World Precision Instruments, Sarasota, FL, USA).
Twenty-four hours after CFA injection or 2, 4, 6, 8, or 24 h after termination of electrocutaneous stimulation, rats were euthanized by CO2 inhalation. Trigeminal ganglia were harvested within 15 min of death, snap-frozen in liquid nitrogen, transferred to pre-weighed Eppendorf-style micro-centrifuge tubes, and stored at −80℃.
Tissue processing
The weight of frozen ganglia was recorded prior to processing. Individual frozen ganglia were transferred onto an aluminium block chilled on a bed of dry-ice, broken in small 1–2 mm pieces using a razor blade and transferred into a chilled Dounce tissue grinder (1 mL volume) with 1 mL of hypotonic buffer added. Tissue was disrupted by 20–30 passes of the glass pestle with the tissue suspension then transferred to a 1.5 mL Eppendorf-style micro-centrifuge tube. Following snap-freezing in liquid nitrogen and thawing on wet ice, a membrane pellet was isolated by centrifugation (20,000 × g for 40 min at 4℃). After removing the supernatant, the pellet was re-suspended in 1 mL hypotonic buffer and again spun (20,000 × g for 40 min at 4℃). Following removal of the supernatant, the membrane pellet was disrupted by addition of lysis buffer (5 µL per mg tissue or a minimum of 150 µL, whichever was greater). Lysis buffer was composed of standard diluent solution, provided with the enzyme-linked immunosorbent assay (ELISA) kit (see below), supplemented with 1.25% (w/v) CHAPS (#220201; EMD Millipore) and protease inhibitor (Protease Inhibitor Cocktail 3, Calbiochem, 1:200 dilution). The lysis solution was transferred into a chilled Dounce tissue grinder (1 mL volume) and the membranes disrupted by 10–15 passes of the tight-fitting pestle. The tubes were stored on wet ice for 1 h with occasional vortexing. Solubilized membrane protein, including oxytocin receptor, was separated from insoluble material by a final spin (20,000 × g for 40 min at 4℃).
Measurement of oxytocin receptor protein levels
Oxytocin receptor protein was measured using a commercial ELISA kit (USCN Life Science, Inc., Wuhan, People’s Republic China) according to manufacturer’s protocol. Assay standard was diluted in lysis buffer to produce a standard curve from which oxytocin receptor content was extrapolated following correction for sample dilution. Briefly, the oxytocin receptor content in the ELISA wells was multiplied by the ratio of the total lysate volume divided by the lysate volume used in the ELISA (obtaining units of pg/mL). This value, in turn, was multiplied by the total volume of lysate (in mL) to obtain the oxytocin receptor content of trigeminal ganglia (in units of pg). Data were analyzed using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test to compare oxytocin receptor concentrations in the trigeminal ganglia of naïve animals to those in inflamed animals (i.e. CFA and shock, respectively). All analyses were conducted at the 0.05 level of statistical significance and used two-tailed tests.
Effect of oxytocin on capsaicin-induced CGRP-release from dural nociceptors
In order to provide evidence for a role of oxytocin in trigeminal pain mechanisms, we examined whether oxytocin could block the release of CGRP. Capsaicin-induced CGRP-release from dural nociceptors was measured using a similar protocol as reported by Reeh et al. (28,29). Rats were injected bilaterally with 50 µL of CFA (DIFCO, Sigma Aldrich) into the vibrissal pads to induce robust inflammation or with vehicle (mineral oil) as a control. Two days later, rats were euthanized by CO2 inhalation, followed by immediate decapitation. Skulls were hemi-sectioned, and the brain tissue removed in order to expose the dura in two (half) skull chambers. Chambers were filled with artificial cerebrospinal fluid (aCSF) containing NaCl 107.8 mM, KCl 3.5 mM, MgSO4*7H2O 0.69 mM, NaHCO3 26.2 mM, NaH2PO4*2H2O 1.67 mM, Na-gluconate 9.64 mM, glucose 5.55 mM, saccharose 7.6 mM, and (added after saturating with CO2) CaCl2 0.025 mM for 10 min to establish baseline CGRP levels (first round). Then new aCSF with capsaicin 10 µM was filled into the hemi-sectioned skulls for 10 min (second round) to activate dural afferents and evoke CGRP release (N = 9). Animals without injections of CFA into the vibrissal pads (N = 8) were also tested in an identical manner, to quantify increased CGRP-release after facial inflammation. In additional experiments, oxytocin (1 µM (N = 7), 10 µM (N = 10), and 100 µM (N = 8)) was added to the aCSF in the first and second rounds to test for inhibitory effects on capsaicin-induced CGRP release. After each round, aCSF was pipetted into individual Eppendorf tubes (1.5 mL; Santa Cruz Biotechnology, Dallas, TX, USA) and stored at −80℃ pending further analysis. CGRP concentrations in aCSF were assayed by ELISA (SPIBio, Bertin pharma, Montigny le Bretonneux, France) according to the manufacturer’s protocol. For each individual dura sample, CGRP concentrations in the first exposure to aCSF with or without oxytocin were measured and set to 1. CGRP concentrations after capsaicin exposure were then normalized to the baseline concentration measured in the first exposure, and tested for differences using Student’s t-tests for the effect of CFA on capsaicin-induced CGRP-release and one-way ANOVA followed by Tukey’s post hoc tests for effects of oxytocin on capsaicin-induced CGRP-release in CFA-pretreated rats, respectively. All analyses were conducted at the 0.05 level of statistical significance and used two-tailed tests.
Results
Oxytocin receptor expression on trigeminal ganglion neurons
Oxytocin receptors are widely present on a large subset of trigeminal ganglion neurons as demonstrated by epifluorescent microscopy (Figure 1(b,e,h). Pre-absorption of the oxytocin receptor antibody with antigens, provided by the manufacturer, completely inhibited fluorescent localization of labelling in cells (data not shown).
Oxytocin receptor co-localizes with calcitonin gene-related peptide (CGRP)-positive neurons in trigeminal ganglion of complete Freund’s adjuvant-treated rats. Immunofluorescence images stained for CGRP are shown in (a), (d) and (g) (red), oxytocin receptor staining is shown in (b), (e) and (h) (green) and co-localization is indicated by yellow in the merged images shown in (c), (f) and (i).
Oxytocin receptor co-expression with CGRP
Of the cells positive for CGRP (N = 174) within the trigeminal ganglion 80% (N = 140) also expressed oxytocin receptors (Figure 1(c,f,i). Pre-absorption of the CGRP antibody with antigens, provided by the manufacturer, completely inhibited fluorescent localization of labelling in cells (data not shown).
Overexpression with inflammation and electrocutaneous stimulation
Mean baseline expression of oxytocin receptor protein in untreated control trigeminal ganglia was 0.13 ± 0.02 pg/mg (mean ± SEM) of ganglion tissue (Figure 2). Inflammation of the TMJ using CFA injections into the TMJ increased oxytocin receptor levels significantly (p = 0.003, one-way ANOVA with Tukey’s post hoc test) to 2.38 ± 1.45 pg/mg (mean ± SEM) compared to controls. Similarly, controlled noxious stimulation of the face of anesthetized rats with 2 h of pulsed electrocutaneous stimulation significantly (p = 0.04, one-way ANOVA with Tukey’s post hoc test) increased oxytocin receptor concentrations compared to controls. This up-regulation was fairly consistent at 2, 4, 6, 8, and 24 h after stimulation (1.1 ± 0.2, 1.3 ± 0.1, 2.0 ± 0.3, 1.6 ± 0.4, and 1.7 ± 0.3 pg/mg; mean ± SEM), indicating that the expression change is rapid and persistent for a considerable time after cessation of the noxious stimulus.
Oxytocin receptor up-regulation in trigeminal ganglia of rats 24 h after inflammation induced by complete Freund’s adjuvant (CFA) or electrocutaneous stimulation (shock), respectively, compared to naïve controls. Measured oxytocin receptor values are expressed as pg/mg tissue. Data displayed as mean ± SEM. *Significant difference (p < 0.05) compared to control (one-way analysis of variance with Tukey’s post hoc test).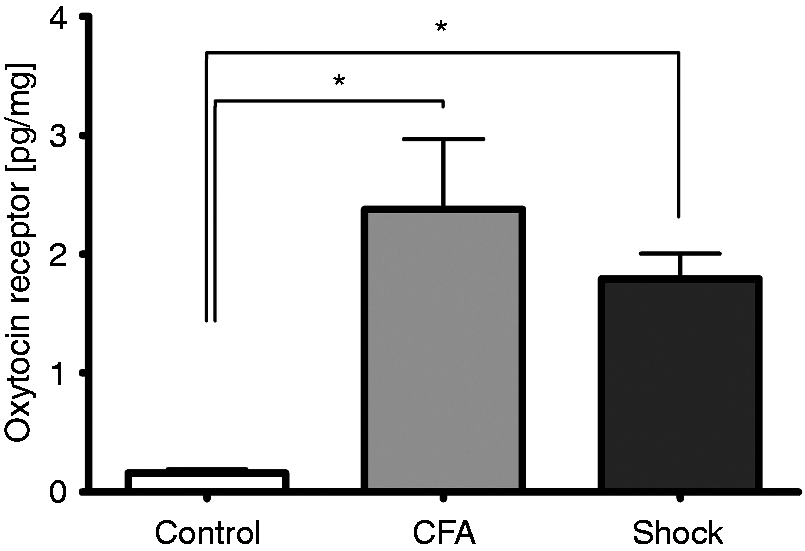
Effect of oxytocin on capsaicin-induced CGRP-release from dural nociceptors
CFA injection into the vibrissa of rats, reliably led to swelling and induration of the vibrissal pad indicating significant inflammation. Baseline concentrations of CGRP from the oxytocin treated dura samples were not different from untreated controls (data not shown). CGRP release from dura after CFA pre-treatment was significantly increased when exposed to capsaicin containing aCSF (11.0 ± 2.1 vs. 19.4 ± 3.8; mean ± SEM, p = 0.04, Student’s t-test, data not shown) compared to controls not injected with CFA. Adding 10 or 100 µM oxytocin to the aCSF, however, significantly reduced capsaicin-induced CGRP release (10.0 ± 1.6 and 9.5 ± 0.8; mean ± SEM, Figure 3) when compared to CFA-pre-treated control rats (19.4 ± 3.8; mean ± SEM, p = 0.026 and 0.023, respectively, one-way ANOVA with Tukey’s post hoc test, Figure 3). The 1 µM oxytocin dose did not significantly reduce CGRP release in CFA-pretreated rats (12.7 ± 3.4; p = 0.32, one-way ANOVA with Tukey’s post hoc test, Figure 3).
Effect of oxytocin on capsaicin-induced calcitonin gene-related peptide (CGRP)-release from dura of complete Freund’s adjuvant-pretreated rats. Relative increase of CGRP concentration after adding 10 µM capsaicin compared to baseline concentration. CTRL: control; OT1, OT10, and OT100: oxytocin 1 µM, 10 µM, and 100 µM, respectively. *Significant difference (p < 0.05) compared to control (one-way analysis of variance with Tukey’s post hoc test).
Discussion
Results of the current experiments demonstrate that oxytocin receptors are expressed in trigeminal ganglion neurons; most of which co-express CGRP, which is indicative of their presence on primarily nociceptive trigeminal neurons. Furthermore, oxytocin receptors are up-regulated on trigeminal neurons following inflammatory and/or noxious stimulation. Finally, activation of these receptors by oxytocin can block the release of CGRP, implicating a role in modulating trigeminal pain.
In addition to expression in the uterus, breast, and other non-neural tissue, oxytocin receptors have been localized in human and rodent brain, brainstem, and spinal cord. A particularly high concentration is found in the lower medulla, in which the trigeminal nuclear pain system is located (30,31). A chromatographic study of cadaverous tissue also indicated the presence of oxytocin in human sensory ganglia, including the trigeminal ganglia (11). Furthermore, the paraventricular nucleus of the hypothalamus sends oxytocinergic projection fibers to the spinal cord (32). These projections have been implicated in the modulation of pain signals, particularly after peripheral nerve injury (33). Thus, the localization of oxytocin and oxytocin receptors suggest an anatomical substrate for oxytocinergic modulation of incoming nociceptive neural traffic in the spinal cord. However, to our knowledge the presence of such a substrate was previously unexplored in the trigeminal system.
CGRP is critical in the relay and modulation of nociceptive signals from the periphery to the central nervous system (CNS) (34). Part of this effect is through an action on secondary neurons in the dorsal horn where, by both direct (35) and indirect (36) mechanisms, CGRP can induce sensitization. CGRP is also critically involved in the pathophysiology of migraine headache, and is likely also important in other craniofacial pain states (37). The significant role CGRP has in trigeminal pain disorders is supported by the efficacy of triptan drugs (e.g. sumatriptan) in treating migraine headache. Among other effects, these compounds attenuate the release of CGRP (38). The clinical efficacy of recently developed CGRP antagonists (39) and monoclonal antibodies specific for this peptide (40) in chronic and high frequency episodic migraineurs further validates the role of CGRP in craniofacial pain. Finally, scalp injections of botulinum toxin (BOTOX®), which is approved for the treatment for chronic migraine has been suggested to act, at least in part, through blockade of CGRP release from trigeminal afferent fibers (41).
Oxytocin produces analgesia when applied to the spinal cord, both in rodents (19,21,22,42,43) and man (44). It has not been clear, however, what the mechanism of this analgesic effect might be. The current results demonstrate that the level of oxytocin receptor expression in the trigeminal system is highly dynamic with craniofacial inflammation increasing the amount of receptor protein by more than ten-fold, 24 h after CFA injection or within 2 h of shock-induced inflammation. Three response elements on the oxytocin receptor gene promoter bind interleukin-6 and are likely responsible for such inflammation-driven upregulation, at least in the CNS (45). These results suggest that inflammation, as occurs with many craniofacial pain states, including temporomandibular disorders, tooth extraction, and, notably, migraine headache, should increase the responsivity of the trigeminal system to oxytocin.
The mechanisms underlying the analgesic effect of oxytocin in the spinal cord likely include presynaptic inhibition of nociceptive primary afferent neurons, possibly through the inhibition of Ca2+ influx into spinal terminals (17). Although the current results do not provide conclusive evidence, the presence of oxytocin receptors on CGRP immunoreactive trigeminal ganglion neurons, and the block of evoked CGRP release provides initial support that presynaptic inhibition does, at least, contribute to the analgesic effects observed. In contrast with our findings, Moreno-Lopez et al. (47) found that, in the spinal dorsal root ganglia, oxytocin receptors are primarily found in cell bodies of non-peptidergic C fibers and not in cutaneous nociceptive terminals, mechanoreceptors or peptidergic afferents, possibly indicating a distinction between the spinal and the trigeminal system. Additionally, a recent report demonstrated that oxytocin blocks acid-sensing ion channels in cultured dorsal root ganglia neurons, suggesting a direct effect on nociceptive transduction (48). In addition to these reports of presynaptic effect, patch clamp studies also support a post-synaptic effect of oxytocin. These studies demonstrated that oxytocin can enhance GABAergic and glycinergic inhibitory transmission on spinal substantia gelatinosa neurons (49). Thus, in addition to our demonstration of blockade of CGRP release from trigeminal afferents, there is evidence for numerous mechanisms through which oxytocin could exert an analgesic effect.
Although the pathophysiology of migraine is only partially understood, it is undisputed that the trigeminal ganglion (50) and meningeal afferents (51) play a key role. However, there are only limited data suggesting an inhibitory effect of oxytocin on migraine. Hoshiyama et al. (23) found significantly lower migraine recurrence rates when comparing breastfeeding (i.e. presumably higher oxytocin levels) to bottle-feeding mothers for at least 6 months post-partum. These findings were corroborated by another study by Sances et al. (24), in which 1 month after delivery of their baby, 100% of bottle-feeding mothers had recurrence of migraine compared to 43% of mothers who were breastfeeding. Similarly, Serva et al. (25) found that breastfeeding was protective for migraine recurrence. In addition, there is a single case series of two patients suffering from an acute migraine headache who had experienced relief after intravenous infusion of oxytocin (52).
The results presented here provide presumptive evidence that oxytocin could act at trigeminal receptors, in a state of upregulation due to inflammation, to block the release of CGRP from nociceptive primary afferent neurons. This effect may occur in concert with other pre- and post-synaptic mechanisms, thereby reducing the intensity and frequency of migraine headaches and potentially of other trigeminal system associated pain conditions.
Key findings
Oxytocin receptors were present on the vast majority of calcitonin-gene related-peptide (CGRP)-positive trigeminal ganglia neurons Oxytocin receptors were upregulated in the trigeminal ganglion after noxious stimulation of the face Exogenous oxytocin blocked capsaicin-induced CGRP release from trigeminal dural afferents
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Daniel Jacobs, Martin S Angst and David C Yeomans are founders and shareholders of Trigemina, Inc. Jordan Mechanic, James Miller, Dean Carson and Conrado Pascual are employees and shareholders of Trigemina, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
