Abstract
Background
The Clinical Trials Subcommittee of the International Headache Society (IHS) recommends that a placebo arm is included in comparative randomised clinical trials (RCTs) of multiple prophylactic drugs due to the highly variable placebo response in migraine prophylaxis studies. The use of placebo control in such trials has not been systematically assessed.
Methods
We performed a systematic review of all comparative RCTs of prophylactic drug treatment of migraine published in English from 2002 to 2014. PubMed was searched using the Cochrane Highly Sensitive Search Strategy for identifying reports of RCTs.
Results
A placebo arm was used in <10% (three of 31) of prophylactic RCTs in migraine. In only 7.1% (two of 28) of the comparative RCTs without placebo was one drug superior to another drug. Thus in 26 RCTs, including one study requiring more than 75,000 patient days, no difference was identified across treatment arms and conclusions regarding drug superiority could not be drawn.
Conclusions
The majority of comparative, prophylactic migraine RCTs do not include a placebo arm. Failure to include a placebo arm may result in failure to demonstrate efficacy of potentially effective migraine-prophylactic agents. In order to benefit current and future patients, the current strong tendency to omit placebo-controls in these RCTs should be replaced by adherence to the guidelines of the IHS.
Introduction
Migraine is a chronic disorder with recurrent headache attacks. The global prevalence of migraine is estimated to be 15% (1,2). Many patients can be treated successfully with current acute, specific treatment, the triptans (5-HT1B/1D receptor agonists) (3). In a very large United States (US) epidemiological study (
Current prophylactic drugs used in migraine, nonsteroidal anti-inflammatory drugs (NSAIDs) (6), antiepileptic drugs (7), β-adrenoceptor blockers (8), antiserotonin drugs (9), and calcium antagonists (10), were with the exceptions of pizotifen and methysergide (10), developed for other indications and then used, often by serendipity, and found somewhat effective in migraine. Subsequently, efficacy was confirmed in randomised, controlled trials (RCTs). Unfortunately, the efficacy of these drugs is relatively low while side effects are frequent. Migraine continues to represent a very large burden of disease worldwide (1,2), and there is a large and unmet need for new prophylactic drugs (11). Evidently, new drugs should be shown to be similar or superior to current drugs in terms of efficacy and tolerability.
Due to highly variable placebo responses in prophylactic migraine RCTs, ranging from 0% to 56% responder rates (12), the guidelines for controlled trials of drugs in migraine of the International Headache Society (IHS) (13) recommend that a placebo arm is included in comparative RCTs of multiple prophylactic drugs in order to determine if an apparent effect of one or more drugs is simply placebo related.
The aim of the present study was to systematically review recent comparative, prophylactic RCTs in migraine in order to assess the adherence to the IHS recommendation of placebo control.
Methods
The study was based on data from a previous review of migraine RCTs from 2002 to 2008 (14), which was further expanded to include studies published until 2014.
We searched PubMed for clinical trials of drug treatment for migraine published from 2002 to 2014.
The search was performed using the Cochrane Highly Sensitive Search Strategy for PubMed (as revised in 2008) (15) and with publication date limits ranging from 1 January 2002 until 31 December 2014, the search string thus being: ‘((‘2002/01/01’[Date - Publication]: ‘2014/12/31’[Date - Publication])) AND ((migraine AND (randomised controlled trial OR randomised OR placebo OR drug therapy OR randomly OR trial OR groups) NOT animals))”. The data search was carried out on 23 September 2015. Search, study selection and data extraction were performed by a single reviewer (AH).
Comparative RCTs (i.e. comparing the efficacy of two or more different drugs) of prophylactic treatment were specifically selected for the present review.
The following were excluded from the review: Studies on acute treatment of migraine, studies of combined pharmacological and non-pharmacological treatment, studies not reported in English, studies not concerning migraine exclusively, studies with non-clinical outcome measures only, studies of induced migraine, and studies of patients under the age of 18 years.
Results
The search retrieved a total of 6316 items. From these 31 comparative, prophylactic trials of acute treatment were excluded from further analysis in this review, while 26 prophylactic trials were included (16–46). Placebo was used in only three of the prophylactic RCTs (9.7%).
In one of these two RCTs the two active drugs, topiramate 100 mg and propranolol 160 mg, were both superior to placebo and not different in efficacy (24). In the other RCT with low doses one active drug, topiramate 50 mg, was superior to the other drug, lamotrigine 50 mg, and placebo, whereas lamotrigine was not superior to placebo (28). In the third study, candesartan 16 mg and propranolol 160 mg were both superior to placebo but not different in efficacy (42).
Overview of the included studies.

D: design; P: parallel group; C: cross-over; Dur: duration (weeks); Pla: placebo; INR: international normalised ratio.
Effect on migraine attacks of flunarizine 5 and 10 mg vs. propranolol for 16 weeks.
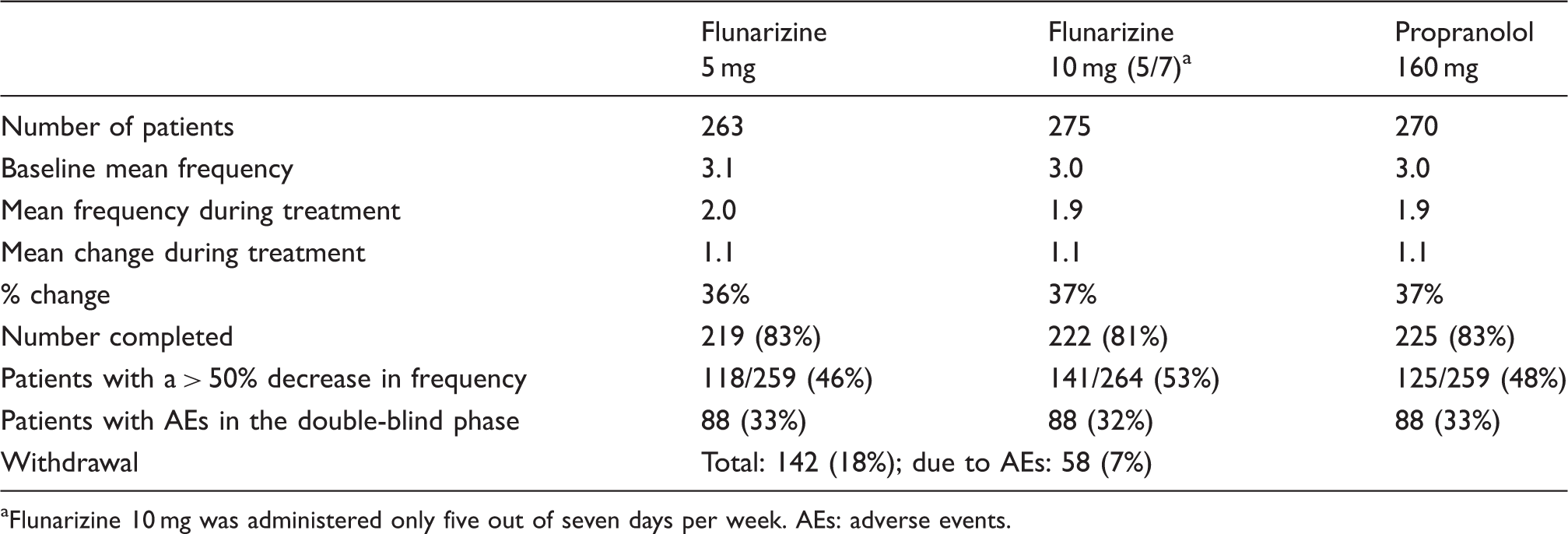
Flunarizine 10 mg was administered only five out of seven days per week. AEs: adverse events.
Discussion
As is clear from this review of comparative, prophylactic RCTs in migraine from 2002 to 2014 most investigators and sponsors, and probably also ethical committees, prefer a trial design with a new drug and a standard drug but without placebo.
World Medical Association Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects (

Citation from the International Headache Society’s guidelines for drug trials in migraine, Third Edition from 2012.

In addition, the choice of doses of the ‘standard drug’, in this case propranolol, varies considerably. Thus in four RCTs in the current review, propranolol doses were: 80 mg (17), 160 mg (23), 40 mg (26) and 80–160 mg (40). In the Cochrane Review propranolol doses of 60 mg to 320 mg were used in RCTs; in 49 of 55 RCTs doses of 120 mg or greater were used and in six RCTS only doses of 60–80 mg were used (48). Only one (49) of these six RCTs was placebo controlled. It was a cross-over study with response in 15/19 with propranolol vs. two of 19 with placebo. The Cochrane reviewer commented on this RCT: ‘small crossover trial with extremely positive results’ (48). This single, small RCT with propranolol 80 is not sufficient to establish small doses of propranolol as standard comparative drugs in prophylactic migraine RCTs. Thus if a non-established dose of an ‘established efficacious drug’ is used in comparative, prophylactic RCTs, it is not only unknown if that dose is effective in the trial population, but also if the dose has any migraine-prophylactic effect at all and therefore, again, placebo control is needed.
The largest prophylactic migraine RCT ever performed (23) is summarised in Table 1. The results exemplify the typical problems with performing even a very large comparative RCT without the inclusion of placebo. The stated aim of this trial ‘was to investigate whether the treatment of migraine could be improved by enhancement of the tolerability of flunarizine without compromising its efficacy as already established in comparison with propranolol’ (23). The basis for the stated presumed comparability between the two drugs was four RCTs (23,50–53) in which propranolol 120–160 mg was found comparable to flunarizine 10 mg for migraine prophylaxis. It is noteworthy that in none of the four RCTs was placebo included. As shown in Table 1, the mean changes (1.1, 1.1, and 1.1) in migraine frequency during treatments (the primary efficacy parameter) were highly similar in the three treatment groups. Also the proportion of patients with adverse events (AEs) during the double-blind phase was remarkably similar (33%, 32%, and 33%). These results do not allow for any firm conclusion. Since it was not demonstrated in the statistical analyses that the three ‘active treatments’ were superior to placebo, the question of any efficacy demonstrated in this patient sample remains open. What was observed in the three treatment groups could merely be due to a placebo effect combined with the ‘natural course of the disease’ (12,54,55). Notably, in the Cochrane Review on propranolol (48) flunarizine and propranolol are described as being equipotent in migraine prophylaxis based on seven comparative RCTs without placebo control. Thus even these Cochrane reviewers seem unaware of the need for placebo control in comparative, prophylactic RCTs. Based on the 666 patients completing the study (see Table 1), we estimate that 75,000 patient days were, in our view, spent ‘in vain’ in this the largest ever prophylactic, migraine RCT (23) without placebo.
Generally, when comparing two drugs without placebo control, there are three possible outcomes: 1) ‘new drug’ is superior to ‘standard drug’, 2) ‘standard drug’ is superior to ‘new drug’, or 3) no significant difference between the drugs. Cases 1) and 2) are less problematic but, due to the highly variable treatment responses in migraine, there is a real risk that none of the drugs would have shown superiority to placebo in that particular trial. In case 3), efficacy of any of the drugs cannot be demonstrated without demonstrating the ‘reactivity of the sample” by including a placebo control. A similar decrease in frequency of migraine in the two treatment groups could, again, simply be due to ‘the natural course of the disease’ (54,55). Inability to demonstrate a statistically significant between-group difference could easily be due to high variability of the response within each group. Inclusion of a placebo arm could reveal superiority to placebo of one drug, but not the other. Therefore it cannot be concluded from a lack of significant difference between drugs that the drugs are ‘equally effective’. Also, when the aim is to show comparability, tolerability for the new drug compared to placebo is important for demonstrating whether the incidence of AEs with the new drug is higher or similar to placebo. In prophylactic RCTs of a single drug, placebo control is, for the same reasons, also crucial in order to determine if the drug has any effect apart from that of placebo. Finally, without the inclusion of placebo, in either case, the data cannot be used in meta-analyses and thus do not add to the scientific evidence on prophylactic treatment of migraine.
The WMA further recommends that any patients who do not receive the best proven treatment should not be subject to additional risks of serious or irreversible harms as a result of not receiving the best proven treatment (Table 3). The risk of such harms during placebo treatment in prophylactic RCTs in migraine is probably negligible since most patients, even up to six months, have a decrease in migraine frequency during placebo treatment (12), most likely partly due to ‘the natural course of the disease’, mentioned above (12,54,55).
In summary, the recommendations against placebo by the WMA, though likely relevant to studies of most disorders, do not apply to migraine prophylactic RCTs for two main reasons: a) migraine prophylactic treatment is characterised by an extremely variable placebo response, and b) placebo treatment in migraine does not cause serious or irreversible harm to patients.
On the contrary, in our view it is not ethical to include patients in RCTs that likely do not benefit actual or future patients, as is the case in 28 of 31 reviewed RCTs in the present study. The total number of days spent, in our opinion, ‘in vain’ by migraine patients in these 28 RCTs was approximately 360,000 and this high number could successfully be used when arguing with, for example, ethical committees for the use of placebo in comparative, prophylactic RCTs in migraine. The argument about the many patient days spent by migraine patients in badly designed RCTs is in our opinion a strong one, and it should be added to the more general arguments for placebo control in prophylactic migraine RCTs presented by the IHS (Table 4).
Clinical implications
In prophylactic migraine drug trials, the true drug effect cannot be established unless one can control for the placebo response. Twenty-eight of 31 comparative prophylactic trials did not include placebo, rendering these studies largely inconclusive. The total number of days spent by migraine patients in these 28 trials was approximately 360,000. Investigators should include placebo in prophylactic migraine trials as recommended by the International Headache Society’s guidelines for drug trials in migraine.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
