Abstract
Objectives
The objectives of this article are to test the feasibility of lumbar puncture (LP) using 25-gauge (G) needles in daily neurological practice and to compare the risk of post-dural puncture headache (PDPH) with four types of needles.
Methods
In a prospective rater-blind study, pros and cons of four different LP needles, the 20G Quincke (20Q), 22G Sprotte (22S), 25G Whitacre (25W) and 25G Sprotte (25S), were evaluated in 394 LPs performed by seven neurologists. The neurologist performing the LP recorded the type and size of needle, intensity of pain, safety, time of the procedure and failure or success. Between five and 15 days later another neurologist, blind to the type of needle used, completed an ad-hoc questionnaire for PDPH.
Results
PDPH developed in 35.9% patients when using a 20Q needle, and in 12.9%, 6.8% and 1.6%, respectively, when using a 22S, 25W or 25S needle. The difference in incidence of PDPH following LP performed with the 20Q needle and the 25S or 22S was statistically significant (p < 0.001 and p = 0.008, respectively) and it approached significance when comparing the 25S and 25W (p = 0.06). As 25W and 25S needles need CSF aspiration, LP requires more time and skill. Pain caused by LP was similar with the four needles.
Conclusion
The use of the 25S needle in diagnostic LP reduces the frequency and severity of PDPH.
Keywords
Introduction
Post-dural puncture headache (PDPH), formerly called post-lumbar puncture headache (1), is a very common complication of lumbar puncture (LP) and is caused by a persistent leak of cerebrospinal fluid (CSF) from the dural puncture site. LP is performed for diagnostic purposes by neurologists and for the injection of anesthetics by anesthesiologists. Different types of needles can be used. They differ in diameter, measured in gauge (G), in the shape of the tip and in the necessity or not of an introducer. The “traumatic” Quincke (Q) needle has a sharp bevel end that cuts the dural fibers whereas the “atraumatic” Whitacre (W) and Sprotte (S) needles have a pencil or an ogival tip, respectively, that spreads out the fibers, causing minor trauma (2). As atraumatic needles cannot cut the skin, muscles and ligamentum flavum, they require an introducer. With 24G or smaller diameter needles, aspiration of CSF is needed because of the very slow flow, especially if the patient is lying on his or her side.
The American Academy of Neurology (AAN) discussed PDPH twice and released ad-hoc guidelines on the prevention of PDPH (PLPHA) in an attempt to modify the traditional way neurologists perform diagnostic LPs. The AAN guidelines in 2000 (3) and its addendum in 2005 (4) recommend the use of a 22G atraumatic needle instead of the traditional 20G Quincke in diagnostic LPs to reduce PDPH. The AAN also highlighted that “smaller needle size is associated with reduced frequency of PLPHA.” Despite the AAN recommendations, the use of the 22G atraumatic needle has not become standard practice in the neurologic community (5,6). Even though atraumatic 22G needles cause less PDPH than cutting needles, PDPH remains a very common complication of LP, ranging from 24.4% to 12.2% (7–9).
The risk of PDPH is substantially reduced by the use of smaller, 24G–26G, non-cutting needles, a long-standing standard procedure among anesthesiologists (10). Although anesthesiologists must inject anesthetics and neurologists must aspirate CSF, the same small type of needles could be used as suggested by three neurological studies using 24G–26G needles: a randomized, double-blind, clinical trial in normal individuals (11) and two studies in Alzheimer patients and controls (12,13). The percentage of PDPH in those studies was 12%, 4% and 0.93%, respectively.
Neurologists are reluctant to change the traditional way of performing LP and to adopt atraumatic needles with an introducer in routine clinical practice. The most common reasons given are: higher cost, greater difficulty, slow flow of CSF requiring aspiration with negative pressure, longer time of the procedure and concerns about patients’ safety (14).
The main goal of the present study was to verify if changing the traditional way neurologists perform LPs can avoid unnecessary morbidity for the patients undergoing LPs. For the first time 25G needles, requiring an introducer and CSF aspiration, were used for LPs during routine neurological clinical practice. This study tested four needles different in diameter and shape of the tip in a rater-blind prospective study in 394 LPs performed by seven neurologists.
Patient population and study design
The study was approved by the ethics committee of San Luigi Hospital, Orbassano, Italy, and all patients signed an informed written consent.
A total of 394 LPs were performed in 376 patients during routine clinical practice by seven neurologists at Centro Riferimento Regionale Sclerosi Multipla (CRESM) in Piedmont. According to the usual clinical practice, before undergoing LP the patients were informed about the risks associated with LP, including PDPH, and about how LP is performed. The patients were blind to the type of needle used, their characteristics and the need for an introducer and for CSF aspiration with 25G needles. Before performing the LP, the patients were asked if and how much pain they expected to feel.
LPs were performed in the morning with the patient sitting or lying on his or her side and the stylet was reinserted before needle withdrawal. Patients were asked to remain recumbent for two hours following LP.
Exclusion criteria were platelet count <80 × 109/l, suspected increased intracranial pressure, and a previous LP in the previous week.
In 282 patients (75.1%) the suspected diagnosis prompting LP was either multiple sclerosis (MS) or clinically isolated syndrome (CIS). The other 94 patients belonged to one of the following subsets (15): non-inflammatory neurologic disease (n = 47; 13.3%), inflammatory neurologic disease (n = 25; 7.2%); peripheral inflammatory neurologic disease (n = 11; 3.2%); and symptomatic “controls” (n = 4; 1.2%).
The neurologist performing LPs filled out the structured data collection form, which included the following information: suspected diagnosis, patient’s position, vertebral space, number of attempts and failures, time spent for LP and CSF collection, ml of CSF drawn, and expected and experienced pain assessed by the Eleven-Point Box Scale (BS11) (16). A neurologist, blind to the type of needle, interviewed the patients for PDPH five to 15 days after the LP. PDPH was defined according to the International Headache Society (http://ihs-classification.org) (1). Its severity was graded according to Lybecker and colleagues (17) as none, mild, moderate and severe PDPH. In detail, mild PDPH is a postural headache that slightly restricts daily activity; the patient is not bedridden at any time during the day and there are no associated vestibular, cochlear, ocular, or musculoskeletal symptoms. The moderate PDPH is a postural headache that significantly restricts daily activities. The patient is bedridden part of the day; associated symptoms may or may not be present. The severe PDPH is a postural headache that results in patient’s desire to stay in bed all day. Associated symptoms are always present. In the questionnaire, PDPH severity was graded as none (value = 0), mild (value 1–2), moderate (value 3–4) or severe (value 5–6). PDPH duration and the medications used were also recorded. The number of red blood cells (RBCs) in the CSF was counted in the first of the tubes.
The study included two phases: In the first phase a 25W needle (Artsana, Grandate, Italy) was introduced into the clinical practice for comparison with the traditional 20Q needle and the two needles were used alternatively; after an interim analysis showing the definitely lower frequency of PDPH with the 25W, only this latter was used. In the second phase Sprotte needles, both 25S and 22S (Pajunk, Geisingen, Germany) in the ratio 2:1 were used. In case of failure the 20Q needle was used. A minority of LPs, 11 cases, were performed with the 22Q because of the unavailability of 25G needles and it was not considered for comparison with the other groups. All the LPs were performed by seven neurologists in the same in-patient unit, the Centro Riferimento Regionale Sclerosi Multipla at San Luigi University Hospital, Orbassano (Italy). The type of needle was established a priori by one of the neurologists (AB), to avoid a choice based on the patients’ risk profile for PDPH or on the neurologist’s experience in performing LPs. The stylet was reinserted after CSF collection (18) and the Quincke needles were placed with the bevel oriented in a parallel/longitudinal direction in order to decrease the incidence of PDPH (19).
Statistics
Continuous data are expressed as medians, with an interquartile range as a measure of variability. Chi-square and Fisher’s test were used for qualitative analysis; the Mann-Whitney test was used for quantitative analysis with Hochberg adjustment for multiple comparisons. Risk factors for development of PDPH were evaluated by a logistic regression model and a univariate estimate of the odds ratios (ORs) was presented along with their 95% confidence interval (95% CI). The level of significance was considered as p < 0.05. Analyses were carried out using R version 3.02.
Results
A total of 394 LPs were included in the study: 376 LPs were performed successfully using one of the five types of needles as follows: 39 with the 20Q, 11 with the 22Q, 62 with the 22S, 133 with the 25W, and 131 with the 25S needle. In 18 LPs two types of needles were used because of failure with the first type. In 18.2% of the patients tapped with 22Q PDPH occurred, but the subset included only 11 patients and it was not considered for further analysis.
Patients and lumbar puncture characteristics by needle type.
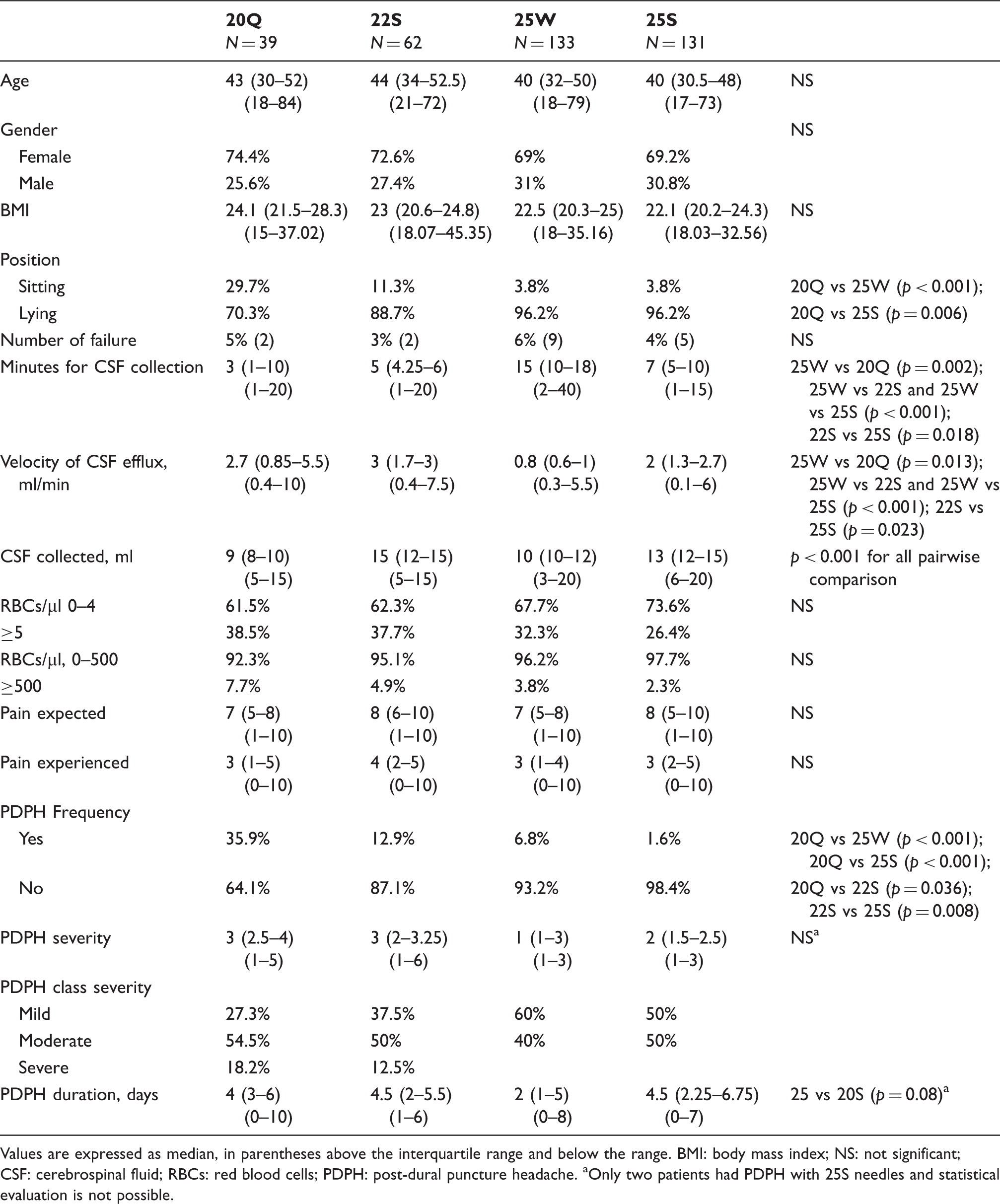
Values are expressed as median, in parentheses above the interquartile range and below the range. BMI: body mass index; NS: not significant; CSF: cerebrospinal fluid; RBCs: red blood cells; PDPH: post-dural puncture headache. aOnly two patients had PDPH with 25S needles and statistical evaluation is not possible.
Women accounted for the majority of patients ranging from 74.4% in the 20Q group to 69.2% in the 25S group without any statistical significance (Table 1).
The body mass index (BMI) spanned from 15 to 45, without statistical difference among the four groups (Table 1).
In the great majority of LPs (70.3% to 96.2%) the patient was lying down; the highest number of patients sitting (29.7%) was observed with the 20Q.
Failure
We considered a failure as occurring when despite several attempts the neurologist was not able to perform the LP with the selected type of needle and he had to use a bigger needle. The 18 failures were so distributed in the groups: two out of 41 (5%) with the 20Q, two out of 64 (3%) with the 22S, nine out of 142 (6%) with the 25W, and five out of 136 (4%) with the 25S. In the 16 failures that occurred with small needles, LP carried out with a bigger needle was successful. No statistical difference was observed among the four groups; however, a learning curve seems to be present as the percentage of failure was 3.5% (seven out of 200 LPs) in the second part of the study (22S and 25S needles), vs 6.0% (11 out of 183 LPs) in the first part of the study (20Q and 25W needles). PDPH was reported in two out of 16 failures. These cases were not taken into account for the evaluation of PDPH prevalence as two different types of needles were used.
Time spent for needle insertion and CSF collection
Unlike cutting needles 20Q or 22Q, small needles 25W and 25S and the atraumatic 22S require the introducer, which takes one to two minutes to be inserted. The time spent for the collection of CSF ranged from one minute to 40 minutes depending on the amount of CSF collected, the position of the patient, the diameter of the needle, the possible aspiration of CSF and the neurologist’s skill. The median time spent with the 20Q was three minutes, shorter than with the 22S (five minutes), 25W (15 minutes) and 25S (seven minutes) (Table 1). The longer time for LP with the 25W and 25S needles is due to the need for CSF aspiration, performed with a 5 ml syringe connected to the needle when this has reached the subarachnoid space. The maneuver allows the collection of 3–5 ml of CSF at a time and it must be repeated to obtain the volume needed for diagnosis and bio-banking. The volume of CSF collected ranged from 3 to 20 ml, with median values 9, 15, 10 and 13 for the 20Q, 22S, 25W and 25S, respectively. The amount of CSF was larger with the Sprotte needles, both 22S and 25S (Table 1).
Traumatic LP
Traumatic LP, that is the presence of RBCs in the CSF, was evaluated using the Nageotte chamber and counting the number of RBCs in the first tube of CSF. The percentage of CSF with a number of RBCs lower than 5/µl ranged from 61.5% to 73.6%, being higher with the small needles, without statistical differences (Table 1). As RBCs lower than 500/µl is the threshold used for proteomic studies (20), the percentage of LPs suitable for those studies ranged from 92.3% to 97.7%, higher in the 25G-needle subgroup but without statistical significance.
Pain expected and experienced during LP
The median expected pain was similar in the four groups, ranging from 7 to 8 in the BS11, and the median experienced pain was definitively lower, ranging from 3 to 4 (p < 0.0001 for all needles). The experienced pain was similar with the four needles, unaffected by the introducer and by CSF aspiration.
PDPH: incidence, severity, days of recovery
PDPH was observed in a total of 33 patients out of 376 (8.8%) so distributed: 14 out of 39 (35.9%) with the 20G, eight out of 62 (12.9%) with the 22S, nine out of 133 (6.8%) with the 25W and two out of 131 (1.6%) with the 25S. The risk of PDPH with 20Q was statistically higher than with all the other needles tested (Table 1); the incidence of PDPH was significantly lower when using the 25S than the 20Q and the 22S (p < 0.001 and p = 0.008, respectively) and lower but without statistical significance with the 25W (p = 0.06). The analysis of the 33 cases of PDPH in the present study showed that the advantage of small needles is also clearly evident when we consider the duration of PDPH and the severity of the pain experienced by the patients. The median intensity of PDPH was lower with both 25G needles, as none of the 11 cases of PDPH was considered severe, whereas 12.5% of 22S-linked PDPH and 18.2% of 20Q-linked PDPH were severe. The median duration of PDPH was longer with the 20Q and 22S than with the 25W, being four, 4.5 and two days, respectively; in the two cases tapped with the 25S PDPH lasted less than 24 hours in one case and one week in the second. Re-admission to the hospital and brain computed tomography (CT) scan were necessary in one patient with severe headache after an LP performed with the 20G needle.
Risk factors for development of post-dural puncture headache.
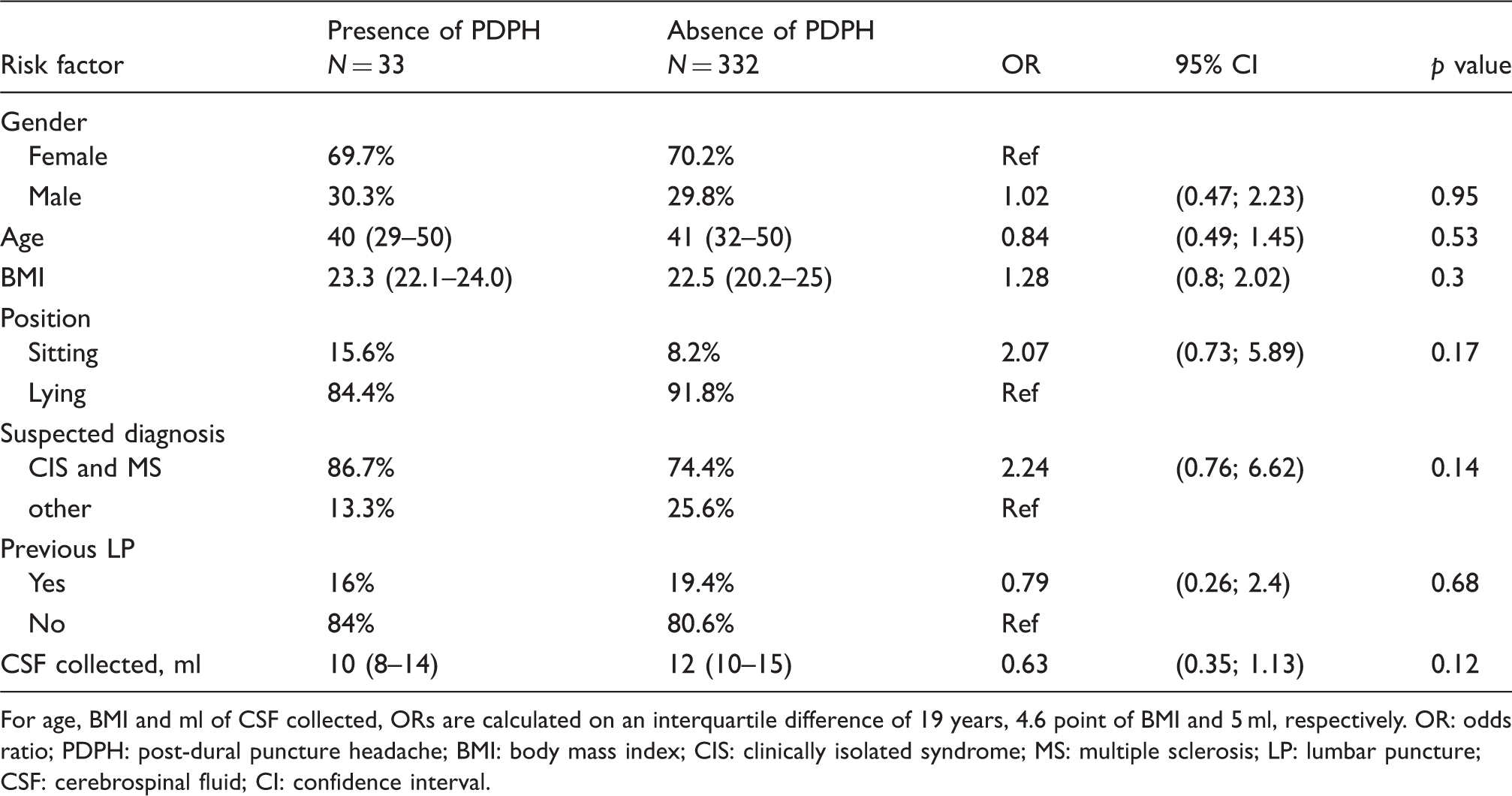
For age, BMI and ml of CSF collected, ORs are calculated on an interquartile difference of 19 years, 4.6 point of BMI and 5 ml, respectively. OR: odds ratio; PDPH: post-dural puncture headache; BMI: body mass index; CIS: clinically isolated syndrome; MS: multiple sclerosis; LP: lumbar puncture; CSF: cerebrospinal fluid; CI: confidence interval.
Discussion
The goals of the present study were to explore the safety and feasibility of needles smaller than 22G, namely 25G, during diagnostic LPs in routine neurological practice and to compare the risk of PDPH of four different types of needles. In the study the use of the atraumatic 25S needle with CSF aspiration reduces the risk of PDPH up to 10-fold. In fact the 22S needle caused PDPH in 12.9% patients, vs only 1.9% of patients with 25S. Once more this study confirms that the diameter of the needle is crucial for the appearance of PDPH: Both 25G atraumatic needles, namely the 25W with the pencil point-shaped tip and the 25S with an ogival tip, were associated with lower occurrence of PDPH than the 20Q and 22S (Table 1).
The comparison between the two 25G needles used in our study indicated that the 25S is less frequently associated with PDPH than the 25W (6.8% vs 1.6%; p = 0.06) and it suggests also that the shape of the tip can play a role in the occurrence of PDPH. This is consistent with two other studies comparing needles with the same diameter but different tip shape and showing that the atraumatic 22S needle was associated with lower PDPH than the 22Q (12.2% vs 24.4%; 19% vs 32%, respectively) (8,9). To the best of our knowledge, the lowest reported percentages of PDPH in diagnostic LPs are 0.93% (13) and 1.6% in the present study, both obtained with small, 24 or 25G, Sprotte ogival-tip needles.
The cases of PDPH in the present study showed that small needles were associated with shorter duration and lower intensity than larger needles as none of the 264 cases of PDPH after LPs performed with small needles was classified as severe, and none required re-admission to the hospital for severe and persistent headache. Many factors have been suggested to cause PDPH (6). In the present study we avoided two of them, by reinserting the stylet (18) and by positioning the Quincke needle parallel to the long axis of the spinal cord (19). Volume of CSF collected, sitting or lying position, previous LP, BMI, age and gender were not associated with a higher risk of PDPH, in agreement with several studies (6,21,22). It must be noted that the present study has a limited capacity to detect risk factors as it included only 33 cases with PDPH and the 95% CIs are wide (Table 2).
This study indicates that 25G needles require more time than traditional ones as they need an introducer to pierce the skin, muscles and ligamentum flavum and they require CSF aspiration. In addition, the neurologist must have greater skill, which can be acquired and improved through experience. In the first part of our study, which compared the 20Q with the 25W, the median time spent with the 25W was 15 minutes, whereas in the second part of the study that compared the 22S and the 25S, the median time with the 25S was seven minutes (25W vs 25S p < 0.001), because the seven neurologists performing LPs became more confident with small needles. The higher skill needed to perform LPs with the 25G is also highlighted by the 14 LPs successfully performed with the 20Q after failure with the 25G.The longer time spent with the 25G is easily counterbalanced by less time spent in the management of less frequent and severe PDPH (5).
Safety is one of the neurologists’ concerns in using 25G needles because aspiration of CSF is required. Aspiration of CSF through a syringe during LP seems to be a dangerous procedure that increases the risk of brain herniation; however, the data collected in this study and in previous ones (12,13) do not support this fear: No patient had discomfort and none required re-admission to the hospital for persisting headache. Brain herniation depends on high intracranial pressure, mainly due to brain tumors, and on the speed of CSF efflux. A CT scan before LP excluded the presence of brain tumor in this study. The speed of CSF efflux with the 25S, evaluated in ml/min, was similar to that with the 20Q and less than with the 22S (Table 1). The neurologist can control the speed of CSF efflux through small needles by changing the degree of aspiration. This is not possible with bigger needles and the efflux depends only on the intracranial pressure.
The risk of damage to nerve root filaments caused by aspiration through the tip of the needle was also ruled out, as the pain referred by patients was similar with all the needles (Table 1). Pain during LP was evaluated by asking the patient to indicate the expected pain before the procedure and the actual experienced pain. The data demonstrated the inconsistency of the popular belief that LP causes “unbearable” pain: The median expected pain was more than 7, in a scale ranging from 0 to 10, and the experienced median pain was between 3 and 4; with all the needles the experienced pain was statistically lower than the expected pain (p < 0.0001). The belief of the neurologists that the use of an introducer increases the patient’s pain is not confirmed by our data, but it must be noted that the diameter of the introducer for the 25G needle is 21G, smaller than the traditional needles.
The present study was not set up to evaluate the cost of the procedure, but other studies have already shown that the use of a needle reducing the risk of PDPH is cost effective (23,24). In our center 25G needles cost 4€ more than 20G needles and they require more time from nurses and neurologists performing the LP. However, these higher costs are easily rewarded by the lower occurrence of PDPH, which is expensive and time consuming. In fact the severity and duration of PDPH was lower with the 25G than with the 22S and 20Q; one out of 39 20G patients with PDPH had to undergo a brain CT scan during re-admission to the hospital at a cost of 500€ a day, while none of the 264 25G patients was readmitted. The rate of re-admission in our study could have been higher if blood patches had been performed in patients with PDPH classified as moderate or severe and lasting two or more days (25). Although the procedure has some adverse effects (26), blood patches have been shown to reduce severity and duration of PDPH (1,25), and we will take them into consideration in future studies.
Although the volume of CSF collected for diagnosis is generally lower than 5 ml, a volume of at least 12 ml is recommended for diagnostic and prognostic tests and for bio-banking (20). Our data show that it is possible to collect up to 20 ml of CSF with every type of needle without increasing the risk of PDPH and the discomfort for the patient.
To test if the introducer increases the percentage of “traumatic” LPs contaminated by blood, the number of RBCs in the first tube of CSF was counted: The data showed that the percentage of LPs containing more than 5 or more than 500 RBC/µl was similar in the four groups of patients and not influenced by the introducer (Table 1). This finding is not surprising as the introducer does not enter the subarachnoid space and the smaller the needle the lower the chance to injure capillaries or venules and to cause leaking of RBCs into CSF.
In the present study the opening pressure was not measured because it was not necessary for the diagnosis. Although the opening pressure can be measured with all the types of needles used (11,27), the 25G needle takes several minutes more than the 20G or 22G needles to reach equilibrium, making small needles not practical when the measurement of opening pressure is important.
We hope our work can convince neurologists that PDPH is, in a great number of cases, an avoidable side effect. Neurologists should implement the use of the 25S needle in everyday clinical practice by trying a small needle first, and in case of failure, the traditional larger needles. As anesthesiologists currently use small needles with an introducer, a short training in an anesthesiology department could help.
The strengths of our prospective study are that we tested a large number of different types of needles in diagnostic LPs and that we proved that it is possible to use a different technique from the one traditionally used by neurologists in clinical practice. The main limitations of the study are that the majority of the patients were young adults affected by MS and that the LPs were carried out in one center only. A study involving patients affected by a wide variety of neurological diseases in a number of centers will establish if the 25S needle is safe and practical in the various clinical settings in which neurologists perform LPs.
In conclusion, this study comparing four types of needles confirmed that PDPH is influenced by the diameter and shape of the needle tip and indicated that the 25S needle is associated with very low frequency of PDPH. Diagnostic LP with the 25S needle is safe and can be performed in routine clinical practice but it requires higher skill than traditional LP. Neurologists can avoid nine out of 10 PDPHs if they change their traditional way of performing LPs.
Clinical implications
We tested the feasibility of lumbar puncture using 25-gauge (G) needles in everyday clinical practice and quantified the risk of post-dural puncture headache (PDPH) of four different types of needles in 394 lumbar punctures performed by seven neurologists. We confirmed that PDPH is influenced by the diameter and shape of the needle tip and demonstrated that the 25 Sprotte needle is associated with very low frequency of PDPH (1.9%). 25 Sprotte needle reduces the risk of PDPH almost 10 times in comparison with 22 Sprotte needle. We hope our work can convince neurologists that PDPH is, in a great number of cases, an avoidable side effect. Neurologists should implement the use of 25S needle in everyday clinical practice by trying a small needle first, and in case of failure, the traditional larger needles.
Footnotes
Funding
This work was supported by Piedmont Health Service (n. 38097/DA2001, 2008) and by Associazione Ricerca Biomedica, Onlus.
Conflicts of interest
None declared.
