Abstract
Objectives
The pathogenesis of idiopathic intracranial hypertension (IIH) is currently unknown and there are speculations about the contribution of some immunologic factors. The aim of this study is to investigate the presence of oligoclonal bands (OCBs) and cerebrospinal fluid (CSF) and/or serum cytokine levels in patients with IIH.
Methods
Patients fulfilling revised diagnostic criteria for IIH were included. Their demographic, clinical, ophthalmologic and laboratory features were examined. Serum and CSF samples were detected by isoelectric focusing and immunoblotting for OCBs. The samples of IIH patients and control groups were investigated by ELISA for cytokine levels.
Results
We detected OCBs in eight (30.77%) patients diagnosed with IIH. There were no other obvious clinical and laboratory differences of IIH profiles between the patients with and without OCBs, but frequency of vision loss was significantly higher in the group with OCBs in comparison to OCB negatives (p = 0.038). Patients with IIH had highly elevated TNF-α, IFN-γ, IL-4, IL-10, IL-12, IL-17 in their sera compared to patients with multiple sclerosis (MS) and healthy controls. Furthermore, all cytokines except TNF-α in the CSF were found significantly higher in IIH patients compared to MS controls.
Conclusion
The presence of OCBs and elevated cytokine levels in IIH patients may support an immunologic background in the pathophysiological pathway of this disorder.
Introduction
Idiopathic intracranial hypertension (IIH), also known as pseudotumor cerebri, is a neurological entity of increased intracranial pressure in the absence of a structural lesion or any other identifiable reason. Headache and visual abnormalities are the main presenting symptoms of elevated cerebrospinal fluid (CSF) pressure. The risk factors are being overweight and of female gender of child-bearing age (1).
The pathophysiology of IIH remains controversial. Several theories, including increased cerebral blood volume, excessive CSF production, reduced CSF absorption and increased venous sinus pressure have been proposed (2–4).
Although pressure dynamics of CSF seem to have a role in the etiology, contribution of inflammatory mechanisms and changes in cytokine levels have been proposed recently (3,5). However, the exact pathophysiological mechanisms of this disorder are still not solved.
Oligoclonal bands (OCBs) are used for the diagnosis of inflammatory central nervous system (CNS) diseases as a biomarker. OCBs are the immunoglobulins derived from clones (usually immunoglobulin G (IgG)) that can be found in CSF and/or serum. The presence of OCBs only in CSF (pattern 2) and both in serum and independently in CSF (pattern 3) both suggest a humoral immune response in the intrathecal compartment (6,7) and is very useful for supporting the diagnosis of multiple sclerosis (MS). The accepted standard method for OCB detection is isoelectric focusing on agarose gels followed by immunoblotting (7,8).
There have been no previous studies systematically looking for the presence of OCBs in IIH, except for a few reports showing increased CSF IgG index or OCBs in small groups of IIH patients (9,10). After detecting OCBs accidentally in two of our patients with clear-cut diagnosis of IIH, we aimed to investigate the presence of OCBs and related clinical and immunological features in a series of consecutive patients diagnosed with IIH.
Methods
Clinical and laboratory findings of patients
All patients admitted to our outpatient clinics and diagnosed with IIH according to the revised diagnostic criteria were included between January 2012 and April 2014 (1). Patients with a detectable reason for increased intracranial pressure were classified as secondary intracranial hypertension and were excluded. Controls included age- and gender-matched 13 relapsing–remitting multiple sclerosis (RRMS) patients and 20 healthy individuals. The study was conducted in a single center and approved by the local ethics committee of Istanbul Medical Faculty (project number: 2011/1441-697). Written informed consent was obtained from all participants.
CSF analysis, magnetic resonance imaging (MRI), complete blood count and blood biochemistry analyses were performed on all patients. To exclude secondary causes of intracranial hypertension, MR venography (MRV) studies, hematological evaluation including levels of antithrombin III, protein C and protein S, antinuclear and antiphospholipid antibodies, lupus anticoagulant, homocysteine, Factor V Leiden and thyroid tests were determined in most of the cases. Detailed medical histories were taken from all participants, and histories of familial Mediterranean fever and Behçet’s disease were questioned as well as use of vitamin A, other drugs and endocrinologic disturbances known to associate with IIH. Furthermore, visual (VEP), somatosensory (SEP), brainstem auditory evoked potentials (BAEP), skin pathergy test and serum protein electrophoresis were done to exclude MS, Behçet’s disease, gammopathies and other systemic inflammatory diseases.
We also investigated current age, age at onset, gender, body mass index (BMI), follow-up duration, prognosis of headache, visual acuity and visual fields. All patients were treated medically or surgically for IIH according to their clinical needs.
Patients were seen at regular neuro-ophthalmological and neurological control visits during the follow-up period. Relapse was defined as recurrence of either: (1) papilledema or (2) symptoms accompanied by raised intracranial pressure.
Investigation of OCBs
OCBs were investigated in the CSF and serum of 26 IIH patients. All CSF samples were obtained for diagnostic purposes after receiving consent and paired CSF and serum samples were analyzed in parallel. For detection of OCBs, isoelectric focusing and immunoblotting were performed on matched serum and CSF sample pairs as described (11,12).
All methods of isoelectric focusing produce one of five internationally standardized distinct patterns (13). Pattern 1 indicates that CSF and serum do not contain OCBs. Pattern 2 discloses the presence of OCBs only in CSF, which indicates the production of intrathecal Ig. Identical bands in serum and CSF plus additional bands in CSF, known as pattern 3, is interpreted as the passage of OCBs into the sera from CSF, also compatible with production of intrathecal IgG. Mirror pattern, named as pattern 4, represents the results of identical bands found in serum and CSF indicating especially systemic immune activation. Pattern 5, in which monoclonal bands are seen, is characterized by systemic paraproteinemia (14). In our study, pattern 1 was defined as OCB negative, i.e. no detectable OCBs in CSF and serum. Patterns 2–5 were designated OCB positive, i.e. OCBs detected in CSF and/or serum (12,13). Patterns 2 and 3 suggested predominantly intrathecal and 4 and 5 suggested mainly systemic IgG production.
All patients with OCBs were also evaluated by an MS specialist to exclude a mild form of MS clinically and neuroradiologically.
Enzyme-linked immunosorbent assay (ELISA) for cytokines
To find further support for the immune hypothesis and describe the immunophenotypes in IIH, levels of major cytokines were measured in serum and CSF samples of IIH patients and controls. Since some of the samples used for OCB detection had not been archived, out of 26 IIH patients whose OCBs status was known, serum samples were available from six OCB-positive (one with pattern 2, one pattern 3, two pattern 4, two pattern 5) and 13 OCB-negative patients, and CSF samples could be obtained from only four OCB-positive (one with pattern 2, one pattern 4, two pattern 5) and 10 OCB-negative patients.
Sera of 13 RRMS patients with or without OCBs (five pattern 1, six pattern 2, two pattern 3) and 20 healthy controls as well as CSF samples of eight RRMS patients with OCBs (seven pattern 2, one pattern 3) were used as controls. CSF samples could not be obtained from healthy controls for ethical reasons.
Serum and CSF tumor necrosis factor-alpha (TNF-α), interferon-gamma (IFN-γ), interleukin (IL)-4, IL-10 IL-12, and IL-17 levels were measured using ELISA kits (Diaclone, Canton, MA, USA) according to the manufacturer’s instructions. Optical density was measured at 450 nm and concentrations were calculated by referring to a standard curve.
Statistics
Statistical comparisons of demographic and clinical features between OCB-positive and -negative IIH patients were performed with Fisher’s exact test, Student’s t-test or Mann-Whitney U test, whereas serum and CSF cytokine levels were compared with analysis of variance (ANOVA) and Tukey’s post-hoc test or Student’s t-test, as appropriate. A p value < 0.05 was considered statistically significant.
Results
Clinical and laboratory features of IIH patients
The clinical and laboratory features of patients with positive oligoclonal band patterns.

BMI: body mass index, BV: blurred vision; CN: cranial nerve; CSF-P: cerebrospinal fluid opening pressure; F: female; JME: juvenile myoclonic epilepsy; L: left; M: male; MRI: magnetic resonance imaging; MRV: magnetic resonance venography; MS: multiple sclerosis; N: normal; NWML: nonspecific white matter lesions; OCBs: oligoclonal bands; PE: papilledema; R: right; TS: transverse sinus; TVO: transient vision obscuration. →denotes changes after therapy with acetazolamide, bold characters denote pathological findings.
Eight OCB-positive patients (seven female, one male) had a mean age of 31.25 ± 6.98 years and follow-up duration of 18.88 ± 8.83 months. All had headache at the time of admission except one, who was referred to our outpatient clinic from the ophthalmology unit during a routine ophthalmologic examination by the finding of papilledema. Additionally, five patients described visual symptoms such as diplopia, blurred vision and transient visual obscurations as seen in Table 1.
The median BMI of the patients was 34.31 ± 7.49 kg/m2 (range: 27.6–48.5). Neurological examination showed papilledema in all patients and additionally right facial nerve palsy in one patient.
MRIs were normal except for nonspecific cortico-subcortical, T2 hyperintense white matter lesions without contrast enhancement in two patients (Figure 1). All MR images of our IIH patients were independently reviewed by an experienced neuroradiologist and these changes were reported as nonspecific findings not consistent with MS.
Non-specific, small cortico-subcortical hyperintense white matter lesions on T2-weighted magnetic resonance images (MRI) of oligoclonal band-positive patients diagnosed with idiopathic intracranial hypertension (patients number 1 (a) and 3 (b)).
Comparison of the clinical and laboratory findings of OCB-positive and -negative IIH patients.

IIH: idiopathic intracranial hypertension; F: female; M: male; CSF: cerebrospinal fluid; OCB: oligoclonal band; BMI: body mass index; MR: magnetic resonance; MRV: magnetic resonance venography.
MRV was performed in seven OCB-positive and 12 OCB-negative patients, ratios were calculated over these values.
Fisher’s exact test.
CSF analysis was normal except for increased CSF opening pressure in all patients (median: 442.5 ± 242.47 mmH2O; range 250–980) (Table 1).
Among all OCB-positive patients, two had no vision loss whereas six had mild to severe vision loss at admission; after medical treatment with acetazolamide vision was normalized in all except one patient, who also had amblyopia leading to vision loss in the right eye. To exclude other diseases with white matter lesions, vision loss, and OCB positivity such as MS, visual-evoked potentials were performed and showed abnormal result in only one patient with decreased visual acuity and severe visual field defect. But this patient’s SEP and BAEP examinations were normal.
History of fibromyalgia (n = 1), insulin resistance and vitamin D deficiency (n = 1), thyroid cancer and hypertension (n = 1), placental abruption (n = 1) and juvenile myoclonic epilepsy (n = 1) were present as comorbid diseases. No other comorbid conditions were found after extensive clinical questioning and investigations.
All patients were treated medically. No relapses were observed during the follow-up except one patient (12.5%), who experienced recurrent headaches after the standard treatment with acetazolamide and needed recurrent lumbar punctures for confirmation of the CSF pressure elevation in the group with OCBs. Overall five of 26 IIH patients had relapses in their follow-up periods (19.2% total relapse rate in the whole group).
Comparison of OCB-positive and -negative IIH patients
No significant differences could be found in regard to IIH profiles between the demographic, clinical and laboratory features of OCB-positive and -negative patients. However, OCB-positive patients showed significantly higher rates of vision loss (p = 0.038) (Table 2).
Cytokine levels
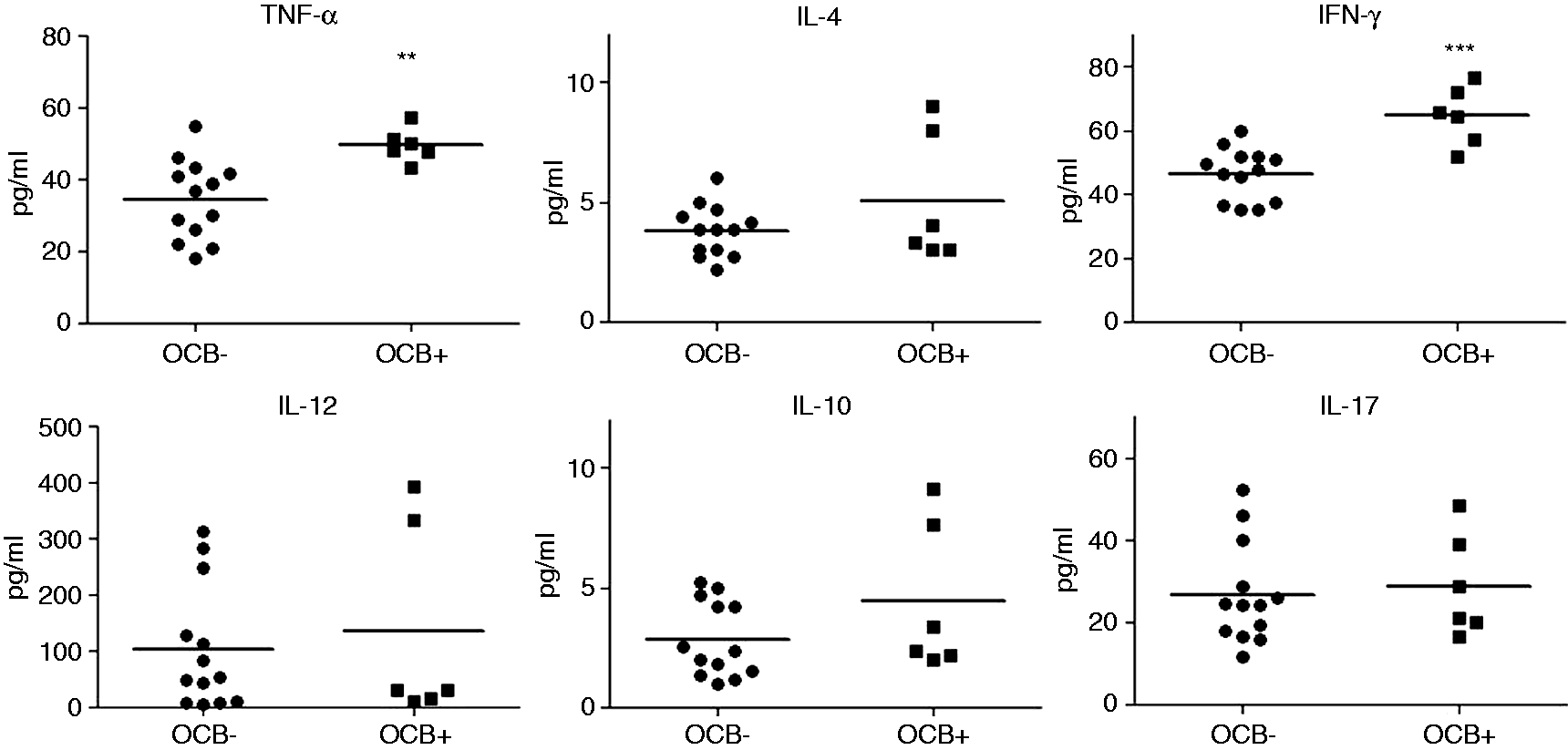
Serum levels of all investigated cytokines were significantly higher than those of healthy controls and RRMS patients. Moreover, MS patients showed significantly higher IFN-γ and IL-17 levels than healthy controls (Figure 2). Likewise, CSF levels of all cytokines except TNF-α were significantly higher than those of RRMS patients (Figure 3). When only IIH patients with known OCB results were compared, OCB-positive patients showed significantly increased serum TNF-α and IFN-γ levels (Figure 4) whereas CSF cytokine levels were comparable among OCB-positive and -negative patients (Figure 5). Analysis of cytokine levels of paired serum and CSF samples from the same patient did not show a statistically significant comparison (data not shown). Also, cytokine levels of the patients did not correlate with their BMI values (data not shown). Moreover, serum and CSF cytokine levels of IIH patients with pattern 2 vs patterns 4 and 5 OCBs did not differ significantly (not shown).
Serum cytokine levels of idiopathic intracranial hypertension (IIH) patients, relapsing–remitting multiple sclerosis (RRMS) patients and healthy controls (HC). Horizontal lines indicate mean values. * p < 0.05, ** p < 0.01, *** p < 0.001 by analysis of variance (ANOVA) and Tukey’s post-hoc test. Cerebrospinal fluid cytokine levels of idiopathic intracranial hypertension (IIH) and relapsing–remitting multiple sclerosis (RRMS) patients. Horizontal lines indicate mean values. * p < 0.05, *** p < 0.001 by Student’s t-test. Distribution of serum cytokine levels of idiopathic intracranial hypertension (IIH) patients according to oligoclonal band (OCB) status. Horizontal lines indicate mean values. OCB–: patients without OCB (n = 13); OCB+: patients with OCB (n = 6). ** p < 0.01, *** p < 0.001 by Student’s t-test. Distribution of cerebrospinal fluid (CSF) cytokine levels of idiopathic intracranial hypertension (IIH) patients according to oligoclonal band (OCB) status. Horizontal lines indicate mean values. OCB–: patients without OCB (n = 10); OCB+: patients with OCB (n = 4).


Discussion
Our results demonstrated that roughly one-third of IIH patients showed various patterns of OCBs. Moreover, we have also shown that IIH patients with OCBs display higher levels of IFN-γ and TNF-α and are more likely to develop vision loss.
There are only a few previous studies with small numbers of patients on the presence of OCBs in IIH. A pioneering study on isoelectric focusing investigated 1007 patients with various neurological diseases and among them nine patients diagnosed with “benign intracranial hypertension” were reported as being negative for local synthesis of IgG by oligoclonal banding, whereas four of them were positive by the Log IgG-Index (9). The diagnostic criteria for IIH were not described in this paper and their methodology was not the same as our current method. They interpreted the finding of local intrathecal synthesis of IgG in IIH as rather unexpected and suggested that such an avenue for immunological mechanisms in IIH would be fruitful given that the pathophysiology of IIH is unknown (9). Another small study found elevated IgG index values in two of the 11 IIH cases and isoelectric focusing was reported to be negative (10). After these studies, OCB investigations in IIH seemed to be forgotten but recently in a study aimed to investigate anti-inward rectifying potassium channel 4.1 antibodies in MS, two patients with IIH in the control group coincidentally showed positive OCBs (15). Thus, our study constitutes the largest prospective series investigating OCBs in IIH.
A few studies have evaluated levels of cytokines in IIH; however, their results showed prominent variations possibly reflecting differing methodologies and patient groups (16,17). In accordance with our findings, a recent study that analyzed the concentrations of 14 different cytokines has shown that IIH patients show higher CSF IL-17, IL-4, IL-2, IL-10 and IFN-γ levels than patients with MS, a hallmark inflammatory disease of the CNS (5), supporting the presence of an intrathecal inflammatory process. Furthermore, in a previous study we identified different IgG binding patterns (i.e. anti-neuronal antibodies) in the sera of several IIH patients in rates comparable to those of encephalitis patients (18), suggesting that inflammatory mechanisms are involved at least in some IIH patients.
Although one of the strongest and most consistent risk factors of IIH is obesity, the involvement of this clear association with IIH pathogenesis has not been unraveled as yet (19). Obesity is recognized as a proinflammatory state and is associated with increased expression of a number of adipokines and cytokines including leptin, IL-6, TNF-α, macrophage chemotactic protein-1 (MCP-1/CCL2) and plasminogen activator inhibitor-1 (PAI-1) (16). It is very interesting to note that OCBs were also determined in a rare, “overweight” syndrome, called “rapid-onset obesity with hypothalamic dysfunction, hypoventilation and autonomic dysregulation syndrome” (20). When reviewed from this aspect, as an inflammatory organ, fat tissue itself could be the main trigger for increased OCB positivity. Therefore, an easy explanation could be that obesity commonly encountered in IIH patients is inducing these generalized cytokine responses through increased release of adipokines and CSF cytokine levels are increasing as a result of passive diffusion of cytokines from serum to CSF. There might be a previously unnoticed correlation between obesity and increased OCB positivity. It could be speculated that obesity might lead to both OCB positivity and IIH emergence by activating some inflammatory pathways. However, currently there are no satisfactory studies related to this hypothesis. Moreover, two major proinflammatory cytokines, TNF-α and IL-17, showed higher CSF than serum concentrations in IIH, arguing against this assumption and indicating the intrathecal presence of cytokine-producing immune cells. Therefore, further investigation of B and T cells in CSF samples of IIH patients is warranted for clarification of the source of cytokines.
Our findings and previous reports implicate a proinflammatory cytokine profile in serum and CSF of IIH patients and thus give further support to the immune hypothesis in IIH. As expected, MS patients showed remarkably increased serum Th1 (IFN-γ) and Th17 (IL-17)-type cytokine responses as compared to healthy controls, while demonstrating relatively comparable Th2-type IL-4 and IL-10 responses. By contrast, levels of all types of cytokine responses were significantly elevated in IIH patients’ serum and CSF samples indicating a non-antigen-driven generalized proinflammatory state. Half of the OCB-positive IIH patients showed pattern 4 or 5 OCB positivity, which is indicative of systemic immune response. Another study analyzing immunoglobulin heavy chain (IGHC) variable region genes of B cells in CSF in 10 patients with IIH using real-time polymerase chain reaction and sequencing techniques has suggested the presence of both T-cell-dependent and T-cell-independent humoral immunity in the CSF of IIH (21). Furthermore, association with immunologic disorders mediated by abnormal humoral immunity, such as systemic lupus erythematosus (SLE) and antiphospholipid syndrome, is well established in IIH (22–25), which may also show a fluctuating course with “remission and relapses” seenin the course of immunological diseases in some patients.
The puzzling lack of knowledge that IIH patients may show patterns 2, 4 or 5 OCBs might create confusion in the diagnosis of IIH cases with nonspecific white matter lesions such as in some of our cases. OCBs shown by isoelectric focusing provide evidence for the occurrence of a humoral immune response, have been in use for more than 20 years in daily neurological practice and establish a part of the diagnostic algorithm of MS patients (6). On the other hand, elevated CSF pressure is extremely rare in MS patients according to our knowledge and it has been reported in only a few case reports (26,27). The surprising pattern 2-type OCB results of our first two index patients diagnosed with definite IIH, having nonspecific white matter lesions of unknown significance on MRI and suspected to have MS by other physicians (Figure 1), led us to prospectively investigate the OCB patterns in IIH patients. Our patients did not have any other symptoms and signs and their evoked potentials were also normal. In addition, experienced ophthalmologists did not report evidence of “bilateral” optic neuritis in these patients, and the visual loss disappeared in a short time after the acetazolamide treatment, which does not resemble an MS course.
It was remarkable that OCB-positive patients showed significantly higher rates of vision loss when compared to OCB-negative patients, although having very similar clinical features typical for IIH and a similar treatment regimen. It is tempting to speculate that vision loss in these patients might have some inflammatory contributions besides an increase in intracranial pressure. In further support of this inflammatory contribution, CSF pressure levels did not seem to correlate with vision loss in IIH.
Although they are from a rather small series from a single center, our findings further suggest the possible contribution of some inflammatory mechanisms in the pathogenesis of IIH and shed light for future research. Moreover, various OCB patterns found in our study indicate both systemic and intrathecal synthesis of IgG during the course of IIH. The key question is whether our findings reflect a disease-causing immunological mechanism or is entirely a coincidental phenomenon caused by non-inflammatory secondary causes such as stagnation of CSF and compression of neural tissue due to elevated CSF pressure. Another important question brought by our study is whether some IIH patients might benefit from immunosuppression or removal of adipokine-producing fat tissue by liposuction. These questions need to be addressed in future prospective studies.
Clinical implications
The presence of oligoclonal bands in idiopathic intracranial hypertension (IIH) may support an immunologic background in this disease. There is an intriguing association of oligoclonal bands with vision loss in our IIH patients.
Footnotes
Funding
This work was supported by the Istanbul University Scientific Research Fund (project no. 5148).
Conflict of interest
None declared.
Acknowledgments
The authors thank Assoc Prof Zeliha Matur for her valuable contributions for the evoked potential investigations. We also thank the participants for taking part in the present study.
