Abstract
Background
Systemic nitroglycerin (NTG) activates brain nuclei involved in nociceptive transmission as well as in neuroendocrine and autonomic functions in rats. These changes are considered relevant for migraine because NTG consistently provokes spontaneous-like migraine attacks in migraineurs. Several studies have suggested a relationship between the endocannabinoid levels and pain mediation in migraine. URB937, a peripheral inhibitor of fatty acid amide hydrolase (FAAH)—the enzyme that degrades anandamide, produces analgesia in animal models of pain, but there is no information on its effects in migraine.
Aim
We evaluated whether URB937 alters nociceptive responses in the animal model of migraine based on NTG administration in male rats, using the tail flick test and the plantar and orofacial formalin tests, under baseline conditions and after NTG administration. Furthermore, we investigated whether URB937 affects NTG-induced c-Fos expression in the brain.
Results
During the tail flick test, URB937 showed an antinociceptive effect in baseline conditions and it blocked NTG-induced hyperalgesia. URB937 also proved effective in counteracting NTG-induced hyperalgesia during both the plantar and orofacial formalin tests. Mapping of brain nuclei activated by NTG indicates that URB937 significantly reduces c-Fos expression in the nucleus trigeminalis caudalis and the locus coeruleus.
Conclusions
The data suggest that URB937 is capable of changing, probably via indirect mechanisms, the functional status of central structures that are important for pain transmission in an animal model of migraine.
Introduction
Numerous lines of evidence suggest the existence of a state of trigeminal sensitization in migraineurs, which results in hyperalgesia, allodynia, and cognitive dysfunction during and between episodes (1,2). Nitroglycerin (NTG) is a vasodilator known to induce spontaneous-like headache attacks in migraine sufferers (3), probably as a consequence of sensitization phenomena (4). Indeed, NTG administration to migraineurs is related to lower temporal summation threshold and increased painful sensation, when compared to baseline, to placebo condition and to controls. This finding suggests that migraineurs bear a vulnerability to develop headache attacks following NTG administration as a specific trait linked to a supersensitivity of the pain system to this drug (5).
Systemic administration of NTG has been used extensively as an animal model of migraine pain since it induces a condition of hyperalgesia in the rat and is associated with the activation of spinal cord and brain structures involved in nociception (6,7). In this model, we have shown that modulation of the endocannabinoid system (ECs) has an antihyperalgesic effect (8–10) and reduces the neuronal activation in the nucleus trigeminalis caudalis (NTC) and area postrema (8). These findings are in keeping with observations of other groups (11,12) to suggest the interaction of ECs with trigeminovascular system activation. The ECs exert a critical control on cerebrovascular tone by interacting with serotonergic transmission, nitric oxide (NO) production, and calcitonin gene-related peptide (CGRP) release (13). Cannabinoid-1 (CB1) receptors have been detected in periaqueductal gray (PAG) matter, rostral ventromedial medulla and NTC, which are potential migraine generators and pain modulators (14).
The hypothesis that the ECs may be dysfunctional in migraine patients has been corroborated also by clinical observations which suggest a potential effect of cannabinoids in the treatment of migraine and in cluster attacks refractory to usual drugs (10). However, biochemical studies providing a scientific basis for the potential efficacy of (endo)cannabinoids in migraine are limited. This is partly due to the psychotropic side effects associated with the use of drugs that mimic the effect of the ECs or that potentiate their activity, whether they are receptor agonists or inhibitors of the degradation process. URB937 is an inhibitor of the activity of the enzyme fatty acid amide hydrolase (FAAH), which degrades the endocannabinoid anandamide. Unlike other FAAH inhibitors, URB937 does not cross the blood-brain barrier (15). URB937 reduces nociceptive behavior in animal models of pain not specific for migraine, through a mechanism that requires elevation of anandamide levels and results in the activation of peripheral CB1 receptors (15). Therefore, URB937 provides a unique pharmacological tool to investigate the mechanisms and sites underlying pain modulation in migraine, prompting, at the same time, new, possibly effective and better-tolerated approaches for the treatment of this common disorder. In the present study, we evaluated the antinociceptive effects of URB937 in animal models of hyperalgesia and neuronal activation associated with NTG administration as a way of probing the potential antimigraine effect of URB937.
Materials and methods
Adult male Sprague-Dawley rats (weight 175 g–200 g) were used. The Helsinki Declaration and the International Association for the Study of Pain (IASP)’s guidelines for pain research in animals were followed (16). Rats were housed in plastic boxes in groups of two with water and food available ad libitum and kept on a 12:12-hour light-dark cycle. All the rats were acclimatized to the test chamber before testing began. Five to eight animals per group were used for the experiments and all procedures were in accordance with the European Convention for Care and Use of Laboratory Animals and were approved by the local animal ethic committee of the University of Pavia (Document n. 2, 2012).
Drugs
NTG (Bioindustria L.I.M. Novi Ligure (AL), Italy) was prepared from a stock solution of 5.0 mg/1.5 ml dissolved in 27% alcohol and 73% propylene glycol. For the injections, NTG was further diluted in saline (0.9% NaCl) to reach the final concentration of alcohol 6% and propylene glycol (PG) 16% and administered at a dose of 10 mg/kg. URB937 was synthesized in the laboratory of Prof Piomelli (17). It was dissolved in 100% dimethylsulfoxide (DMSO) and injected intraperitoneally at a dose of 1 mg/kg (15). DMSO was administered in a volume of 1 ml/kg body weight (18).
For the plantar formalin test (pFT), a 100 µl volume of 1% formalin (formaldehyde diluted in 0.9% saline) was injected intraplantarly. For the orofacial formalin test (oFT), a 50 µl volume of 1.5% formalin (formaldehyde diluted in 0.9% saline) was injected into the right upper lip, just lateral to the nose.
Experimental groups
Experimental plan and experimental groups.

NTG: nitroglycerin in saline, 6% alcohol, 16% propylene glycol; bNTG vehicle: saline, 6% alcohol, 16% propylene glycol; DMSO: dimethylsulfoxide; cURB937 diluted in DMSO. TFT: tail flick test; pFT: plantar formalin test; oFT: orofacial formalin test.
Tail Flick Test (TFT)
Experimental groups: NTG vehicle (N = 6); NTG (N = 7); NTG + DMSO (N = 5); NTG vehicle + DMSO (N = 5); NTG vehicle + URB937 (N = 5); NTG + URB937 (N = 8).
All rats were acclimatized to the test chamber 30 minutes before testing. Rats in this subgroup were tested for latency of reflex tail withdrawal (TFT) from a high-intensity light beam, which was considered as a measure of physiological phasic pain. The test was performed with a test instrument (Ugo Basile, model 7360, Varese, Italy) that allowed automatic recording of tail-flick latency to radiant heat with a temperature of 50℃. Latency at each evaluation was calculated as the mean of three measurements in three different parts of the tail. A cutoff limit of exposure corresponding to 20 seconds was set to avoid tissue damage. Each animal was placed on the recording platform of the instrument where it was kept under slight, painless restraint, with its tail positioned on the radiant heat window. The movement of the tail from window of the beam of light to hit a sensor (latency in seconds) was automatically registered (7). Following the TFT, each rat was sacrificed with a lethal dose of chloral hydrate.
pFT
Experimental groups: NTG vehicle (N = 6); NTG (N = 8); NTG + DMSO (N = 6); NTG vehicle + DMSO (N = 7); NTG vehicle + URB937 (N = 6); NTG + URB937 (N = 5).
Rats were acclimatized to the test chamber 30 minutes before testing. One animal at a time was placed into a Plexiglas observation chamber (10 × 20 × 24 cm) with a mirror (45-degree angle) positioned to permit unhindered observation of the animal paws. A 100 µl volume of 1% formalin solution was injected subcutaneously into the center of the plantar surface of the right hindpaw with slight restraint (19). A 26-gauge needle connected to a 1 ml syringe was used and the solution was delivered as rapidly as possible while the animal was immobilized. Each rat was then replaced in the box, the clock was started and nocifensive responses were recorded for a period of 60 minutes.
Nocifensive behavior was quantified by counting the total number of flinches and shakes occurring for one-minute periods from one to five minutes (Phase I) and then for one-minute periods at five-minute intervals during the period from 15 to 60 minutes (Phase II) after the formalin injection. Phase I is generally considered the result of chemical activation of nociceptors, while Phase II reflects the inflammatory reaction and central processing. Flinches/shakes were readily discriminated and were characterized as rapid and brief withdrawal movements or flexions of the injected paw. Following the pFT, each rat was sacrificed with a lethal dose of chloral hydrate.
oFT
Experimental groups: NTG vehicle (N = 8); NTG (N = 8); NTG + DMSO (N = 7); NTG vehicle + DMSO (N = 7); NTG vehicle + URB937 (N = 7); NTG + URB937 (N = 7).
Each animal was placed in an observation box at least 30 minutes before formalin injection to allow the acclimatization in the new environment. The box of observation was a 30 × 30 × 30-cm glass chamber with mirrored sides. A camera, recording animal behaviors for off-line analysis, was located at a distance of 50 cm from the box to provide a clear view of each rat.
The subcutaneous injection of formalin (1.5%, 50 µl) was administered into the right upper lip. Immediately after formalin injection, each animal was placed into the observation box and its behavior was recorded for 45 minutes (20).
Pain-related behavior was quantified by counting the seconds the animal spent grooming the injected area with the ipsilateral forepaw or hindpaw in both Phase I and Phase II of the test. The observation time was divided into 15 blocks of three minutes each for the time course analysis. The face rubbing time was quantified by counting the time (seconds) of face rubbing recorded during Phase I (0–6 minutes after formalin injection) and during Phase II (12–45 minutes after the formalin injection). Following the oFT, each rat was sacrificed with a lethal dose of chloral hydrate.
c-Fos expression
Experimental groups: NTG vehicle (N = 6); NTG (N = 6); NTG + DMSO (N = 10); NTG vehicle + DMSO (N = 5); NTG vehicle + URB937 (N = 5); NTG + URB937 (N = 9).
Rats in this subgroup were anaesthetized with a lethal dose of anesthetic (chloral hydrate) and perfused transcardially with saline and 270–300 ml of ice-cold 4% paraformaldehyde four hours after NTG or vehicle administration. Brains were removed, post-fixed for 12 hours in the same fixative and subsequently transferred in solutions of sucrose at increasing concentrations (up to 30%) during the following 72 hours. All brains were cut at 50 µm on a freezing sliding microtome. c-Fos expression was measured using the free floating immunohistochemical technique with a rabbit polyclonal antiserum directed against c-Fos protein (residues 4–17 of human c-Fos). Coronal sections were incubated for 48 hours at 4℃ with the c-Fos antibody (1:1000; Oncogene, Cambridge, MA, USA). After thorough rinsing in potassium phosphate-buffered saline (PBS) containing Triton X-100, sections were processed with the avidin-biotin technique, using a commercial kit (Vector Labs, Cambridgeshire, UK). Nuclei positively stained for c-Fos were visualized with nickel-intensified 3′,3′-diaminobenzidine tetrahydrochloride (DAB). After staining, sections were rinsed with potassium PBS, mounted onto glass slides, air dried and coverslipped.
Statistical evaluation
An a priori power analysis was conducted to determine the minimal sample size needed to obtain a statistical power of 0.80 at an alpha level of 0.05. In our previous study (7) evaluating the difference in nociceptive response in the second phase of the plantar formalin test (total number flinches/shakes) between rats injected with NTG and rats injected with vehicle (NTG vehicle), we calculated a standardized effect size of 23.8 for this variable. The a priori power analysis estimated a sample size of at least six rats for the experimental group. Therefore, in this study we used 6 to 10 rats per experimental group for TFT, pFT, oFT and c-Fos expression (Table 1).
The effects of treatments on the latency of the TFT were evaluated by means of the Wilcoxon rank-sum test (baseline vs post-treatment). A probability level of less than 5% was regarded as significant.
For pFT the total number of flinches/shakes evoked by the formalin injection were counted separately for Phase I and for Phase II. For oFT, the time spent (in seconds) in face rubbing was counted separately for Phase I and for Phase II. The data were tested for normality using the Shapiro-Wilk and Kolmogorov-Smirnov (K-S) normality tests and considered normal. Therefore, the differences between groups were analyzed by the one-way analysis of variance (ANOVA) followed by Tukey’s Multiple Comparison Test. A probability level of less than 5% was regarded as significant.
For c-Fos expression, cell counts of individual nuclei were made from every sixth section throughout their rostrocaudal extent for each rat and its control. In order to avoid differences related to the asymmetrical sectioning of the brain, c-Fos-positive cells were counted bilaterally (three sections for each nucleus and the mean value obtained from the two sides was used for the statistical analysis). Image analysis was performed by an investigator unaware of the experimental design, using an AxioSkop 2 microscope connected to a computerized image analysis system (AxioCam MR5) equipped with dedicated software (AxioVision Rel 4.2) (Zeiss, Oberkochen, Germany). Differences between groups were analyzed by Kruskal-Wallis test followed by Dunn’s Multiple Comparison Test. A probability level of less than 5% was regarded as significant.
Results
Evaluation of the model when using DMSO as vehicle
During the TFT, the treatment with the NTG vehicle or NTG vehicle + DMSO did not show a significant effect when compared with baseline levels. Conversely NTG, either used alone or in association with DMSO, induced hyperalgesia, as demonstrated by the significant reduction in the latency of the tail flick when compared with baseline levels (Figure 1).
Effect of NTG and DMSO during the tail flick test.
At the pFT, DMSO induced an increase in nocifensive behavior during Phase I, but not during Phase II (Figure 2). NTG administration significantly increased nocifensive behavior only in Phase II, in agreement with our previous report (21). DMSO did not influence the hyperalgesic effect of NTG on Phase II.
Effects of NTG and DMSO in the plantar formalin test.
At the oFT, DMSO administration (NTG vehicle + DMSO) caused a significant decrease in face rubbing elicited by the formalin injection during both phases of the test, when compared to the NTG vehicle alone (Figure 3). Furthermore, DMSO in association with NTG produced a significant reduction in nocifensive behavior in Phase II (NTG + DMSO group vs NTG group, p < 0.05), which suggests that DMSO has an antihyperalgesic effect. Notwithstanding the observed antihyperalgesic effect of DMSO, NTG administration, alone or with DMSO, significantly increased Phase II nocifensive behavior, when compared to either the NTG vehicle or the NTG vehicle + DMSO group, thus showing for the first time that NTG induces trigeminal hyperalgesia and confirming the validity of the model even with use of DMSO as a vehicle.
Effects of NTG and DMSO in the orofacial formalin test.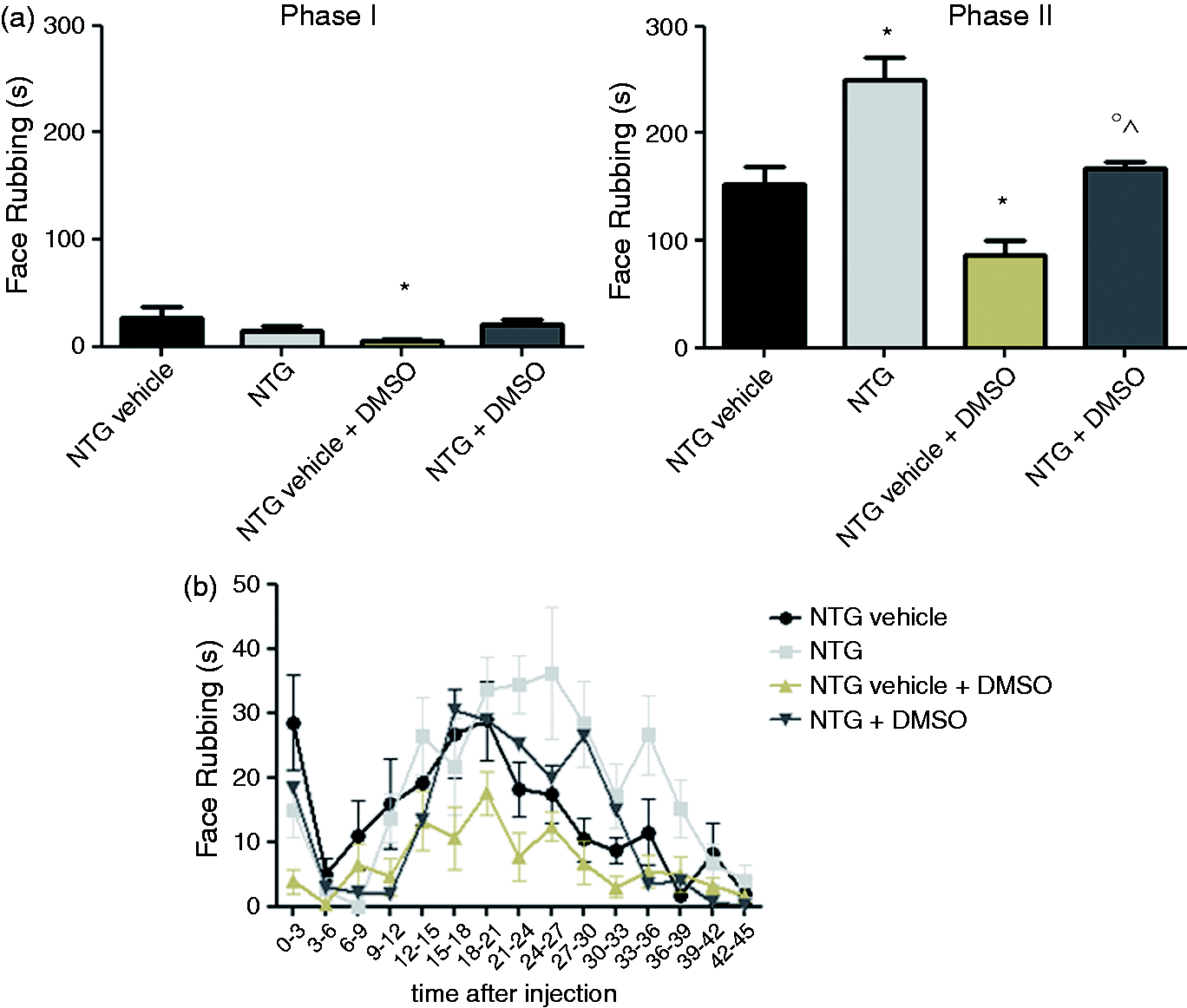
Effect of URB937 during the TFT
URB937 administered after NTG vehicle caused a significant increase in tail flick latency when compared to baseline value, suggesting an antinociceptive effect (Figure 4). When URB937 was administered after NTG injection, the inhibitor prevented NTG-induced hyperalgesia, since no significant difference was reported when compared with baseline levels.
Effect of URB937 during the tail flick test.
Effect of URB937 on the pFT
No differences were observed between groups as regards Phase I of the test (Figure 5). URB937 (NTG vehicle + URB937 group) significantly inhibited nociceptive behavior induced by the formalin injection during Phase II of the test, when compared to the NTG vehicle + DMSO group, thus confirming previous results by others (15). Furthermore, when administered in association with NTG, URB937 significantly decreased the total number of flinches/shakes observed in the NTG + DMSO group in Phase II of the test (NTG + URB937 group vs NTG + DMSO group p < 0.05).
Effect of URB937 during the plantar formalin test.
Effect of URB937 during the oFT
No differences were observed between groups as regards Phase I of the test (Figure 6). As regards Phase II, NTG vehicle + URB937 administration did not inhibit the face rubbing induced by the formalin injection when compared to the NTG vehicle + DMSO group (Figure 6); by contrast, when administered in association with NTG, URB937 significantly decreased the face rubbing when compared to the NTG + DMSO group.
Effect of URB937 during the orofacial formalin test.
c-Fos expression
Systemic NTG administration alone or with DMSO induced significant c-Fos expression in the paraventricular nucleus of the hypothalamus (PVH), the central nucleus of the amygdala (AMI), the ventrolateral column of the periaqueductal grey (PAG), locus coeruleus (LC), parabrachial nucleus (PAB), nucleus trigeminalis caudalis (NTC) and nucleus tractus solitarii (NTS), when compared to NTG vehicle or NTG vehicle + DMSO groups, respectively (Figure 7 and Table 2). No significant differences in c-Fos expression were seen between NTG vehicle + DMSO and NTG vehicle. NTG vehicle + URB937 administration did not induce any significant c-Fos expression in the brain nuclei under evaluation, while when it was administered three hours after NTG, URB937 significantly reduced NTG-induced c-Fos expression in the NTC and LC.
Micrograph of representative sections of the locus coeruleus (LC) and nucleus trigeminalis caudalis (NTC) of rats treated with nitroglycerin and DMSO (NTG + DMSO) (a), NTG vehicle + DMSO (b) and NTG + URB937 (c) killed by means of perfusion four hours after NTG or NTG vehicle. 4V: fourth ventricle; DMSO: dimethylsulfoxide. Expression of Fos protein in specific brain areas (data are expressed as mean + SEM) in the experimental groups. Kruskal-Wallis test followed by Dunn’s Multiple Comparison Test: ap < 0.05 vs NTG vehicle; NTG vehicle + DMSO; bp < 0.05 vs NTG + DMSO. NTG: nitroglycerin in saline, 6% alcohol, 16% propylene glycol; DMSO: dimethylsulfoxide; PVH: paraventricular nucleus of the hypothalamus; AMI: central nucleus of the amygdale; PAG: ventrolateral column of periaqueductal gray; LC: locus coeruleus; PAB: parabrachial nucleus; NTC: nucleus trigeminalis caudalis; NTS: nucleus tractus solitarius.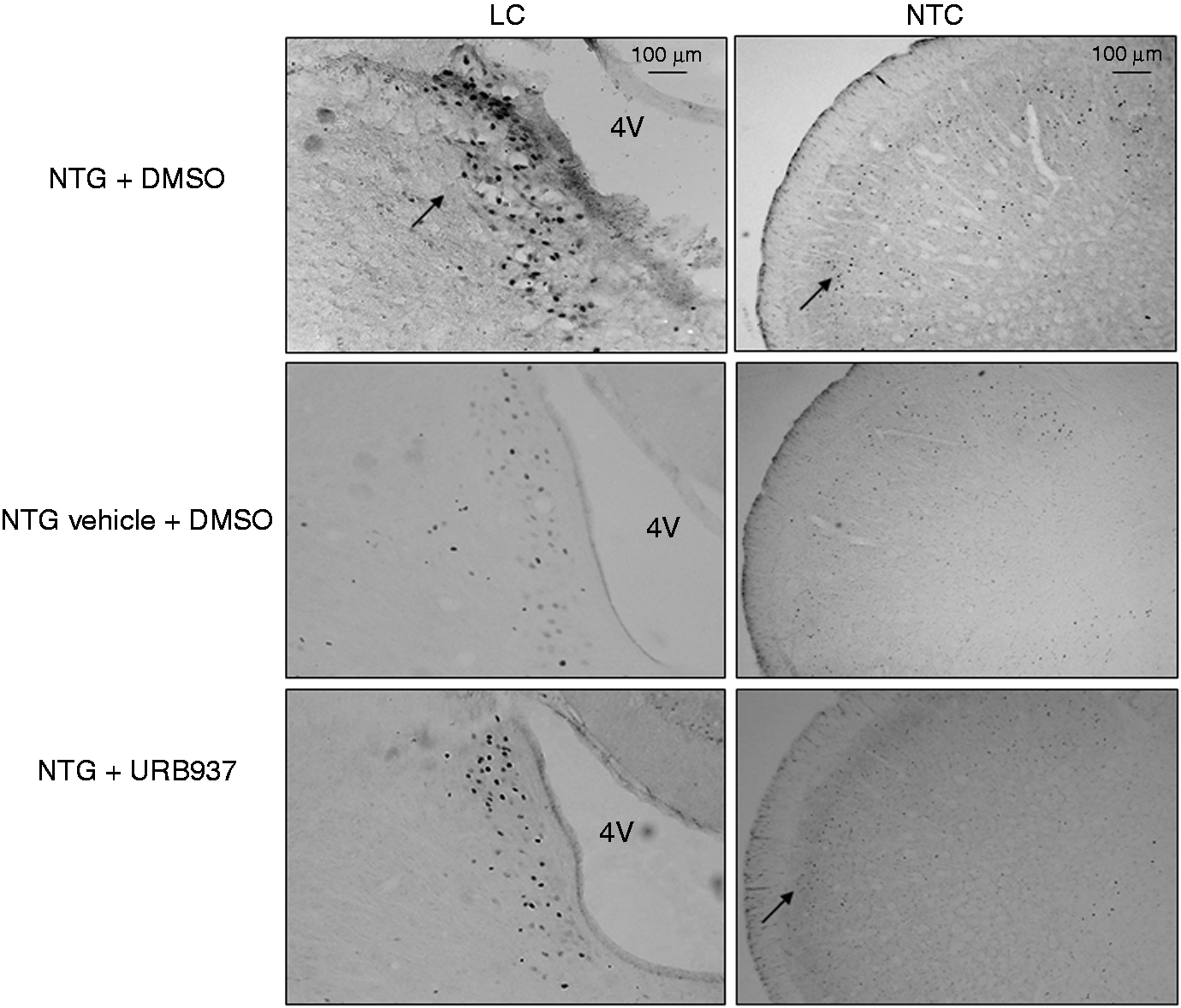

Discussion
The antinociceptive actions of cannabinoids are well established, but their potential therapeutic use continues to be hindered by their central psychotropic side effects. Stimulation of CB1 receptors by cannabinoid agonists has been associated with memory loss, sedation and dependence (22). In particular, modifications of synaptic plasticity by activation of CB1 receptors may affect signal processing as well as learning and memory in different regions of the brain (22). Enhancing endocannabinoid tone has been proposed as an alternative means of activating cannabinoid receptors without concomitant overt psychotropic effects associated with potent synthetic cannabinoid receptor agonists. FAAH is an intracellular serine hydrolase that catalyzes the cleavage of several bioactive endogenous fatty acid amides, such as anandamide, while the hydrolysis of 2-arachidonoylglycerol is primarily operated by monoacylglycerol lipase (MAGL). Several studies have reported that inhibition of FAAH or MAGL produces analgesia and reduces inflammation in models of acute inflammatory pain (23,24), but there is no information on their effects in migraine. The antinociceptive effects of FAAH inhibitors positively correlated with increased central nervous system (CNS) endocannabinoid levels (25) and they might also share some of the adverse effects of cannabis. In this frame, URB937, a peripheral inhibitor of FAAH (15), seemed particularly promising in terms of safety as well as of potential efficacy since it was shown that the substance caused analgesia in diverse animal models of pain via the reduction in anandamide deactivation only in peripheral tissues.
Previous reports (6) have suggested that NTG induces hyperalgesia via complex mechanisms that involve also sensitization of the trigeminovascular system at the meningeal level via the activation of NO synthesis. Based on experimental evidence of the antinociceptive action of endocannabinoids and their role in the modulation of trigeminovascular system activation (10), it was hypothesized that the ECs may be dysfunctional in migraine patients, thus suggesting the potential use of endocannabinoid-based agents in the treatment of migraine and in cluster attacks refractory to usual drugs.
Therefore, the objective of the present study was to investigate the potential antinociceptive effect of URB937 in animal models of hyperalgesia induced by NTG administration. The model based on the effects of neuronal activation and hyperalgesia induced by NTG administration is generally recognized as a reliable animal model of migraine (6) and it has been used over the years to test the antihyperalgesic effects of different drugs (8,26).
Our findings indicate that peripheral inhibition of FAAH caused via systemic administration of URB937 is capable of causing analgesia during the TFT (with or without NTG), of counteracting formalin-induced hyperalgesia in the hindpaw and to attenuate NTG-induced hyperalgesia in the hindpaw and in the face. These findings are in agreement with previous data regarding the ability of URB937 to modulate spinal nociceptive processing during the pFT (15). The antinociceptive effect of URB937 during the TFT is entirely novel and suggests that URB937 may interfere with the normal processing of nociception. However, it is worth noting that, in our experimental paradigm, the latency in each animal was calculated as the mean of three measurements in three different parts of the tail (initial, middle and terminal part of the tail). We hypothesize that the heat reaches the nociceptors more rapidly at the tip of the tail, where the skin is thinner, but not in the initial or middle part of the tail. Therefore, it is possible that the exposure to radiant heat emission has induced local damage in the tissue and, thus, a local release of anandamide by peripheral nociceptors.
The most important finding in the present study is the inhibitory effect of URB937 on NTG-induced hyperalgesia in the oFT, which is not associated with an inhibitory effect of URB937 on formalin-induced nocifensive behavior in the same region. This “dissociated” effect suggests a possible specificity of action of URB937 in cephalic, trigeminally mediated pain. The existence of differences in pain generation and modulation between the extracephalic and cephalic areas is further suggested by the observation regarding the opposite direction of the effect of DMSO on Phase I of the pFT and oFT (Figures 2 and 3). Additional studies are necessary to elaborate more contextualized hypotheses.
Since URB937 acts only peripherally, we hypothesize that the effects observed in the present study are possibly related to the maintenance of a higher concentration of local anandamide released by neurons localized in the injured peripheral tissues (hindpaw, upper lip, tail) (27).
Therefore, in particular during the pFT and oFT, anandamide might activate CB1 receptors or desensitize vanilloid type 1 receptors (TRPV1) expressed in primary sensory neurons, therefore reducing the transmission of pain to central areas (15,28), probably via the inhibition of the release of NO (29) or of neuropeptides from primary afferents at the spinal (30) and probably trigeminal level. NO is involved in the development and maintenance of hyperalgesia in the periphery and probably also at central sites. Indeed, activation of neuronal NOS may induce spinal (and/or supraspinal) synaptic plasticity with a prolonged increase in synaptic length (31).
Several reports suggest that NTG, as an “NO donor,” may exert its hyperalgesic effect through peripheral mechanisms (32), via activation of NOS and calcitonin gene-related peptide (CGRP) expression at the meningeal level (32,33). This would in turn induce a condition of sensitization, which explains the intensification of nocifensive behavior observed in particular during the oFT. Indeed, the present study shows for the first time that systemic administration of NTG alone or with DMSO enhances pain in the trigeminal area and that URB937 treatment reduces it.
Our experimental paradigm seems thus to identify another possible peripheral site of action for URB937, represented by the dura mater, where anandamide may be released from the terminal of meningeal nociceptors and from trigeminovascular endings after NTG injection to modulate the trigeminovascular system. This is further supported by the demonstration that infusion of NTG promotes a delayed and robust increase in the mechanosensitivity of meningeal nociceptors with a time-course resembling the development of the delayed migraine headache (34). URB937-induced protection of anandamide signaling in the dura might activate CB1 receptors or desensitize the TRPV1 receptor on peripheral trigeminovascular endings or meningeal nociceptors to block NTG-induced neuronal inflammation and sensitization, via inhibition of NO and/or CGRP action, thus reducing nocifensive behavior (32,35). This is in keeping with data by Akerman et al. (11), which demonstrated that anandamide is able to inhibit dural blood vessel dilation brought about by electrical stimulation, CGRP, capsaicin or NO, and is also in keeping with data from our group (8), which showed that peripherally administered anandamide inhibits NTG-induced nocifensive behavior in the same model used here. Additionally, anandamide might reduce the formation of NO by inducible nitric oxide synthase (iNOS) in macrophages located in the meninges (33) via CB1 or CB2 receptors activation (36). However, since the CB1 receptors are present in the smooth muscle of cerebral arteries and other vascular beds (37), it is possible that they are also represented in the smooth muscle of dural arteries. Therefore, it is intriguing to speculate that peripheral anandamide via these or other receptors may inhibit NO release in the smooth muscle of dural arteries, indirectly reducing trigeminal hyperalgesia. This is also true for TRPV1 receptors (38). Future studies will be aimed at evaluating whether CB1 and/or CB2 receptor activation contribute to URB937-induced analgesia.
The antihyperalgesic effect of URB937 in the trigeminal area in vivo was paralleled by the inhibition of NTG-induced neuronal activation in NTC and LC, two brain nuclei that play an important role in nociception and in NTG-induced hyperalgesia, likely mediated via the sensitization of the trigeminovascular system (6). The finding is in keeping with the hypothesis that URB937 exerts its effect at the meningeal level within the trigeminovascular system, a system that has direct connections with TNC and indirect connections with LC.
According to this line of reasoning, it seems reasonable to hypothesize that the inhibition of NTG-induced increase in c-Fos expression in the NTC and LC observed following URB937 administration is likely to represent an indirect effect, which is secondary to the reduction in pain transmission from peripheral areas to the CNS. In particular, the inhibition of activation of peripheral afferents following URB937 administration might reduce the nociceptive signal to second-order neurons and increase c-Fos transcription in the NTC. On the other hand, trigeminal peripheral fibers innervate the dura mater but also cerebral blood vessels, to create, together with LC projection, the anatomic and functional substrate for neurovascular coupling, a function that is fundamental for brain homeostasis and that is likely altered in migraine brain (39). Thus, it is possible that URB937 causes a reduction of NTG-induced neuronal activation in the LC indirectly, via inhibition of the trigeminovascular action on cerebral vessels. The LC is the main provider of noradrenergic projections to intracranial vessels, which makes it one of the major players when considering migraine as a disorder of neurogenically-mediated change in cerebral blood flow (40). Previous studies showed that noradrenergic projections from the LC are closely associated with cerebral arterioles and capillaries (41) and also directly affect cerebral blood flow (42). We have previously shown (43) that, in the dura, NTG administration induces a biphasic response represented by an initial decrease in blood flow followed by a significant increase. At variance, in the cortex NTG causes only an increase in blood flow. Pretreatment with inhibitors of NOS, L-NG-nitroarginine methyl ester (L-NAME) or 7-NI prevents NTG-induced increase in blood flow in both districts (43). Further studies are needed, which include the essay of anandamide in the tissues of interest and the evaluation of intracranial (dural and cerebral) blood flow in order to provide evidence to support or refute this hypothesis.
Conclusions
The present findings show that URB937 is effective in preventing pain in an animal model of migraine. Its mechanism of action is peripheral, but capable of changing, probably via indirect mechanisms, the functional status of central structures that are important for pain transmission and modulation.
Article highlights
URB937, a peripheral inhibitor catabolizing enzyme fatty acid amide hydrolase (FAAH), is effective in counteracting nitroglycerin (NTG)-induced hyperalgesia, a model of migraine pain. URB937 also significantly reduces NTG-induced c-Fos expression in the nucleus trigeminalis caudalis and the locus coeruleus, probably via indirect mechanisms. Modulation of the endocannabinoid system at the peripheral level may be important in the mediation of pain associated to migraine.
Footnotes
Funding
This work was supported by a grant from the Italian Ministry of Health to “C. Mondino” National Neurological Institute (Ricerca Corrente 2012).
Conflict of interest
None declared.
