Abstract
Background
Trigeminal sensitization represents a major mechanism underlying migraine attacks and their recurrence. Nitroglycerin (NTG) administration provokes spontaneous migraine-like headaches and in rat, an increased sensitivity to the formalin test. Kynurenic acid (KYNA), an endogenous regulator of glutamate activity and its analogues attenuate NTG-induced neuronal activation in the nucleus trigeminalis caudalis (NTC). The anti-hyperalgesic effect of KYNA analogue 1 (KYNA-A1) was investigated on animal models specific for migraine pain.
Aim
Rats made hyperalgesic by NTG administration underwent the plantar or orofacial formalin tests. The effect of KYNA-A1 was evaluated in terms of nocifensive behavior and of neuronal nitric oxide synthase (nNOS), calcitonin gene-related peptide (CGRP) and cytokines expression in areas involved in trigeminal nociception.
Results
KYNA-A1 abolished NTG-induced hyperalgesia in both pain models; NTG alone or associated to formalin injection induced an increased mRNA expression of CGRP, nNOS and cytokines in the trigeminal ganglia and central areas, which was reduced by KYNA-A1. Additionally, NTG caused a significant increase in nNOS immunoreactivity in the NTC, which was prevented by KYNA-A1.
Conclusion
Glutamate activity is likely involved in mediating hyperalgesia in an animal model specific for migraine. Its inhibition by means of a KYNA analogue modulates nNOS, CGRP and cytokines expression at peripheral and central levels.
Introduction
Migraine pathophysiology is not fully understood, although it has become evident over the years that migraine attacks result from a vicious cycle where trigeminovascular system activation causes dural neurogenic inflammation in a predisposed ‘terrain’ characterized by the dysfunction of the pain control system. This dysfunction favors the activation of the trigeminal nerve on one side, leading, on the other, to its sensitization as a consequence of the repeated release of inflammatory mediators at the dural level. Numerous lines of evidence suggest the existence of a condition of trigeminal sensitization in migraineurs, which can be detected as hyperalgesia and allodynia during and between episodes (1). Among the mediators of migraine-associated hyperalgesia, nitric oxide (NO) has been implicated in the periphery as well as at central sites. The systemic administration of nitroglycerin (NTG), an NO donor, provokes spontaneous migraine-like attacks (2), and the facilitation of pain transmission at the spinal level only in migraine patients (3), probably as a consequence of a sensitization phenomenon (4). Analogously, in the rat, NTG induces a condition of hyperalgesia detectable as an increase in the nocifensive response to the plantar and orofacial formalin tests (5,6). Another neurotransmitter that has been implicated in the mediation of hyperalgesia, with mechanisms that may be relevant for migraine, is glutamate. Glutamate dilates intracranial and extracranial blood vessels through activation of N-methyl-D-aspartate (NMDA) receptors, leading to NO release (7). Furthermore, it plays a key role in trigeminal nociception, as suggested by the presence of different types of glutamate receptors in the nucleus trigeminalis caudalis (NTC) (8). The role of glutamate in migraine is also suggested by clinical evidence showing increased levels in the platelets and plasma of migraineurs (9). Kynurenic acid (KYNA) is a product of normal metabolism of the amino acid L-tryptophan formed from L-kynurenine. KYNA, through the antagonism of NMDA receptors (10), and its analogues may exert modulatory effects in many neuropathological conditions, including migraine pain.
KYNA crosses the blood–brain barrier (BBB) very poorly, so that has stimulated the synthesis of several new KYNA analogues, prodrugs or KYNA amides able to cross the BBB. In particular, it is also worth noting that the KYNA analogue 1 (KYNA-A1), N-(2-N,N-Dimethylaminoethyl)-4-oxo-1H-quinoline-2-carboxamide hydrochloride, shows a more marked inhibitory effect than KYNA upon NTG-induced neuronal activation of second-order sensory neurons in the NTC (11). Moreover, KYNA-A1 reduces the number of cells immunoreactive for neuronal NOS (nNOS), calcitonin gene-related peptide (CGRP) release and calmodulin-dependent protein kinase II alpha (CamKIIα) expression in the same nucleus (12,13).
The aim of this study was to investigate the role of KYNA-A1 in modulating spinal and trigeminal hyperalgesia induced by NTG, a well-known animal model of migraine, through the evaluation of KYNA-A1 modulation of the nocifensive response to plantar and orofacial formalin tests (5). Additionally, in order to gain new insights on KYNA-A1 effect upon the trigeminal hyperalgesia mechanisms, our attention was focused specifically on changes in nNOS, CGRP and cytokine expression in specific cerebral and peripheral areas involved in sensitization.
Material and methods
Adult male Sprague-Dawley rats (weight 175–200 g) were used. The IASP’s guidelines for pain research in animals were followed (14). Rats were housed in plastic boxes in groups of two with water and food available ad libitum and kept on a 12:12 hours light-dark cycle. All procedures were in accordance with the European Convention for Care and Use of Laboratory Animals and were approved by the Italian Ministry of Health (Document number 2, 2012).
Drugs
Nitroglycerin (NTG) (Bioindustria L.I.M. Novi Ligure (AL), Italy) was prepared from a stock solution of 5.0 mg/1.5 mL dissolved in 27% alcohol and 73% propylene glycol. For the injections, NTG was further diluted in saline (0.9% NaCl) to reach the final concentration of alcohol 6% and propylene glycol (PG) 16% (used as NTG vehicle). The KYNA-A1 was synthesized in the laboratory of Dr Ferenc Fülöp (Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University of Szeged) and was freshly prepared daily by dilution in saline (90 mg/ml) (12).
Experimental design
Rats were assigned to the plantar or orofacial formalin test. For both types of formalin test we foresaw the following four treatment groups, each formed by 6–12 animals: 1) Control; 2) KYNA-A1; 3) NTG and 4) KYNA-A1+NTG.
Vehicle or NTG (10 mg/kg) were injected intraperitoneally (i.p.) and rats were exposed to plantar or orofacial formalin test four hours later (5,6). The latency of four hours was selected in agreement with our previous observations, as it corresponds to the timing of maximal neuronal activation induced by NTG (5,6).
Group 4 (KYNA-A1+NTG) was formed by two subsets of animals that were randomized to one of the following treatment schedules: a) in order to investigate the inhibitory effect of KYNA-A1 on NTG-induced hyperalgesic responses, a dose of 300 mg/kg was administered i.p. 30 minutes before NTG/vehicle administration; b) in order to evaluate the potential abortive effect of KYNA-A1 on NTG-induced trigeminal hyperalgesia, in another experimental group of rats KYNA-A1 (300 mg/kg i.p.) was administered three hours after NTG/vehicle.
Rats were assigned to formalin test type and treatment groups according to a randomization list to ensure blinding to treatments of the researchers who performed the behavioral testing. Animals were acclimatized to the test chamber 30 minutes before testing. Following nociceptive tests, to decrease pain or distress in the animal, each rat was sacrificed with a lethal dose of anesthetic (Zoletil 100, 300 mg/Kg, intramuscular).
Plantar formalin test
One animal at a time was placed into a plexiglass observation chamber (10 × 20 × 24 cm) with a mirror (45° angle) positioned to permit unhindered observation of the animal’s paws. A 100 µl volume of 1% formalin solution (formaldehyde diluted in 0.9% saline) was injected subcutaneously into the center of the plantar surface of the right hind paw with slight restraint (15). A 26-gauge needle connected to a 1 ml syringe was used, and the solution was delivered as rapidly as possible while the animal was immobilized.
Pain-related behavior was quantified for one hour by counting spontaneous flinches and shakes of the injected paw: over 60 second periods for the first five minutes and thereafter following four minute pauses, for one minute periods up to the hour. Phase I was defined as the period from one to five minutes, phase II was defined as the period from 15 to 60 minutes inclusive. Flinches/shakes were readily discriminated and were characterized as rapid and brief withdrawal movements or flexions of the injected paw.
Orofacial formalin test
The box of observation was a 30 × 30 × 30 cm glass chamber with mirrored sides. A camera, recording animal behaviors for off-line analysis, was located at a distance of 50 cm from the box to provide a clear view of each rat.
A 50 µl volume of 1.5% formalin solution was injected subcutaneously into the right upper lip. Immediately after formalin injection, each animal was placed into the observation box and its behavior recorded for a 45-minute period (16). The pain–related behavior was quantified by counting the seconds spent by the animal in grooming the injected area with the ipsilateral fore- or hind-paw counted separately for phase I (0–6 minutes after formalin injection) and phase II (12–45 minutes after formalin injection) of the test. The observation time was divided into 15 blocks of three minutes each for the time course analysis.
CGRP, nNOS, TNF-alpha and IL-6 mRNA expression following the orofacial formalin test
Primer sequences.

CGRP and nNOS protein expression in the NTC following the orofacial formalin test
CGRP and nNOS protein expression was evaluated separately on each side of the NTC 60 minutes after the orofacial formalin injection. Rats were anaesthetized and perfused transcardially with saline and 270–300 ml of ice-cold 4% paraformaldehyde. The CSC and medulla were removed, post-fixed for 12 h in the same fixative and subsequently transferred into solutions of sucrose at increasing concentrations (up to 30%) during the following 72 h. All samples were cut at 50 µm on a freezing sliding microtome. CGRP and nNOS protein expression was evaluated using the free-floating immunohistochemical technique (6). For CGRP we used an anti-rabbit antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA) at a dilution of 1:3200. For nNOS we used an anti-goat antibody (Abcam, Cambridge, UK) at a dilution of 1:1000. CGRP/nNOS immunoreactivity was visualized with the peroxidase substrate kit DAB (3′3′-diaminobenzidine tetrahydrochloride) (Vector Laboratories, Burlingame, CA, USA).
Experimental groups, timing of treatments/tests and parameters evaluated
Experimental paradigm used for the plantar formalin test.

Experimental paradigm used for the orofacial formalin test (KYNA-A1 30 minutes before NTG or NTG vehicle injection).

Experimental paradigm used for the orofacial formalin test (KYNA-A1 3 hours after NTG or NTG vehicle injection).
Statistical evaluation
An a priori power analysis was conducted to determine the minimal sample size needed to obtain a statistical power of 0.80 at an alpha level of 0.05. On the basis of our previous studies evaluating the total number of flinches/shakes in phase II of the plantar formalin test, between rats injected with NTG and rats injected with its vehicle, we calculated a standardized effect size of 1.62 for this variable (GPower 3.1.9.2), estimating a sample size of at least six rats for each experimental group. However, since the orofacial formalin test shows an intergroup variability, we have used a maximum of 12 rats. All data were tested for normality using the Shapiro-Wilk and KS normality tests and considered normal. The differences between groups or between ipsilateral and contralateral sides were analyzed by one-way or two-way analysis of variance (ANOVA) followed by Newman-Keuls Multiple Comparison Test or Bonferroni post-hoc test. Data are expressed as mean ± SEM. For the plantar and orofacial formalin tests, the nocifensive behavior was evaluated separately for phase I and for phase II. For mRNA expression, results were analyzed using the ΔCt method to compare expression of genes of interest with that of GAPDH. Optical density (OD) values were taken to indicate the expression of CGRP in NTC as suggested by the previous report (18). For nNOS immunoreactive neurons, we counted those exhibiting cytoplasmic staining in the NTC using an AxioSkop 2 microscope (Zeiss).
Results
Plantar formalin test
NTG administration significantly increased nocifensive behavior in phase II compared to NTG vehicle, in agreement with our previous findings (5). No differences were observed between groups as regards phase I of the test. In phase II, KYNA-A1 administered 30 minutes before NTG vehicle did not influence formalin-induced nocifensive behavior per se, but it prevented NTG-induced hyperalgesic behavior, as suggested by the observed significant decrease in the total number of flinches/shakes in phase II of the test. Data are shown in Figure 1.
Effects of KYNA-A1 on nitroglycerin-induced hyperalgesia in the plantar formalin test. In panel a, the histograms illustrate the total number of flinches and shakes per phase of the test. No significant effect was observed in phase I. In phase II, Nitroglycerin (NTG) administration significantly increased the total number of flinches/shakes compared to the CT group (Control group) and to the KYNA-A1 group. Pretreatment with KYNA-A1 counteracted NTG-induced hyperalgesia. Panel b illustrates the time course of the nocifensive behavior in the different groups. Data are expressed as mean ± SEM. Two-way analysis of variance (ANOVA) was followed by Newman-Keuls Multiple Comparison Test: **p < 0.01 versus the CT group and versus the KYNA-A1 group; *** p < 0.001 versus the NTG group.
Orofacial formalin test
NTG administration significantly increased the time spent in face rubbing during phase II of the orofacial formalin test compared to the CT group. No differences were observed between groups in phase I of the test. In phase II, KYNA-A1 administered 30 minutes before or three hours after NTG vehicle did not show any effect on the face rubbing behavior induced by formalin injection per se, but it prevented NTG-induced hyperalgesia, as suggested by the attenuation of face rubbing behavior. Data are shown in Figure 2.
Effects of KYNA-A1 on nitroglycerin-induced hyperalgesia in the orofacial formalin test. Panel a shows the effects of KYNA-A1, administered 30 minutes before nitroglycerin (NTG). Panel b shows the effects of KYNA-A1, administered three hours after NTG. The histograms illustrate the total time of face rubbing (in seconds) during Phases I and II. The respective time course is illustrated under the histogram plot. No significant effect was observed in phase I. In phase II, NTG administration significantly increased the time of face rubbing compared to the CT group (Control) and to the KYNA-A1 group. KYNA-A1 counteracted NTG-induced hyperalgesia when administered both before and after NTG. Data are expressed as mean ± SEM. Two-way analysis of variance (ANOVA) followed by Newman-Keuls Multiple Comparison Test: **p < 0.01 versus the CT group and KYNA-A1 group; °°°p < 0.001 and °°p < 0.01 versus the NTG group.
mRNA expression of nNOS, CGRP and cytokines during the orofacial formalin test
Formalin injection per se (CT group) induced an increase in mRNA expression of all the investigated genes in all areas under evaluation, when compared to rats injected with saline into the right upper lip (data not shown).
Medulla pons
We did not detect any significant differences in this area between the CT and KYNA-A1 groups. NTG administration caused a marked and significant increase in the expression of mRNA of all genes compared to the CT group. KYNA-A1 did not influence the formalin-induced increase in mRNA levels of genes in the areas under investigation, but it prevented the increase induced by NTG administration in mRNA levels of all genes. Data are shown in Figure 3.
mRNA expression of nNOS, CGRP and cytokines in the medulla-pons area following the orofacial formalin test. We did not detect any significant differences between the medulla-pons of the CT and KYNA-A1 groups. Conversely, nitroglycerin (NTG) induced a marked increase in the expression of mRNA of all genes compared to the CT group. KYNA-A1 administration prevented the increase induced by NTG administration in mRNA levels of all genes. Data are expressed as mean ± SEM, two-way analysis of variance (ANOVA) followed by Newman-Keuls Multiple Comparison Test: *p < 0.05 and ***p < 0.001 versus the CT and KYNA-A1 groups; °°°p < 0.001 versus the NTG group; §§p < 0.01 and §§§p < 0.001 versus the KYNA-A1 group; ##p < 0.01 versus the CT group.
Cervical spinal cord (CSC) and Trigeminal Ganglion (TG)
NTG administration induced a marked and significant increase in the expression of mRNA of all genes as compared to the CT group. KYNA-A1 prevented this increase. When analyzing differences between the formalin-injected versus the non-injected side, we did not find any significant difference in nNOS gene expression in any of the experimental groups. At variance with this, we detected a significant difference between sides in CGRP and cytokines mRNA expression in both areas, with more expression on the formalin-injected side (Figures 4a and 5a).
mRNA expression of nNOS and CGRP in the cervical spinal cord (CSC) and trigeminal ganglia (TGs) following the orofacial formalin test. Nitroglycerin (NTG) administration caused a significant increase in nNOS, and CGRP mRNAs in both areas. The observed increase was inhibited by KYNA-A1 administration. Panel a: no significant difference in nNOS gene expression was reported between sides of CSC of all experimental groups, while CGRP gene expression was more marked on the injected side of the NTG group. KYNA-A1 inhibited both nNOS and CGRP expression on both sides. Panel b: a significant increase in the expression of both nNOS and CGRP mRNAs was found in the TG ipsilateral to the formalin injection compared to the contralateral side in the NTG group. KYNA-A1 inhibited the mRNA increase of both genes only in the TG ipsilateral to formalin injection. mRNA expression of cytokines in the cervical spinal cord (CSC) and trigeminal ganglia (TGs) following the orofacial formalin test. Nitroglycerin (NTG) administration caused a significant increase in IL-6 and TNF-alpha mRNAs in both areas. The observed increase was attenuated by KYNA-A1 administration. Panel a: a significant increase in IL-6 mRNA expression was observed in the CSC only on the formalin-injected side of the NTG group. KYNA-A1 administration inhibited this increase. Panel b: a significant increase in IL-6 and TNF-alpha mRNAs was observed bilaterally in the TGs in the NTG group, being more marked on the ipsilateral side. KYNA-A1 administration significantly inhibited the observed formalin-induced increase in the NTG group.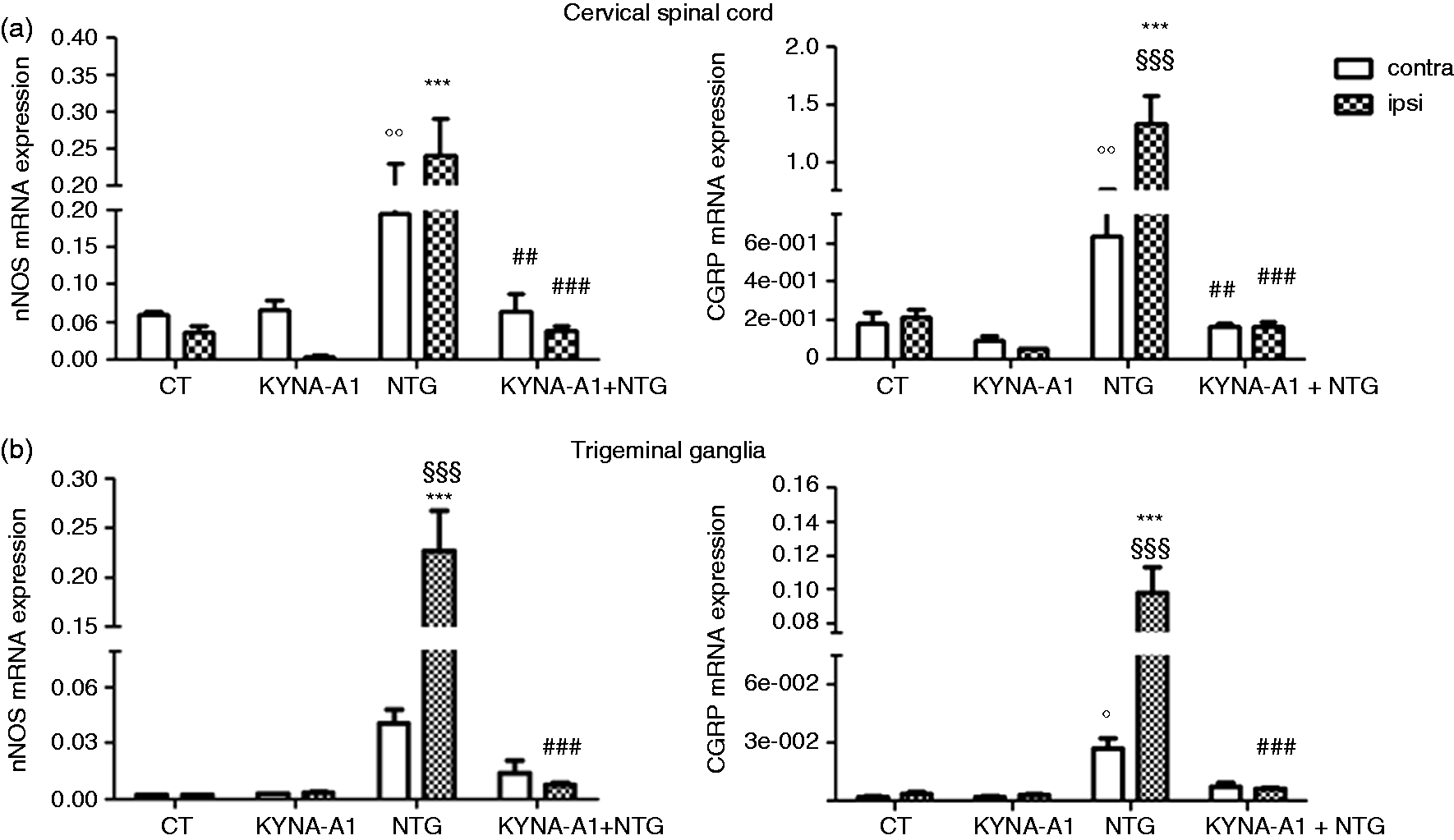
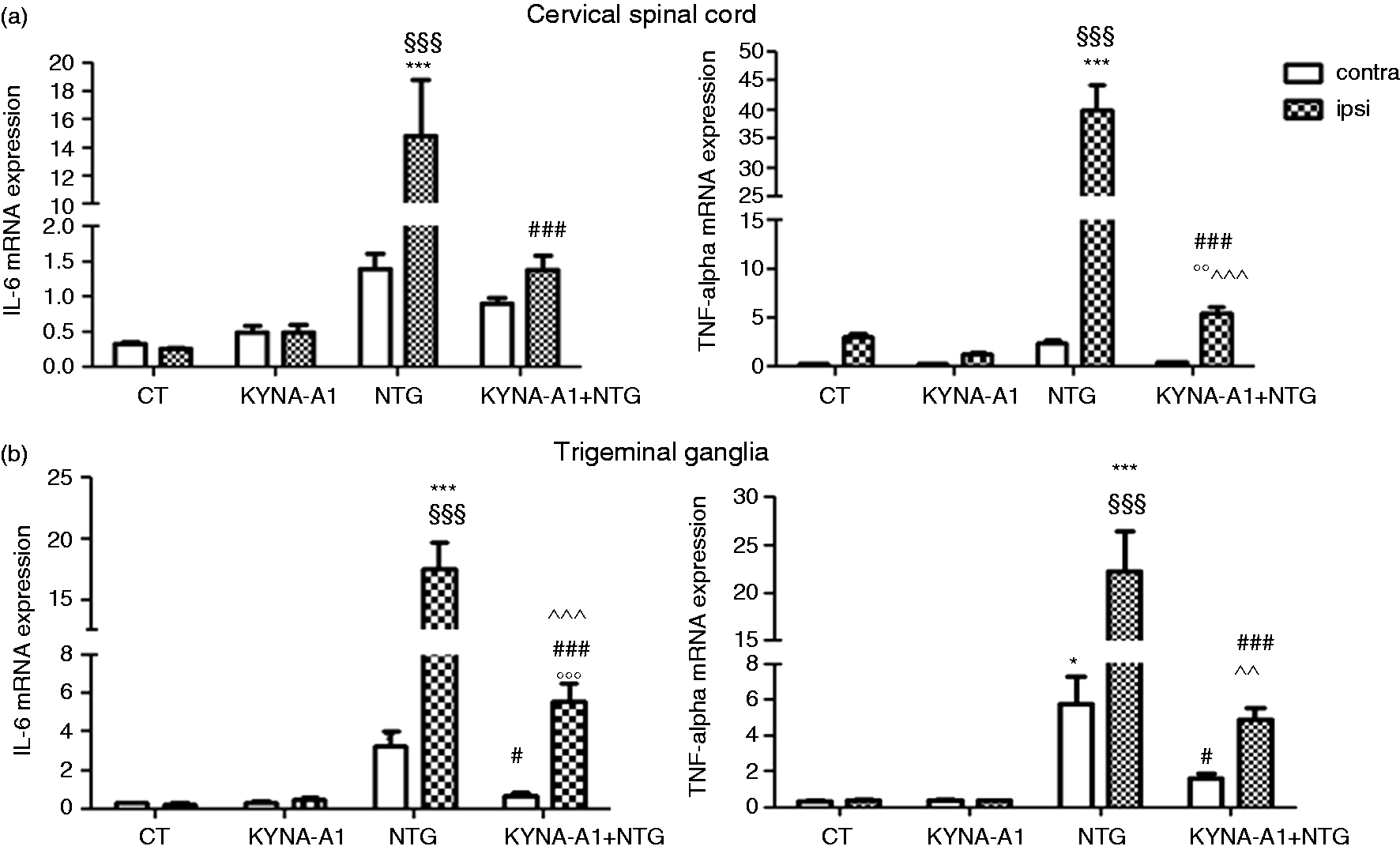
A significant increase in IL-6 mRNA and TNF-alpha expression was observed in the CSC only on the formalin-injected side of the NTG group. KYNA-A1 administration inhibited this increase. Cytokines mRNA expression increased on both TGs of NTG-injected rats. KYNA-A1 administration significantly inhibited the observed formalin-induced increase in the NTG group. Data are shown in Figures 4b and 5b.
Protein expression of CGRP and nNOS in the NTC during orofacial formalin test
No difference was found in the OD of CGRP fibers between both sides of the NTC following formalin injection in all experimental groups. NTG administration did not induce any significant changes in CGRP staining in the NTC compared to the CT group.
nNOS-positive cells and fibers were visible on both sides of NTC in all experimental groups. No significant difference in nNOS-immunopositive cells between the two sides was reported one hour after formalin injection. NTG administration induced a significant increase in nNOS protein expression on both sides of the NTC compared to the CT group. This increase was partially but significantly attenuated by KYNA-Al pretreatment. Data are shown in Figure 6.
Expression of CGRP and nNOS in both sides of the nucleus trigeminalis caudalis (NTC) following orofacial formalin injection. The histograms illustrate optical density (OD) of the CGRP immunoreactive fibers (a) and the number of nNOS positive nuclei (b) within the bilateral NTC. NTG injection induced a significant increase in nNOS protein expression in both sides of the NTC compared to the CT group, while no difference was found in the OD of CGRP fibers. KYNA-A1 pretreatment prevented the NTG-induced increase in nNOS positive cells in the NTC. No significant difference in nNOS-immunopositive cells/CGRP staining was reported between the two sides in any of the experimental groups. Data are expressed as mean ± SEM, two-way analysis of variance (ANOVA) followed by Bonferroni post-hoc test, *p < 0.05 versus the CT group; #p < 0.05 versus the NTG group.
Discussion
Several lines of evidence suggest the presence of a state of central and peripheral sensitization in migraine subjects. Allodynia and hyperalgesia, the clinical correlate of sensitization, are related to activation of NMDA receptors in the dorsal horn of the spinal cord (19). Activation of these receptors, which increase calcium conductance, leads to protein kinase activation, the release of serotonin, bradykinin and prostaglandins, NOS activation and, consequently, NO synthesis.
In this study, we used the model of NTG-induced hyperalgesia at the spinal and trigeminal level (5,6) to investigate the effects of KYNA-A1. Additionally, we investigated the targets and mediators modulated by the compound in the peripheral and central areas involved in trigeminal nociception.
Characterization of nitroglycerin induced-hyperalgesia at the trigeminal level
NTG-induced hyperalgesia in the orofacial formalin test was associated with an increased nNOS and CGRP mRNA expression in central areas containing the NTC. In the NTC, we detected an increase in nNOS immunoreactivity and a tendency toward a decrease in the staining of CGRP fibers. These findings are in agreement and expand on previous studies (18) to suggest that NTG administration induces a rapid release of CGRP from fibers originating in the NTC, which, at four hours, is already recovering as a consequence of the compensating activation of the Calca gene (12,18). This is in agreement with other studies that showed an increase in CGRP release from the TGs neurons during neurogenic inflammation or after NO donor treatment with activation of Calca gene transcription (20,21). Also at the peripheral level, in the TGs we observed an increase in nNOS and CGRP mRNA, but this was more marked on the formalin-injected side, suggesting that the phenomenon was related to both types of stimuli used in our model, NTG and formalin, but more to the latter. These findings show that systemic NTG can induce transcriptional changes in nNOS and CGRP expression, important mediators in the initiation and maintenance of sensitization (22), and this induction takes place at both peripheral and central areas. These data are in agreement with previous data that demonstrated an up-regulation of CGRP and nNOS immunoreactivity in primary trigeminal afferents of rats (23), and an increase in NOS-positive second order neurons (24). Peripheral effects of NTG infusion were also seen as a time-dependent increase in CGRP and nNOS expression in the dura mater (25). At variance, Eberhardt et al. (26) showed that NTG infusion did not change CGRP mRNA levels in the TGs. It seems likely that these conflicting results are related to the different doses of NTG used by the various groups, the highest dose (10 mg/Kg, i.p.) probably being more likely to have transcriptional effects, as suggested by the activation of CamKIIα in the NTC of rat (13).
Cytokines are involved in the development and maintenance of sensitization of peripheral nociceptors and second order nociceptive neurons. A correlation between tissue levels of cytokines and hyperalgesia has been observed in several painful states (27). In particular, a significant increase in pro-inflammatory cytokines levels was found in the TGs and in spinal trigeminal nuclei after capsaicin injection in the eyebrow regions (28), thus confirming a role for inflammation in migraine pathogenesis. Here, we report a significant increase of IL-6 and TNF-alpha mRNA expression following NTG administration in the CSC ipsilateral to the formalin injection, while cytokines gene expression significantly increased in both TGs, though more markedly on the formalin injected side. This suggests a contribution of NTG in the amplification of the formalin-induced nociceptive response, via additional release of cytokines (30). Probably, NTG-induced nNOS expression (18,22,31) in turn could induce the expression of pro-inflammatory cytokines (32), thus prompting glial and neuronal responses to sustained trigeminal system activation (33).
Characterization of the KYNA-A1 effect upon NTG-induced hyperalgesia in the orofacial formalin test
KYNA-A1 pretreatment inhibits NTG-induced hyperalgesia in the hindpaw, but more importantly for migraine, also in the trigeminal area. The findings obtained suggest that the state of hyperalgesia induced by NTG, likely related to the increased availability of NO at the trigeminovascular level or at central sites (i.e. the NTC) (31), is to some extent mediated by the activation of glutamate receptors. Previous studies suggested that KYNA amides exert their effect, similarly to KYNA (34), via an interaction with glutamate activity, although no direct evidence was reported. However, one possibility is that KYNA amides can dissociate into KYNA (35), which is known to block the glycine site of NMDA receptors (26). Though in previous studies MK801 has shown an anti-nociceptive effect on phase II of the plantar formalin test, in this study KYNA-A1 showed a significant anti-hyperalgesic effect in the same phase only in the presence of NTG. Given the known anti-nociceptive effects of NMDA antagonists, the absence of the effect of KYNA on formalin-induced behavior in rats not pre-treated with NTG was somewhat surprising. Although KYNA-A1 has been extensively studied, its exact mechanisms of action have not yet been fully clarified. KYNA-A1 is considered a prodrug that produces KYNA, an intermediate in the tryptophan catabolic pathway that exerts pharmacological actions as an NMDA receptor antagonist. One possible explanation is that NTG may modulate the activity of kynurenine aminotransferase II (KAT II) (36), the enzyme that converts L-Kinurenine to KYNA, to induce alterations in central KYNA and thus glutamate levels. A decrease in KAT II activity has indeed been associated with increased cerebral glutamate levels and thus NMDA activation (37). In addition, the increased NMDA activation could interfere with opioid receptor function (38). Anatomical studies, ultrastructural analyses, and electrophysiological data have indeed suggested interactions between opioid receptors and NMDA receptors. Thus, it is also possible that the anti-hyperalgesic effects of KYNA-A1 are related to modulation of the opiod receptor function (39), which could be altered after NTG administration, probably via an increased nNOS activity (40).
A role for NMDA receptors in central sensitization has been suggested and linked to nNOS activation. Administration of NMDA agonists to the paw of rats causes thermal and mechanical hyperalgesia, which is blocked by nNOS inhibitors (41). Conversely, spinal cord injury-induced nNOS expression is attenuated following NMDA antagonist treatment (42). At the trigeminal level, the effect of KYNA-A1 upon NTG-induced hyperalgesia is associated with the modulation of the transcriptional activation of genes related to the synthesis of CGRP, nNOS and pro-inflammatory cytokines in TGs and in central areas. KYNA-A1 also prevented the increase in nNOS proteins in the NTC observed following NTG administration. These observations suggest that the KYNA-A1 effect may take place both at peripheral and central levels. A role for glutamate in sensory modulation at peripheral levels has been suggested by Kung et al. (43), although Eftekhari et al. (44) have demonstrated that, in rat TG, neurons expressing CGRP do not contain glutamate as a cotransmitter. It is noteworthy that Fos-positive neurons in the NTC co-localize especially with the NMDA receptor and with nNOS, confirming that NMDA receptor activation in response to trigeminal pain may mediate neuronal activation involved in nociception. Reciprocal interactions between CGRP and NO/NOS have been demonstrated in previous studies (45), and a co-expression of CGRP and nNOS was found in the majority of NOS positive neurons in the TG (23). It seems therefore possible that KYNA-A1 may act primarily via the interaction with nNOS expression, which in turn influences sensory fibers’ activation and the availability of mediators such as CGRP.
KYNA-A1 administration also proved able to reduce mRNA expression of cytokines. This effect likely related to the inhibition of nNOS expression at central and peripheral levels, and thus to the reduction in CGRP release. In agreement with this interpretation, it has been shown that cytokines may be modulated in trigeminal pain by nNOS under inflammatory conditions (46). Inhibition of nNOS induced by spinal cord injury was associated with a marked reduction of neutrophil infiltration, NF-κB activation and cytokine levels following MK801 pre-treatment (42).
Collateral findings
It is noteworthy that in baseline condition, formalin injection per se significantly increased mRNA expression of all the investigated genes in all areas under evaluation compared to orofacial saline injection (data not shown). Tissue injury causes the release of various inflammatory molecules, from the damaged endothelial and inflammatory cells that activate primary sensory neurons to produce persistent pain. These peripheral changes in turn induce the release of additional mediators in the spinal cord, such as NO and cytokines (47). NO may induce calcium influx – possibly via membrane depolarization or cGMP-dependent calcium channels – activate multiple protein kinases and promote CREB activation, which is implicated in the transcriptional regulation of several genes. The primary signaling pathway involved in the initiation and amplification of inflammatory responses is the one that leads to activation of nuclear factor (NF)-kB transcription factors, which represents a critical regulatory mechanism of nNOS and cytokine expression. During inflammatory responses, NF-kB cooperates with the mitogen-activated protein kinases (MAPKs). MAPKs are important for intracellular signal transduction and contribute to pain sensitization through distinct molecular and cellular mechanisms (48). This complex set of mediators likely represents the framework on which NTG impacts to further amplify nociceptive responses in our model.
Conclusions
Our study shows that KYNA-A1, a modulator of glutamate activity, counteracts NTG-induced trigeminal hyperalgesia at peripheral and central levels. This effect is likely mediated by a primary action on nNOS expression and the consequent modulation of CGRP and cytokine release at peripheral and central sites. KYNA-A1 therefore qualifies as a potential therapeutic option in migraine treatment.
Article highlights
KYNA-A1 pretreatment abolished NTG-induced hyperalgesia in the trigeminal area in an animal model of migraine. NTG-induced hyperalgesia in the trigeminal area was associated with increased transcriptional activity of CGRP, nNOS and cytokines in the trigeminal ganglia, cervical spinal cord and medulla-pons, which was blocked by KYNA-A1 injection. KYNA-A1 is a potential therapeutic target for migraine treatment.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by a grant from the Italian Ministry of Health (Ricerca Corrente 2015) to the IRCCS C. Mondino (C.T.)
