Abstract
Background
Visual aura is present in about one-third of migraine patients and triggering by bright or flickering lights is frequently reported.
Method
Using migraine with visual aura patients, we investigated the neurochemical profile of the visual cortex using magnetic resonance spectroscopy. Specifically, glutamate/creatine and GABA/creatine ratios were quantified in the occipital cortex of female migraine patients.
Results
GABA levels in the occipital cortex of migraine patients were lower than that of controls. Glutamate levels in migraine patients, but not controls, correlated with the blood-oxygenation-level-dependent (BOLD) signal in the primary visual cortex during visual stimulation.
Conclusion
Migraine with visual aura appears to disrupt the excitation-inhibition coupling in the occipital cortex.
Introduction
Migraine is intimately linked to the visual system, associated with visual auras, photophobia and visual triggering of attacks. Halogen bulbs, flicker or bright sunlight are well-described triggers for migraine attacks in addition to more standard triggers such as tiredness or stress (1). A recent study has suggested that visual stimuli may not be a reliable migraine provocant, but may be dependent on the nature of the stimulus used and the state of the patient (2). The vulnerability of the visual system in patients with migraine may reflect deficits in excitatory-inhibitory balance, with several studies of rare genetic forms of migraine affecting both glutamatergic and gamma-aminobutyric acid (GABA)-ergic synapses (3). Disturbances in excitatory-inhibitory balance may also predispose the cortex to cortical spreading depression (CSD), a phenomenon thought to underlie migraine aura (4).
Proton magnetic resonance spectroscopy (1H-MRS) can be used to quantify a number of neurochemicals within a specific region of the brain, and current MRS protocols permit the detection of GABA and glutamate peaks in addition to metabolites that are found in higher concentrations (5). No previous studies have succeeded in quantifying GABA levels in migraine patients, but studies have suggested that there may be abnormalities in glutamatergic cycling. Prescot et al. (2009) (6), using linear discriminant analysis, suggested a different pattern of N-acetyl-aspartate (NAA) and glutamine concentrations in migraine patients compared to controls. In a second study, Siniatchkin et al. (2012) (7) showed that young migraine with aura patients had significantly higher glutamate + glutamine (Glx)/creatine (Cr) ratios than healthy individuals. Furthermore, while application of excitatory (anodal) transcranial direct current stimulation (tDCS) to the visual cortex in healthy individuals caused an increase in Glx/Cr ratio and inhibitory (cathodal) stimulation caused a decrease, both types of tDCS caused a decrease in Glx/Cr ratio in migraine patients. Finally, a recent paper by Gonzaález de la Aleja (8) showed an increase in the ratio of occipital glutamate:glutamine. Taken together these findings suggest disruption of normal glutamate turnover and function without changes in glutamate levels.
The comprehensive review by Reyngoudt et al. (9) highlights the lack of studies that recruited homogeneous patient types with control groups matched for age and gender, meaning that MRS results to date are difficult to interpret. Here, we scanned female patients with migraine with visual aura (vMA) and age- and gender-matched controls using an MRS sequence that allowed accurate quantification of GABA, glutamate and glutamine.
Methods
Twenty-six females were recruited to the study conducted under ethics approval from the Staffordshire NHS Research Ethics Committee (11/WM/0088), and all participants provided informed written consent. Thirteen individuals (mean age 33 ± 8 years) with vMA (citing visual stimuli as potent migraine triggers) were recruited via clinics in Oxford, personal contacts and via an advertisement placed on the “Migraine Trust” website. All potential participants were clinically assessed by a migraine clinician (MZC) to ensure that they met the eligibility criteria for the study: revised International Headache Society criteria for migraine with aura; predominant aura was visual; and visual stimulation a strong self-reported trigger for migraine episodes.
Thirteen female controls, with no history of migraine or other primary headache disorder, were recruited via email advertisement and through personal contacts (mean age 30 ± 6 years). The migraine characteristics of the patients are shown in SI Table 1.
A single imaging session was performed at the Oxford Centre for Functional Magnetic Resonance Imaging of the Brain (FMRIB) using a Siemens 3 T Verio scanner with a 32-channel head coil. The scan room was dark and participants wore light-diffusing goggles. The first scan was a whole-head structural scan (magnetization prepared rapid gradient echo, repetition time (TR) = 11.2 ms, echo time (TE) = 4.68 ms, resolution 1 × 1 × 1 mm3). MRS used a single voxel manually located to cover V1 bilaterally. The spin echo full intensity acquired localized spectroscopy (SPECIAL) sequence (10) was employed using the following standard parameters: volume of interest (VOI) = 20 × 20 × 20 mm3, TR/TE = 4000 ms/8.5 ms, number of averages = 128. The short echo-time single voxel acquisition enables simultaneous detection of many metabolites, including glutamate and GABA (10,11).
Standard pre-processing to correct for motion and frequency drift was applied using in-house scripts as described previously (12), and all spectra used for analysis had a linewidth <6 Hz (average 4.8 Hz; no group difference). Metabolite quantification was performed using LCModel (Provencher, 1993), and all metabolites were fitted with Cramer-Rao lower bounds of <20%. As is customary, all neurotransmitter levels are presented as a ratio to total creatine (Cr + PCr), a simultaneously acquired reference metabolite. For each participant the T1-weighted structural image was segmented using FAST (FMRIB’s automated segmentation tool) (13), and the relative gray matter, white matter and cerebrospinal fluid contributions to the MRS voxel was calculated and used to correct MRS values for gray matter volume (14).
Blood-oxygenation-level-dependent (BOLD) activity in the primary visual cortex was quantified in two functional magnetic resonance imaging (fMRI) experiments. In experiment 1, participants were presented with diffuse light for 120 seconds followed by 60 seconds in the dark. This was repeated three times, and “light onset” was defined as the first 30 seconds of diffuse light in each repeat. In experiment 2 participants were presented with 15-second blocks of either “high-” (20% and 50%) or “low-” (2% and 5%) contrast flickering checkerboard stimuli or a uniform gray background with a fixation cross. Participants were explicitly asked to keep their eyes open for the duration of both fMRI experiments. Two controls were excluded from the checkerboard experiment, as the mirror was not correctly positioned.
Results
Spectra of sufficient quality to determine GABA and glutamate levels were obtained from 11 individuals in each group. In line with previous work, we did not find any difference in glutamate levels between patients and controls (Figure 1(c), t(21) = 0.85; Magnetic resonance spectroscopy (MRS) data were acquired from a voxel centered on the calcarine sulcus as shown in (a). An example spectrum with creatine, glutamate and NAA peaks indicated is shown in (b). Concentrations of glutamate and GABA normalized to creatine in both groups are shown in (c) and (d), respectively.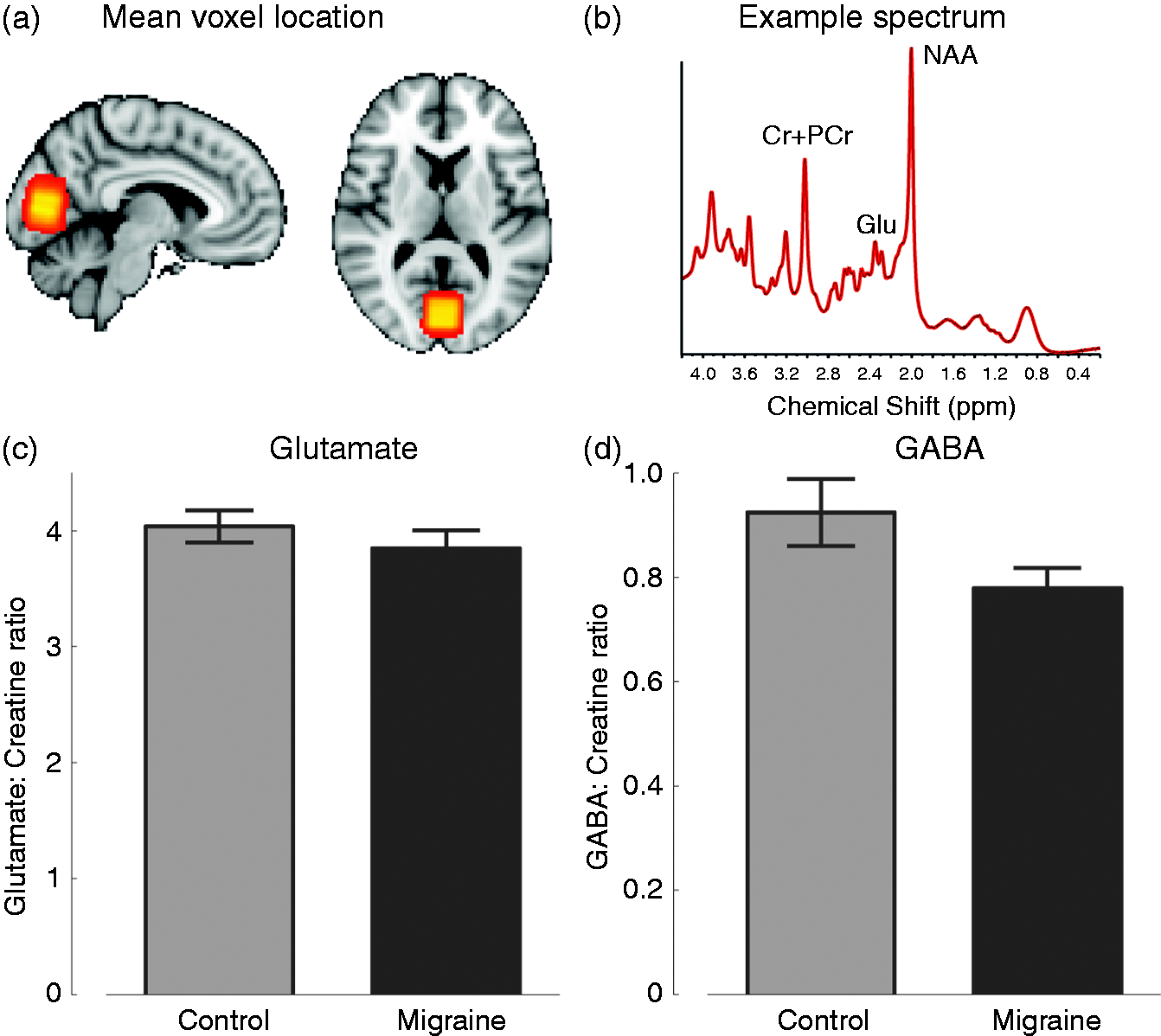
Occipital glutamate correlates with BOLD signal in migraine patients, but not controls
To determine whether there was any relationship to visual activation in the occipital cortex, BOLD responses to two different types of light stimulation were correlated to levels of glutamate and GABA. Figure 2(a) shows the relationship of glutamate level with “light onset,” which was not significant in migraine patients ( Image (a) shows the correlation between the response to the onset of diffuse, dim light in V1 and normalized occipital glutamate concentration. Filled black circles are migraine patients and open circles are control participants. The black line shows the least squares fit to the migraine patients and the gray line shows the fit to the controls. Images (b) and (c) show the correlations of glutamate with the V1 responses to low-contrast and high-contrast flickering checkerboards, respectively. In both cases there is a significant correlation for migraine patients, but not controls.
There was no correlation between BOLD signal and GABA concentration in either migraine or control participants in any visual condition. This remained the case even when glutamate concentration was partially correlated out of the calculation.
Discussion
This homogeneous population of vMA patients shows decreased GABA levels in the occipital cortex relative to matched controls. While resting GABA levels have been linked to orientation discrimination, gamma oscillation frequency and BOLD activation in the visual cortex of healthy controls (14,15), there was no such relationship in the control individuals in this study. This could be due either to small participant numbers or to a difference in sensitivity in GABA detection between the SPECIAL sequence and the Meshcher-Garwood Point Resolved Spectroscopy (MEGA-PRESS) used previously.
MRS-GABA has been suggested to be a measure of extra-synaptic tonic GABAergic activity (5), and therefore the reduced GABA levels observed here in migraineuses could most easily be interpreted as a reduction in local inhibition. This finding is consistent with several studies suggesting occipital hyper-excitability in migraine (16). However, we note that in our patients, BOLD responses to light onset and checkerboard stimulation were not increased relative to controls, suggesting any change in excitability may be subtle, complex and perhaps not homogenous across the occipital cortex.
Consistent with at least one previous study (8), we found no difference in glutamate levels between migraineurs and controls. However, we demonstrated a significant positive relationship between occipital glutamate levels and the BOLD response to visual stimulation in our migraine patients, a relationship that was not seen in the controls. This finding provides further support for the hypothesis of disrupted excitatory-inhibitory coupling in migraine (3). A close relationship between BOLD signal and glutamate levels has not previously been demonstrated in the visual cortex of migraine patients or controls, but has been shown in the anterior cingulate cortex in major depression (17).
Total glutamate, as assessed by MRS, reflects glutamate’s role as a metabolite as well as a neurotransmitter. Significant changes may occur in glutamatergic transmission in migraineurs, which drives the correlation between the BOLD signal and glutamate in this group, but is not visible using this technique. However, this interpretation remains speculative. Although a causal relationship remains to be established, our data suggest that, in addition to GABA, glutamate may be a key driver of occipital activation in migraine with aura patients. Owing to time constraints, we did not acquire data from an additional voxel in another cortical region, so it is not possible to determine whether the observed changes are confined to the occipital lobe or are more widespread. Nevertheless, disturbance of glutamate metabolism and neurotransmission is also supported by genetic studies (18). The extent to which our findings are aura specific or more generally migraine related will require further studies comparing migraine with aura and migraine without aura patients. This could also address the important question of whether altered neurochemical coupling is a feature of light sensitivity or visual aura.
In conclusion, a homogenous group of vMA patients show reduced GABA levels and enhanced glutamate-cortical activation coupling in the occipital cortex, which may predispose patients to visual auras and migraine triggering by visual stimuli. It is notable that topiramate, a first-line migraine prophylactic, is a glutamate receptor antagonist (19), enhances GABA activity (20) and increases brain GABA (21), although this is not the case for all migraine prophylactics. Future studies should investigate the causal relationship between these neurotransmitters and cortical activity in migraine through pharmacological manipulation of GABA and glutamate signaling. Nevertheless, the observed change in excitatory-inhibitory coupling is an important signature of the migraine brain and highlights therapeutic opportunities.
Key findings
Female patients with migraine with visual aura and visual sensitivity show decreased gamma-aminobutyric acid (GABA) in the occipital cortex. Occipital glutamate/creatine ratio correlates with blood-oxygenation-level-dependent (BOLD) activation levels in the primary visual cortex of migraine patients but not controls.
Footnotes
Funding
This work was funded by the Medical Research Council (UK), the Royal Society and The John Fell Fund.
Conflict of interest
None declared.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
