Abstract
Background
Preventive medication is indicated for many migraine patients, but is used in relatively few. The aim of the present study was to evaluate the efficacy of acetyl-l-carnitine as a prophylactic drug in migraine patients.
Methods
A single-center, randomized, triple-blind, placebo-controlled, crossover study was carried out. Men and women, age 18–65 years, with episodic migraine but otherwise healthy, were recruited mostly through advertisements. After a four-week run-in-phase, 72 participants were randomized to receive either placebo or 3 g acetyl-l-carnitine for 12 weeks. After a four-week washout, treatment was switched. The primary outcome was days with moderate or severe headache per four weeks. Secondary outcomes were days with headache, hours with headache, proportion of responders (>50% reduction in migraine days from baseline) and adverse events.
Results
In the complete case analyses, no statistically significant differences were found between acetyl-l-carnitine and placebo in severe or moderate headache days per month (3.0 versus 3.1, p = 0.80), headache days per month (5.1 versus 5.2, p = 0.73) or for the other secondary outcome measures.
Conclusion
In this triple-blind crossover study no differences were found in headache outcomes between acetyl-l-carnitine and placebo. Our results do not provide evidence of benefit for efficacy of acetyl-l-carnitine as prophylactic treatment for migraine.
Trial registration: EUDRACT (2012-001624-36), ClinicalTrials.gov (NCT01695317).
Introduction
Migraine is ranked seventh highest among specific causes of disability globally (1). Prophylactic medication is indicated for many migraine patients, but is used in relatively few, e.g. because of limited therapeutic effects, side effects or contraindications of existing drugs (2,3). There is thus an urgent need for new prophylactic drugs with few side effects that can be used by most migraine sufferers (3).
One substance that may fulfill these requirements is acetyl-l-carnitine, which is available as a dietary supplement, and which also occurs naturally in plants and animals. Acetyl-l-carnitine stimulates lipid-mediated energy production in the central nervous system (4), and has an impact on several neurotransmitter pathways, including serotonergic, noradrenergic and dopaminergic pathways important in migraine pathophysiology (5).
Over the last few decades, acetyl-l-carnitine has been evaluated in placebo-controlled trials for a large number of diseases (6–12), including neurological disorders such as Alzheimer’s dementia and amyotrophic lateral sclerosis (13–15). Several clinical studies have indicated analgesic effects of acetyl-l-carnitine in different types of painful neuropathies (16). Interestingly, acetyl-l-carnitine has been shown to be effective as a migraine prophylactic in one single-blind, randomized, parallel trial in children. Unfortunately, this study has been published only in an abstract (17).
The aim of the present study was to evaluate the efficacy of acetyl-l-carnitine as a prophylactic drug in adult migraine patients.
Materials and methods
The study was designed as a placebo-controlled, triple-blind, crossover trial, with a four-week run-in-phase (baseline period), and two 12-week treatment periods with a four-week washout period in between. It was conducted at the neurological outpatient clinic of St. Olav’s University Hospital between April 2013 and July 2014. Participants were recruited either from patients referred to the clinic or among those who contacted the study nurse in response to advertisements in newspapers or on websites.
The study followed the International Headache Society (IHS) guidelines for trials on prophylactic medication in migraine (18,19). The primary efficacy variable was number of days with moderate or severe headache, lasting ≥ 4 hours or being treated with the patient’s usual medication (“migraine days”) per four weeks (19). Secondary measures were: days with headache; hours with headache; headache intensity on days with headache; doses of analgesics; doses of triptans; number of days of sick leave; number of responders (≥ 50% decrease in migraine days compared with baseline) (18). Adverse event (AE) measures were the occurrence of AEs (yes/no), the level of discomfort due to AEs (scale 1–5), and the number of AEs occurring during treatment periods.
Inclusion criteria were: age 18–65 years; signed informed consent; migraine with or without aura (18) or chronic migraine (19); ≥ 2 migraine attacks per month during the last three months before inclusion, and ≥ 2 migraine attacks during the four-week baseline period documented in the diary, debut of migraine ≥ 1 year prior to inclusion, and before the age of 50; body mass index (BMI) between 18 kg/m2 and 35 kg/m2, and no medication-overuse headache during the last three months, defined as headache >14 days/month combined with overuse of simple analgesics in monotherapy >14 days/month or triptans or combined medications ≥ 10 days/month.
Exclusion criteria were: headache other than migraine not distinguishable from the migraine attacks; chronic tension-type headache or other headache occurring on ≥ 15 days/month; pregnancy, nursing or no use of contraceptives in fertile women; history of angioneurotic edema; diabetes or previous or present history of asthma or vascular disease, including arterial claudication; decreased hepatic or renal function; psychiatric illness and/or Hospital Anxiety and Depression Scale (HADS) anxiety score ≥ 11 or HADS depression score ≥ 11; use of daily migraine prophylactics within four weeks prior to start of study; use of ≥ 3 prophylactic drugs against migraine during the last five years; consistent failure to respond to any acute migraine medication; alcohol or illicit drug dependence; previous use of acetyl-l-carnitine; BMI <18 kg/m2 or > 35 kg/m2; and individuals requiring detoxification from acute medication.
During a screening examination by a neurologist, a thorough history was taken to ascertain compliance with inclusion and exclusion criteria. All participants received a neurological examination that also included measurements of height, weight, blood pressure (BP) and pulse. Throughout the whole study, patients were instructed to keep a headache diary recording relevant attack variables, as well as AEs or other new health-related conditions. At inclusion and all follow-up visits, the patients were asked to fill in the HADS questionnaire.
The four-week baseline period without prophylactic medication before randomization was used to ascertain whether patients had ≥ 2 attacks per month. If not, the baseline period was extended by four weeks, and if there were ≥ 4 attacks during the whole eight-week period, the patient was included. After the baseline period, there was a further visit to evaluate whether the patient could be included, before randomization and dispensing of the study drug for the first period.
Seventy-two patients were randomized in blocks of 18 to one of two possible treatment sequences. The study drug consisted of tablets of either acetyl-l-carnitine 500 mg or placebo. Because the optimal daily doses for investigating acetyl-l-carnitine for adult migraine patients were unknown, the selection of daily dose in the present study was based on a review of double-blind, randomized trials evaluating acetyl-l-carnitine versus placebo in the treatment of mild cognitive impairment and mild Alzheimer’s disease (varied between 1.5 g and 3 g/day) (20). In week 1, the patient took three tablets each day (1.5 g), and during weeks 2–12, six tablets (3 g). The active drug and placebo were prepared by an external producer (Kragerø Tablettproduksjon AS). The placebo tablets (500 mg dextrose and citric acid) were made with a similarly acidic taste to that of active drug and were packaged in identical bottles in order to ensure a blinded design.
After two weeks of each treatment period, all patients were contacted by telephone by the study nurse to check for AEs and compliance with the protocol (Figure 1). If there were AEs after increasing the dose, the dose could be reduced to three tablets per day for the rest of the period. A few days after finishing the first treatment period of 12 weeks, the patients visited the doctor for evaluation (Figure 1). Adherence was checked again, and study drugs for the next period were also handed out. Approximately one week after the second treatment period, there was a final visit (Figure 1).
Study design.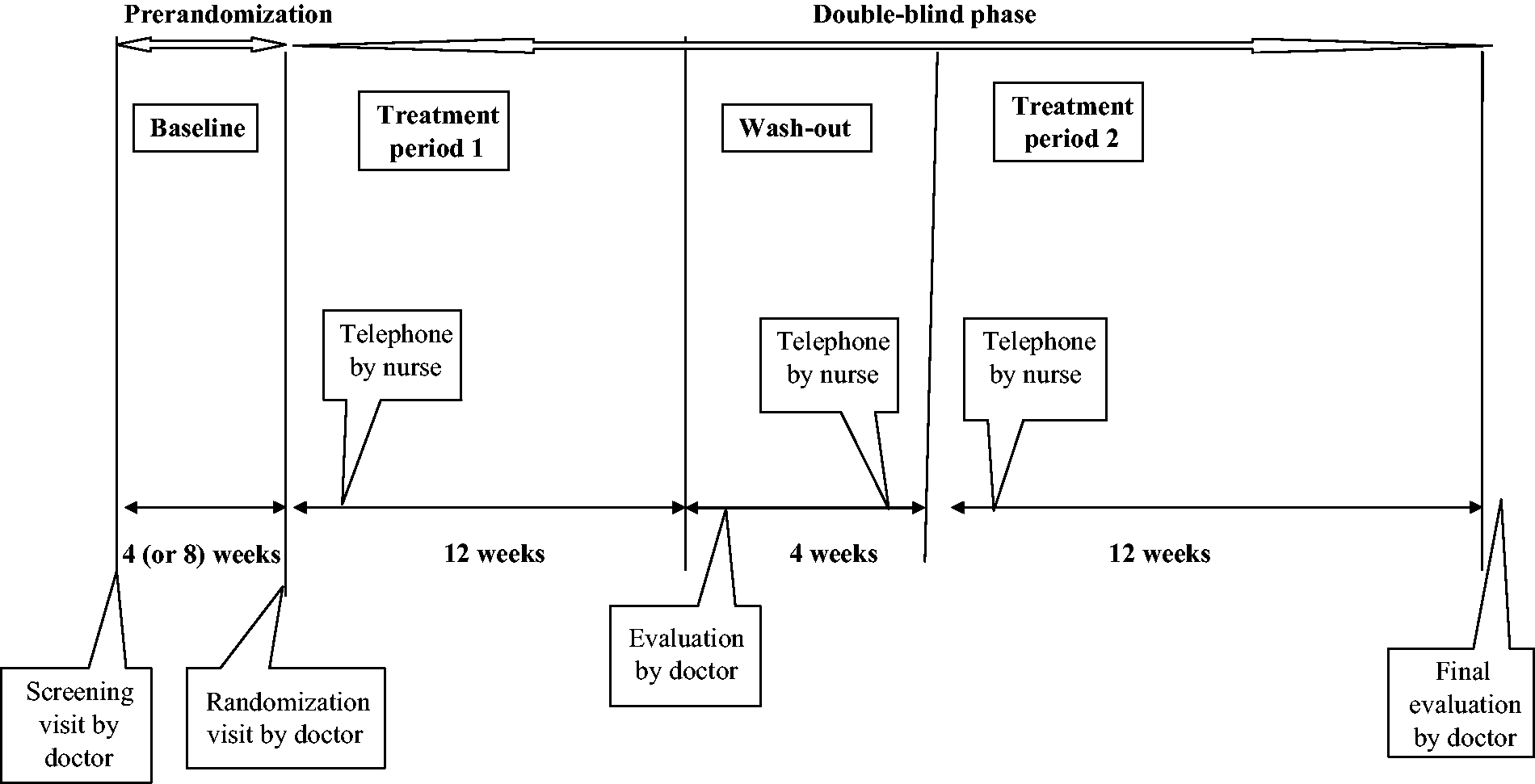
AEs were recorded in the headache diary (free text, one line for each day), and enquired about during telephone interviews and visits using an open-ended question about side effects and other health-related complaints.
Adherence (completion per protocol; PP) was defined as having taken the tablets ≥ 8 weeks in both periods, and was measured using self-reports and pill counts. For individuals to be included in the complete case (CC) analyses, drugs had to be used ≥ 4 weeks in each period. The remaining participants were defined as dropouts. Intention to treat (ITT) analyses (not specified in the protocol) were also performed. AEs were reported in all patients who had taken at least one tablet.
The study was approved by the Regional Committee for Ethics in Medical Research (REK-Midt) (from which the full trial protocol can be accessed on request: rek-midt@medisin.ntnu.no), the Norwegian Data Inspectorate, and the Norwegian Medicines Agency.
Randomization, blinding and data handling
Participants fulfilling criteria for randomization were consecutively given a randomization number (1–72) assigning them to one of the two treatment sequences (Figure 2) according to a computer-generated list, premade by the company producing the drugs, but unknown to participants, clinicians and the statistician. Randomization numbers were preprinted on study medication labels, and information about the medication in each period for each participant was placed in a sealed envelope. Hence, in the event of serious AEs (SAEs), it was possible to unblind. The envelope was kept in a limited-access area.
Flow of participants through the study (figures refer to number of patients).
Data entry was performed by the study nurse. After completion of the study, the data file, together with the unopened envelopes containing randomization codes, were handed over to personnel at the Unit for Applied Clinical Research at the Norwegian University of Science and Technology who decoded the data and returned the file with each treatment type having a code (D or S). A predetermined statistical protocol had been written for the analysis of the primary and secondary efficacy variables, and the statistician (TS) performed analysis of this file without knowing the treatment type. This was not revealed until final tables with coded efficacy data had been completed (triple-blind study). A few data entry errors were detected during the blinded analysis, and these were corrected before the final unblinding.
The study was monitored by personnel at the Unit for Applied Clinical Research with experience and formal training in monitoring clinical trials.
Statistical methods
The predetermined hypotheses were: H1: Acetyl-l-carnitine is better than placebo (superiority analysis). H2: Placebo has fewer AEs than acetyl-l-carnitine (superiority analysis).
A power analysis before study start indicated that 60 evaluable patients would give >80% power to detect a difference between acetyl-l-carnitine and placebo of 0.5 SD (approximately 1.3–1.7 headache days/month based on our previous data) for the main efficacy variable (3,21). Expecting a dropout rate of 15%–20%, 72 patients would have to be included.
All statistical tests were between treatment periods, and did not include baseline data (except for the analyses of responder rate). In accordance with the predetermined statistical protocol, H1 was tested with Wilcoxon’s paired signed rank test in the CC group. A secondary analysis was performed in the PP group. In order to perform the ITT analyses for the main outcome, cases with no data or insufficient data for period 2 were also imputed with period 1 data, if available, or baseline data. A sensitivity analysis for number of responders, replacing missing observations with either “non-responder” or “responder” status, was also performed.
An AE reported several times during one treatment period was counted only once.
The differences in responder rate and AE proportions were tested with a standardized normal deviate.
Analysis of period effects was not included in the protocol since a long washout was used to minimize any crossover effects and the study was not powered to provide precise estimates of periodicity. However, a post-hoc period analysis was performed with repeated-measures analysis of variance (ANOVA) for imputed ITT and CC data sets, while mixed-model ANOVA was used for complete non-imputed data.
SYSTAT v11 (Systat Software Inc, Chicago, IL, USA) was used with two-sided level of significance <0.05.
Results
Of the 72 patients randomized to treatment, one was recruited at the outpatient clinic while the remaining patients contacted the study nurse in response to advertisements. A patient flow diagram is shown in Figure 2.
Background variables of the whole group, the complete case (CC) population, per protocol (PP) population, and dropouts (DO).

Patients who dropped out of the study (n = 8), were partial completers (n = 3), or who reduced dose in one period (n = 3).
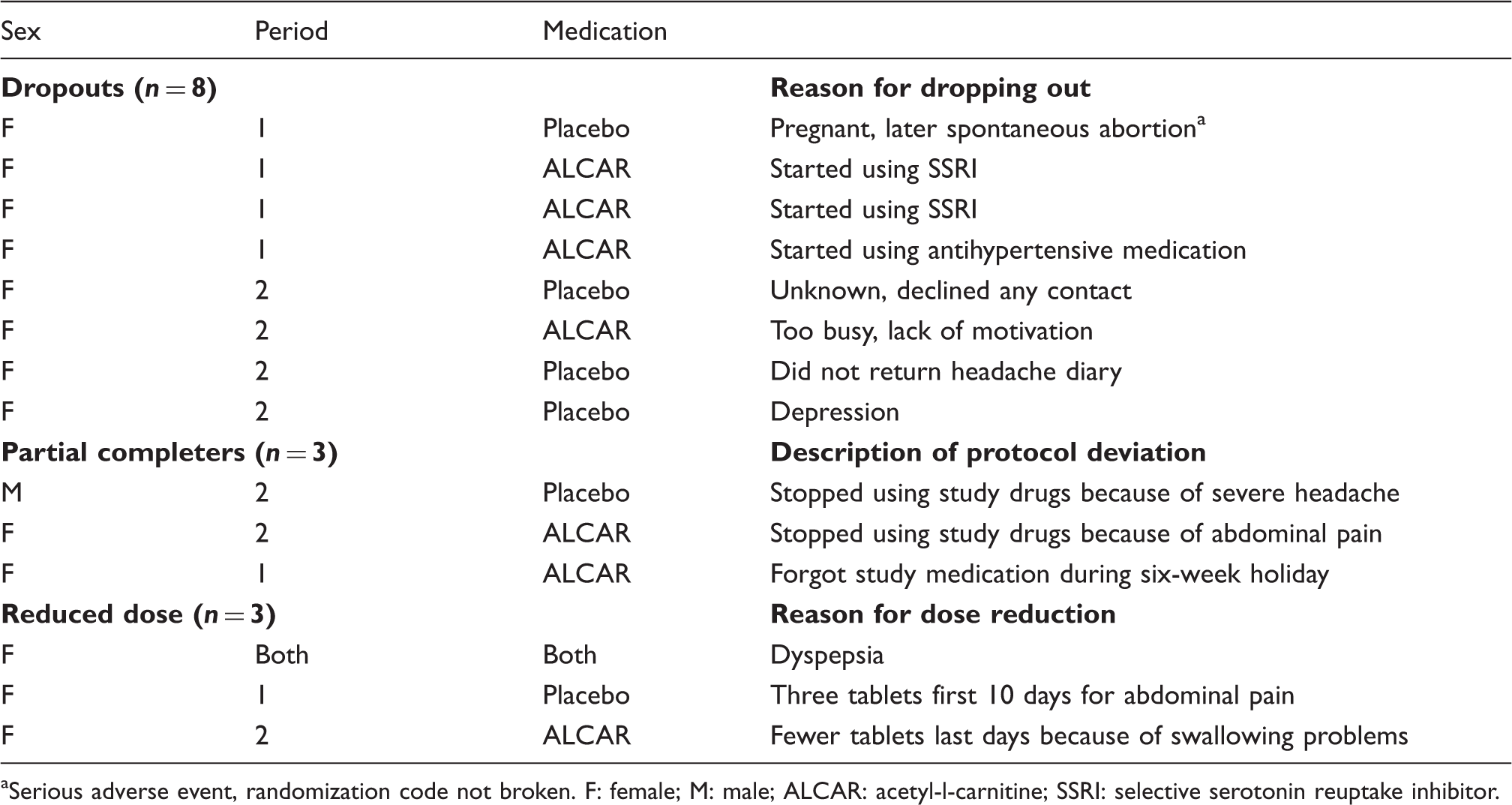
Serious adverse event, randomization code not broken. F: female; M: male; ALCAR: acetyl-l-carnitine; SSRI: selective serotonin reuptake inhibitor.
Diary-based efficacy variables and adverse event in different periods, complete case (CC) analysis.
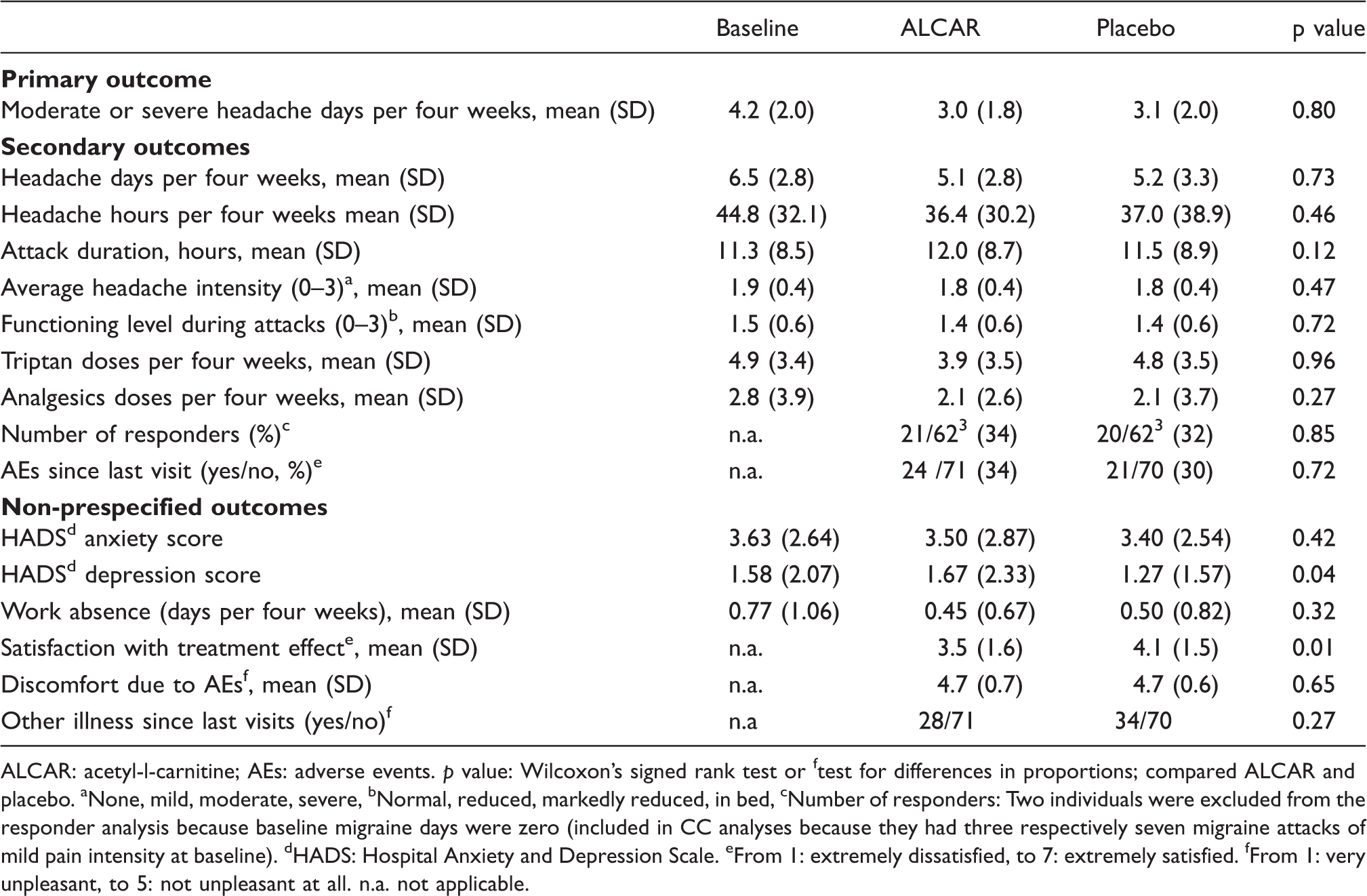
ALCAR: acetyl-l-carnitine; AEs: adverse events. p value: Wilcoxon’s signed rank test or ftest for differences in proportions; compared ALCAR and placebo. aNone, mild, moderate, severe, bNormal, reduced, markedly reduced, in bed, cNumber of responders: Two individuals were excluded from the responder analysis because baseline migraine days were zero (included in CC analyses because they had three respectively seven migraine attacks of mild pain intensity at baseline). dHADS: Hospital Anxiety and Depression Scale. eFrom 1: extremely dissatisfied, to 7: extremely satisfied. fFrom 1: very unpleasant, to 5: not unpleasant at all. n.a. not applicable.
Adverse events (AEs) (number of patients reporting the AE at least once during the treatment period).

ALCAR: acetyl-l-carnitine.
Post-hoc repeated-measures ANOVA for the main outcome variable showed no significant period effect for ITT (F = 0.66, p = 0.42) or CC analysis (F = 0.11, p = 0.73) and no significant drug × period interaction (ITT: F = 0.05, p = 0.82 and CC: F = 0.06, p = 0.81). ANOVA cell means showed no trends toward periodicity or non-homogeneity (Levene test p = 0.82 and 0.56, respectively). Also, the mixed-model ANOVA for the complete non-imputed dataset (n = 72) did not add any new information (drug F = 0.026, p = 0.87; period F = 1.04, p = 0.31 and drug × period F = 0.02, p = 0.88).
Discussion
In this triple-blind, crossover study, acetyl-l-carnitine was not superior to placebo in the prevention of migraine. Hence, our results do not provide evidence for efficacy of acetyl-l-carnitine in migraine prophylaxis.
Strengths of this study include its relatively strict accordance with IHS guidelines for migraine prophylaxis studies (18,19), as well as the use of predetermined hypotheses and statistical methods, and blinded statistical evaluation.
The crossover design is accepted as a valid study design in the IHS Guidelines (18), although it has some inherent limitations. The present study lacked sufficient power to estimate potential carry-over and period effects. However, these were probably minimized and largely eliminated by the use of four-week washout periods and balanced crossovers.
One possible limitation of the study is that only a single dosage was examined. An optimal daily dose of acetyl-l-carnitine has not been established (20). We cannot rule out the possibility that doses higher than 3 g per day may have had an effect on migraine. The fact that there were few side effects in the present study suggest that doses higher than 3 g might have been tolerated by most of the patients.
Another potential problem with crossover studies is high dropout and low adherence to protocol, particularly in studies like the present one, which lasted almost eight months for each patient. However, in this study, the dropout rate was relatively low (11%) and the proportion of PP completers high (85%), probably because of close follow-up by the study nurse.
The absence of any beneficial effect of acetyl-l-carnitine may possibly be explained by a relatively low frequency of headaches among the patients included. The mean number of days with moderate or severe headache per month in the screening visit was lower in the present study than in our previous study on candesartan and propranolol for migraine prophylaxis (3.6 versus 4.8) (3).
Another possible limitation of the study is that information about randomization was kept in opaque sealed envelopes in a limited-access area. This method may in principle imply a risk of subverting randomization (22). However, because SAEs did not occur, we did not need to unblind.
It should be emphasized that 88% of the patients were women. As a consequence, generalization of the study results to men should be performed with caution.
No difference was found between acetyl-l-carnitine and placebo for the main headache outcome measures and for most secondary measures, with the exception of the non-prespecified outcome that placebo was significantly better than acetyl-l-carnitine regarding satisfaction with treatment effect. We note that the placebo group also showed a small but significant difference in the non-prespecified outcome “HADS depression score” compared to the acetyl-l-carnitine group, indicating that placebo improved mood. In contrast, several randomized clinical studies have demonstrated efficacy of acetyl-l-carnitine in patients with depression (23,24). This contrasting finding may illustrate the importance of a blinded statistical evaluation.
In conclusion, this triple-blind crossover study did not provide evidence for efficacy of acetyl-l-carnitine as a prophylactic treatment for migraine.
Key findings
We conducted a single-center, randomized, triple-blind, placebo-controlled, crossover study. It was determined that 3 g of acetyl-l-carnitine was not superior to placebo in the prevention of migraine. The study did not provide evidence for efficacy of acetyl-l-carnitine as a prophylactic treatment for migraine.
Author contributions
Knut Hagen and Trond Sand had full access to all data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Study concept and design: Hagen, Stovner, Sand
Acquisition of data: Hagen, Stovner, Linde, Tronvik, Brenner, Gravdahl
Statistical analysis: Sand
Drafting of the manuscript: Hagen, Sand, Stovner, Linde, Tronvik, Brenner, Engstrøm, Gravdahl, Sonnewald, Helde
Funding
This work was supported partly by the Department of Neuroscience, NTNU, and with research funding from NTNU and St. Olav’s Hospital.
Conflicts of interest
Knut Hagen has nothing to declare.
Lars Jacob Stovner has received speaker’s fees from GlaxoSmithKline.
Mattias Linde has nothing to declare.
Eiliv Brenner has nothing to declare.
Gøril Bruvik Gravdahl has nothing to declare.
Morten Engstrøm has nothing to declare.
Ursula Sonnewald has nothing to declare.
Grethe Helde has nothing to declare.
Erling Tronvik reports grants from the Liaison Committee between the Central Norway Regional Health Authority (RHA) and the Norwegian University of Science and Technology (NTNU), personal fees from Allergan, and grants from Allergan, outside the scope of the current work.
Trond Sand has nothing to declare.
Footnotes
Acknowledgment
We thank the Unit for Applied Clinical Research at NTNU for assistance with randomization and monitoring of the study.
