Abstract
Acetylsalicylic acid (ASA) in combination with metoclopramide has been frequently used in clinical trials in the acute treatment of migraine attacks. Recently the efficacy of a new high buffered formulation of 1000 mg effervescent ASA without metoclopramide compared to placebo has been shown. To further confirm the efficacy of this new formulation in comparison with a triptan and a nonsteroidal anti-inflammatory drug (ibuprofen) a three-fold crossover, double-blind, randomized trial with 312 patients was conducted in Germany, Italy and Spain. Effervescent ASA (1000 mg) was compared to encapsulated sumatriptan (50 mg), ibuprofen (400 mg) and placebo. The percentage of patients with reduction in headache severity from moderate or severe to mild or no pain (primary endpoint) was 52.5% for ASA, 60.2% for ibuprofen, 55.8% for sumatriptan and 30.6% for placebo. All active treatments were superior to placebo (P < 0.0001), whereas active treatments were not statistically different. The number of patients who were pain-free at 2 h was 27.1%, 33.2%, 37.1% and 12.6% for those treated with ASA, ibuprofen, sumatriptan or placebo, respectively. The difference between ASA and sumatriptan was statistically significant (P = 0.025). With respect to other secondary efficacy criteria and accompanying symptoms no statistically significant differences between ASA and ibuprofen or sumatriptan were found. Drug-related adverse events were reported in 4.1%, 5.7%, 6.6% and 4.5% of patients treated with ASA, ibuprofen sumatriptan or placebo. This study showed that 1000 mg effervescent ASA is as effective as 50 mg sumatriptan and 400 mg ibuprofen in the treatment of migraine attacks regarding headache relief from moderate/severe to mild/no pain at 2 h. Regarding pain-free at 2 h sumatriptan was most effective.
Keywords
Introduction
Despite the common use of acetylsalicylic acid (ASA) in the acute treatment of migraine attacks there are only few well-controlled studies on the efficacy of ASA given as the single drug. In the past most studies were performed on the action of ASA coadministered with metoclopramide (MCP) (1–4). However, whereas ASA is available over-the-counter (OTC), metoclopramide is a prescription drug. The dose of ASA or equivalent used in these studies varied from 650 mg to 900 mg and was thus lower than the single-dose of 1000 mg currently recommended (5). Newly developed high buffered effervescent ASA tablets with an acid neutralization capacity of 15 mEq provide dissolved citrates which promote gastric emptying and thereby enhance the onset of the analgesic effect of ASA (6, 7).
Using the aforementioned effervescent tablets of ASA 1000 mg a significant difference in response rates compared to placebo was observed in the acute treatment of migraine attacks (8). In this study the effervescent placebo reduced nausea and vomiting to the same degree as active drug. Therefore, it is reasonable to assume that the anti-nausea and anti-emetic effects are attributed to the high buffering capacity of the dissolved citrates provided. However, there still is a substantial need to confirm this and to further confirm the efficacy of oral acetylsalicylic acid effervescent 1000 mg compared to placebo by using a different study design. Until now, oral acetylsalicylic acid in combination with metoclopramide is as effective as a triptan (2, 3). A particular aim of this study was to investigate the efficacy of these drugs on accompanying symptoms of a migraine attack. The present study addressed several questions:
Is 1000 mg effervescent ASA as effective as a specific migraine agent like sumatriptan 50 mg for the treatment of migraine attacks?;
Is 1000 mg effervescent ASA as effective as ibuprofen 400 mg as a nonprescription nonsteroidal anti-inflammatory agent, which was recently approved by the FDA for the treatment of migraine (9)?;
Does 1000 mg effervescent ASA influence the associated symptoms of a migraine attack?.
Patients and methods
Patients
The study was conducted in 16 centres in Germany, Italy and Spain. Patients were recruited and treated between March 2002 and October 2002. The trial was implemented in accordance with the rules and regulations of the German Drug Law, the International Conference on Harmonization Good Clinical Practice (ICH GCP) standards and in accordance with the Principles of the Declaration of Helsinki after approval by local ethics committees. Patients (18–65 years) were eligible for the study if they gave informed consent in writing and if they met the International Headache Society (IHS) diagnostic criteria for migraine with or without aura (10). Patients were required to have a history of migraine of at least 1 years and between 1 and 6 attacks per month. Patients having at least one of the following criteria were excluded: participation in a study during 4 weeks prior to the start of the study; all other types of headache, including tension-type headache; hypersensitivity to acetylsalicylic acid, salicylates, ibuprofen, nonsteroidal anti-inflammatory drugs or sumatriptan; peptic ulceration or gastric bleeding; haemorrhagic diathesis; disorders of kidney, liver, lung, heart, or brain function; neurological disorders; hypertension; coronary heart disease and/or history of myocardial infarction; pregnant or lactating women or women of childbearing age not using contraception; drug or alcohol abuse and prohibited concomitant medication.
Study design
The trial was a double-blind, placebo-controlled, randomized, three-fold crossover, multicentre study in outpatients. All patients were treated with a single-dose of each of the three treatments or the related placebo: two tablets effervescent ASA 500 mg or matching placebo, 1 capsule ibuprofen 400 mg or matching placebo, 1 tablet sumatriptan 50 mg or matching placebo. A double-dummy design was used. ASA placebo was an effervescent low buffered tablet. Due to blinding thin gelatin capsules containing sumatriptan tablets were matched with identical capsules containing inactive substance. The encapsulated sumatriptan was demonstrated to be equivalent to the commercial tablet based on in vitro dissolution conducted in deionized water (data on file). In vivo pharmacokinetic comparison of encapsulated sumatriptan and sumatriptan standard tablets performed by Milton et al. (11) showed equivalence of encapsulated sumatriptan. Patients treated three migraine attacks during a study period of 3 months per patient. Treatment order was assigned by a predetermined randomization code. Patients were instructed to leave a minimum of 48 h between consecutive study treatments to ensure that a new attack and not a migraine recurrence was treated. Each dose of study medication was to be taken within 6 h of headache onset. The condition had not to be improving at the time of treatment. Medication should only be taken when pain was at least moderate or severe on a 4-point headache severity scale (0 = no, 1 = mild, 2 = moderate, 3 = severe). Patients were allowed to remedicate with any medication of their choice (‘escape medication’) at any time during the course of the study. However, they were encouraged to wait until 2 h after taking study medication. Ergots and triptans were not permitted until 12 h after study medication. Use of ‘escape medication’ was recorded over the treatment period. A diary card was given to the patients at the screening visit and the investigator instructed the patients how to complete the diary card. During each treated migraine attack patients recorded in the diary date and time of onset of symptoms and of intake of medication; presence or absence of aura; pain intensity and nausea just before the intake of study medication, and at 0.5, 1, 1.5 and 2 h postdosing on a 4-point verbal rating scale (VRS: 0 = no, 1 = mild, 2 = moderate, 3 = severe); presence or absence of vomiting, photophobia and phonophobia at the same time points; global assessment of medication on a categorical scale (poor, moderate, good, excellent), time of use of ‘escape medication’; time when headache disappeared; return of attacks within 24 h. In addition, adverse events were documented over the treatment period.
Evaluation of efficacy and safety
The primary efficacy analysis was based on the number of patients with headache relief from grade 3 or 2 to grade 1 or 0 for 1000 mg effervescent ASA and placebo 2 h after administration of study medication. Secondary endpoints were defined as efficacy of 1000 mg effervescent ASA, 50 mg sumatriptan and 400 mg ibuprofen as measured by comparison of 50 mg sumatriptan vs. placebo, 400 mg ibuprofen vs. placebo, and by comparison of the active substances for the number of patients with reduction in headache from severe or moderate to mild or no at 2 h after administration of study medication. Other secondary endpoints were efficacy of the three active treatments by comparison with placebo and by comparison of the active substances for the number of patients pain-free (grade 0) at 2 h. Furthermore the number of patients with reduction in nausea intensity from severe or moderate to mild or no nausea within 2 h after intake of study medication, the number of patients with an improvement of accompanying symptoms vomiting, photophobia and phonophobia within 2 h after intake of study medication, the number of patients using escape medication 2 h after administration of study medication and the frequency of headache recurrence within 24 h after intake. Finally patients’ global assessment of medication at 2 h after dosing and adverse events was recorded.
Sample size and statistical analysis
Sample size calculation was based on assumed response rates of 55% (ASA), 55% (ibuprofen), 60% (sumatriptan) and 40% (placebo). A patient was defined as responder if pain intensity 2 h after intake of study medication changed from severe or moderate to mild or no pain. In order to detect a significant difference between the treatments (P = 0.05, two-sided, power = 90%) 148 patients per treatment were required. Taking into consideration a block size of 12 patients, which includes each medication 9 times, 198 patients should complete the trial according to the protocol. Assuming a dropout rate of approximately 30%, the final study size had to include a minimum of 288 patients.
Hierarchical statistical 2-step test procedure was performed using a χ2-test. First the overall null hypothesis of no differences between any of the four treatments was tested. Subsequently only if statistically significant difference (P < 0.05) was detected the following treatment comparisons were tested for statistical difference: acetylsalicylic acid vs. placebo, sumatriptan vs. placebo, ibuprofen vs. placebo, sumatriptan vs. acetylsalicylic acid, ibuprofen vs. acetylsalicylic acid, ibuprofen vs. sumatriptan. Data from the secondary efficacy criteria were analysed descriptively. Statistical analysis was based on a 5% probability for type I error. In the event of significant differences, the limit probability P-value was specified.
Results
Study population
Three hundred and fifty-six patients were randomized (Germany 164, 6 centres; Italy 118, 6 centres; Spain, 74, 4 centres), of whom 43 did not take study medication. One treated patient did not return the diary and was excluded from efficacy analysis. Therefore the intention-to-treat (ITT) population consisted of 312 patients who took at least one dose of study medication and provided efficacy assessments. Thereof, 120 patients showed major protocol violations, resulting in a per-protocol (PP) population comprising 192 patients. The most frequently observed protocol violations were drug intake later than 6 h after onset of the migraine attack and discontinuation of the study before all three attacks had been treated. The safety analysis was based on 313 patients. The demographic details of ITT patients for the four treatment groups are listed in Table 1. Of the 312 ITT patients 222 were treated with ASA, 212 with ibuprofen, 226 with sumatriptan and 222 with placebo. No baseline characteristic differences between treatment groups were found. Nineteen percent of the patients were male and 81% female. Mean age was 38 years. 79% of patients reported a history of migraine without aura.
Baseline characteristics of patients for the four treatment groups (ITT population)

Years since first diagnosis.
Efficacy
The primary endpoint of the present trial was the comparison of the percentage of patients with a reduction in headache severity from severe or moderate to mild or no pain after 2 h of drug intake after ASA and placebo. Results for this endpoint as well as the percentage of patients pain-free after 2 h are shown in Fig. 1. After 2 h 52.5% of patients receiving ASA vs. 30.6% of patients treated with placebo recorded mild or no pain. The corresponding numbers for pain free were 27.1% and 12.6%, respectively. Both differences are statistically significant (P ≤ 0.0001). The efficacy data of ibuprofen (60.2% and 33.2%, respectively) and sumatriptan (55.8% and 37.1%, respectively) also show significant differences to placebo (P < 0.0001). The 95% confidence intervals for all treatment were tight and very similar showing post hoc adequate study power. The statistical comparison of the active treatments showed no significant differences between treatments with the exception of the comparison ASA vs. sumatriptan for percentage of patients pain-free at 2 h (P = 0.025). However, whereas all ITT results could be confirmed by the PP analysis the latter differences were not confirmed by the PP analysis. The PP percentages for reduction of headache severity from moderate or severe to mild or no pain at 2 h were: ASA, 59.6%; ibuprofen, 66.9%; sumatriptan, 61.0%; placebo, 34.9%. For the criterium pain-free at 2 h the numbers were: ASA, 35.3%; ibuprofen, 39.3%; sumatriptan, 41.8%; placebo, 16.1%.

Pecentage of ITT patients (responders, mean and 95% confidence intervals) with reduction in headache severity from severe or moderate to mild or no pain at 2h (□) and percentage of patients (responders) pain-free at 2h ( ). All active drugs are superior to placebo.
). All active drugs are superior to placebo.
The time course of headache improvement up to 2 h after intake of study medication in terms of patients with a reduction in severity from severe or moderate to mild or no pain is shown in Table 2 and Fig. 2. The highest responder rates obtained at 0.5 h and 1 h were achieved with ASA treatment, followed by ibuprofen. At 0.5 h only ASA was statistically significant different from placebo (P = 0.0104). Ibuprofen and sumatriptan were only at 1 h significantly different from placebo. At 1.5 h and 2 h after intake of study drug, the rates of treatment responders were comparable between the three active treatments. The highest rate of treatment responders at 2 h was achieved by ibuprofen, followed by sumatriptan and ASA. Statistical analysis for a period effect showed a P-value of 0.542. Therefore there was no positive effidence for a period effect.

Time course for number of patients (%) with reduction in headache severity from severe or moderate to mild or no pain at 0.5, 1.0, 1.5 and 2.0 h (ITT population). • ASA 1000, ▪ ibuprofen 400, □ sumatriptan, ○ placebo.
Number of patients (%) with a reduction in headache severity from severe or moderate to mild or no pain at 0.5, 1.0, 1.5 and 2.0 h (ITT population)

significantly different from placebo (P < 0.05).
The number and percentages of patients who recorded to be headache free are shown in Table 3 and Fig. 3. At 0.5 h, no therapeutic effect on the rate of patients with complete remission was observed in any of the treatments. At 1.0 h, the highest rate of patients was reached with ibuprofen (10.1%), followed by ASA (6.4%) and sumatriptan (5.4%). At 1.5 h, the percentages of patients with complete relief of headache actually did not differ between the active treatments but were clearly higher in comparison with placebo (P < 0.05).
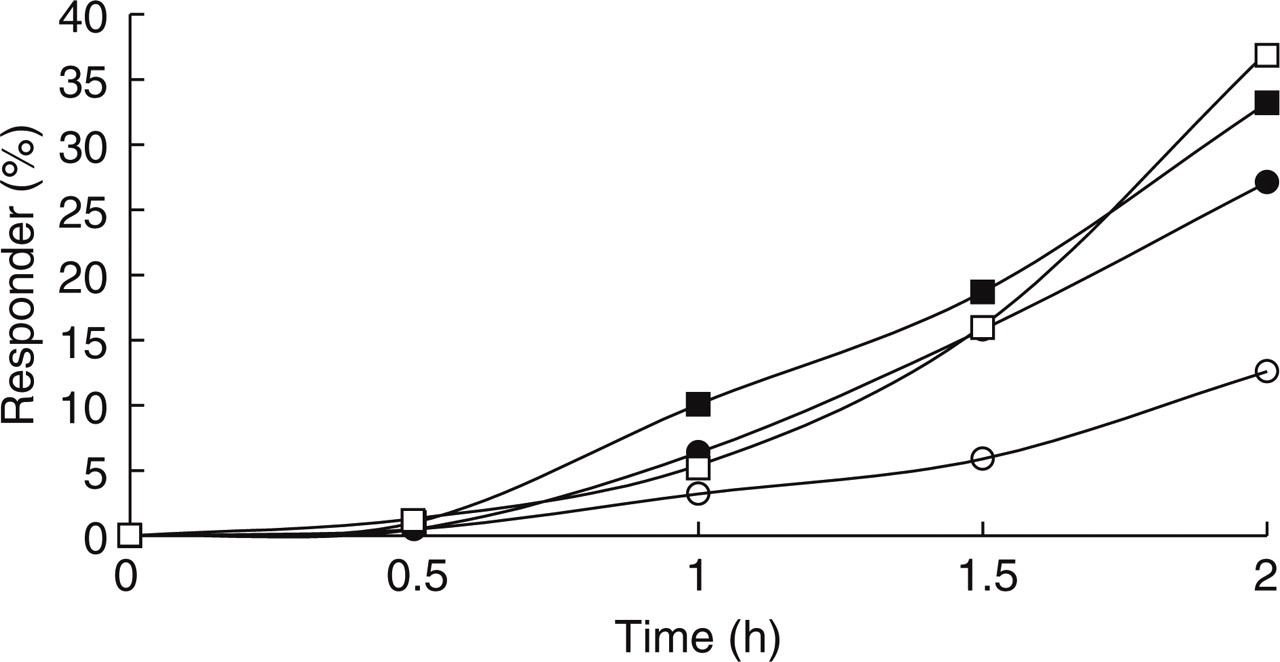
Time course for number of patients (%) with a complete remission of headache at 0.5, 1.0, 1.5 and 2.0 h (ITT population). • ASA 1000, ▪ ibuprofen 400, □ sumatriptan, ○ placebo.
Number of patients (%) pain free at 0.5, 1.0, 1.5 and 2 h (ITT population)

significantly different from placebo (P < 0.05).
In this study three different placebos have been used. Whereas in the above description the combined placebo response have been presented, in Table 4 the treatment-related placebo-adjusted response of the active treatments for the criterium reduction in headache severity from severe or moderate to mild or no pain are shown. The therapeutic gain of ASA was 27.8%, of ibuprofen 23.5% and of sumatriptan 25.8%. The differences between treatments are not statistically significant. The resulting number-needed-to-treat (NNT) for all active treatments was 4 (Table 4).
Placebo-adjusted active treatment response for the criterium headache reduction from severe or moderate to mild or no pain, therapeutic gain and numbers-needed-to-treat (NNT) (ITT population), CI = confidence interval

Occurrence of nausea was recorded in the diaries throughout the 2-h observation phase after each treated attack. No treatment differences regarding the number of patients with a reduction in nausea intensity from severe or moderate at baseline to mild or no nausea between 0 and 2 h was found (Fig. 4). The percentages of patients with an improvement of nausea at 2 h were similar in all active treatment groups (ASA: 64%; ibuprofen: 66%; sumatriptan: 65%), but slightly lower in the placebo group (56%). However, there were no statistically significant differences between the treatments.

Time course for number of patients (%) with reduction in nausea intensity from severe or moderate to mild or no nausea at 0.5, 1.0, 1.5 and 2.0 h (ITT population). • ASA 1000, ▪ ibuprofen 400, □ sumatriptan, ○ placebo.
The incidence of accompanying symptoms such as photophobia, phonophobia and vomiting was evaluated separately for patients reporting the symptoms at baseline (Table 5). Photophobia and phonophobia was reported in about 65% and 58%, respectively, of the patients in each treatment group at baseline. Vomiting was reported less frequently and occurred in about 16% of all patients. After 2 h of drug intake, the rates of patients with remission of photophobia and phonophobia in patients on ASA, ibuprofen and sumatriptan were between 62% and 70% and were significantly superior to placebo (46% and 49%, respectively). With regard to vomiting patients on ASA, ibuprofen and placebo reported remission rates of 94%, 97% and 91%, respectively, whereas the remission rate for sumatriptan was 77%. However, remission rates of vomiting are calculated from a low baseline frequency.
Number of patients and percentage of all patients reporting an accompanying symptom at baseline, number of patients reporting an accompanying symptom at 2 h, and number of patients and percentage of patients with accompanying symptoms at baseline with remission of the symptoms after 2 h (ITT population)

significantly different from placebo (P < 0.05).
In case of insufficient treatment effect patients were allowed to take rescue medication. Headache severity just prior to escape medication did not show differences between the treatments. 45%, 41% and 41% of patients treated with ASA, ibuprofen and sumatriptan took escape medication, but more patients treated with placebo (66%) used escape medication (P < 0.0001). The percentage of patients without any recurrent migraine attack within 24 h after intake of the study drug was around 80% in all treatment groups (ASA 80%, ibuprofen 81%, sumatriptan 77%, placebo 82%).
Global assessment analysis was based on percentages of subjects in each group assessing the medication to be excellent or good. The highest rate of patients was observed with sumatriptan (42.9%), followed by ibuprofen (38.2%) and ASA (32.5%). All active treatments clearly differed from placebo (16.7%) (P ≤ 0.0001). The difference between sumatriptan and ASA was also significant (P = 0.0238), but could not be confirmed by the PP analysis (ASA: 40.5%; sumatriptan: 48.6%; P = 0.1842).
Safety
The total incidence of adverse events (AE) in the ASA (16%) and ibuprofen group (12%) was comparable to placebo (14%) while it was slightly higher in the sumatriptan group (20%) (Fig. 5). Incidence rates of AE in the treatment group were not influenced, by the order in which the drugs were taken. Adverse events considered by the investigator to be drug-related were reported in 4.1% of the patients on ASA and 4.5% of the patients on placebo treatment. The respective incidence rates for ibuprofen and sumatriptan were also comparable to placebo with 5.7% and 6.6%, respectively (Fig. 5). None of the potentially related adverse events reported in the ASA and ibuprofen groups occurred in more than 1% of patients in each group. Two serious adverse events were reported. One patient experienced a renal colic after having treated a migraine attack with ASA. In the investigator's opinion there was no causal relationship to the study drug. One patient was hospitalized due to a perforation of a duodenal ulcer after having taken ibuprofen. The investigator assessed causal relationship to the study drug to be possible. Both events resolved completely.

Adverse events (safety population, n= 313). Total adverse events (▪); drug-related averse events (□) were investigstor attributed.
Discussion
This crossover trial showed, that effervescent acetylsalicylic acid (ASA, 1000 mg) is as effective as a specific migraine drug such as sumatriptan (50 mg) for the acute treatment of migraine attacks. Our study used ASA without an antiemetic such as metoclopramide and achieved similar results as earlier comparative trials using ASA (900 mg) plus metoclopramide (10 mg) (2). In the study by Tfelt-Hansen (2) headache improvement 2 h after intake of study medication was observed in 57% and 43% of patients treating 2 attacks with ASA plus metoclopramide, while we observed a success rate of 52.5% for ASA without metoclopramide. The two study populations were comparable as shown by headache improvement observed with sumatriptan which was 53% and 55% with 100 mg sumatriptan in the study of Tfelt-Hansen, while we observed headache improvement in 55.8% of patients after 50 mg of sumatriptan. 50 and 100 mg sumatriptan have a similar efficacy as shown by direct comparisons (12) or meta-analysis (13). A direct comparison of ASA plus metoclopramide compared with ASA alone using modern trial design and large patient numbers, however, needs to be done.
In this study no significant differences between the three treatments 1000 mg ASA effervescent, 400 mg ibuprofen and 50 mg sumatriptan regarding the therapeutic gain could be observed. Also the NNTs do not differ between ASA, sumatriptan and ibuprofen. For all treatments NNTs of 4 have been found, which is in the range of 2–5 which is considered as indicative of high efficacy with analgesics in acute pain (14). However, for pain-free, which is currently the recommended efficacy parameter by the IHS, a higher efficacy of sumatriptan has been found. The placebo rates differed in the different treatment groups between 24.7 and 36.7%. This is most probably due to chance. Unblinding due to the formulation used (capsule vs. effervescent tablet) should have resulted in a higher placebo rate in the sumatriptan arm compared to the other two treatments. This was, however, not the case.
Another interesting aspect is, that triptans so far failed to show a clear superiority over analgesics such as acetylsalicylic acid (2, 3), ketoprofen (15), diclofenac-K (16) or tolfenamic acid (17). In most of these trials this was true for headache response after 2 h. With the exception of our trial, however, triptans were more effective than analgesics or NSAIDs for the parameter pain-free after 2 h.
This study in addition showed, that ASA is as effective as ibuprofen in terms of improving headache and autonomic disturbances. The percentage of responders to ibuprofen is comparable with earlier studies in migraine (9, 18, 19). Although our study failed to show a statistically significant difference in improvement of nausea and vomiting, for photo- and phonophobia the 3 active drugs were statistically significantly superior to placebo.
All other secondary endpoints confirmed the results of the primary analysis, namely that ASA is equivalent to sumatriptan and ibuprofen and that all three active drugs are superior to placebo. All active drugs were well tolerated.
In this study encapsulation of the comparator drug sumatriptan was done. This is a routine practice in randomized double-blind clinical trials. Equivalence between encapsulated sumatriptan and the marketed form was shown by in vitro dissolution studies. An in vivo bioequivalence study showing equivalent pharmacokinetic of the two forms was not performed. However, in this study we found evidence of pharmacodynamic bioequivalence of the encapsulated sumatriptan. For headache response at 2 h and pain-free at 2 h, both absolute and placebo-subtracted responder rates were similar to those obtained in past studies (13).
In conclusion, 1000 mg effervescent acetylsalicylic acid without metoclopramide is as effective as 50 mg sumatriptan or 400 mg ibuprofen for the acute treatment of migraine attacks and offers high efficacy and good tolerability. Analysing the pain-free response 50 mg sumatriptan was more efficacious than 1000 mg effervescent acetylsalicylic acid.
Footnotes
Acknowledgements
The study was funded by Bayer AG Germany. The protocol was developed jointly by Bayer and the principal investigator. The PI and the members of the study group had unrestricted access to all study data.
