Abstract
Background
Cluster headache (CH) is a debilitating disorder characterized by unilateral, severe pain attacks with accompanying autonomic symptoms, often waking the patient from sleep. As it exhibits strong chronobiological traits and genetic studies have suggested a link with the hypocretin (HCRT) system, the objective of this study was to investigate HCRT-1 in CH patients.
Methods
Cerebrospinal fluid HCRT-1 concentration was measured in 12 chronic and 14 episodic CH patients during an active bout, and in 27 healthy controls. The patients were well characterized and clinical features compared to the HCRT concentration.
Results
We found significantly lower HCRT levels both in chronic (
Conclusions
This is the first report of significantly reduced HCRT concentrations in CH patients. We speculate that decreased HCRT may reflect insufficient antinociceptive activity of the hypothalamus. The mechanism of the antinociceptive effect of HCRT is not known and requires further investigation. This study supports the hypothesis of a connection between arousal regulation and CH.
Introduction
Efforts to understand the pathological mechanisms behind the highly disabling cluster headache (CH) have intensified over the course of the past 15 years. Alas, the difficulty in studying a disorder characterized by transitory, extremely severe pain attacks with autonomic symptoms and accompanied by agitation has hindered significant progress. CH is a well-defined primary headache disorder (1,2) typified by many nightly attacks and is considered a chronobiological disorder; the daily attacks often occur at predictable hours and patients go through periods of remission and exacerbation, shown to follow daylight (3). Imaging studies have implicated the hypothalamus in CH pathology (4,5).
Hypocretin (HCRT)-1 and -2 (also known as orexin A and B) are hypothalamic neuropeptides. HCRT-1 and -2 are involved in a plethora of functions including maintenance of arousal, regulation of sleep, homeostatic functions, and pain (6). Genetic studies implicate HCRTs in CH as a
We hypothesize that the HCRT system is involved in the pathophysiology of CH and that reduced HCRT neuronal activity may lead to destabilization of nociceptive input. The aim is to measure the HCRT-1 concentration in CSF from CH patients during an active bout to investigate whether decreased levels of HCRT reflects activity of CH and to characterize disorder subgroups, rhythmicity and underlying mechanisms of CH with respect to the concentration of HCRT.
Patients and methods
Patients
Patients. Patient characteristics and clinical details. Clusters/year and cluster duration are not shown for chronic patients as these patients have attacks almost every day.
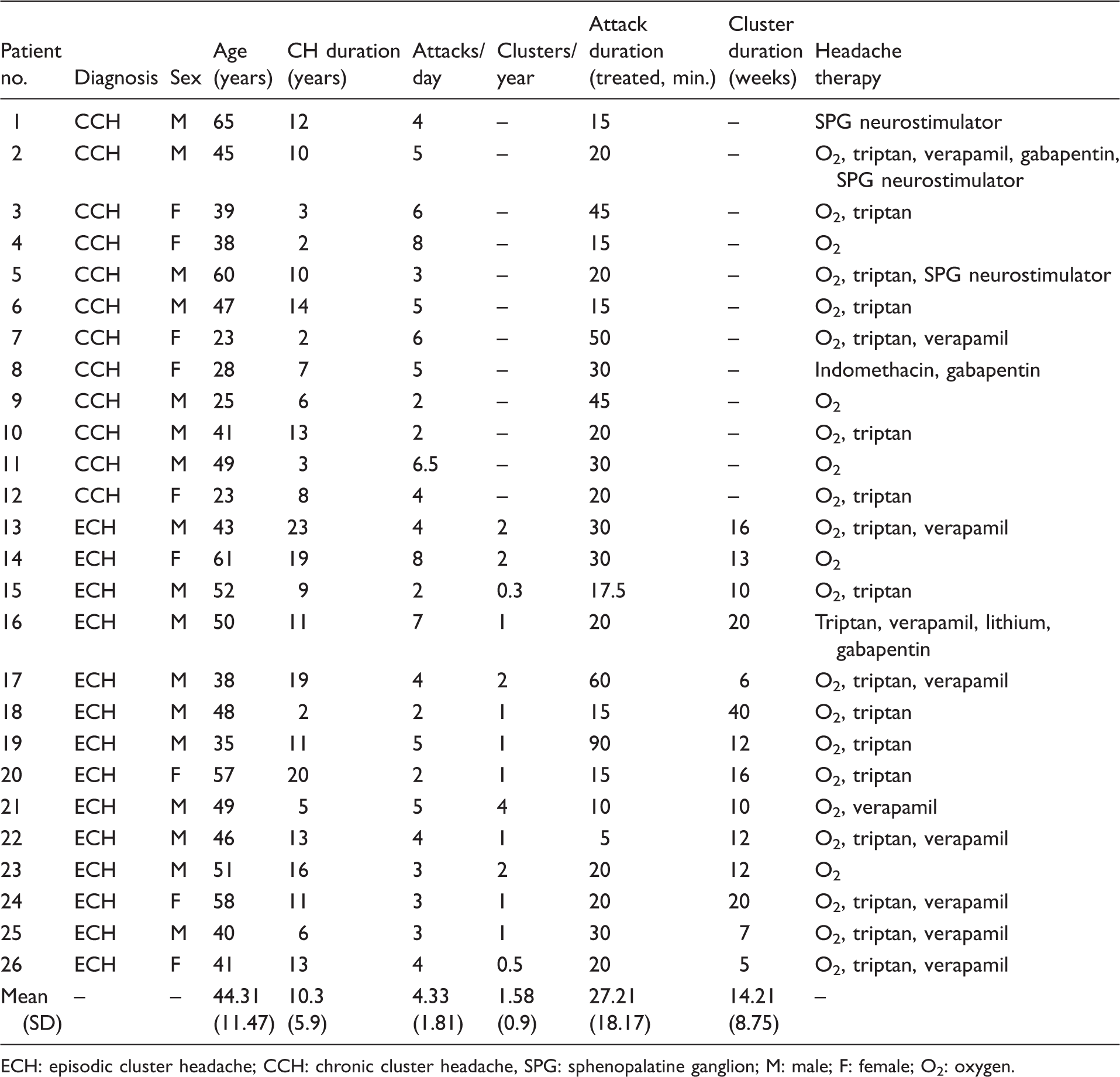
ECH: episodic cluster headache; CCH: chronic cluster headache, SPG: sphenopalatine ganglion; M: male; F: female; O2: oxygen.
Clinical characterization of the patients’ headache disorder was based on an extensive, physician-conducted, semi-structured interview based on a questionnaire. The questionnaire covered many aspects of the patients’ lives including headache characteristics, habits and sleep quality. All patients were investigated with polysomnography (PSG) and multi-sleep latency test (MSLT) according to American Association of Sleep Medicine (AASM) standards (15,16) during the active phase of the headache disorder. Exclusion criteria were other chronic primary or secondary headaches and serious somatic or psychiatric illness. If patients were taking prophylactic medication, this was kept stable for at least seven days prior to sample collection. Patients would attempt to treat their attacks using oxygen but were allowed to use other medication such as injectable or nasal triptans, as a rescue. One patient was examined but later excluded as it was discovered he had an acute herpes simplex infection.
Controls
From a prior study 27 healthy controls without headache-, sleep- or other neurological disorders were included (17). Twelve of the participants were recruited through an advertisement for healthy volunteers (www.forsoegsperson.dk) and clinical and neurological examination by a physician was conducted. The remaining 15 participants were referred for subjective sleep-related complaints but were concluded as healthy by experienced sleep specialists (SK, PJ) by interviews, normal clinical and neurological findings, PSG and MSLT.
Measurement of CSF HCRT-1
CSF (10 ml) was collected in the attack-free state between 8:00 a.m. and 12:00 noon, cooled on ice and stored at −80℃ within 30 minutes until analysis. HCRT-1 was analyzed in crude CSF by radioimmunoassay from Phoenix Pharmaceuticals (Belmont, CA, USA). We used the same methodology as in a previous study (17). All samples were blindly measured in duplicate and the mean of the results was calculated. The standard curve range was 10–1280 pg/ml. Assay quality was monitored by the internal positive control sample included in the assay kit. Furthermore, the intra-assay coefficient of variation was estimated in each assay and if the value was >5% the samples were reanalyzed. Furthermore, one external reference sample of pooled CSF from healthy individuals with a HCRT-1 concentration of 163 pg/ml was included in each assay to determine the inter-assay variability, i.e. the precision of results between different assays. The CSF samples were analyzed monthly over a period of two years and the inter-assay coefficient of variation was determined as 9.9%. Finally, a sample from the previous assay was included in each assay to estimate the day-to-day variability. The mean deviation between two repeated measurements was 6.6%. The external reference CSF sample was originally donated by Dr. E. Mignot, Stanford Center for Sleep Sciences and Medicine, Stanford University, USA. Accordingly, the CSF concentration of HCRT-1 in all measurements was adjusted to the level of CSF HCRT-1 defined at Stanford University. Previously used groupings of CSF intervals for HCRT-1 concentrations (low (≤110 pg/ml), intermediate (>110 to ≤200 pg/ml), and normal (>200 pg/ml)) were not strictly applied in this sample as these are most relevant in the diagnosis of narcolepsy (11,18,19).
Statistical analysis
SAS 9.3 was used for statistical analysis. Values of
Rhythmicity and HCRT. CH-index, rhythmicity, attack timing, MSLT and HCRT-1. CCH-patients scored significantly higher on the CH-index compared to ECH (

ECH: episodic cluster headache; CCH: chronic cluster headache; CSF: cerebrospinal fluid; HCRT-1: hypocretin 1; MSLT: multi-sleep latency test.
Results
Twenty-six patients (12 CCH and 14 ECH), all in active bout, were included. Clinical characteristics of patients and controls are provided in Table 1. Table 2 provides individual HCRT-1 concentrations for each patient. CSF samples had normal cell counts and glucose and protein concentrations. Opening pressure, where available (
Diagnostic subgroups
Summary statistics. Controls were younger (
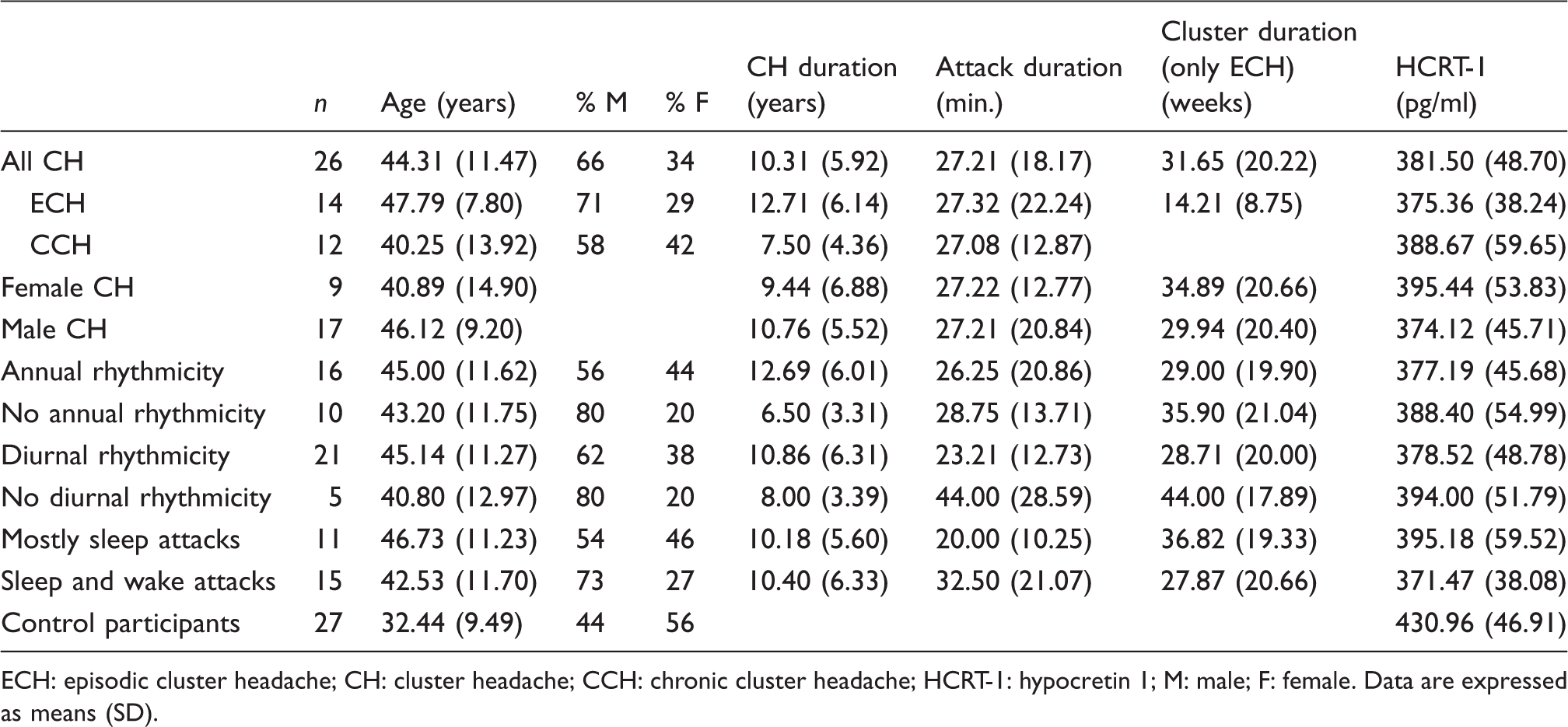
ECH: episodic cluster headache; CH: cluster headache; CCH: chronic cluster headache; HCRT-1: hypocretin 1; M: male; F: female. Data are expressed as means (SD).
Attack timing
There was no significant difference in HCRT-1 levels between patients reporting annual (377.19 and 388.40 respectively,
Discussion
We found the concentration of HCRT-1 in CSF to be significantly reduced in a clinically well-characterized population of CH patients compared to healthy controls. Clinical research in the association between CSF HCRT-1 levels and headache is sparse and data are available only from a few small studies. In idiopathic intracranial hypertension no difference was found between patients (
Mechanisms of action
The effects of HCRT are mediated by dopaminergic, histaminergic, noradrenergic, and serotonergic neurons (23–25). HCRT neurons project to the locus coeruleus, raphe nuclei, nuclei within the hypothalamus, the dorsal and ventral horns, motor nuclei of the brainstem, the limbic regions of the brainstem and the neocortex (26) and are involved in sleep-wake, feeding, arousal and autonomic regulation (27). Normal function of HCRT is crucial for regulating sleep and wakefulness as demonstrated by the devastating effects the disappearance of hypothalamic HCRT nuclei and neurons has in narcolepsy (26). A role similar to the switching or gating role the neuropeptide plays in sleep has been suggested for headache (25). Since CH patients do not suffer loss of wakefulness or cataplexy (indeed, during attacks patients experience increased locomotion), it is unlikely that their symptoms are a result of transient, total dysfunction of the entire HCRT system. Rather, the observed lower HCRT levels may reflect a transient and reversible dysfunction of part of the system, or an overall lowered function of the entire system, perhaps with short-lasting, compensatory surges during attacks.
HCRT-1 and -2 activation in the posterior hypothalamus differentially modulates nociceptive dural inputs to the trigeminal nucleus caudalis (TNC) (28). In rats, systemically administered HCRT-1 inhibits nociceptive responses of TNC neurons to electrical stimulation of the dura mater surrounding the middle meningeal artery (29). The hypocretinergic trigeminal modulatory effect is further evidenced by its inhibition of neurogenic dural vasodilation (30). Also, activated trigeminovascular afferents result in excitation of hypothalamic neurons (31), some of which produce HCRT in the lateral and posterior hypothalamic nuclei.
On a spinal level, dysfunction of part of the hypocretinergic system may affect HCRT neurons projecting to lamina I and II of the dorsal horn, involved in the processing of pain. Second-order neurons are directly excited by hypocretinergic projections, but also
In this setting, speculatively, CH patients may exhibit a vulnerable HCRT system. During sleep there is an inhibition of a part of the HCRT system from the GABAergic neurons in the ventrolateral preoptic nucleus. These fluctuations could theoretically explain the predilection the attacks have for occurring during this stage of arousal.
A syndrome of transient or permanent HCRT deficiency?
A dysfunctional HCRT system could partly explain the stereotypical phenotype of CH patients. It has been demonstrated that HCRT plays a role in addictive behaviors and HCRT neurons project to areas associated with nicotine response (34). This association is especially interesting since, anecdotally, CH patients experience nicotine craving after attacks and present as a higher proportion of heavy smokers (35).
Additionally, ictal restlessness is an important diagnostic criterion and a clinical finding distinctively associated with CH. High levels of HCRT have been demonstrated to induce hyperlocomotion via D1 and D2 receptors in rats (36) and physical activity with an emotional component has been shown to increase the concentration of HCRT in CSF in dogs (37). CH hyperlocomotion could be the result of the aforementioned hyperactivity of a compensatory descending mechanism. In a study of attack timing, 71% of daytime attacks occurred while the patient was physically relaxed (38), perhaps during physiologically lowered HCRT levels, in an already dysfunctional HCRT system.
Methodological considerations
This is the first study to report a statistically significant reduction of HCRT-1 in a large sample of well-characterized CH patients. As mentioned, CH is a notoriously difficult disorder to study as the transient nature of the clusters and the agitation requires a high level of patient cooperation and logistical coordination. Measuring HCRT in CSF collected by spinal tap may not reveal subtle fluctuations in the hypothalamic region, and it is impossible to measure acute and/or small changes in neuropeptide concentrations in CSF.
Our patients were not unmedicated. Although their dosing was kept stable, their treatment (mainly verapamil and lithium) could theoretically influence HCRT levels. However, based on previous reports we consider this less likely as HCRT-1 levels are generally considered treatment robust (11). Lastly, it is highly desirable to repeat the lumbar punctures on ECH patients when they are outside of bout to establish if the change in HCRT is transitory. This is not an easy undertaking as there are important ethical considerations and patients are not as motivated to participate in such studies outside of bout.
Conclusion
We found significantly reduced concentrations of HCRT-1 in CSF in a large group of well-characterized CH patients compared to healthy controls, indicating that the HCRT system is involved in CH pathology. The exact role of HCRT in CH is not clear, but we speculate that a periodic deficiency of descending modulation of pain or a dysfunctional modulation of the activity in other brainstem nuclei may be involved. HCRT is actively involved in the regulation of sleep and further study of the connection between nightly CH attacks, sleep architecture and HCRT may provide valuable insight into an intriguing and powerful regulatory neuropeptide system.
Footnotes
Key Findings
Hypocretin (HCRT) is lower in cluster headache (CH) patients than healthy controls but remains within normal limits.
Lower HCRT levels may reflect altered function of the hypothalamus and may be responsible for altered pain processing in these patients.
Overall, the findings of lower HCRT levels in CH patients support a hypothesis of central dysfunction in CH.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
Acknowledgments
We thank Birte Kofoed for her skillful technical assistance with the HCRT assays.
