Abstract
Background and aim
M.R. present address: PAIN, National Institutes of Health, Bethesda, MD, USA
Methods
Patients and controls completed questionnaires and structured interviews composed of new and previously validated parts including the Pittsburgh Sleep Quality Index (PSQI) and Morningness-Eveningness Questionnaire (MEQ). Patients were characterized by a CH index, a unified measure of headache burden.
Results
A total of 275 CH patients and 145 matched controls were included. The most common trigger was sleep (80%) and a relationship between clusters and daylight was identified. Of the patients, 82.2% reported diurnal and 56% annual rhythmicity. Patients reported impaired sleep quality (PSQI) (p < 0.0001) and an inverse relationship between time passed since last attack and sleep quality was identified (p < 0.0001). The CH index was positively related to the PSQI (p < 0.0001).
Conclusion
Diurnally, CH exhibits a relationship with night-time and annually with daylight hours. Patients’ sleep quality is reduced compared with controls. Results suggest a complex relationship as sleep quality improves between clusters, but remains pathological.
Introduction
Cluster headache (CH) is an extremely painful headache disorder that exhibits striking chronobiological features. In its classical description the regularity of the attacks and the features that set it apart from other primary headache disorders have seeded many interesting studies and results (1,2). CH typically presents in younger males as severe attacks of side-locked, unilateral headache with accompanying autonomic symptoms (3,4). The attacks are described to be predominantly related to nocturnal sleep and follow specific diurnal and annual rhythms that often afford a certain degree of predictability (5).
Despite an increase in knowledge of the severe pain attacks, the relation to sleep-wake regulation and management, there are numerous unresolved issues in disease pathophysiology and the relation to circadian and intricate sleep-wake associations (6). A genetic link to the hypocretin system (7), a relationship between CH and sleep apnea (8), and a particular temporal relationship with rapid eye movement (REM) sleep (9) have all been suggested. However, it appears that the association between CH and sleep apnea may not exist (10) and the attacks have recently been shown to occur in both non-REM (NREM) and REM sleep (10,11). Thus, the link between CH and sleep is complex as is also evident by the expansive anatomical and physiological overlap between sleep and headache (12).
The first studies investigating CH chronobiology uncovered a unique diurnal and annual periodicity (1,13) that have later been corroborated to an extent (5,14). The annual rhythms seem to follow a biphasic pattern, but results are diverging as to a possible relation with the solstices or equinoxes. Diurnal rhythms may also be subject to local cultural influences (5,13,14) but the exact mechanisms are unclear. According to hypotheses the hypothalamus is intricately involved as demonstrated in both clinical investigations (15,16) and by the chronobiological features themselves (5). In CH, particularly the possible roles and close interaction between the suprachiasmatic nucleus and the posterior hypothalamus, shown to be exclusively activated during CH attacks (15), are fascinating.
The aim of this study was, through descriptive epidemiology, to investigate self-reported sleep quality, chronotype, trigger factors and chronobiological features in a Danish population of CH patients. Further, we aimed to investigate whether possible poor sleep quality was confined to the cluster periods or universally present in this patient group.
Methods
A questionnaire composed of already validated sections (Pittsburgh Sleep Quality Index (PSQI), Morningness-Eveningness Questionnaire (MEQ)) and new questions developed by authors MB and RJ were sent to patients and controls. The questionnaire as a whole was assessed by an expert panel including 10 senior headache experts (unpublished data). They were asked to evaluate the questions: Are the instructions clear, is the questionnaire coherent, are the questions relevant, is the questionnaire logically divided into parts, and are the questions formulated clearly. The questionnaire was also tested with a randomly selected population of 10 CH patients representing different ages, genders and diagnoses (episodic (ECH) and chronic (CCH)), and comments and suggestions were implemented. The Capital Region of Denmark Ethical Committee approved the study (H-2-2012-016).
Questionnaire
The paper questionnaire contained 362 questions in seven sections: headache diagnosis, headache burden, treatment, sleep, work, lifestyle habits, and physical activity. It was estimated that it took the patients roughly one hour to complete the questionnaire. Following completion of the questionnaire, answers were verified and ambiguities ruled out by a structured in-person or telephone interview conducted by a physician or a trained medical student (MB, NL, AP). If the patient or control invited to participate did not respond within 21 days, he or she was contacted again by letter and/or telephone and encouraged to complete the questionnaire. Only questionnaires for which all questions were answered were used. Only part of the data obtained by this study are available here as the remainder will be made available in subsequent publications.
The PSQI (17) is a validated (18) 19-item measure of subjective sleep quality during the past month. The sum of seven component scores provides a global score, a higher value reflecting poorer sleep quality. A global score greater than 5 yields a high sensitivity and specificity in distinguishing “good and poor sleepers” (17). To assess daytime performance as a function of sleep quality, the Epworth Sleepiness Scale (19) (ESS) was used.
The MEQ (20) uses 19 multiple-choice items to chronotype patients into five categories: definite morning, moderate morning, intermediate, moderate evening, definite evening. The tool has been validated in middle-aged populations (21). Chronotype reflects at what time of the day a person is active or inactive, often reduced to sleeping habits only, i.e. “lark” or “owl.”
Regarding rhythmicity, patients were asked to report the hours of the day or the months of the year where attacks or clusters were most likely to occur. They also had the option to report no rhythmicity. Patients were asked if they smoked currently and could answer “yes, daily,” “yes, sometimes (less than one cigarette or equivalent per day),” “no, but I used to smoke daily,” “no, but I used to smoke sometimes (less than one cigarette or equivalent per day),” or “no, I have never smoked.” They were also asked for how many years they have smoked regularly, if they stopped, when, and finally how many cigarettes, cheroots, cigars or grams of pipe tobacco they smoked on average a day.
Patients
Patients diagnosed with ECH or CCH were recruited from a register of present and former contacts at the Danish Headache Center. Additionally, a notice was posted in the newsletter and website of the Danish patient organization for cluster headache. Other headache clinics in Denmark were contacted and patients receiving treatment at these clinics were referred to the study in case of interest.
Patients were included if they: were between 18 and 65 years old, had been diagnosed with ECH or CCH (International Classification of Headache Disorders, second edition (ICHD-II) criteria) and were able to tell CH attacks apart from other types of headache. Patients were excluded if they: had been diagnosed with another primary or secondary chronic headache (≥14 days/month) or did not speak and understand Danish. All patients’ diagnoses were verified by a headache specialist. Apart from the dichotomy of episodic/chronic offered by existing diagnostic criteria, patients were characterized according to self-reported annual (months of the year during which clusters/bouts occur) and diurnal (hours of the day when individual attacks occur) attack patterns, whether sleep was a trigger and according to a CH index (described below).
Controls
Controls were recruited via notices posted on the Internet and in work places and sport clubs in the Capital Region of Denmark. Controls were matched for sex and age based on means. Controls were required to be between 18 and 65 years old and headache free (≤1 day of headache/month). They were excluded if they had severe symptoms of sleep disorders.
Statistical methods
Analysis of variance (ANOVA) was used for comparison between more than two groups and Bonferroni corrections were applied in the analysis of PSQI. Chi-square test was used to calculate differences in gender composition, smoking status, MEQ groups, consumption of sleep medication and laterality. Wilcoxon rank sums were used for non-parametric data. Linear regression was used to describe the association between daylight hours for the months of the year and corresponding cluster occurrence as well as the ESS, CH index and PSQI. Two-sample t-test was used to compare means of two groups. A p value <0.05 was considered statistically significant. SAS 9.4 for MS Windows was used for all statistical analysis.
A CH index was calculated by the following equation: (attacks per day ×hours per attack) × (days per cluster × clusters per year) and is a measure of the area under the curve – the total time/year that the patient has CH. In this specific calculation, for CCH patients, number of clusters/year was set at one and cluster duration was set at 365 days. The motivation for calculating this index is to provide one number that reflects headache burden, as this is sometimes not completely clear, taking into consideration clusters, cluster duration, attacks and attack duration. Further, we wanted to characterize the patients beyond the dichotomy of episodic/chronic as some episodic patients in fact may experience far more headache than some chronic. Body mass index (BMI) was calculated from self-reported height and weight (BMI = kg/m2). Tobacco pack-years was calculated as number of cigarettes (or equivalent) smoked daily/20 × number of years smoking.
Results
A total of 275 patients and 145 controls completed the questionnaire and interview. In total, 481 patients and 266 controls were invited (Figure 1). The response rate was 57.2% for patients and 54.5% for controls.
Flowcharts for the recruitment of patients and controls.
Demographics
Demographic distribution of patients and controls.

Data are reported as means (SD) or as a percentage. ECH: episodic cluster headache; CCH: chronic cluster headache; BMI: body mass index; F: female; M: male. ap < 0.05.
Clinical characteristics
Clinical characteristics of the patient population.
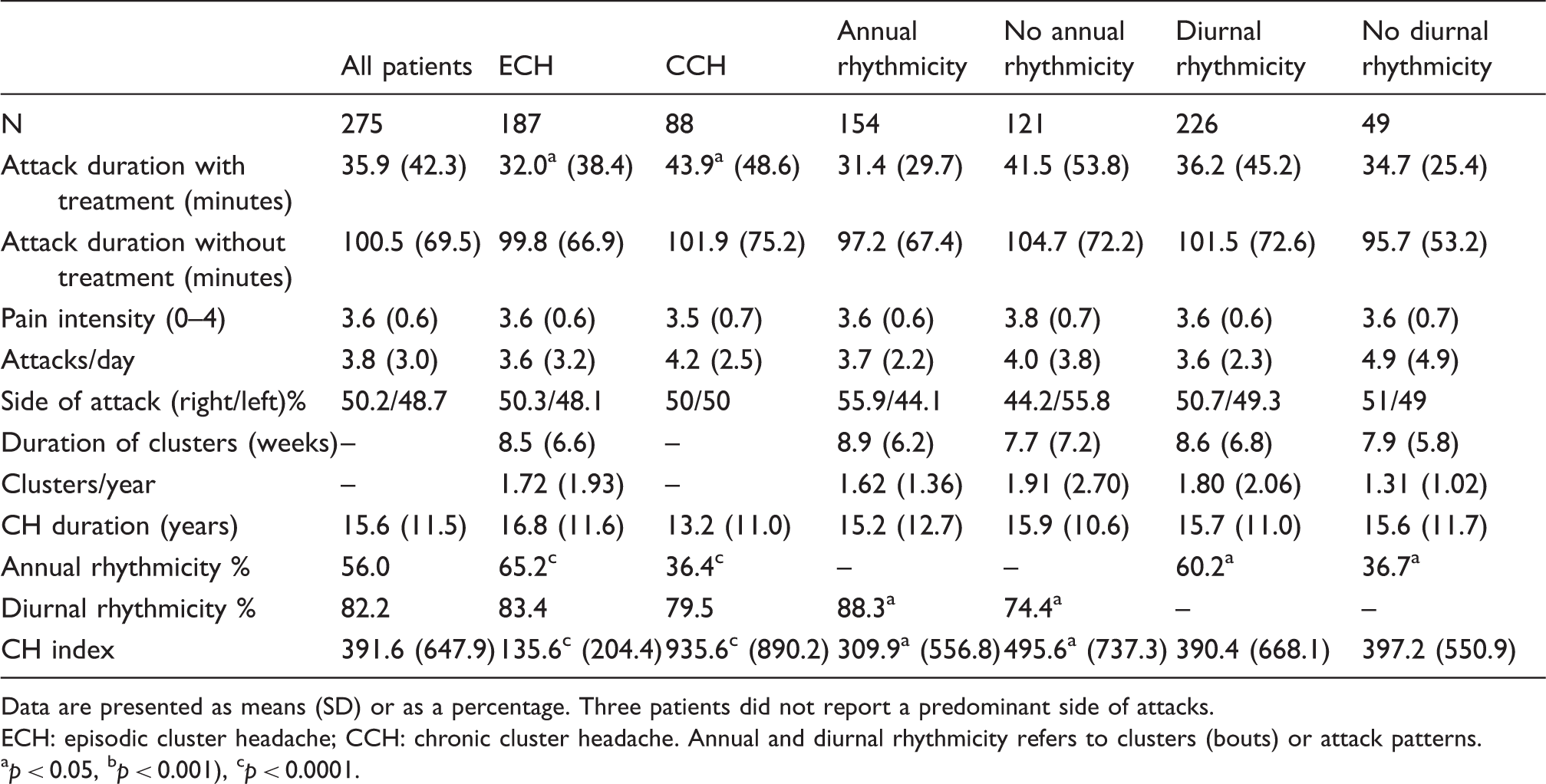
Data are presented as means (SD) or as a percentage. Three patients did not report a predominant side of attacks. ECH: episodic cluster headache; CCH: chronic cluster headache. Annual and diurnal rhythmicity refers to clusters (bouts) or attack patterns. ap < 0.05, bp < 0.001), cp < 0.0001.
Trigger factors
Percentage of patients reporting a given chrono-dependent trigger factor.
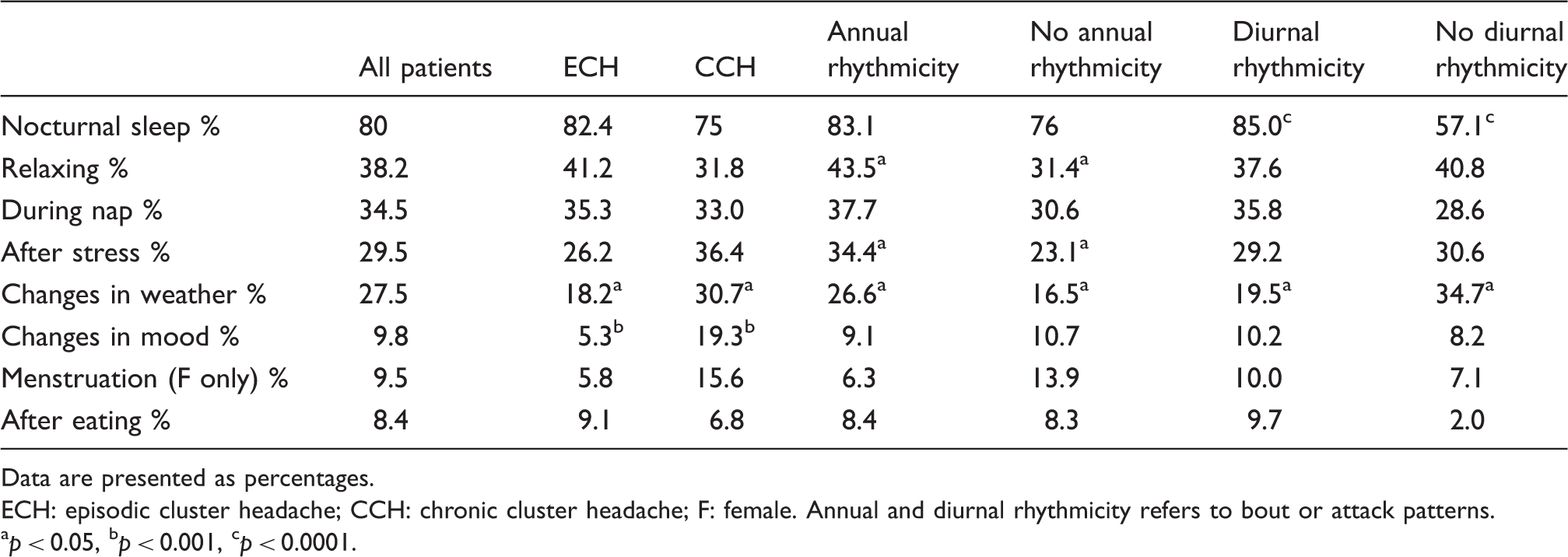
Data are presented as percentages. ECH: episodic cluster headache; CCH: chronic cluster headache; F: female. Annual and diurnal rhythmicity refers to bout or attack patterns. ap < 0.05, bp < 0.001, cp < 0.0001.
Diurnal and annual rhythmicity
A total of 82.2% of patients reported diurnal and 56% annual rhythmicity. By far the most commonly reported hour for attacks to occur was 02:00 (Figure 2). Times when the fewest attacks were reported were 09:00, 12:00 and 18:00 hours. The month during which clusters (ECH) or worsening of attacks (CCH) was least likely to occur was June (Figure 3). November was the month when most patients reported clusters or worsening. In a linear regression model the number of daylight hours (for central Denmark) for each month of the year was strongly associated with the number of patients reporting cluster or worsening of cluster during that month (p = 0.0002, R2 = 0.76). Dividing into ECH and CCH subtypes, the pattern was very similar with increased frequency from August to March.
Diurnal rhythmicity. Patients with diurnal rhythmicity (n = 226) and the self-reported distribution of their attacks during the 24 hours of the day. Annual rhythmicity. The number of patients (n = 154) reporting the most likely months for clusters of attacks (ECH), or worsening of attacks (CCH) to occur (bars, left y-axis). Daylight hours (gray line, right y-axis) are for the city of Odense, central Denmark. A linear regression model for the results is significant (p = 0.0002). ECH: episodic cluster headache; CCH: chronic cluster headache.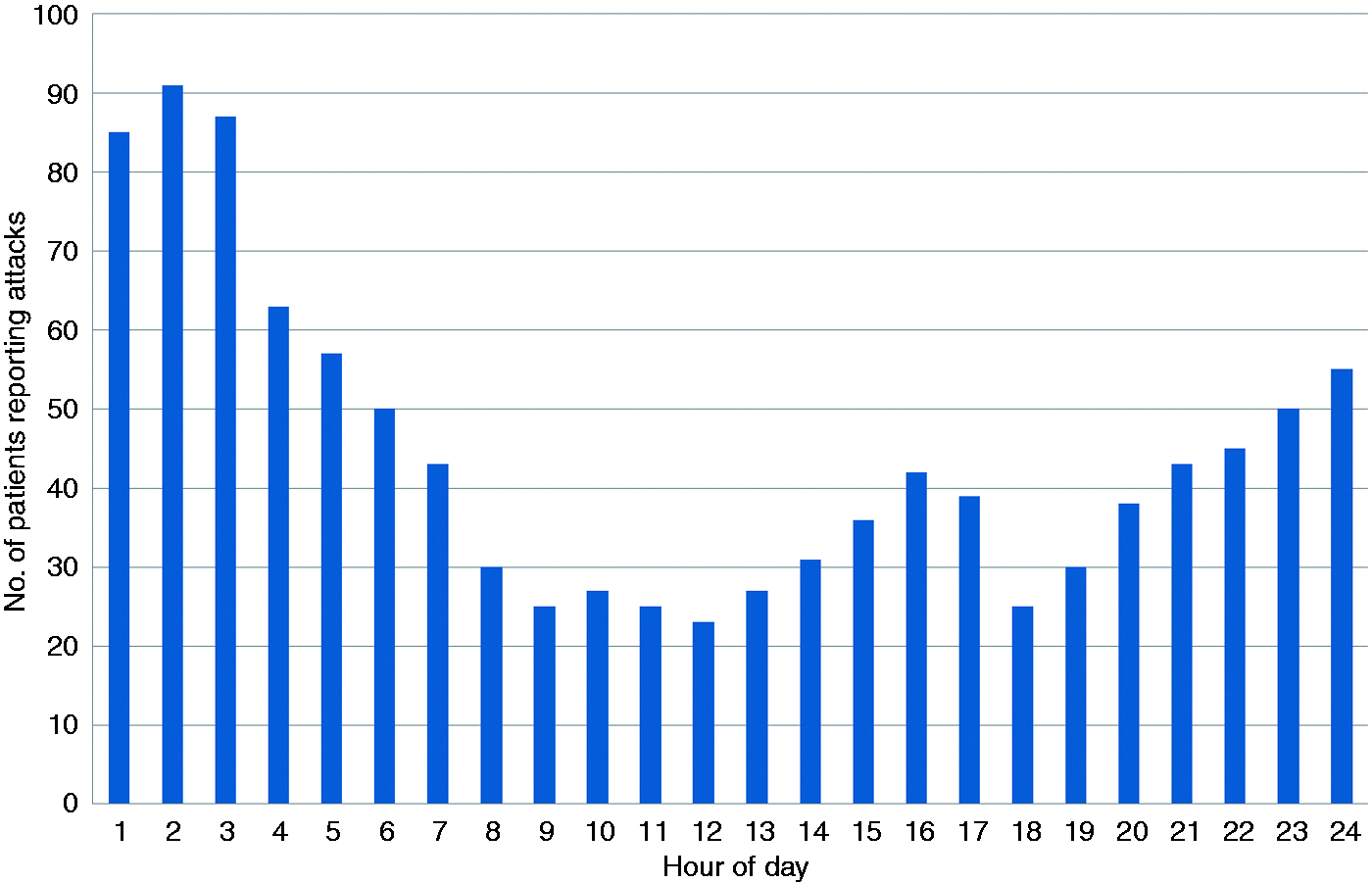

PSQI
Sleep quality and chronotype.

PSQI and MEQ according to diagnosis, rhythmicity of attacks, sleep trigger and sex. ECH: episodic cluster headache; CCH: chronic cluster headache; PSQI: Pittsburgh Sleep Quality Index; MEQ: Morningness-Eveningness Questionnaire. Annual and diurnal rhythmicity refers to bout or attack patterns. ap < 0.05, bp < 0.001, cp < 0.0001.

PSQI scores and time passed since last attack in patients (Groups 1–5) and healthy controls (Group 6). Values are presented as 25th percentile low, 75th percentile high (boxes), ±1.5 interquartile range (whiskers), mean (diamond), median (horizontal line), outliers (circles). PSQI: Pittsburgh Sleep Quality Index.
MEQ
There was no difference between patients and controls in the distribution into the five MEQ categories (p = 0.0899) (Table 4). There was a tendency for patients to present relatively fewer in the moderate and definite morning categories and more in the moderate and definite evening compared to controls, indicating more owls than larks among patients. However, the vast majority of patients and controls were in the intermediate (53.1%, 51% respectively) or moderate morning (31.6%, 38.6%, respectively) category. Neither MEQ score nor MEQ category showed any association with annual or diurnal rhythmicity, particular triggers or the CH index.
Discussion
We report results from a large, well-characterized population of CH patients and matched controls. Focusing on sleep and rhythmicity, our main findings relate to trigger factors, rhythmicity and sleep quality:
By far the most frequently reported trigger factor of CH attacks was nocturnal sleep, especially in patients reporting diurnal rhythmicity. Annual rhythmicity was more pronounced in ECH as compared to CCH, but one-third of CCH patients still experience yearly, rhythmic fluctuations in the activity of the disorder. Furthermore, those reporting either annual or diurnal rhythmicity were more likely to also report the other. CH attacks exhibited a striking diurnal and annual rhythmicity; the latter showed a close association with daylight hours. CH patients had greatly reduced sleep quality. Sleep quality improved with the passage of cluster-free intervals but approached healthy controls’ only after more than a year and still remained pathological. There was no difference in chronotype composition between CH patients and controls.
Clinical characteristics
We did not find the same dominance of right-sided attacks as one previous study (60%) (22). Our results are more in line with those of Rozen and Fishman (5), who found 49% right sided, 44% left sided and 3% bilateral. We also found longer attack duration in CCH patients compared with ECH. This might reflect that CCH patients in general are less responsive to treatment and may present more treatment-refractory cases (23).
The frequency of diurnal rhythmicity appears to be the same in both CCH and ECH but, as might be expected, we found that CCH patients are less likely to report that their headaches vary with the seasons. Despite this tendency, about one-third of CCH patients still reported annual rhythmicity, suggesting that the activity of the disorder in these patients is by no means stationary. As would also be expected, the CH index was much higher in CCH patients than ECH. It was also higher in patients not exhibiting annual rhythmicity but was unaffected by diurnal rhythmicity. This may be influenced by the fact that annual rhythmicity is more frequent in ECH than CCH. Interestingly, it seems that diurnal and annual rhythmicity seem to follow each other since exhibiting one makes it more likely that the patient also exhibits the other. Whether the type of rhythmicity really represents a specific phenotype or underlying mechanism is unknown but should be investigated in future studies.
Trigger factors
By far the most common trigger factor was nocturnal sleep (80%). In the questionnaire we differentiated between nocturnal sleep and daytime napping and interestingly only one-third of the patients reported napping as a trigger. Relaxing (38%) was also frequently reported as a trigger, which is in line with an earlier study that found 71% of daytime attacks occurred during a state of physical relaxation or napping (24). We also found that patients exhibiting diurnal rhythmicity were more likely to report sleep as a trigger factor. This is particularly interesting from a pathophysiological perspective because strong chronobiological rhythmicity must be expected to involve hypothalamic functions, the hypothalamus serving as the body’s main pace- and rhythm generator (25). Likewise, sleep regulation partly involves the hypothalamus and the present results could be taken as indirect proof that in those patients with strong chronobiological features, sleep and the pain attacks are more tightly interwoven, one affecting the other more strongly.
Rhythmicity
Fifty-six percent of our patients reported annual rhythmicity. Previous reports on this topic in CH exist and our results are in disagreement with these to an extent; however, it is also apparent that no two previous studies have found exactly the same results. Four studies including 70 to 1134 patients exist (1,5,13,14). Manzoni et al. found no prevailing time patterns of cluster periods in their patient population but the three latter studies identified two peaks of onset; Kudrow in July and January, Rozen and Fishman in April and October and Ofte et al. in spring and autumn. We did not find two peaks; rather we found a trough—a period when clusters are less likely to occur. While the existing studies suggest that it may be a problem of synchronization between external light cues and internal pacemakers, ours suggest that it may be the presence of longer days, as in its maximum in June in Denmark, at latitude 56 degrees, which prevents penetration of clusters. There may be several possible factors that play a role, for example, sunlight exposure, amount and quality of sleep, seasonal affective disorders or entirely different and perhaps unknown chronobiological rhythms. However, further research is needed to determine how seasons affect CH. It may be theorized that the distance from the equator affects the manifestation of cluster periods. The four prior studies were all conducted at different latitudes (North 30, 66 degrees) but no clear pattern has emerged. Studies of populations closer to the equator might provide some insight. Presently, results indicate that clusters may follow the solstices (1), the equinoxes (5,14) and lastly, in the present paper, day-time hours.
A total of 82% of our patients reported that their attacks exhibit diurnal rhythmicity, which is similar to the Rozen and Fishman study (5). We also found the same peak of attack incidence at 02:00 hours as they did. Remarkably, according to the Rozen and Fishman study there were also three troughs: at 08:00, 11:00 and 17:00 in the course of the day, which is close to those we found at 09:00, 12:00 and 18:00 (Figure 3). Ofte et al. found that 59% of Arctic patients reported diurnal rhythmicity and that attacks were most likely to occur between 24:00 and 04:00 (14). In an Italian population the most likely hour for patients to report attacks was 14:00 hours (13). It seems likely that the diurnal rhythms are highly influenced by culture and the daily rhythms that predominate in a region. There are indications that manipulating bio-rhythms might influence the occurrence of cluster attacks (26,27) but evidence is not overly convincing and further research is needed. Particularly, a better understanding of brainstem and (posterior) hypothalamic neuronal circuits may provide insight into CH pathology. It seems that the best explanation for the high incidence of attacks during the night is what the patients also report: sleep. Finally, it should be remembered that different factors may influence attack rhythmicity in individual patients. Some may be particularly vulnerable to factors adversely affecting sleep, others alcohol and so on.
Sleep and chronotype
Self-reported sleep quality in patients compared to controls was greatly reduced. Further, we have shown that sleep quality is positively related to the time passed since the last attack, finding a clear pattern of continuous improvement as time progressed since the last attack. However, even those patients for whom it had been more than a year since their last attack still suffered from significantly worse sleep quality than our controls. One interesting detail, not revealed by the present results, is whether sleep quality worsens before cluster onset as may be suggested by one very recent case report (28).
The fact that sleep quality is dependent on the amount of time that has passed since the patient suffered his or her last attack, and incrementally improves when the cluster is over, indicates that the mechanism that causes headache attacks is not ever-present as is the mechanism that produces poor sleep. This suggests that there is a connection between sleep and CH that goes beyond the direct disturbance of sleep caused by the attacks themselves and perhaps involves circuits in the hypothalamus and brainstem. This may be further corroborated by the fact that patients not reporting sleep as a trigger still had an elevated PSQI score. It cannot be concluded that absence of a sleep trigger means that the patient does not have nocturnal CH attacks. However, it would still be expected that these patients’ sleep quality would be much better, yet this is certainly not the case.
The strong chronobiological features of CH may suggest that the chronotype of CH patients is somehow different from controls’. We found no evidence of such a relationship employing the MEQ; however, this does not exclude the possibility that a particular chronotype may be more prevalent in CH patients. Prospective sleep diaries and actigraphy studies may show different results.
Methodological considerations
The present study presents strong data of a high quality. Diagnoses were made by headache specialists and participants were interviewed by trained medical staff to ensure correctness of the answers. Via the structured interview ambiguities could be resolved and the fact that the present study was conducted in a hard-copy fashion ensured that no patients (e.g. those without Internet proficiency) were excluded.
However, in order to amass the information the questionnaire turned out to be somewhat lengthy. This was noted in the testing phase; however, the authors felt that it was preferable to work with one large questionnaire rather than several small, separate studies. It may have resulted in some potential participants choosing not to answer but our response rates were similar to other epidemiological health care studies (29).
Studying CH presents certain unique problems. For example, there are dilemmas relating to the episodic-chronic classification, e.g. how a patient who is attack free on prophylactic medication, but has attack penetration when medication is tapered, should be classified. Further, the fact that patients may be cluster free for months or even years means that it may be difficult to recall features, if a long time has passed since the last cluster.
We stratified the patients beyond the classic episodic-chronic dichotomy in two ways; by rhythmicity and CH index. The motivation for this was the shortcomings in the existing distinction, especially with regards to research. Episodic patients may, according to current criteria, remain episodic, even with cluster periods lasting most of the year. In contrast, one single, sporadic CH attack a month will classify a patient as chronic. Furthermore, as evidenced by the present results, rhythmicity is a prominent feature of ECH but also of CCH. With our knowledge of the (chronobiological) functions of the hypothalamus and our understanding of CH pathology, it seems prudent to distinguish patients according to this feature.
Conclusion
In a large population of well-characterized CH patients we discovered several remarkable features concerning trigger factors, annual and diurnal rhythmicity and sleep quality. Sleep is a certain trigger of CH attacks and we demonstrate a clear temporal relationship between the attacks and sleep. However, since sleep quality improves slowly and incrementally only after the attacks subside, we conclude that the relationship between sleep and CH goes beyond a simple, direct temporal relationship. In addition, we identified a strong relationship between daylight hours and cluster occurrence. While sleep is greatly affected and individual attacks and clusters show a striking periodicity, we found no difference in patient and control chronotype composition. Whether presence/absence of rhythmicity really represents a specific phenotype or underlying mechanism is still unknown and should be investigated in future studies.
Clinical implications
Sleep is the most important trigger factor in cluster headache (CH) and patients should be screened for sleep-related problems, even outside of the bout. Information about the chronobiological features, especially the association between clusters and the amount of daylight, should be included in patient education as it may help patients understand and plan around their headache. Even outside of their cluster periods, CH patients are greatly affected by poor sleep quality and investigations of possible sleep disturbances may be indicated.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
