Abstract
Background
Cognitive difficulties contribute to patients’ disability during migraine attacks and have been overlooked in migraine research. Neuropsychological studies performed during attacks have produced inconsistent findings due to design differences and limitations.
Objective
Our objective is to document changes in cognitive performance of migraine patients during migraine attacks with a comprehensive battery of cognitive/behavioral tests, while controlling for potential confounders.
Method
A prospective two-period, randomized, cross-over study compared within-subject neuropsychological evaluation in two conditions—during a naturally occurring untreated migraine attack and a headache-free period.
Results
Thirty-nine patients with episodic migraine (37 females, average 38 years old) were included and 24 completed the study. Participants performed worse during the attack in the majority of cognitive tests, compared to the headache-free status, and significantly so in word reading speed (p = 0.013), verbal learning (p = 0.01), short-term verbal recall with (p = 0.01) and without (p = 0.013) semantic cueing and delayed recall with (p = 0.003) and without (p = 0.05) semantic cues. Differences found were unrelated to age, gender, literacy, condition order, interval between evaluations, anxiety, pain intensity or duration of the attack.
Discussion
Cognitive performance decreases during migraine attacks, especially in reading and processing speed, verbal memory and learning, supporting patients’ subjective complaints. These findings suggest the existence of a reversible brain dysfunction during attacks of migraine without aura, which can relate specifically to migraine or be a consequence of acute pain processing by the brain.
Introduction
Migraine is a disabling disease with a significant impact in global health (1). Attacks of migraine without aura are complex biological phenomena with distinct clinical manifestations that include headache, gastrointestinal symptoms and dysfunctional modulation of sensory inputs (light, sound, smell or movement) (2,3).
Additional attack-related symptoms, such as cognitive difficulties (4–8), are often reported by patients and probably reflect brain function changes during attacks. Cognitive and mental symptoms are significant contributors to patients’ disability (6) and may not be relieved by effective abortive medication (9). Difficulties most often reported by patients relate to different cognitive domains such as sustained and divided attention, concentration, planning, judgment, initiative, processing speed, language and memory (7,10). These may persist beyond the pain phase, up to the following day (9), as 80% of patients report mental tiredness, asthenia, depressed mood and concentration difficulties (11,12) after the attack.
Cognitive dysfunction has probably been overlooked in migraine research, since data about its impact on disease-related disability are scarce and it has seldom been evaluated as a therapeutic outcome (13,14).
Existent data on neuropsychological performance during migraine attacks are difficult to summarize because of a paucity of published studies, often based on small samples, having different designs and using distinct neuropsychological measures, often targeting specific cognitive domains (10,13–22). The profile of cognitive impairment is inconsistent, varying from dysfunction documented in several cognitive domains (attention, processing speed, working memory, calculation, visuo-motor processing, visual and verbal memory) (10,13,14,16–18,22) to normal performance (19–21). In addition, there is lack of control for factors that influence cognitive performance in general, such as mood changes (14,16,18,19,21) concomitant drug treatments, substance abuse or the practice effect of repeated neuropsychological testing. Migraine has its own potential confounders that also need to be considered, such as the presence of aura (a cortical phenomenon that might influence visual processing and speed (18,19)), photophobia (that might interfere with tasks requiring prolonged staring at a computer screen) and the effects of pharmacological agents, either migraine treatments or migraine-triggering drugs.
Our study was designed to evaluate cognitive changes during naturally occurring untreated migraine attacks, compared to a headache-free status. We selected a comprehensive battery of cognitive and behavioral tests targeting different cognitive domains that have been identified as disturbed on at least one previous study, and we controlled for anxiety, depression, pharmacological effects, presence of migraine aura and for the practice effect bias.
Participants and methods
Population
Otherwise healthy episodic migraine patients were recruited consecutively at a headache outpatient clinic, both from first and from follow-up visits. Inclusion criteria were: a) age between 20 and 65 years; b) ≥4 years of formal education; c) diagnosis of episodic migraine according to the International Classification of Headache Disorders, second edition (ICHD-II) (23); d) ≥1 attack/month of migraine without aura in the three months preceding inclusion; e) living/working near or at the study center (allowing an evaluation within 60 minutes of an established migraine attack); e) written informed consent. Exclusion criteria included the co-existence of any other headache type, chronic migraine, medication overuse, exclusive or very frequent attacks of migraine with aura, history of alcohol or drug abuse, and any medical or psychiatric disorder requiring daily treatment. In order to minimize potential cognitive effects of pharmacological agents, the only allowed daily medications were oral contraceptives and migraine prophylactics. The study protocol was approved by the Hospital and Faculty of Medicine Ethics Committee. There was no financial compensation for the volunteers.
Study design
Study design consisted of a randomized, two-period cross-over study requiring two evaluations of the same patient in two different conditions: condition 1, during an untreated spontaneous attack of migraine without aura (migraine attack—M); condition 2, during a headache-free period that was not treatment-induced (baseline—B) with a minimum of 72 hours elapsed since the last attack. Evaluation order was randomized, half the patients having the first evaluation while headache free (B→M), while the other half was first evaluated during the attack (M→B). A minimum interval of one month between evaluations was required. This design had the purpose of controlling for the practice effect and minimizing the need to determine the expected magnitude of improvement in neuropsychological testing between such short-term repeated evaluations (24,25). Assessments took place during working hours and within 24 hours of the onset of the attack. Attacks occurring at night or during weekends, attacks with aura and attacks treated with abortive medication in the previous 12 hours were not eligible. ICHD-II criteria (23) for migraine without aura and absence of aura in the previous 48 hours were re-checked immediately before cognitive assessment in the evaluation during an attack.
Individuals were excluded if they did not have an eligible attack within a two-year period after recruitment. At the end of a five-year enrollment period, the authors decided to terminate the study despite the fact that the initial goal of evaluating 50 participants had not been met.
Study protocol
Recruitment and inclusion of study participants were performed by headache specialists, who verified study criteria and carried out a standard clinical evaluation, including previous medical history and physical examination. After informed consent was obtained, data were collected and included verification of the ICHD-II diagnosis, gender, age, education years, disease duration, attack frequency, duration and intensity, and use of prophylactic treatment and other current treatments. Migraine impact was evaluated with the Headache Impact Test (HIT)-6 (26) and the Migraine-Specific Quality of Life (MSQoL) (27) questionnaires. Depressive symptoms and anxiety were quantified with the Zung Depression scale (28) and the State-Trait Anxiety Inventory (STAI) (29) as depression and anxiety are frequent comorbidities of migraine (30) and can influence neuropsychological performance (31). Attack evaluation included questions about current attack features as well as about pain intensity, which was scored before testing with a 10-point visual analog scale (VAS).
Neuropsychological battery.

Test description, reliability and practice effects.

Statistical analysis
Statistical analysis used SPSS v20. Frequencies and means ± standard deviations were used for descriptive statistics. Two-sample t-tests were used to compare baseline patient variables between randomization groups. The effect of the exposure to a migraine attack on the test scores, controlling for a learning effect, was tested with two-sample Student’s t-tests of the difference between groups in the change in test scores from the first to the second evaluation (51). A learning effect was tested with standard t-tests of the difference between groups in the change in test scores from the evaluation during an attack to the baseline evaluation. An exposure-by-period interaction was tested with standard t-tests of the difference between groups in the sum of the two evaluations of each individual. Data were tested for normality with the Shapiro-Wilk test and the test scores with non-normal distribution were analyzed with the nonparametric Wilcoxon-Mann-Whitney rank-sum test, instead of t-tests. The Hochberg step-up procedure (52) was adopted to account for multiple testing and to provide strong control of the type I error at the 5% significance level. Reported p values are adjusted for multiplicity using the Bonferroni correction (53). For each test showing a significant difference between the two evaluations, we used multiple linear regression to evaluate the effect of the following variables on the identified difference: gender, age, literacy, time between evaluations, anxiety, pain intensity and duration of the attack. The power of this study is 70% to detect a difference in test scores between evaluations greater than 0.8 standard deviations, at the two-sided 0.05 significance level.
Results
Population and study flow
Fifty-eight individuals were randomized, 27 to have their first evaluation while headache free (B→M) and 31 to have the first evaluation during a migraine attack (M→B). There were 34 non-completers (six withdrew from the study, 13 did not have an eligible attack within two years and 15 completed only one of the evaluations). Twenty-four patients (12 from each group) were able to complete the study (Figure 1).
Study flow and sample.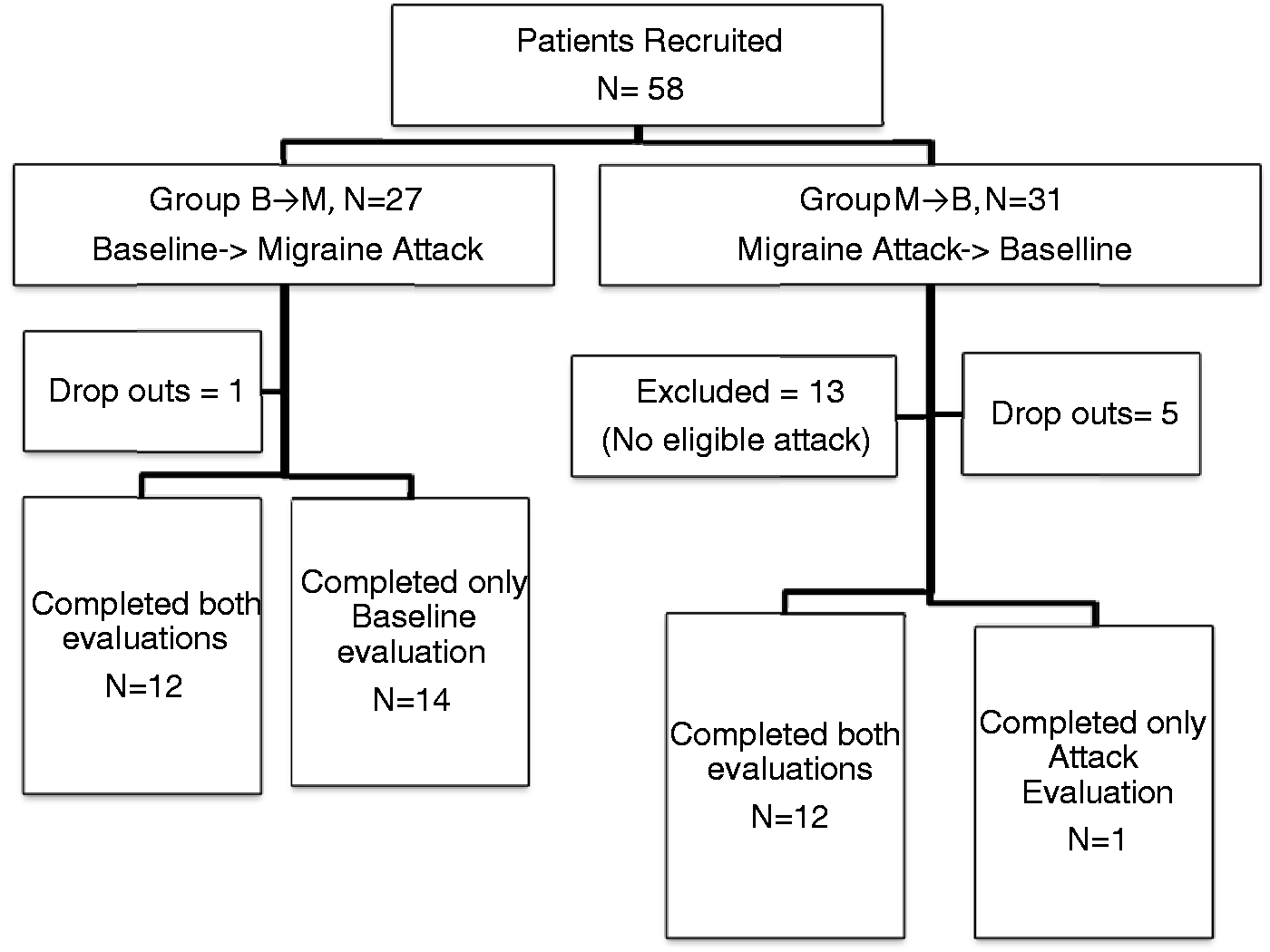
The study population consists of 24 patients, all right-handed but one. Table 2 shows the baseline characteristics of the patients. Four (17%) had migraine with and without aura (three visual, one visual and somatosensory). The sample had an average of 38.0 ± 11.6 years of age (range 21 to 63), 19.3 ± 12.1 years of education (range four to 22) and average disease duration of 19.3 ± 12.1 years (range three to 57). Average Zung score was 48.7 ± 8.0 (range 35 to 79) and eight patients had scores corresponding to mild depression but none was severely depressed.
On inclusion, the majority of patients (45.8%) had one to four attacks monthly, most (42%) lasting four to 24 hours and of moderate to severe intensity (100%). Seven participants (29%) were receiving migraine prophylactics (two propanolol, two propanolol and amytriptiline, two amytriptiline and one topiramate and amytriptiline). Migraine impact was moderate to high, with an average HIT-6 score of 63.3 ± 7.3 (range 45 to 76) and an MSQoL score of 75.6 ± 14.1 (range 43 to 95).
Patient characteristics.

B: baseline; M: migraine; HIT-6: Headache Impact Test; MSQoL: Migraine-Specific Quality of Life instrument; SD: standard deviation; m: mean.
Attack and baseline cognitive performance analysis
At the time of the studied attack, patients had experienced an average of 3.1 ± 2.5 (range 0 to 10) attacks in the previous month, lasting an average of 33.2 ± 29.6 hours (range 30 minutes to 96 hours). Ninety-two percent of the previous attacks were of moderate to severe intensity. During this attack, two patients (8%) experienced vomiting and 15 (62.5%) photophobia, and mean VAS pain intensity was 5.7 ± 1.6 (range 3 to 9.5). Average duration of symptoms at the time of the evaluation was 8.2 ± 8 hours (range 30 minutes to 28 hours, median five hours 45 minutes).
Average uncorrected raw scores of neuropsychological tests in each evaluations and mean difference of raw scores between evaluations (baseline-migraine).

SD standard deviation; SEM: standard error of the mean; CLVT: California Learning Verbal Test; WMS-III: Wechsler Memory Scale; STAI: State Trait Anxiety Inventory. aWilcoxon-Mann-Whitney rank-sum test, otherwise Student’s t-test; bp < 0.01 and cp < 0.05 after adjustment for multiple comparisons.
Significant differences were observed in Stroop word reading (p = 0.013), California Verbal Learning Test (CVLT) total learning (p = 0.01), CVLT short-term recall with (p = 0.01) and without (p = 0.013) semantic help, and delayed recall with (p = 0.003) and without (p = 0.05) semantic help (Figure 2). The anxiety state was higher during the attack (p = 0.003) but trace anxiety showed no difference. There was evidence for a learning effect in Stroop word reading (p = 0.009) but no evidence that the learning effect was different between groups (p for interaction = 0.28).
Differences in test performance of baseline versus during a migraine attack.
The effect of the clinical (independent) variables (gender, age, literacy, time interval between evaluations, anxiety, pain intensity and duration of the attack) on the differences found between the two evaluations of the Stroop reading, CVLT total learning, CVLT short-term free and cued recall, CVLT delayed free and cued recall were analyzed with multiple linear regression. Pain intensity during the attack was found to influence CVLT short-term free recall (p = 0.008) but no other variable influenced the differences found in the tests (online Supplementary Table S1).
Discussion
This study showed that migraine attacks are associated with a nominal performance decline in the majority of neuropsychological tests in relation to headache-free periods. The only exception were measures with a ceiling effect and those, such as the visual reproduction test, that are sensitive to practice (54). The decreased test performance during migraine was significant in two tests—the word reading task of the Stroop test (measuring processing speed and reading) and the CVLT (measuring learning and memory). The observed decline in performance was unrelated to the order of and time between evaluations, anxiety state and attack duration; only pain intensity was found to influence the short-term free recall of the CVLT, a measure related to the retrieval process (55).
This cognitive impairment seems related to the attack itself, giving objective support to patients’ subjective complaints (7,10,19) and corroborating previous studies that have demonstrated reversible dysfunction in processing speed, working memory, visual-spatial processing, alertness/fatigue (10,14,16,19,56), immediate and sustained attention, verbal learning (17,56) and inhibition (56). The predominant involvement of processing speed, learning and memory could suggest a preferential dysfunction of the prefrontal and temporal cortices and/or frontal subcortical white matter during migraine attacks, an observation supported by functional imaging findings during migraine attacks showing changes in the cingulated and pre-frontal cortex (57) insula and the temporal lobe (39).
The observed decline in word reading speed of the Stroop test might be related to an impaired automatic process of attending to the lexical features of words or reduced processing speed. Visual abilities, such as low-contrast sensitivity in older individuals, has also been linked to slower performance on word reading (58) yet visual factors are an unlikely explanation of these findings in our sample of young, otherwise healthy migraine without aura patients. The two other subtests of the Stroop test did not show a significant decline in performance. This suggests that conscious process of color naming, which involves the discrimination and retrieval of names, attention and verbal response was maintained, as was selective attention and the ability to suppress an automatic response to the interference task and seems to be consistently correlated to activation of the anterior cingulated cortex (60). Additional support of the functional integrity of pre-frontal cortical systems was the adequate performance on the remaining tests directed to executive functions such as set shifting, divided attention, mental flexibility, processing speed and verbal working memory.
Decline in episodic memory tasks was consistent across the retrieval measures of the CVLT test, reflecting a verbal learning and retrieval impairment, tasks supported by a large network involving the hippocampus, medial temporal lobe and prefrontal (dorsolateral) cortex (61,62) and connectivity between the thalamus and the putamen and striatum (63). The additional memory tests performed, which targeted famous faces naming and recognition (semantic memory), logical and visual memory, were not selectively impaired, which may relate to lower relative sensitivity of these tests (64) in which a higher level of contextual information is presented. Another possible explanation is a higher resistance of the anterior and dorsal lateral prefrontal cortices function during migraine, as those are specifically involved in the context-related retrieval process (65).
Our data therefore favor the involvement of temporal (58) or subcortical nuclei during the migraine attack, over pre-frontal or the cingulate cortices (57).
The mechanism by which cognitive impairment occurs during migraine without aura attacks is speculative. It may relate to a cortical spreading depression-like phenomena occurring during migraine without aura, shown to decrease cerebral blood flow (CBF) in the occipital, temporal and parietal lobes.
Cognitive dysfunction in migraine can also be secondary to influences of subcortical structures, namely the raphe nuclei and its cortical serotoninergic projections (for example, the orbitofrontal cortex, precentral gyrus, temporal pole, insula and somatosensory area). An alternative subcortical candidate is the thalamus, which is activated during attacks in humans (57) and whose activity was shown to control cortical states and influence perception, learning and cognition in animal studies (66).
An important issue that is not answered by this study design is whether these findings are specific to migraine or could be simply induced by the cognitive processes related to the pain experience. A wide range of neuropsychological and imaging changes have been described in chronic pain populations that support an impairment in attention, executive and general cognitive function, as a consequence of long-term pain-related neurochemical and neuroplastic brain changes (67). These data cannot be inferred to episodic pain, despite there being some evidence for interictal brain changes in migraine (68). Studies on the effects of acute pain in healthy volunteers have consistently documented hemodynamic responses in the primary somatosensory (SI) cortex (a pain-processing area), bilateral insula and second somatosensory (SII) cortices (regions involved in somatosensory integration that is influenced by attention), bilateral thalamus and brainstem (discriminative and arousal pain responses and descending pain modulation) but also other higher cognitive relay areas such as the anterior cingulated cortex (involved in the cognitive-attentional response to pain and anticipation of pain) and the dorso-lateral prefrontal (DLPF) and posterior parietal cortices (related to cognitive aspects of pain processing) (69). Involvement of these areas during pain processing could be the reason for decline in certain neuropsychological tests, in particular in tasks requiring attention (70), although this is not consistent with the cognitive profile shown in this study.
A recent functional magnetic resonance imaging (fMRI) study on migraine was able to demonstrate enhanced functional connectivity of the anterior temporal pole with pain-related cortical structures involved in acute pain processing, which suggests that the pain-processing mechanism of the migraine attack may not be entirely identical to experimental acute pain. In particular, this study was able to demonstrate hyperexcitability of the anterior temporal pole in the interictal phase of migraine that increased during the attack (58), giving support to our observation of pain intensity influencing some measures of the CVLT.
Another argument against the effect of pain by itself is the fact that cognitive subjective symptoms are reported during the prodrome of the migraine attack, before pain arises (71).
The main strengths of this study are its design, which dealt with most bias and confounders: the migraine population that was free of psychiatric co-morbidities and medication overuse, the use of an extensive neuropsychological battery covering the main cognitive domains and the control for multiple testing in the analysis. Studies with neuropsychological evaluation of migraine patients during attacks imply motivating patients to come into the office during an untreated attack and keeping them cooperative during one-hour testing. For that reason, as well as because of photophobia, it was important that the tests were paper and pencil based instead of computerized (which may be more sensitive to reaction times). Interaction with the examiner was used to improve task engagement and to ensure adequate compliance levels throughout the evaluation. However, this made blinding impossible and did not allow the use of more accurate tests to measure reaction speed.
The most important limitation of this study was the difficulty of assessing patients during the attack: Eighteen individuals were excluded or dropped out because they were not able to be evaluated during an attack; only 12 (46%) of the patients first evaluated at baseline managed to return during the attack, while only one patient first evaluated during an attack did not return for the second, baseline evaluation. The time lapse between evaluations in both groups was also quite different; the delay of the group starting with the attack evaluation almost doubled the delay of the other group. These facts had implications in the interpretation of the results and an impact in final sample size that, although in line with the sample sizes from previous studies (10,14,17–21), was insufficient to ensure adequate statistical power to attain significant statistical differences in some tests. Our difficulty in ensuring the actual participation of patients during the attack was perhaps influenced by the lack of financial compensation of participants, as evaluations were possible only during working hours. We also acknowledge that some cognitive aspects could have been better detailed. In particular, a more comprehensive executive testing would be preferable to study frontal lobe dysfunction and the inclusion of measures of crystallized intelligence would help in identifying the cognitive profile of dysfunction, such as a dissociation decline between fluid and crystallized intelligence, with potential implications in actual cognitive performance. Poor effort could have influenced neuropsychological scores obtained in this study, as we have not included a measure of effort in our battery. Finally, as stated earlier, the amplitude of estimated practice effect was not taken into consideration since it was minimized by the design of the study that allowed all participants to gain practice with the tests at first exposure.
Our study supports existing literature that reports neuropsychological and neurophysiological evidence of reversible brain dysfunction occurring during the migraine without aura attack that probably underlies migraine-related cognitive impairment (72). These findings are crucial in supporting patients’ claims of attack-related cognitive impairment and have clinical implications in relation to working and learning abilities during attacks and also on the evaluation of response to acute migraine treatment. In the research setting, it may help in the enlightening the nature of brain dysfunction in migraine. More studies are needed to determine if the attack-related cognitive impairment is specific to migraine pathophysiology or is simply related to acute pain processing by the brain.
Clinical implications
Reversible cognitive impairment is documented during migraine without aura attacks, supporting patients’ subjective complaints. Cognitive impairment is a symptom of the migraine attack that can be explained by reversible cortical dysfunction. Cognitive impairment contributes to attack-related disability and should be considered a target of migraine attack management.
Footnotes
Funding
This work was supported by the Portuguese Headache Society-Tecnifar investigation grant. The design, conduction, management, analysis and interpretation of the data of this study were performed by the authors without any financial compensation. Preparation, review and approval of the manuscript were also performed by the authors without any financial compensation. The grant was used to support the neuropsychologists’ work (neuropsychological evaluations) that was related to data collection and database preparation.
Conflicts of interest
None declared.
Note
Raquel Gil-Gouveia had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
