Abstract
Chronic migraine is associated with abnormalities in the periaqueductal grey that may be progressive. The condition is also associated with a greater degree of impairment in cortical processing of sensory stimuli than episodic migraine, perhaps due to more pervasive or persistent cortical hyperexcitability These findings fit with the model of migraine as a spectrum disorder, in which the clinical and pathophysiological features may progress over time. This progression may result from changes in nociceptive thresholds and ensuing central sensitization caused by recurrent migraine in susceptible individuals. This may lead to changes in baseline neurological function between headaches, evident not only in electrophysiological and functional imaging studies, but also as psychological and somatic complaints that occur after years of episodic migraine. From current research and migraine models, a conceptualization of chronic migraine is emerging in which relatively permanent and pervasive central changes have occurred that warrant novel and tolerable treatments.
Introduction
Migraine is a debilitating primary headache disorder that is ranked by the World Health Organization as 19th among all causes of years lived with disability (1). Since there are no biological markers for migraine, diagnosis is based on clinical history. Episodic migraine is characterized by recurrent headaches that last 4–72 h, with pain typically described as unilateral, pulsating, moderate or severe, aggravated by routine physical activity and associated with nausea and/or photophobia and phonophobia. The diagnosis of chronic migraine is made when headaches occur on ≥ 15 days/month, where at least half meet criteria outlined for episodic migraine or are treated with an acute migraine specific medication (ergotamine or triptans) (2–4).
Important advances in the pathogenesis and pathophysiology of migraine have been made over the past several decades. Structural lesions in the brains of migraineurs have been reported, although their clinical relevance is uncertain. White matter lesions are more common in migraineurs than in non-migraineurs and have been correlated with increased frequency of migraine (5). Recently, there have been reports of grey matter abnormalities in migraine (6–9), particularly in the areas of visual processing (10) and executive functioning (11).
However, the persistent lack of clinically relevant structural abnormalities in the brains of migraineurs (12) has led to a focus on functional alterations in the brain. In several variants of familial hemiplegic migraine, mutations have been identified in genes that code for P/Q type calcium channels (CACNA1A) or sodium-potassium pumps (ATP1A2) (13, 14) and lately the voltage gated sodium channel (15). These mutations may result in increased synaptic concentrations of glutamate, leading to neuronal hyperexcitability (13) and subsequent cortical spreading depression (16). Cortical spreading depression is likely to be the basis for migraine aura, as evidenced by both animal and human studies (16, 17). Although the link between cortical spreading depression and migraine pain is not definitely established, the ability of selected preventive medications to inhibit this phenomenon in the laboratory has suggested that inhibition of cortical spreading depression may provide a model system in which to test migraine-preventive pharmacotherapies (16).
Functional imaging and neurophysiological studies continue to provide insight into the pathophysiology of migraine. These techniques include positron emission tomography (PET), perfusion weighted imaging, magnetoencephalography (MEG), functional magnetic resonance imaging (fMRI) and blood oxygenation level-dependent (BOLD)-fMRI. Using BOLD-fMRI, changes consistent with cortical spreading depression have been observed, beginning in the extrastriate cortex and progressing to the occipital cortex during spontaneous visual aura in several migraineurs (18). These results are supported by a controlled study using MEG, in which visually triggered aura was associated with MEG-direct current shifts typical of those observed during cortical spreading depression (19). PET techniques have been used to examine brain regions that are activated by migraine pain, although exact anatomical localization is not possible. Using this technique, a 1995 study reported activity in the dorsal raphe nuclei, periaqueductal grey and locus coeruleus during spontaneous migraine (20). Subsequent studies using fMRI, which allows more precise anatomical localization, have identified activity in the red nucleus and substantia nigra during spontaneous migraine-related visual aura and visually triggered migraine (21, 22). Although these areas are best known for their roles in motor function, they are also involved in sensory processing and pain (23–25).
The majority of functional studies have been directed at understanding the pathophysiology of aura and episodic migraine. However, it is also critical to investigate chronic migraine, because it is frequently refractory to treatment and is extremely disabling. Individuals with chronic migraine exhibit disability scores that are nearly twice as high as those with episodic migraine and miss significantly more work, school, housework and social/leisure activities (26). Chronic migraine typically evolves from episodic migraine and is frequently associated with acute pain medication overuse (26). Over a period of months or years, episodic headaches increase in frequency and decrease in overall intensity, and the headache pattern includes fewer typical migraines (27, 28). It is of interest to determine how the differences between episodic and chronic migraine are manifested in cortical and brainstem neurophysiology. In the following section, we examine the findings of neurophysiological and imaging studies that have attempted to differentiate episodic from chronic migraine with the goal of understanding the functional correlates of chronic migraine in the brain. We then consider these findings in the context of models designed to explain the evolution of episodic migraine into chronic migraine.
Functional correlates of episodic and chronic migraine
Magnetic suppression of perceptual accuracy
Magnetic suppression of perceptual accuracy (MPSA) is a test that examines the effects of transcranial magnetic stimulation on visual perception. This technique has been used as an index of cortical excitability in migraine (29). In the MPSA test, individuals are shown a series of three letters (trigrams) flashed briefly on a computer screen. Each trigram is followed by an interval that varies from 40 to 190 ms, at which time a single magnetic pulse is delivered to the occipital skull via a stimulation coil.
In normal participants, MPSA profiles show a U-shaped function, in which the accuracy of reporting the letters is good at short (40 ms) and long (190 ms) intervals, but no better than chance for mid-range (100 ms) intervals (Fig. 1). In contrast, individuals with migraine show a flattened function, whereby perceptual accuracy is relatively maintained at the 100-ms and other mid-range intervals (Fig. 1) (29, 30). Perceptual accuracy differences are also evident between those with episodic and chronic migraine. In chronic migraine, the accuracy of letter reporting is not measurably influenced by transcranial stimulation of the visual cortex at any of the intervals from 40 to 190 ms (Fig. 1) (29, 30). In episodic migraine, the accuracy of letter reporting is decreased at the mid-range intervals and most pronounced at the 100-ms interval, but not as much as in normal individuals (Fig. 1). Thus, there appears to be a continuum of cortical excitability in migraineurs, with episodic migraine associated with more cortical excitability than controls, but chronic migraine associated with yet a greater degree of cortical excitability than either episodic migraine or controls (29).
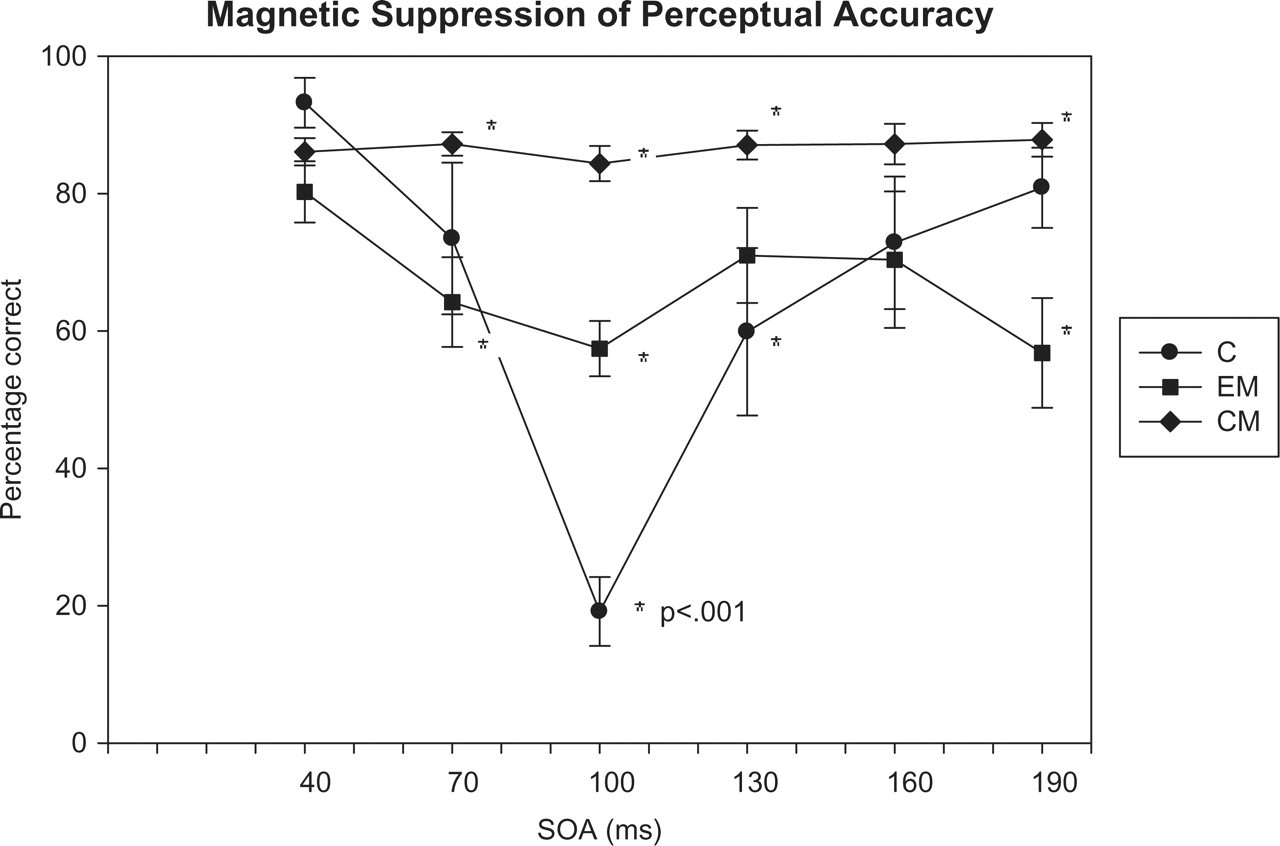
Magnetic suppression of perceptual accuracy profiles for individuals with chronic migraine (CM), episodic migraine (EM) and controls (C). The percentage of trigrams correctly identified is plotted as a function of stimulus onset asynchrony (SOA) or the time in milliseconds between the appearance of the letter trigram and the delivery of the transcranial magnetic pulse. Bars show standard errors. Reprinted from Cao et al. (22) with permission.
The neural basis for suppression of perceptual accuracy in the MSPA test is believed to be the preferential activation of inhibitory neurons by a high-intensity transcranial magnetic stimulus at the 100-ms time point (31). In patients with migraine, it may be more difficult to suppress perception at the mid-range intervals because of increased baseline cortical excitability, which may be caused by impaired intracortical inhibitory mechanisms (32). The greater degree of suppression observed in chronic migraine compared with episodic migraine may be due to more pervasive or persistent cortical hyperexcitability.
Functional imaging techniques
Functional imaging techniques have been used to determine the activity of various brain regions in individuals with migraine, either during or between attacks. We used PET to examine cerebral glucose metabolism (a marker of cellular activity) in 10 individuals with chronic migraine (30). These individuals were subjected to brain scans during the interictal period using 18F-fluorodeoxygluose (FDG) PET (30). We found increased cerebral metabolism in the pons and right temporal cortex and decreased metabolism in several areas bilaterally in the medial frontal, parietal and somatosensory cortices, as well as bilaterally in the caudate nuclei. These findings suggest that normal inhibitory capacity of the cortex is reduced in chronic migraine (30).
Several studies have examined cerebral activation in episodic migraine (20, 33). In a PET study, increased blood flow was noted in the cingulate, auditory and visual association cortices and in the brainstem during the migraine (20). This study noted activation over several planes, slightly lateralized to the left (contralateral to the headache), anterior to the cerebral aqueduct and posterior to the corticospinal tract and cranial nerve nuclei, possibly localized to the locus coeruleus and dorsal raphe nuclei (20). Following injection of the acute migraine medication sumatriptan, which induced complete relief from headache and phono- and photophobia, only the brainstem remained activated. In a subsequent PET study of evoked migraine, activation of the pons (a brainstem structure) was noted both during and following an attack, compared with the pre-migraine baseline (33). However, other structures, including the anterior cingulate, bilateral insula, bilateral cerebellar hemispheres, prefrontal cortex, putamen and rostral medulla, were active during the migrainous attack, but not after it was fully controlled with sumatriptan. This study also found that the side of the pons that was activated corresponded to the laterality of the migraines. Thus, these authors suggest that lateralization of pain in migraine may be due to lateralized dysfunction of the brain, as manifested in the dorsal pons (33).
Only a few studies have examined cerebral activation in chronic migraine (34, 35). In a group of 16 chronic migraineurs who were overusing analgesics, the bilateral thalamus, orbitofrontal cortex, anterior cingulate gyrus, insula/ventral striatum and right inferior parietal lobule showed reduced metabolism using 18FDG-PET, whereas the cerebellar vermis showed increased metabolism (34). All of these areas returned to near-normal glucose uptake after analgesic withdrawal, except the orbitofrontal cortex, which showed a further metabolic decrease. Another PET study has examined the effects of electrical stimulation of the suboccipital cortex via implanted bilateral electrodes in chronic migraineurs (35). Results showed significant changes in regional cerebral blood flow in the dorsal rostral pons, anterior cingulate cortex and cuneus that were correlated with pain scores, and in the anterior cingulate cortex and left pulvinar that were correlated with stimulation-induced paraesthesia scores. Taking into account earlier findings, the authors concluded that the dorsal rostral pons may be a locus of neuromodulation by suboccipital stimulation (35).
Our group has compared the activation of brainstem nuclei in episodic and chronic migraine (36). In this study, we examined episodic migraineurs between attacks and chronic migraineurs during attacks. High-resolution MRI was used to map iron homeostasis as an indicator of brain function; measures included the relaxation rates R2, R2′ and R2∗. Results showed a significant increase in R2′ and R2∗ values in the periaqueductal grey of both migraine groups compared with the control group, but no difference between episodic and chronic migraine groups. We found positive correlations between duration of illness and R2′ for both migraine groups. In the red nucleus and substantia nigra, the chronic migraine group showed a significant decrease in R2′ and R2∗ values compared with the episodic migraine and control groups (36). From these data, we concluded that iron homeostasis in the periaqueductal grey is persistently and progressively impaired in migraineurs, perhaps caused by repeated migraine attacks.
Summary of electrophysiological and functional imaging findings
A summary of electrophysiological and functional imaging findings in episodic vs. chronic migraine is shown in Table 1. Taken together, these findings suggest that certain features of brain dysfunction may be persistently manifest in individuals with chronic migraine, but only during and/or following attacks in those with episodic migraine. In the Magnetic Suppression of Perceptual Accuracy test, those with episodic migraine show an intermediate level of failure to suppress the perceptual images when the magnetic stimulus is applied 100 ms after the visual stimulus, whereas those with chronic migraine show near-complete failure to suppress these images. Similarly, selected brain regions such as the pons may be overactive during an attack in episodic migraine, but may be continuously overactive in chronic migraine. The significant decreases in the MRI values R2′ and R2∗ in the substantia nigra and red nucleus noted in chronic migraine are probably attributable to hyperoxia associated with head pain during an attack and thus may be more frequent in chronic migraine (36). All individuals with chronic migraine in the MRI study were experiencing headache at the time of the scan, whereas all of the episodic migraineurs were between headaches (36). Thus, the electrophysiological and functional imaging evidence suggests a spectrum of migraine in which changes observed in episodic migraine are more pronounced than those in chronic migraine (Table 1).
Summary of electrophysiological and functional imaging findings in episodic vs. chronic migraine

Current models of migraine
As initially suggested by Mathew in 1982 (37), migraine is currently conceptualized (Fig. 2) as a continuum or spectrum disorder, in which episodic migraine may or may not evolve into chronic migraine (38, 39). Approximately 3% of individuals with episodic migraine progress to chronic migraine over the course of a year (38). The clinical evolution appears to occur gradually over months or years, with some individuals progressing from infrequent attacks (2–104 headache days per year) to frequent episodic attacks (105–179 headache days per year), to chronic migraine (38). However, the transition is not always towards greater headache frequency, but may also manifest as a transition to a more episodic pattern of headaches (38, 40).

Overview of features associated with episodic and chronic migraine.
Despite the spectrum conceptualization of migraine, important distinctions between episodic and chronic migraine exist (Fig. 1). The burden of chronic migraine is substantially greater than that of episodic migraine, as evidenced by disability scores (26), ability to work and engage in everyday activities (26) and emergency department visits (41). Furthermore, treatment of chronic migraine is frequently unsuccessful, and no therapies are currently approved by the United States Food and Drug Administration for this condition. Furthermore, pharmacotherapies currently used for treatment of chronic migraine are typically accompanied by intolerable side-effects (42). Because of these distinctions, as well as the pathophysiological differences noted below and the functional correlates observed in electrophysiological and imaging studies, it is critical to focus on chronic migraine as a unique condition, even though its relationship to episodic migraine—primarily as a predisposing condition—is acknowledged.
A number of studies have attempted to define the factors associated with the progression of episodic to chronic migraine. Non-modifiable risk factors include female sex, lower socioeconomic status and marital status (unmarried associated with higher risk) (40). Modifiable risk factors for progression include obesity, snoring, other pain syndromes, previous neck or head injury, stressful life events, caffeine intake and acute headache medication use (40). Sleep disorders, psychopathology (especially anxiety and depression) and gastrointestinal disorders also occur more frequently in those with chronic migraine than in those with episodic migraine (40, 43, 44).
Two recent models of migraine incorporate neurological changes as a central tenet of migraine progression (38, 39). Bigal and Lipton postulate that physiological progression of migraine may be indicated by changes in nociceptive thresholds and pain pathways (allodynia and central sensitization) (38). These authors also note that anatomical progression of migraine may be evident as stroke, white matter lesions or lesions outside the brain (38). Cady and colleagues emphasize the changes in baseline neurological function between episodes of headache (39). These authors note that depression, anxiety, non-head pain, fatigue, gastrointestinal disorders and other somatic complaints that occur after years of episodic migraine may serve as an indicator of chronic transformation (39). In support of these models, chronic migraine has been associated with altered cortical processing of pain (45), frontal lobe dysfunction (46) and vascular malformation in the brainstem (47).
A related finding in migraine research that may have implications for chronification is the observation of cutaneous allodynia. Burstein and colleagues reported that 79% of 44 individuals with migraine seen at a medical centre demonstrated a pain response to non-noxious stimuli during a migraine attack (48). Later studies have confirmed this finding and noted its occurrence in individuals with episodic as well as chronic migraine (38, 49, 50). One study found that individuals who met the criteria for episodic migraine with aura or chronic migraine showed a higher frequency of cutaneous allodynia than episodic migraineurs without aura (49); another study reported a tendency of chronic migraineurs to have more severe cutaneous allodynia than episodic migraineurs, although the study did not appear to separate migraineurs with and without aura (38).
Cutaneous allodynia in migraine is believed to represent sensitization of second-order brainstem trigeminal neurons (48), referred to as central sensitization. It has been suggested that chronic migraine may be due to the chronic sensitization of central pain pathways caused by repeated attacks of migraine (38, 50). Cutaneous allodynia has been associated with refractoriness to triptans (51), and chronic migraineurs frequently do not obtain adequate relief from triptans. However, cutaneous allodynia has also been described in episodic migraine. It will be important to determine the prevalence of cutaneous allodynia in the general population of episodic migraineurs, as opposed to the clinical population that has been studied to date, as the latter may exhibit more severe or frequent migraines that lead them into professional consultation. Another possibility is that chronic migraineurs exhibit more persistent or severe central sensitization than those with episodic migraine. One study has found that several individuals with transformed migraine who were tested on headache-free days exhibited cutaneous allodynia on those days, as well as their headache days (50). Another study found that 29% of individuals with chronic migraine exhibited severe cutaneous allodynia, in contrast to 20% of individuals with episodic migraine (38). These findings may indicate more persistent or pervasive central sensitization in chronic migraine. Coupled with observations of lower pain thresholds in chronic migraine than in episodic migraine (52) and abnormal cortical processing of cutaneous nociceptive input (45, 53), these results support the pathophysiological progression of migraine that involves disruption of central pain mechanisms.
Implications
Clinical, neurological and functional studies of chronic migraine are increasingly suggestive of a pathophysiological state in which the brain exhibits enduring and pervasive alterations, in contrast to the intermittent changes noted in episodic migraine during attacks. As such, chronic migraine is characterized by neurological alterations that are evident even in the absence of headache—as the baseline condition—or as more extreme or severe changes. Because of the disability associated with chronic migraine and the substantial interference of this condition with everyday activities, it is critically important to understand and attempt to prevent chronification and to also to devise effective, tolerable treatments.
Given the differences noted between episodic and chronic migraine in central pain processing, treatments are likely to differ—a concept supported by clinical experience. With episodic migraine, abortive medications such as the triptans are often effective. The potential for chronification suggests that the frequency of headaches should be closely monitored in patients with risk factors for progression, with the goal of preventing the development of chronic migraine (54). For patients who have already developed chronic migraine, novel treatments that do not force patients to choose between pain relief and serious side-effects must be sought (42). We cannot presume that treatments effective for episodic migraine will necessarily be effective for chronic migraine. Investigations into the efficacy of migraine treatments are complicated by a number of factors (54); however, rigorous clinical studies are essential for establishing efficacy. As research continues to progress in these areas, the unique entity of chronic migraine is increasingly recognized as a critical target for effective intervention that has the potential to reduce patient suffering substantially.
Footnotes
Acknowledgements
The author acknowledges that the professional medical writing assistance with this manuscript was funded by Allergan, Inc.
