Abstract
Objective
To quantify photophobia in visual snow syndrome (VSS), a debilitating migraine-associated visual disturbance manifesting with continuous “TV snow-like” flickering dots in the entire visual field and additional visual symptoms, such as photophobia.
Methods
Photophobia was compared between 19 patients with VSS and 19 controls matched for age, sex, migraine and aura using the Leiden Visual Sensitivity Scale (L-VISS).
Results
Patients with VSS had an increased L-VISS-score compared to matched controls [(22.2 ± 5.9 vs. 4.4 ± 4.8; ANOVA, factors VSS and comorbid migraine: Main effect for VSS (F = 100.70; p < 0.001), but not for migraine (F < 0.01; p = 1.00) or the interaction (F = 1.93; p = 0.16)]. An L-VISS-score of 14 identified VSS with a sensitivity and specificity of 95% (Receiver Operating Characteristic analysis, 0.986 ± 0.014, p ≤ 0.001).
Conclusion
Patients with VSS suffer continuously from photophobia at a level similar to chronic migraineurs during attacks. Although migraine and VSS share dysfunctional visual processing, patients with VSS might be more severely affected.
Introduction
Patients with visual snow syndrome (VSS, ICHD-III A1.4.6) suffer from continuous TV-static-like tiny flickering dots in the entire visual field; that is, visual snow (VS) and additional visual symptoms, such as palinopsia and photophobia (1,2). The syndrome is highly disabling due to the continuous presence and the lack of treatment options (3). Its pathophysiology is unclear, with some overlap with migraine and aura (2). The clinical picture of VS, palinopsia, enhanced entoptic phenomena and photophobia (2), hypermetabolism of the lingual gyrus (4), and increased latency of N145 in visual evoked potentials (5) suggest impairment of higher order visual cortex function, although there is also evidence of a more proximal dysfunction (6).
Photophobia, that is, “normal” light causing discomfort in the eye or head (7), is a hallmark not only of VSS (2), but also of migraine attacks (1), where it can be attributed to a dysfunction of the extra striate visual cortex (8), or the thalamus (9), or both. Studying photophobia in VSS might improve our understanding of the common basis of VSS and migraine with possible implications for therapy in the future. Here, we used the validated Leiden Visual Sensitivity Scale (L-VISS) to investigate whether the extent of photophobia differs between VSS patients and controls.
Patients and methods
The study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of the Ludwig Maximilians University Munich (227-15). All patients gave written informed consent.
Subject recruitment
Subjects were recruited from 2015 to 2017 using advertisements in social media with support from the self-help group “Eye on Vision Foundation” (www.eyeonvision.org). After being contacted by patients, we explained the study over the phone and assessed the inclusion criteria (age ≥18 years and presence of VS syndrome, subtype black and white) (2) and the exclusion criterion (history of illicit drug use). Eligible patients were invited to a visit at our out-patient clinic, where they all had a neurological exam and standard visual evoked potentials (VEPs) to exclude anterior visual pathway pathology. Controls were recruited from the outpatient headache clinic or among co-workers from our hospital. They were matched for age, sex, migraine and aura (1), and did not have VS. We did not specifically address other visual symptoms and did not perform VEPs in controls. In patients with comorbid migraine, frequency of headache was approximated using Migraine Disability Assessment part A (MIDAS-A) (10), and severity was assessed using MIDAS or Headache Impact Test-6 (HIT-6) (11), or both. Headache diaries were not used.
Measurement of visual sensitivity
Visual sensitivity was measured using Leiden Visual Sensitivity Scale (L-VISS), a nine-item, five-point Likert-type response scale validated in migraine patients (12,13). Subjects with comorbid migraine had been free of headache for at least 48 hours before filling in the questionnaire. No subject reported having a migraine attack within the 48 hours following study participation.
Statistical analysis
Statistical analysis was done using IBM SPSS Statistics Version 25 (IBM Corp., released 2017, Armonk, NY). Statistical significance was assumed at p ≤ 0.05 (two-tailed). Normal distribution was assessed using Kolmogorov-Smirnov test. Group characteristics were compared using chi-square test, t-test, and Mann-Whitney U test as appropriate. Group differences in L-VISS-score were assessed with ANOVA, using VSS and comorbid migraine as factors. For the comparison of pre-specified subgroups stratified for migraine and aura comorbidity, t-test was used. Values are expressed as mean ± standard deviation (SD), or median (25th; 75th percentile). Cut off values to differentiate between VSS and controls were calculated using Receiver Operating Characteristic (ROC) curve analysis.
Data availability statement
Anonymized data will be shared by request from any qualified investigator.
Results
Subjects
Characteristics of study population including additional visual symptoms in patients with VSS (not present in control population).

One patient had isolated migraine aura without headache.
HIT-6: Headache Impact Test (VSS: n = 18, controls: n = 16); MIDAS: Migraine Disability Assessment Score (VSS: n = 18, controls: n = 19); MIDAS-A: MIDAS part A (number of headache days per 3 months; VSS: n = 18, controls: n = 19); BFEP: blue field entoptic phenomenon.
Visual sensitivity
Patients with VSS had an increased L-VISS-score compared to age-, sex-, migraine- and aura-matched controls (22.2 ± 5.9 vs. 4.4 ± 4.8, Figure 1(a)). In ANOVA, there was a main effect of VSS (F = 100.70; p < 0.001), but not of comorbid migraine (F < 0.01; p = 1.00), and no interaction between the two (F = 1.93; p = 0.16). After stratification for the presence of migraine and aura, all VSS subgroups had increased L-VISS-scores compared to the respective control group: No comorbid migraine (VSS vs. controls: 24.3 ± 7.6 vs. 2.2 ± 3.7; T = 6.4; df = 10; p < 0.001), comorbid migraine without aura (22.3 ± 4.1 vs. 4.0 ± 2.7; T = 7.7; df = 8; p < 0.001), comorbid migraine with aura (20.1 ± 5.8 vs. 6.1 ± 5.9; T = 4.8; df = 14; p < 0.001; Figure 1(b)).
Enhanced visual sensitivity in visual snow syndrome (VSS) compared to matched controls (C).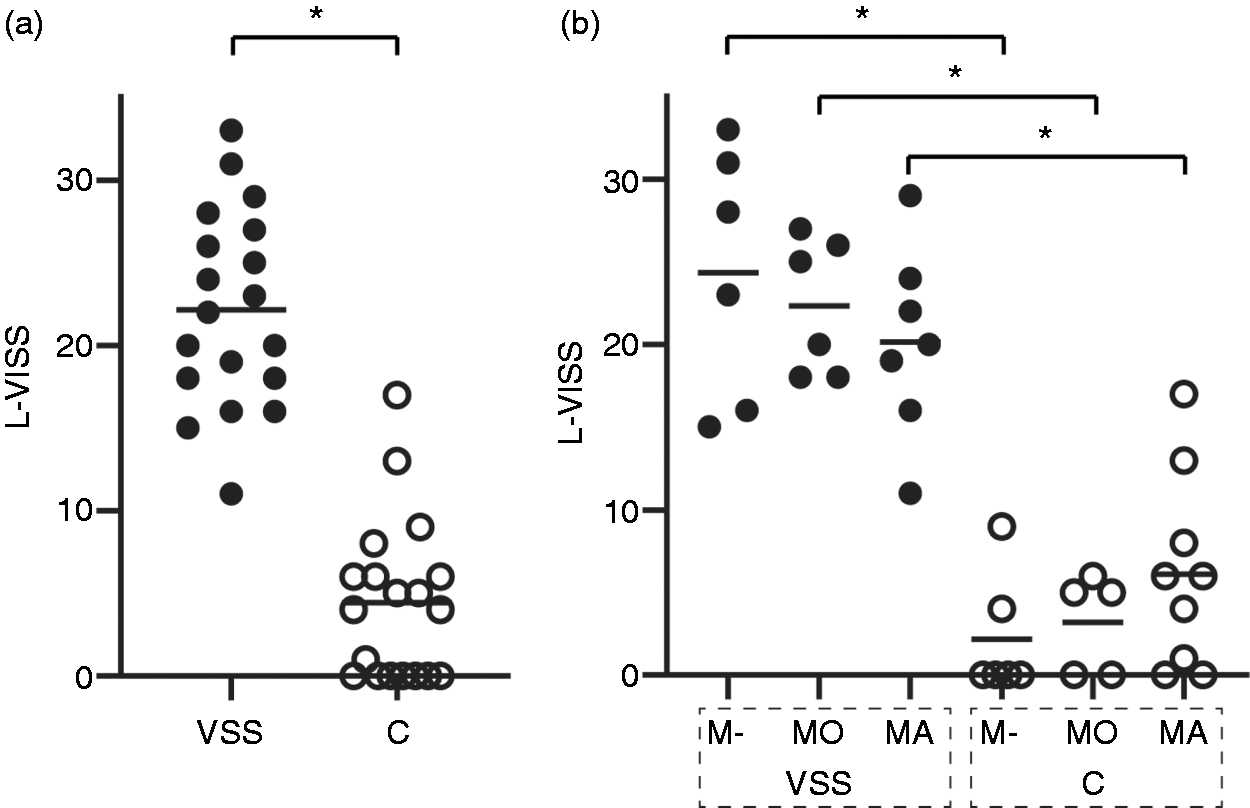
Receiver Operating Characteristic (ROC) analysis between VSS and controls showed an area under the ROC curve of 0.986 ± 0.014 (p ≤ 0.001). An L-VISS-score of 14 points differentiated best between both groups with a sensitivity and specificity of 95% each.
Discussion
Patients with VSS have increased visual sensitivity measured with the Leiden Visual Sensitivity Scale (L-VISS) independent of comorbid migraine and aura. The L-VISS is a self-report instrument developed to quantify visual sensitivity to light (photophobia) on an almost continuous linear scale. It has excellent internal consistency, test-retest reproducibility, and correlates well with established psychophysical tests (pattern glare and light discomfort test), making it an important method for quantifying visual sensitivity (12).
While the L-VISS-score in our control group (4.4 ± 4.8) was in the range of healthy controls (3.6 ± 2.8) and interictal migraineurs (11.3 ± 5.4) from the Perenboom study (12), patients with VSS (22.2 ± 5.9) almost reached values found in patients with chronic migraine with aura during the attacks (25.8 ± 7.9) (12). Since all subjects with comorbid migraine were studied in the interictal phase, a bias due to assessment of VSS patients during migraine attacks is highly unlikely. Further, the group differences were not driven by migraine severity since both groups were similarly affected by migraine according to HIT-6 and MIDAS scores. The extent of sensitivity to light in patients with VSS reaches a level similar to that of migraineurs during their headache attacks, when patients typically withdraw from daily routine into dark places. The relevant difference is that photophobia is continuous in VSS and restricted to attacks in migraine. This study quantifying one of the major symptoms of VSS is relevant for daily clinical practice. It relates the suffering of patients with VSS to the suffering of migraine and emphasises the impact of VSS on visual function.
A previous study demonstrated that comorbid migraine worsens VSS by increasing the risk of having additional visual symptoms, such as palinopsia or photophobia (4). This might suggest that VSS patients with comorbid migraine might also have higher levels of photophobia than VSS patients without migraine. This hypothesis was not confirmed by our data showing that the visual sensitivity measured in L-VISS is excessive in VSS independent from comorbid migraine or aura. Instead, comorbid migraine might increase the risk of having photophobia in VSS (4), but, when present, the suffering from visual sensitivity is severe but uncoupled from migraine. This is important for our understanding of the interaction of migraine and VSS. Migraine might increase the risk of developing VSS with additional symptoms, but VSS may not follow a migrainous modulation. This is supported by VSS symptoms being constant without fluctuations during migraine attacks (2), and by the tenacious resistance to migraine pharmacological therapy (3).
The mechanism of VSS remains enigmatic. In migraine, photophobia has been attributed to thalamic or thalamocortical dysfunction (9) and to hyperactivity or hyperresponsiveness of the visual cortex (8). In migraine, such hyperresponsiveness can be reduced by the migraine prophylactics topiramate (14) and beta-blockers (15) or by the antidepressant fluoxetine (16). In our study, only patients with VSS were on migraine prophylaxis for the treatment of VSS. Assuming a correlation between hyperresponsiveness and L-VISS score (12), this would result in a reduction of L-VISS. In contrast, L-VISS score was increased in VSS, suggesting that a bias from treatment with migraine prophylactics is unlikely. For VSS, hypermetabolism of the visual association cortex (4), alterations of visual evoked potentials (5), and a potential thalamocortical dysrhythmia (17) suggest involvement of mechanisms similar to migraine. Understanding how these mechanisms are differently conducted in both conditions, and how the primary visual cortex is involved (6), might be necessary to generate hypotheses on how to treat VSS.
The limitations of this study are the small sample size in the subgroups stratified for comorbid migraine and the approximation of migraine burden by using scales instead of headache diaries. The prevalence of VSS is unknown and so is the sex distribution. Patients were recruited based on their interest in participating without aiming at a male-female ratio typical for migraine. Therefore, it remains to be determined if there is a bias resulting from the male predominance in our study. Although excessive visual sensitivity in VSS is independent from comorbid migraine, the direct effect of comorbid migraine or aura on visual sensitivity in VSS could not be assessed and would require future studies using larger numbers of subjects, ideally without migraine prophylactic therapy.
Conclusion
Increased sensitivity to light in patients with visual snow syndrome is independent from comorbid migraine or aura. The suffering from light sensitivity is continuous in VSS and in the range of photophobia during migraine attacks. The uncoupling of light sensitivity from migraine might explain the failure of migraine preventives in VSS.
Clinical implications
This study quantifies photophobia in patients with visual snow syndrome. Photophobia is continuous at a level comparable to migraineurs during attacks, emphasizing the disability caused by visual snow syndrome. Excessive photophobia in visual snow syndrome is independent from comorbid migraine or migraine aura.
Footnotes
Author contributions
Conception and design of the study: OE, CJS. Acquisition and analysis of data: OE, RR, CJS. Drafting of a significant portion of the manuscript: OE, RR, AS, CJS.
Acknowledgements
We thank all patients who have taken part in the study. The study was supported by the self-help group for visual snow (Eye On Vision Foundation) by communicating the study to patients.
Ethics or Institutional Review Board approval
The study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of the Ludwig Maximilians University Munich (227-15). All patients gave written informed consent.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: OE reports grants from Friedrich-Baur Foundation during the conduct of the study. RR reports personal fees and/or other from Allergan, Novartis, Teva Pharmaceuticals, Hormosan outside the submitted work. AS reports personal fees from Allergan, Bayer, Sanofi, Desitin, Electrocore, Eli Lilly, Teva Pharmaceuticals, and grants from German Research Council, Kröner-Fresenius Foundation, Ludwig-Maximilian University, and Friedrich-Baur Foundation outside the submitted work. CJS reports grants from Deutsche Migräne- und Kopfschmerzgesellschaft, Eye on Vision Foundation, Baasch Medicus Foundation during the conduct of the study; personal fees from Novartis, Eli Lilly, Allergan, Almirall, Amgen, and personal fees and other from Teva Pharmaceuticals outside the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Deutsche Migräne- und Kopfschmerzgesellschaft (www.dmkg.de): grant to OE and CJS, Eye on Vision Foundation (![]() ): grant to OE and CJS, Baasch Medicus Foundation: grant to CJS, Friedrich-Baur Foundation: grant to OE and CJS.
): grant to OE and CJS, Baasch Medicus Foundation: grant to CJS, Friedrich-Baur Foundation: grant to OE and CJS.
