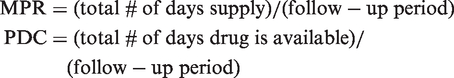Abstract
Background
Chronic migraine (CM) is a disabling disorder characterized by ≥15 headache days per month that has been shown to significantly reduce quality of life. Migraine-prevention guidelines recommend preventive medications as the standard of care for patients with frequent migraine. The aim of this study was to assess adherence to 14 commonly prescribed oral migraine-preventive medications (OMPMs) among patients with CM.
Methods
Retrospective claims analysis of a US claim database (Truven MarketScan® Databases) was queried to identify patients who were at least 18 years old, diagnosed with CM, and initiated an OMPM (antidepressants, beta blockers, or anticonvulsants) between January 1, 2008 and September 30, 2012. Medication possession ratios (MPR) and proportion of days covered (PDC) were calculated for each patient. A cutoff of ≥80% was used to classify adherence. The odds of adherence between OMPMs were compared using logistic regression models.
Results
Of the 75,870 patients identified with CM, 8688 met the inclusion/exclusion criteria. Adherence ranged between 26% to 29% at six months and 17% to 20% at 12 months depending on the calculation used to classify adherence (PDC and MPR, respectively). Adherence among the 14 OMPMs was similar except for amitriptyline, nortriptyline, gabapentin, and divalproex, which had significantly lower odds of adherence when compared to topiramate.
Conclusion
Adherence to OMPMs is low among the US CM population at six months and worsens by 12 months.
Introduction
Migraine is an episodic neurological disorder affecting 12% of Americans and millions worldwide (1). Women are three times more likely to be affected, with prevalence peaking between the ages of 25 and 55 years (2). The frequency of headache days are used to classify two main subtypes of migraine: episodic and chronic. Episodic migraine (EM) has been generally characterized by the presence of up to 14 headache days per month. Chronic migraine (CM) is defined as ≥15 headache days per month with ≥8 days per month meeting criteria for migraine and/or for which a migraine-specific medication was used (e.g. triptans or ergot), for more than three months (3). In general, the prevalence of CM is typically estimated at 1.4% to 2.2% among adults worldwide and approximately one out of 100 adults in the United States (US) (4). CM is a disabling disorder that has been shown to significantly reduce quality of life and leave many patients unable to perform daily activities (5,6). It has also been associated with significant resource utilization including increased health care provider visits, emergency department visits, and diagnostic testing (7,8).
Patients with frequent headaches, such as those suffering from CM, may benefit from oral migraine-preventive medications (OMPMs). Guidelines by the American Academy of Neurology (AAN) and the American Headache Society recommend first-line oral prophylaxis with propranolol, timolol, amitriptyline, divalproex, or topiramate (9). However, data suggest that OMPMs require adherence to daily dosing regimens in order to adequately reduce the burden of migraine (10). Adherence has been identified as a significant health problem in the US and it is estimated that non-adherence is associated with more than $290 billion in additional health care costs per year (11). Low adherence has been shown to cause difficulty in treating numerous chronic conditions and non-adherence has been directly linked with poor health outcomes in hypercholesterolemia, hypertension, intestinal disease, and sleep disorders (12,13).
Adherence to OMPMs has been studied in EM patients using a variety of methods. A recent systematic review describes the methods that have been applied in estimating adherence to OMPMs (14). This review highlighted two studies that investigated adherence to oral migraine medications using claims databases. In one of the studies, Lafata et al. reported adherence to OMPMs among 2517 migraine patients from a single health plan in Michigan between 2000 and 2001 (15). In the second study, Berger et al. used claims data from 2003–2005 to investigate adherence to OMPMs among 5187 migraine patients sampled from a database that included more than 1.6 million privately insured individuals from the US (16). At the time of either of these studies CM was not yet operationally defined, and hence claims data could not distinguish between CM and EM. To date, no studies have examined adherence to OMPMs among the CM population using real-world data.
The aim of this study was to describe the CM population using a US insurance claims database and to assess adherence to OMPMs among this population between January 1, 2008 and September 30, 2012.
Methods
Data were collected from Truven Health MarketScan® Research Databases (2007–2012), which contain inpatient, outpatient, and pharmacy claims for patients covered by commercial, Medicare Supplemental, and Medicaid insurance plans in the US (17). The inpatient and outpatient claims databases include procedure and visit level details from medical claims such as International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM) diagnosis codes, Current Procedural Terminology medical procedure codes, dates of service, and variables describing the financial expenditures both of the patient and his or her insurance plan. The pharmacy claims database provides prescription dispensing details that include National Drug Code and generic identifier of the drug dispensed, date dispensed, quantity and days’ supply, and payments made for each claim. A separate eligibility and demographics file provides additional information about each individual such as age, gender, insurance plan type, employment status and classification, geographic location, and enrollment status by month. All patient-level identifiers are encrypted to protect patient privacy, and compliance with the Health Insurance Portability and Accountability Act of 1996 (HIPAA) was ensured by the data administrator, Truven Health Analytics. As the dataset was created by a third-party analyst and confirmed to be HIPAA compliant, the University of Washington Human Subjects Division Institutional Review Board (IRB) determined that the study protocol did not meet the federal definition of “human subjects research” and was exempt from IRB review.
Sample selection
Individuals 18 years of age and older who were diagnosed with CM and who initiated an OMPM between January 1, 2008 and September 30, 2012, were selected. The beginning of the look-back period was chosen because the diagnosis code specific to CM (346.7) was added with the 2008 revision of the ICD-9-CM (18). The analysis included the following commonly prescribed OMPMs: tricyclic antidepressants (TCAs) amitriptyline and nortriptyline; selective serotonin reuptake inhibitors (SSRIs) citalopram, sertraline, fluoxetine, and paroxetine; serotonin-norepinephrine reuptake inhibitor (SNRIs) venlafaxine; beta blockers (BBs) propranolol, metoprolol, nadolol, and atenolol; and anticonvulsant medications gabapentin, topiramate, and divalproex. Decisions regarding which medications to include in the analyses were based on current migraine-prevention guidelines and previously published studies of survey data in CM patients and claims data in EM patients that detail the most commonly used OMPMs among patients with migraine (9,16,19). The index date was established by the first pharmacy claim for an OMPM filled among patients with a diagnosis of CM.
Patients taking more than one OMPM on the index date or patients who did not have continuous enrollment for six months prior to the index date and 12 months post-index date were excluded from the analysis. In an effort to control for confounding resulting from the use of the OMPMs included in this analysis for treatment of chronic conditions other than migraine, the following patients were excluded: patients whose first pharmacy claim for a BB was within 12 months after a diagnosis of congestive heart failure (ICD-9-CM 398.91, 402.01, 402.11, 402.91, 404.01, 404.03, 404.10, and 404.XX) or hypertension (ICD-9-CM 401.XX and 405.XX), whose first pharmacy claim for an antidepressant was within 12 months after a diagnosis of depression (ICD-9-CM 290.21, 292.84, 296.XX, 298.0, 300.4, 309.0, 309.1, and 311.0), or whose first pharmacy claim for an anticonvulsant was within 90 days of a seizure diagnosis (ICD-9-CM 345.XX).
Adherence measurement
Adherence is defined as the extent to which a patient follows prescribed directions with respect to timing, dose, and frequency of a medication (19,20). There are a variety of ways to estimate adherence, including biological assays, pill counts, electronic monitoring, claims data analysis, or patient reports using surveys or interviews (21). Claims database analyses are the most commonly used methods, as large studies can be conducted quickly and at relatively low cost (22). Medication possession ratio (MPR) and proportion of days covered (PDC) are two well-cited calculations used to estimate adherence from claims (23,24). Each calculation uses a different method for summing the number of days a person is considered to be on therapy. The number of days on therapy is then divided by the total number of days during follow-up. Although MPR is more commonly cited, recent literature suggests that PDC may possibly be a more accurate method for estimating adherence (25). Although cut-points for classifying adherence using MPR and PDC are arbitrary, current evidence points to 80% as an optimal cut-point for either of these methods (26).
To ensure that our conclusions were not influenced by the choice of a particular calculation, adherence to OMPMs were estimated using both MPR and PDC equations. A fixed follow-up of six and 12 months (183 and 365 days, respectively) was established as the denominator in each formula, which are as follows:
Claims with days’ supply of zero were excluded. The last fill days’ supply was adjusted to include only the days up to the end of the follow-up. Because of overlapping days’ supplies, MPR calculations could generate values higher than 1. For cases where MPR was greater than 1, values were truncated at 1. Adherence rates were reported as the proportion of patients who have ≥80% MPR or PDC at six and 12 months, stratified by each medication.
Statistical analyses
Demographic and clinical characteristics were analyzed using descriptive statistics. Continuous data were reported using means and standard deviations (SDs) while categorical data were reported using counts and proportions. Bivariate analyses, using t-tests for continuous data and chi2 tests for categorical data, were conducted to compare patient demographics by the OMPM initiated as well as adherence status. Unadjusted and adjusted comparisons of adherence among the various OMPMs were made using logistic regression models. The dependent variable was adherence (defined by MPR or PDC ≥80%) and the primary independent variable was the OMPM medication specified in the claims data. Decisions about which covariates to include in the adjusted models were made using clinical, epidemiological, and statistical methods. Clinically, each covariate was considered for its potential association with adherence to determine whether adjusting for that covariate would be necessary. This was performed using targeted literature reviews and the clinical expertise of the authors. The covariates were also analyzed using a causal pathway framework in order to determine their relevance to CM management and to the outcome of adherence. Lastly, the baseline descriptive statistical results, as well as the results from the comparison between adherent and non-adherent patients, were used to analyze each covariate and assess whether the differences in these covariates were significant within our sample. The final decision to include or exclude each covariate in the model was informed by the composite of these three evaluations.
Because of the number of hypotheses being tested, a sensitivity analysis was performed by setting the alpha level for statistical significance for the regression models at 0.01 (99% confidence interval) using the Bonferroni correction. Statistical analyses were conducted using statistical software package STATA 13 (College Station, TX) (27).
Results
Demographics
Of the 121 million unique lives represented in the MarketScan databases between 2007 and 2012, 74,870 patients were identified with a CM diagnosis between 2008 and 2012, of whom 36,720 (49%) initiated an OMPM. A total of 8688 patients met the inclusion/exclusion criteria and were thus included in the analyses. Figure 1 describes the patient selection process in detail. The majority of patients were female with a mean age of 40 years (SD = 12). A majority of patients were working full time (59%) and were enrolled in a preferred provider organization (PPO) health insurance plan (58%). Patients from all regions of the US were included, with the largest proportion from the South (41%) and the smallest from the Northeast (12%). Approximately 84% of patients resided in a metropolitan area during the analysis period. The CM patients in this sample had significant comorbidities, of which the most common were headache disorders other than migraine (54%), cancer (22%), hypertension (18%), depression (18%), sleep disorder (11%), and gastroesophageal reflux disorder (11%). Table 1 provides a complete list of baseline characteristics and comorbidities stratified by the therapeutic class of the OMPM initiated. Online Supplementary Appendix 1, reports baseline demographics and comorbidities by individual OMPM. Bivariate analyses showed significant variation among the OMPMs with respect to baseline demographics and all comorbidities except allergic rhinitis, congestive heart failure, irritable bowel syndrome, and renal failure.
Patient selection process. Baseline demographics and clinical characteristics. COBRA: Consolidated Omnibus Budget Reconciliation Act; GERD: gastroesophageal reflux disease; HMO: health maintenance organization; MSA: metropolitan statistical area; n: number; POS: point of service; PPO: preferred provider organization; SD: standard deviation. Employment status and employee classification refer to the employee who is enrolled in the plan.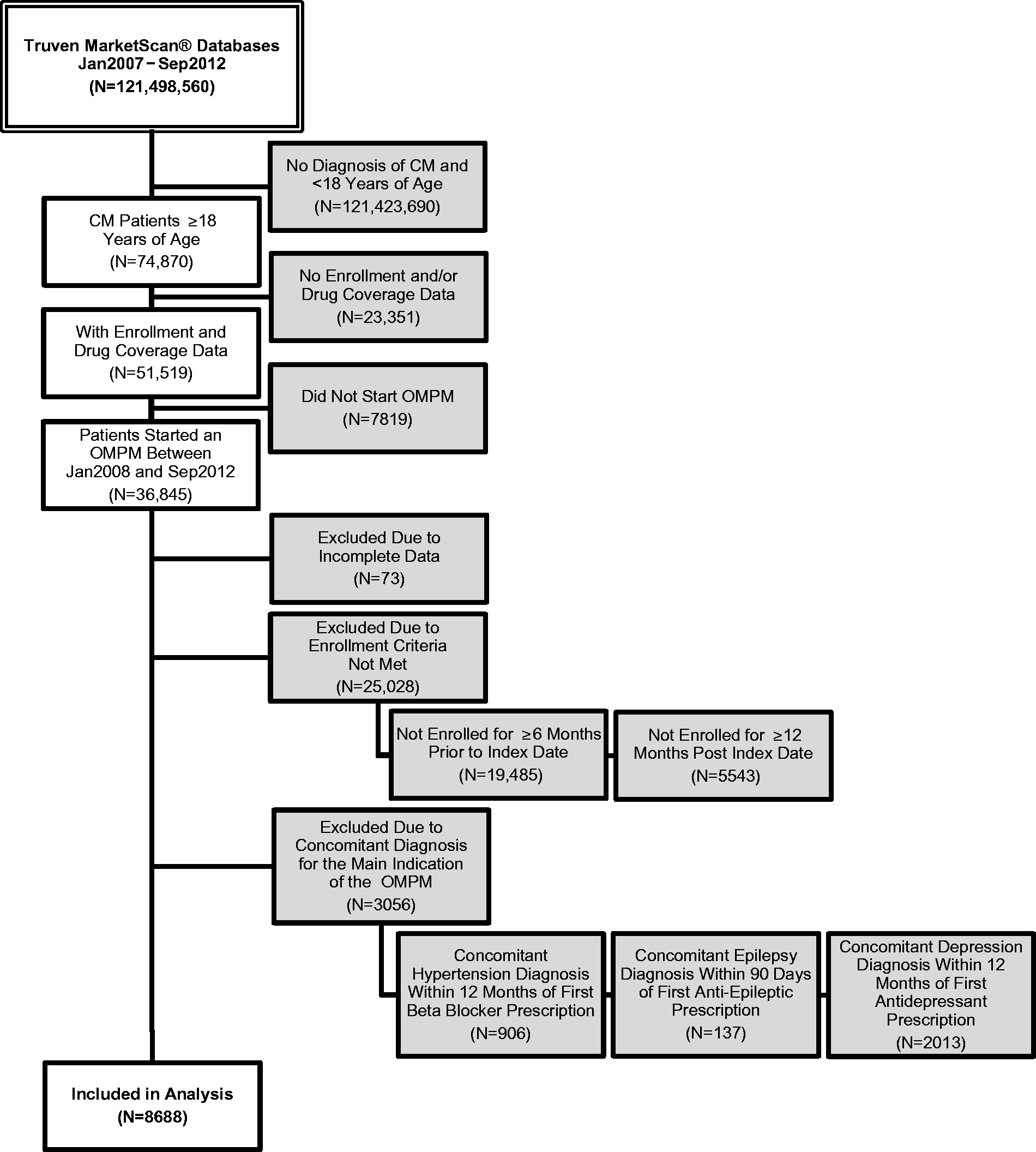

Adherence
Adherence was generally low among all OMPMs, with approximately 29% of patients adherent at the end of six months and 20% at 12 months when using ≥80% MPR to classify adherence (Figure 2). Using ≥80% PDC to calculate adherence rates yielded even lower estimates, with 26% and 17% remaining adherent at six and 12 months follow-up, respectively. An additional figure, available (online Supplementary Appendix 2), features adherence rates for each individual OMPM at six and 12 months. Bivariate analyses of demographics between adherent and non-adherent groups (online Supplementary Appendix 3) revealed that, except for sex and total out-of-pocket cost, demographics differed significantly among adherent and non-adherent patients. However, these differences were small between the two groups and not clinically meaningful for almost all of the variables. One variable that was consistently different (and possibly clinically meaningful) was the difference in proportion of patients with PPO and HMO. The adherent group, regardless of time horizon or calculation methodology, had a larger proportion of primary beneficiaries enrolled in PPO plans while the non-adherent group had a larger proportion of primary beneficiaries enrolled in HMO plans.
Rate of adherence to OMPMs, by class, among insured patients with chronic migraine. OMPM: oral migraine-preventive medications; MPR: medication possession ratio; PDC: proportion of days covered.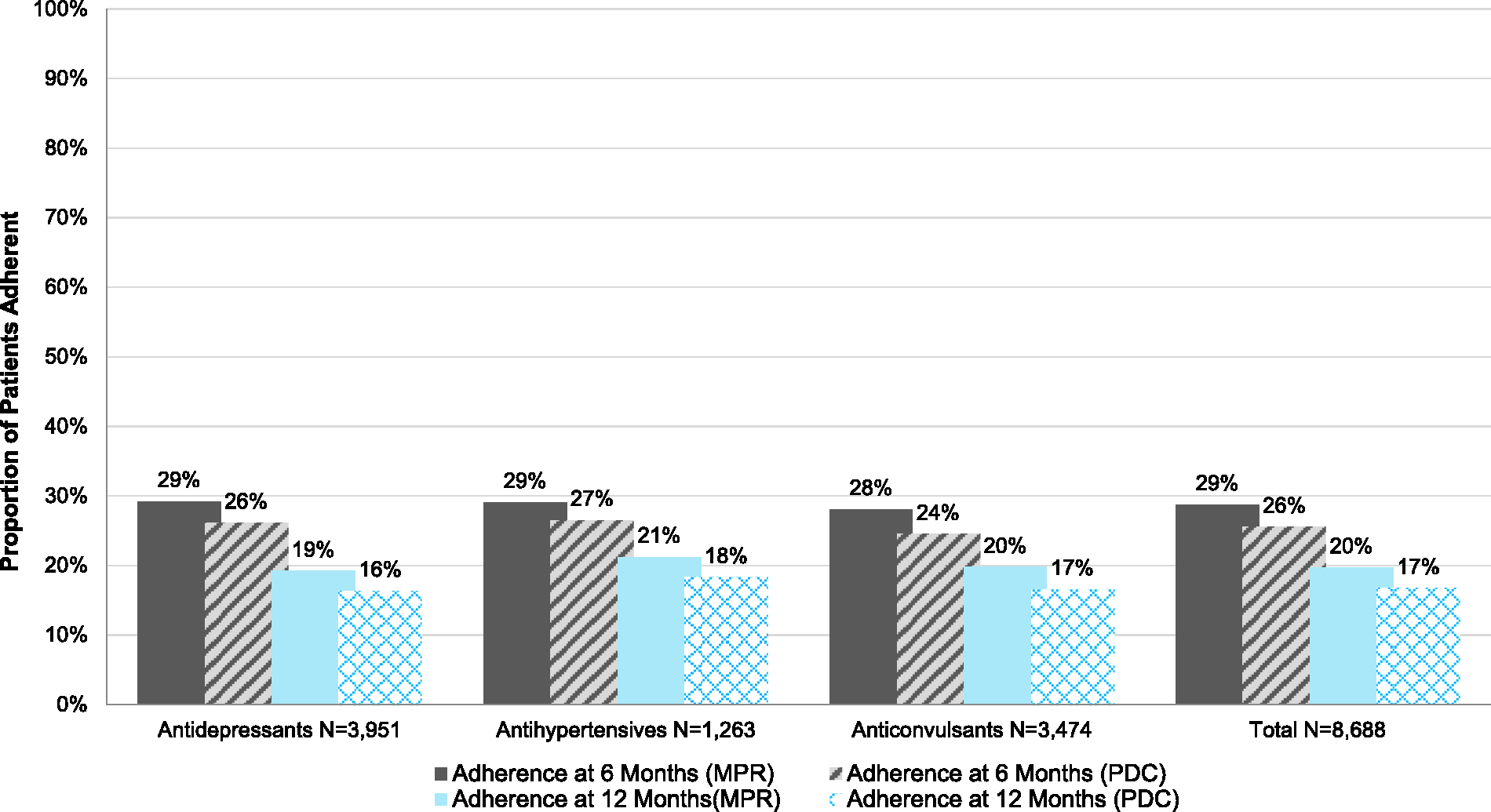
Odds of adherence at 6 months, stratified by oral migraine-preventive medication and method of calculation.

Adjust: adjusted; CI: confidence interval; MPR: medication possession ratio; PDC: proportion of days covered; Ref.: reference drug; SNRI: serotonin-norepinephrine reuptake inhibitor; SSRI: selective serotonin reuptake inhibitor; TCA: tricyclic antidepressants; Unadjust: unadjusted.
Adjusted for demographics: age, sex, employee status, employee classification, region, metropolitan area vs. rural, plan type, total prescription cost; and comorbidities: anxiety, bipolar, cancer, coronary heart disease, depression, diabetes, epilepsy, hypertension, headache, sleep disorder.
Odds of adherence at 12 months, stratified by oral migraine-preventive medication and method of calculation.

Adjust: adjusted; CI: confidence interval; MPR: medication possession ratio; PDC: proportion of days covered; Ref.: reference drug; SNRI: serotonin-norepinephrine reuptake inhibitor; SSRI: selective serotonin reuptake inhibitor; TCA: tricyclic antidepressants; Unadjust: unadjusted.
Adjusted for demographics: age, sex, employee status, employee classification, region, metropolitan area vs. rural, plan type, total prescription cost; and comorbidities: anxiety, bipolar, cancer, coronary heart disease, depression, diabetes, epilepsy, hypertension, headache, sleep disorder.
Discussion
In this study, we analyzed the adherence rates of 14 commonly prescribed OMPMs among the US CM patient population. This analysis was conducted using data from a large US health care database that included commercial, Medicare Supplemental, and Medicaid data from inpatient, outpatient, and pharmacy claims. Regardless of the medication used, adherence to OMPMs was generally poor both at six and 12 months of follow-up. Although the reasons for non-adherence are not captured in claims data, previously published studies suggest that low adherence may be attributed to a number of factors, including side effects and/or lack of efficacy of the OMPM (14,19,28). Furthermore, treatment guidelines by the AAN also point out that only a handful of the OMPMs available actually have good clinical evidence of their efficacy in preventing migraine headaches. Medications such as gabapentin and fluoxetine, for example, are not recommended for the use of migraine prevention because of “inadequate or conflicting data,” yet we see both of these medications prescribed in our current sample as well as in previous studies (9,16,19).
There has been some debate regarding the methods used to calculate adherence from claims data. It has been suggested that PDC is more accurate than MPR because it does not overestimate adherence. In our study, adherence rates were calculated using both methods. We found that PDC resulted in slightly lower estimates of adherence both at six and 12 months, suggesting that MPR may indeed overestimate adherence. Although some of the outcomes varied slightly, overall the two methods produced similar results in our regression analyses.
While our study is the first to investigate adherence among patients with CM, the results may still be compared to those reported for EM patients from similar published studies. A comparison of our results to those reported by Berger et al. (2012) (16) and Lafata et al. (2010) (15) revealed equivalent OMPM adherence rates as described by Berger (29% at six months for both), but considerably higher adherence rates, as reported by Lafata (56% at 12 months compared to 20% in the present study). These results are surprising, as we would expect better adherence among patients with CM compared to patients with EM. Episodic migraine patients may reject the idea of taking a pill every day independently from the occurrence of their attacks, because they may feel it is unnecessary (29,30). We would expect such an objection to be less common among patients with CM because of the chronic nature of their condition, which would theoretically result in higher rates of adherence. In addition, patients with CM in our study had a larger burden of comorbidities compared to EM patients in these previously published studies. Since OMPMs have indications for many of these comorbidities, in theory, patients with CM should have better adherence since the OMPMs may be providing relief of the comorbid disease symptoms as well; however, our findings indicate otherwise. Alternatively, the findings from our comparison may be reflective of the lack of efficacy associated with OMPMs in preventing migraine headaches. Patients with more frequent headaches would be more likely to notice treatment failure and be quicker to react to it than patients with EM, which would account for these findings.
Our regression analysis models were used to compare the odds of adherence among the various OMPMs. These analyses suggest that several of the OMPMs were associated with significantly lower odds of adherence when compared to the referent OMPM, topiramate. Lafata et al. conducted similar analyses and found that adherence did not vary significantly among the studied medications in their sample (15). This incongruity may be due to their significantly smaller sample size and limited generalizability to regions outside of southeast Michigan.
Since CM is a chronic disease, data suggest that OMPMs should be taken daily in order to adequately prevent migraine attacks. Therefore, it may also be reasonable to compare adherence rates among patients with CM with those of other chronic conditions requiring similar daily oral medication therapy. Yeaw et al. used a national database of medical claims to compare adherence among different chronic diseases over a 12-month follow-up period using PDC to calculate adherence (11). Findings of Yeaw’s study ranged from a 35% adherence rate for overactive bladder medications, to 72% for oral glycemic control medications, indicating that adherence across all six chronic conditions reported in this study was higher, compared to our finding of 17% adherence (using the comparable PDC calculation) for patients with CM at 12 months of follow-up.
As is the case with large insurance claims database analyses, our study has several important limitations that must be considered when interpreting the results. First, our analyses were based on a number of assumptions. For example, once the medication was picked up from a pharmacy, we assumed that the patients actually ingested their medication as directed by their physicians or at least as indicated by the refill patterns used in the analyses. In addition, since days’ supply was used to calculate adherence, there was an assumption that the physician prescribed the medication correctly and that those directions were correct for the indication of migraine prevention. Furthermore, our analyses assumed that the ICD-9-CM codes contained within the database for each patient accurately described their migraine diagnosis and comorbid conditions. Lack of the latter may lead to misclassification, which is an important limitation in observational studies. Further research should be conducted to validate the use of the ICD-9-CM code for identifying CM patients.
The MarketScan databases have additional limitations. Claims do not, for example, provide any information on disease severity. CM encompasses patients who experience ≥15 headache days per month; however, a patient who experiences 15 headache days per month compared to someone who has 30 headache days per month may have very different attitudes and behaviors related to his or her migraine-preventive medication treatment. Each headache episode may also vary in severity among patients, a fact that is not captured in claims and thus not represented in this dataset. Lastly, MarketScan does not collect patient socioeconomic data such as education, income, race, and smoking status, which are important covariates often included in regression analyses. Although every effort was made to adjust for possible confounding, we were limited to only those variables provided by the database and thus residual confounding most certainly exists.
Finally, our study is the first to use a claims-based analysis to characterize patients with CM and assess OMPM adherence in this population. The sample used in the analysis is representative of commercial, Medicare Supplemental, and Medicaid populations of the entire US and may be generalizable to regions therein. However, this study reflects an insured population and thus the results may not be applicable to uninsured individuals. Additionally, by excluding patients with a disease diagnosis other than CM (e.g. depression if on an antidepressant) and those who received two OMPMs on the initial fill, our results are not necessarily generalizable to all CM patients. However, this represents only a small proportion of our sample and would likely not affect our overall conclusions.
Conclusion
Adherence to OMPMs is low among the US CM population at six months and declines even further by 12 months. Adherence is an important component to successful oral medication therapy and our findings highlight a large gap in care for this highly burdened population. Among the 14 OMPMs analyzed, amitriptyline, nortriptyline, gabapentin, and divalproex were shown to have lower odds of patient adherence at either follow-up interval when compared to topiramate. However, the data presented herein are insufficient to guide treatment on their own; appropriate OMPM selection must be made on a case-by-case basis that takes into account other important characteristics such as safety and efficacy. The therapy decision must ultimately rest with the physician and the patient.
Clinical implications
Regardless of medication used, adherence was low among oral migraine-preventive medications. Amitriptyline, nortriptyline, gabapentin, divalproex, and propranolol have lower odds of adherence when compared to topiramate. Results from these analyses highlight a large gap in therapy for patients who are suffering from chronic migraine; however, further research to understand reasons for non-adherence and to develop strategies to improve adherence are needed.
Footnotes
Conflicts of interest
Dr Hepp is an employee of Allergan Inc. Dr Dodick has received consulting honoraria from Allergan Inc, Amgen, Alder, Arteaus, Pfizer, Colucid, Merck, ENeura, NuPathe, Eli Lilly & Company, Autonomic Technologies, Ethicon, J&J, Zogenix, Supernus, and Labrys; receives honoraria from Sage Publishing as editor of Cephalalgia (Dr Dodick was not involved in the editorial handling of this manuscript); has received honoraria/royalties for publishing with WebMD, UptoDate, Oxford University Press, Cambridge University Press; and has received honoraria for speaking engagements/lectures at IntraMed, Synergy, Universal Meeting Management, Sun Pharma, Starr Clinical, Decision Resources, American Academy of Neurology, American Headache Society, Laval University, Western Virginia University Foundation, Texas Neurological Society, and the Canadian Headache Society. Dr Gillard is an employee of Allergan Inc. Dr Varon is an employee and stockholder of Allergan Inc. Drs Hansen and Devine have nothing to declare.
Funding
This work was supported by Allergan Inc.
Acknowledgment
The authors acknowledge Amy Kuang, PhD, Allergan Inc, for editorial support on this manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.