Abstract
Background
High-frequency repetitive transcranial magnetic stimulation of the left dorsolateral prefrontal cortex (rTMS-DLPFC) is an effective treatment for depression. Preliminary studies indicated beneficial effects of rTMS-DLPFC on pain relief in patients treated for depression, and in patients with chronic migraine.
Methods
In this randomized, double-blind, parallel-group, single-center, proof-of-principle clinical trial, we tested the hypothesis that 23 sessions of active rTMS-DLPFC delivered over eight weeks would be feasible, safe and superior to sham rTMS to decrease the number of headache days in 18 patients with chronic migraine without severe depression. Per-protocol analysis was performed.
Results
rTMS-DLPFC applied over eight weeks was feasible and safe in patients with chronic migraine. Contrary to our primary hypothesis, the number of headache days decreased significantly more in the sham group than in the group treated with active rTMS-DLPFC at eight weeks. Average decrease in headache days was >50% in the sham group, indicating a powerful placebo response. Pain intensity improved in both groups to a similar extent.
Conclusions
Positive results of M1 stimulation in other studies, and the absence of significant benefits of active high-frequency rTMS of the DLPFC in the present study, point to M1 as a more promising target than the DLPFC, for larger trials of noninvasive brain stimulation in patients with chronic migraine.
Introduction
Chronic migraine is a disabling primary headache that typically affects 1.4%–5.4% of the general population (1,2). The only evidence-based therapeutic options are topiramate and onabotulinum toxin (3). Medications used for off-label treatment of chronic migraine are often associated with intolerable side effects.
A neuromodulation strategy that has been preliminarily evaluated for treatment of migraine is repetitive transcranial magnetic stimulation (rTMS) (4). High-frequency rTMS of the left dorsolateral prefrontal cortex (DLPFC) is effective to treat depression (5,6), a condition present in up to 80% of persons with chronic migraine (7,8). Preliminary studies have also supported a beneficial role of rTMS-DLPFC in anxiety disorders (9).
Prefrontal dysfunction is a plausible mechanism in the pathogenesis of chronic migraine. Consistent with this concept, performance in tests of prefrontal function is compromised in patients with chronic migraine (10). In addition, anecdotal migraine improvement and amelioration of self-rated pain were reported in patients treated for depression with left rTMS-DLPFC (11). Decrease in the number of migraine attacks was reported after 12 sessions of active left rTMS-DLPFC, primarily administered as add-on treatment to drug prophylaxis in patients with chronic migraine, compared to sham rTMS (12). It remained to be investigated whether a prolonged course of rTMS-DLPFC would be effective in patients not taking prophylactic drugs that potentially interfere with cortical excitability (13). In addition, whether treatment effects would be restricted to improvement in pain or would extend to benefit affective states and disability remained unknown.
We investigated the hypothesis that 23 sessions of active rTMS-DLPFC would be feasible, safe and superior to sham rTMS to decrease the number of headache days, pain intensity, disability as well as symptoms of depression and anxiety in patients with chronic migraine without severe depression, not taking drugs known to interfere with cortical excitability.
Methods
Participants
Exclusion criteria.

The protocol was approved by the Albert Einstein Israelite Hospital’s Ethics Committee (CEP 08/828) and conformed to ethical standards as described in the Declaration of Helsinki. All patients provided written informed consent to participate. The protocol was registered in clinicatrials.org (NCT01496950).
Baseline measures
After undergoing a telephone screening interview, each eligible subject was instructed to fill out a headache paper diary for four weeks (lead-in phase), to check the ability to properly log information in the diary and to confirm that criteria for chronic migraine were fulfilled.
Randomization, allocation concealment and blinding
Patients were randomly assigned in blocks by the principal investigator with a basic random number computerized generator in a 1:1 ratio to receive a total of 23 sessions of either active or sham rTMS within eight weeks. During the first four weeks, 15 sessions were administered, as previously described for the treatment of depression (5,11). Patients were allowed to continue their usual medical treatments, including stable doses of migraine preventive drugs (restricted to calcium-channel blockers or β adrenergic antagonists).
To ensure anonymity, information about randomization and rTMS procedures was kept in electronic format in password-protected files, accessed only by the researcher who performed rTMS. Patients were informed that they would be randomized to either active or placebo treatment, but were not aware of group assignment or of the study hypothesis. They were also instructed about alternative prophylactic treatments for chronic migraine. The neurologist and the psychologists who evaluated outcomes at baseline and throughout the protocol were blinded to group assignment.
rTMS interventions
All interventions were performed in the TMS laboratory at Hospital Israelita Albert Einstein. In each treatment session, the optimal site of motor stimulation of the left hemisphere was defined as the location where TMS elicited the largest motor-evoked potentials (MEPs) in the right first interosseous dorsalis (FDI) with surface electrodes. The signal was amplified and filtered (10 Hz to 2 kHz) with an electromyography (EMG) and evoked potential measuring unit (MEB-9104 J, Nihon Kohden, Japan). For determination of resting motor threshold (rMT) (15), one TMS pulse was delivered at random intervals of 5±2.5 seconds.
In both groups, 10 Hz rTMS was administered with a figure-eight coil (MC B-70, outer diameter 100 mm, max dB/dt31 kT/s near the coil surface) at 110% of the right FDI rMT with a biphasic MagPro X100 (Alpine Biomed). All patients underwent head magnetic resonance imaging scans (MRIs). We used the MRI-based correction described by Avery et al. (5) to derive an estimated rMT. If the corrected intensity of stimulation surpassed safe intensities of stimulation based on the measured rMT, we opted to keep intensity of stimulation as 110% rMT. The intensity was adjusted in only one patient in the active group, in 13/23 sessions of treatment. In all other patients, intensities were 110% rMT throughout the protocol.
In each session, 32 rTMS trains were administered (train duration 5 seconds, intertrain interval 30 seconds, total of 1600 pulses per session), as previously described for treatment of depression (5,11).
In the active group, the TMS coil was tangentially positioned to the scalp, 5 cm anteriorly to the optimal site for the right FDI in the left hemisphere with the intersection of both wings at a 45-degree angle with the midline, as performed in depression trials (5,6). In the sham group, the coil was held perpendicularly to the vertex.
After the eight weeks of treatment had been completed, patients were asked whether they thought they had been treated with active or placebo rTMS.
Outcome measures
Primary outcome measures
The primary feasibility outcome was in compliance with the interventions. Compliance was measured as number of planned sessions/number of attended sessions × 100 (%).
The primary safety outcome was the proportion of adverse events in the active and sham groups. After each session of treatment, researchers asked all of the participants about adverse events.
The primary efficacy outcome was the number of headache days in the past four weeks, as registered in the headache diary. This outcome was assessed by a neurologist at baseline, after four weeks and after eight weeks of treatment.
Secondary outcome measures
Secondary outcomes were: average pain intensity in the past four weeks, Migraine Disability Assessment (MIDAS) (16), Beck Depression Index (BDI) (17) and State Anxiety Inventory (STAI) scores (18). All secondary outcomes were assessed at baseline, week 4 and week 8. Pain intensity and MIDAS scores were evaluated by a blinded neurologist, and the other outcomes by psychologists.
Statistical analysis
Characteristics of the patients in each of the groups were compared with parametric tests for normally distributed data and with nonparametric tests if otherwise. Compliance with treatment in each group was compared with Mann-Whitney tests. Frequencies of adverse events were compared with Fisher's exact tests. This was a “hypothesis-generating” project and for this reason sample size was not formally determined.
All other endpoints were evaluated using generalized estimating equations (GEE) models. GEE is used to analyze correlated data, particularly when analysis of variance assumptions are not met (19). A Poisson model with a logarithmic link function and an autoregressive correlation matrix was fitted to all outcomes except for “headache days.” For “headache days,” a Poisson model with an identity link function and an autoregressive correlation matrix was used. Two-tailed
Results
Between October 2008 and December 2011, 228 subjects with headache were screened for the study and 18 were included. Figure 1 shows the flow of individuals through the study. Characteristics of the patients are given in Table 2. All participants were women. None of them used any prophylactic drugs within the four weeks prior to inclusion or during the protocol. None of the patients fulfilled criteria for major depression. Patients reported using analgesics/nonsteroidal anti-inflammatory drugs (NSAIDs), triptans, ergot alkaloids or mixed analgesics to treat pain, during the protocol. None of the patients reported using opioid drugs within the four weeks before or throughout the study.
Flow diagram of patients through the trial. The protocol was announced in local media and patients with headache from the community were screened for eligibility. Patients' characteristics. SCID: Structured Clinical Interview for Diagnostic and Statistical Manual for Mental Disorders (SCID-DSM IV). Unpaired Fisher's exact test. Mann-Whitney test.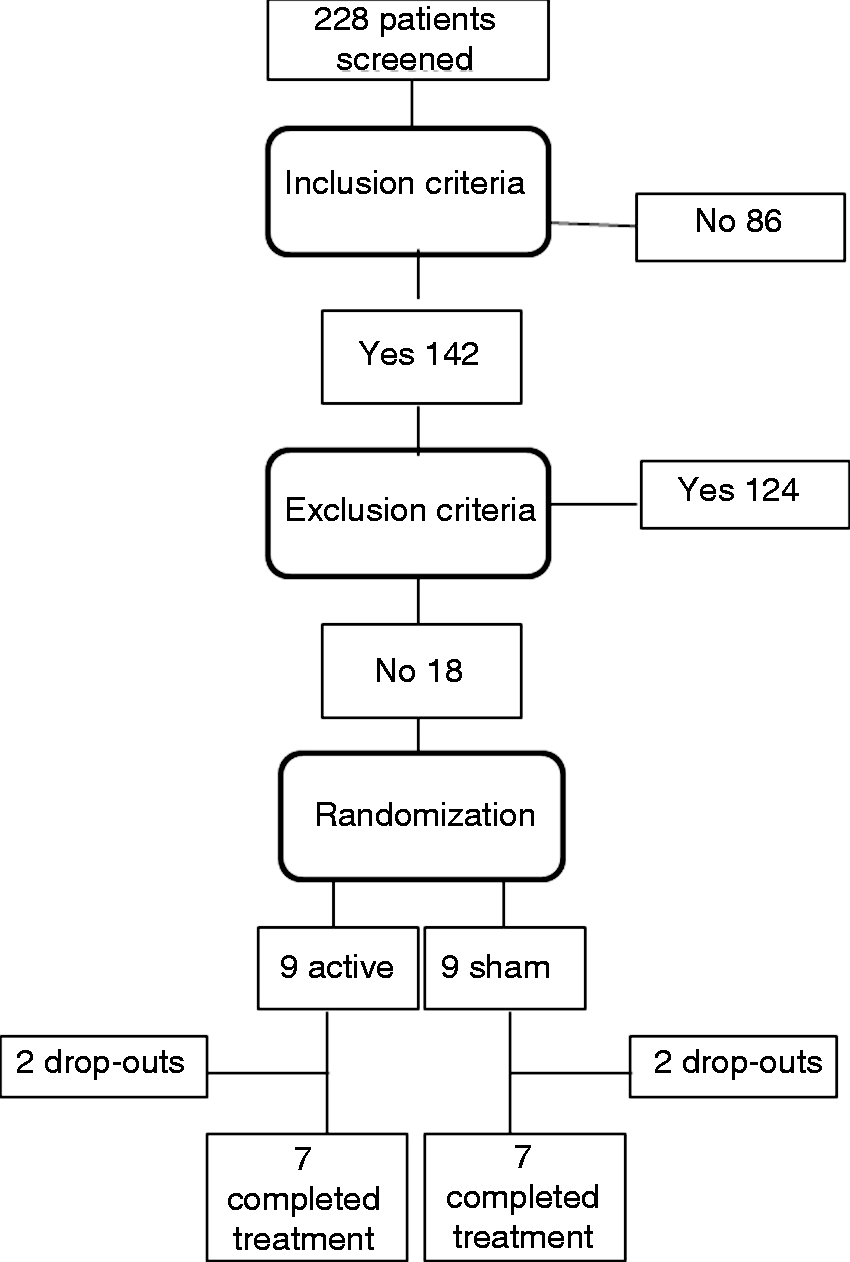

After their participation in the protocol ended, one subject in the active group and one in the sham group reported that they thought they had received sham treatment. All other subjects said they believed that active stimulation had been performed.
Primary outcome measures
Feasibility and safety
Two subjects in the active group discontinued the study: Both considered the rTMS intervention to be too painful and dropped out after three sessions. Both complied with neurological evaluations but not with psychological assessments at week 4. One of them also reported hypesthesia in the territory of the ophthalmic division of the left trigeminal nerve (V1) after the third session of treatment. In this patient, the position of the coil to stimulate the left DLPFC was very near the forehead because of the anatomy of her skull. The neurological examination showed tactile and pain hypesthesia on the left V1. The symptom subsided one week later. The other patient reported that, in addition to pain from the procedure, she felt that “it was difficult to concentrate” during the first session of treatment. Two other patients in the active group, and none in the sham group, reported the same symptom during rTMS.
Two subjects in the sham group discontinued the study: One, after five sessions, reported that personal reasons impeded compliance with the protocol's schedule. She was the only one in the sham group who, when debriefed, reported to believe that sham treatment had been performed, and did not attend any of these assessment sessions after drop-out. The other subject decided to drop out, justifying her decision by the lack of noticeable improvement after 14 sessions of rTMS. This subject complied with neurological and psychological evaluations at week 4. Except for drop-outs, all subjects completely filled out the headache diaries.
There were no significant differences in compliance with the sessions of treatment between the sham (78± 28%) and active (73±23%) groups (
Worsening or onset of headache during rTMS or local pain under the coil were reported by seven of nine (78%) patients in the active group and three of nine subjects in the sham group (33%) (
Number of headache days
Data from 14 patients were included in the analysis because two patients in the sham group and two patients in the active group dropped out and did not comply with treatment or all evaluations up to eight weeks.
GEE revealed a significant effect of GROUP (χ2(1)=7.13; Absolute differences (mean, standard errors) compared to baseline, in the number of days with pain (a) and pain intensity (b), after four and eight weeks of treatment. *
Considering these results, and that pain during TMS was more frequent in the active group, we investigated whether the lack of improvement in the number of headache days in the active group could be due to the fact that patients might have counted days in which pain occurred during TMS as a “headache day” in the diary. Headache days from all patients in the active group were pooled, and post-hoc analysis showed that headache days were significantly more frequent when TMS was not administered (61% of all headache days) compared to days when TMS was administered (39%; χ2=32.8;
Secondary outcome measures
Beck Depression Inventory, State and Trait Anxiety Inventories, as well as Migraine Disability Assessment (MIDAS) scores (per-protocol analysis).

Median (range).
In regard to pain intensity (
Two patients in the active group did not comply with psychological evaluations at four or eight weeks. Therefore, 12 patients were included in the analyses of BDI, Trait and State Anxiety scores (Table 3).
For BDI scores, GEE showed a significant effect of GROUP (χ2=5.14;
GEE did not reveal significant effects of GROUP (χ2=0.33;
GEE did not reveal significant effects of GROUP (odds ratio (OR)=0.11;
GEE showed significant effects of GROUP (χ2=13.5;
Discussion
High-frequency rTMS-DLPFC applied over eight weeks was feasible and safe in patients with chronic migraine. Active rTMS was less well tolerated than sham rTMS, but drop-outs were comparable in the two groups.
Contrary to our primary hypothesis, the number of headache days did not decrease significantly more in the active group than in the group treated with sham rTMS-DLPFC. Average decrease in headache days by 58.1% in the sham group, indicating a powerful placebo response, similar to responsiveness reported for migraine treatment with sham acupuncture (20,21). Pain intensity improved in both groups at week 8.
Rates of discomfort in the active group were higher than those reported for patients submitting to active rTMS-DLPFC for depression (headache, 39%; pain or discomfort, around 40%) (22,23). Discomfort in the sham group was also higher than that described in patients treated with sham rTMS in depression trials (headache, 16%; pain or discomfort, 15%) (22). In a recently published study, all patients with episodic or chronic migraine submitted to high-frequency rTMS of M1 reported pain in association with the procedure (24). Reasons behind rTMS painfulness are unclear. It is possible that patients with chronic migraine have lower thresholds for headache or neck pain during the procedure.
The lack of significant improvement in the number of headache days in the active group contrasts with results reported by Brighina et al. (12). Differences between the two studies can explain this apparent discrepancy. First, rTMS frequency and intensity were different. Second, while the number of headache days was the primary outcome in the present study, the number of headache attacks was the main outcome in the study of Brighina et al. (12). Disagreements in results obtained for these two outcomes after administration of the same treatment have been previously reported and are likely due to variable duration of headache episodes (25). It has been argued that the number of headache days is a more standardized measure than the number of headache attacks.
Third, 10/11 patients in the study performed by Brighina et al. (12) used various prophylactic medications during the protocol, and therefore improvement in the active group may have reflected greater responsiveness to prophylaxis. Alternatively, use of prophylactic drugs may interact with rTMS and therefore lead to dramatically different results due to state-dependent mechanisms (26). Finally, the magnitude of the placebo effect in the sham group was much lower in the study of Brighina et al. (12) compared to the present study.
The placebo response permeates all therapeutic interventions in medicine and has been defined as the effect elicited by a therapeutic procedure lacking specific activity over the treated condition. Effects of a given intervention thus represent the interactive sum of specific treatment effects and the nonspecific placebo response (27). It is known that dopamine-mediated mechanisms underlie some placebo effects and that placebo rTMS administered with the same paradigm used in the present study increases activation in the dorsal and ventral striatum in patients with Parkinson's disease (28). Furthermore, sham rTMS delivered to the vertex, as in the present study, has led to a decrease in the number of pain attacks in patients with episodic migraine (29). The scalp sensory stimulation during TMS may set a favorable stage for placebo analgesia by augmenting the patient's expectation and by inducting striatal dopamine release (30).
The subjective nature of pain intensity renders this outcome more difficult to interpret and may be related to changes in the affective dimension of pain perception rather than to the presence or absence of pain, evaluated by the number of headache days. Comparable effects of active and sham rTMS on disability may reflect amelioration of perception of pain.
The floor effect represented by the relatively mild symptoms of depression, and the lower compliance with psychological evaluations compared with neurological evaluations, limit interpretation of the results of improvement in BDI scores in the sham group (
Analgesic overuse was present in two-thirds of the patients in the sham group. Even though at baseline there was no statistically significant difference between the active and sham groups, analgesic overuse was more frequent in the active group, and the lack of significant differences in baseline characteristics may be due to the relatively small sample size. Triptans can change cortical responsivity evaluated by intensity dependence of cortical auditory evoked potentials (31). Still, patients with chronic headache related to triptan overuse have no significant change in cortical inhibition, while those overusing NSAIDs have increased cortical silent periods measured with single-pulse TMS, suggesting enhanced cortical inhibitory mechanisms (32). We cannot rule out the hypothesis that analgesic overuse interacted with rTMS and placebo effects in the present study, and that different results would be obtained if none of the patients included in this study had analgesic overuse. Further studies are necessary to clarify the role of rTMS and placebo interventions for treatment of analgesic overuse, a challenging condition diagnosed in up to 80% of patients attending specialized headache clinics (33).
This study has some limitations. The sample size was small, as usual in proof-of-principle rTMS studies. Per-protocol analysis was performed, given the exploratory nature of the study. Drop-outs and absences in psychological evaluations limited interpretation of secondary outcomes. The patients included in the study were not widely representative of patients with chronic migraine because they had relatively mild symptoms of depression, while it is known that depression is a frequent comorbidity. The profile of our patients is a consequence of excluding individuals using antidepressant drugs to avoid confounding factors associated with state-dependent rTMS effects.
Conceptually, the few TMS pulses (typically, 20–30 per session, 460–690 over eight weeks) applied to M1 for rMT determination are not expected to have after-effects or ameliorate pain. Conversely, single sessions of 120–500 pulses of low-frequency rTMS of M1 administered acutely decreases perception thresholds (34), and high-frequency rTMS of M1 (500 to 21,000 pulses) has successfully reduced chronic pain (35). Beneficial effects of active M1 stimulation on pain intensity were also reported in a proof-of-principle study (36) that randomized 13 patients with chronic migraine to either anodal or sham transcranial direct current stimulation during four weeks. Importantly, significantly greater improvement in pain frequency was recently reported in patients with either episodic or chronic migraine free of prophylactic drugs, submitted to active high-frequency rTMS of M1, compared to sham (24).
Currently, therapy for chronic migraine is faced with the challenges of costs and adverse events of medications or interventions. Clinical trials are costly and time consuming. Proof-of-principle studies are crucial to generate hypotheses and to plan larger clinical trials, maximizing their chances of success in showing evidence in favor of novel, more effective treatments. Positive results of M1 stimulation in other studies, and the absence of significant benefits of active high-frequency rTMS of the DLPFC in the present study, point to M1 as a more promising target than the DLPFC for larger trials of noninvasive brain stimulation in patients with chronic migraine.
Clinical implications
In patients with chronic migraine and without depression, active repetitive transcranial magnetic stimulation (rTMS) of the left dorsolateral prefrontal cortex over eight weeks was not superior to sham rTMS. The strong placebo effect observed after sham rTMS was comparable to effects reported for sham acupuncture. Active rTMS was less well tolerated than sham rTMS, but drop-outs were comparable in the two groups and no serious adverse events were observed.
Footnotes
Acknowledgments
We thank André Amorim, Lais Lois, Mariana S. Moraes and Marta O. S. de Freitas for patient recruitment. We thank Inara Siqueira for technical assistance.
Conflict of interest
None declared.
Funding
This work was supported by the Albert Einstein Israelite Hospital Teaching and Research Institute/Instituto de Ensino e Pesquisa do Hospital Israelita Albert Einstein (grant number IEP.PE.08-0122), and the Brazilian National Council for Scientific and Technological Development/Conselho Nacional de Desenvolvimento Científico e Tecnológico (grant number 472407/2007-4). The funding sources had no roles in study design, data analysis or interpretation.
