Abstract
Background
Hemiplegic migraine (HM) is a rare type of migraine with aura that involves motor weakness. Data on conventional and advanced neuroimaging findings during prolonged attacks of HM are limited, particularly in children.
Case
A 13-year-old-female with a history of migraine had a typical attack of HM characterized by right-sided hemiplegia, deterioration of vigilance and paraphasia. MRI performed 3 hours after hemiplegia onset revealed normal diffusion tensor imaging (DTI) sequences, but perfusion weighted imaging (PWI) showed a large area of hypoperfusion within the left cerebral hemisphere and susceptibility weighted imaging (SWI) demonstrated a matching area with prominent, hypointense draining sulcal veins. Magnetic resonance angiography (MRA) revealed subtle narrowing of the left middle cerebral artery. The neuroimaging abnormalities completely resolved 24 hours after the attack onset.
Conclusion
Multiple conventional and advanced MRI techniques including SWI play a key role in an HM attack to (1) exclude acute arterial ischemic stroke and (2) further understand the pathophysiology of HM.
Keywords
Introduction
Hemiplegic migraine (HM) is a rare subtype of migraine with aura as defined by the International Classification of Headaches (1). HM may occur as a sporadic or familial disorder, with the latter often caused by mutations in membrane channels (CACNA1A, ATP1A2 or SCN1A) (2). The aura associated with HM attacks is by definition characterized by motor weakness, but is often associated with sensory, visual or language disturbances as well. Rare subsets of patients may also experience more severe symptoms such as transient or permanent cerebellar ataxia, encephalopathy or even coma.
Data on neuroimaging during attacks of HM are scant, especially in children (3–9). Neuroimaging studies done during or after a HM attack may be normal (2), but hyperintense signal abnormalities and cortical swelling on T2-weighted and fluid-attenuated inversion-recovery (FLAIR) images (5,6) as well as hypoperfusion on perfusion weighted imaging (PWI) (10) and vasoconstriction on magnetic resonance angiography (MRA) (11) have been reported in a few pediatric patients.
We report on acute and follow-up diffusion tensor imaging (DTI), PWI, susceptibility weighted imaging (SWI) and MRA findings in a 13-year-old female with HM to highlight the role of multiple conventional and advanced MRI techniques in this disorder.
Case report
A 13-year-old female with a recent diagnosis of migraine involving right sensory and visual aura associated with left-sided headache presented to the emergency room of our tertiary care pediatric hospital with an acute developing right-sided hemiplegia. Initially, she developed pain that started in the middle of her nose and extended to her left forehead and cheek. Within 5 minutes, she could not lift her right arm, her mouth became uneven and she could not talk. In triage, 20 minutes later, the patient was oriented to person and place, but had a right-sided hemiplegia and facial droop. She could speak but was making paraphasic errors (‘knee’ for ‘knuckle’), which she was aware of, and her pain had progressed to the entire left side of her head. She was actively vomiting.
Initial brain MRI was obtained on a 3.0 T scanner (Skyra, Siemens, Erlangen, Germany) about 3 hours after onset of symptoms. Standard departmental stroke imaging protocol including T1-weighted, T2-weighted and FLAIR imaging, axial DTI and apparent diffusion coefficient (ADC) maps, axial echo-planar dynamic susceptibility weighted contrast enhanced perfusion imaging (DSC-PWI) with post-processed mean transit time (MTT), relative cerebral blood flow (rCBF) and relative cerebral blood volume (rCBV) maps, axial SWI and axial time-of-flight (TOF) MRA with volumetric 3D reconstruction was applied.
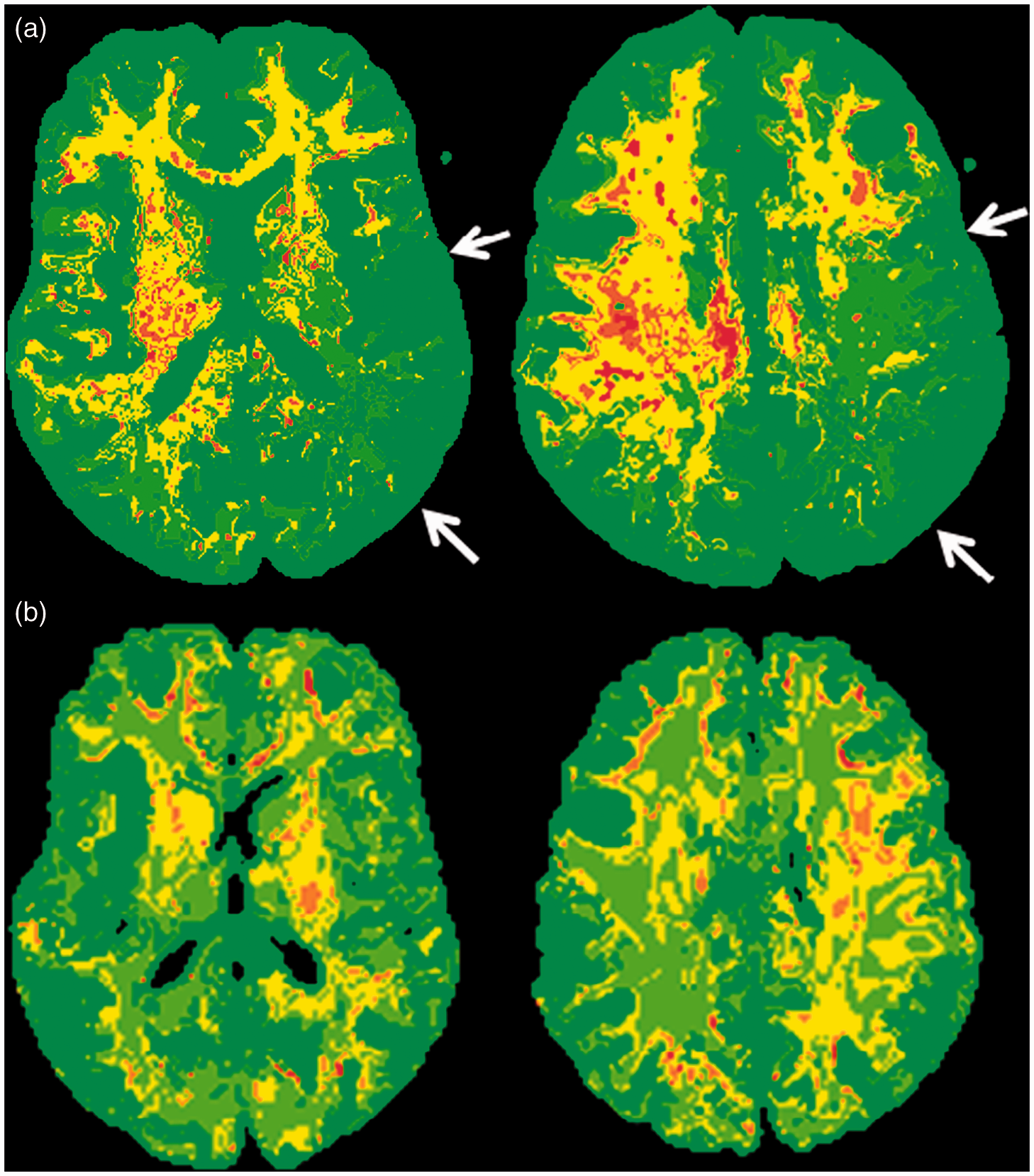
T1- and T2-weighted, FLAIR and post-contrast T1-weighted images did not show signal abnormalities or pathological contrast enhancement (Figure 1). The qualitative evaluation of trace of diffusion and ADC maps did not show areas with restricted diffusion thereby excluding an acute arterial ischemic stroke (Figure 1). The MTT and cerebral blood flow (CBF) maps on the DSC-PWI demonstrated decreased CBF and increased MTT, respectively, within the complete left cerebral hemisphere, representing hypoperfusion (Figure 1). The minimal intensity projection (minIP)-SWI image showed prominent, hypointense sulcal veins within a matching cerebral region suggesting the presence of relatively increased concentration of intravascular deoxyhemoglobin and/or slow blood flow (Figure 1). The 3D-TOF MRA showed mild decrease in caliber of the left MCA in comparison with the right, possibly secondary to vasospasm (Figure 1). Post-processing of the DTI data revealed a relative increase in the diffusivity in the area of decreased CBF, predominantly in the white matter, which resolved on follow-up imaging (Figure 2).
A 13-year-old female with hemiplegic migraine underwent brain magnetic resonance imaging (MRI) 3 hours after onset of right-sided hemiplegia. (a, b) Axial T2-weighted images show normal signal intensity in the brain parenchyma. (c, d) Axial trace of diffusion and (e, f) axial apparent diffusion coefficient (ADC) map demonstrate no evidence of diffusion restriction. (g, h) Axial minimal intensity projection (minIP)-susceptibility weighted imaging (SWI) image shows prominent hypointense sulcal veins in the left cerebral hemisphere (arrows). (i, j) Axial mean transit time (MTT) map shows increased mean transit time suggesting hypoperfusion and decreased cerebral blood flow in the left cerebral hemisphere (arrows). (k, l) Maximum intensity projection (MIP) reformat of time of flight (TOF) magnetic resonance angiography (MRA) of the Circle of Willis demonstrates patent vessels with subtle narrowing of the left middle cerebral artery (MCA) (arrowhead). Post-processed diffusion imaging data shows relative increase in the apparent diffusion coefficient (ADC) or diffusivity of the white matter in the area of decreased cerebral blood flow (CBF) in the left cerebral hemisphere during the hemiplegic migraine (HM) attack (arrows on a), which normalized on follow-up (b).
The patient was treated with a migraine ‘cocktail’ and within 24 hours of onset, her symptoms completely resolved and she was discharged home with a normal neurological examination. A follow-up brain MRI at 24 hours was normal.
Discussion
SWI is increasingly used in pediatric and neonatal neuroimaging for the evaluation of hemorrhage, ischemic stroke, vascular malformations, traumatic brain injury, brain tumors, congenital infections and neurodegenerative disorders (12,13). This case demonstrates the substantial role of SWI in understanding the neurophysiology of HM in better detail.
The migraine aura is generally indicative of a reversible cerebral cortical dysfunction that is most probably caused by cortical spreading depression (14). Cortical spreading depression is characterized by a neuronal excitation followed by a prolonged inhibition of the neuronal activity (15). In animal models, mutations in CACNA1A, ATP1A2 and SCN1A have been shown to increase neuronal excitability and reduce the threshold for cortical spreading depression (2,16,17). In a Cacna1a knockin migraine mouse model, a disrupted balance of cortical neurotransmission with an increased excitatory glutamate release was reported (18). Glutamate release is known to activate neuronal N-methyl-d-aspartate (NMDA) and α-amino-3-hydroxy-5-methylisoxazole-4-propionic acid (AMPA) receptors resulting in neuronal hyperactivity and hypermetabolism. During this phase, neuronal tissue requires higher oxygen consumption resulting in higher oxygen extraction fraction (OEF) and prominent, hypointense veins on SWI as seen in our patient and previously reported in five children with typical HM attacks (7,9). In all patients, the SWI findings resolved spontaneously. Transient, prominent, hypointense veins on SWI have been shown in other patients with migraine with aura (19,20).
Cortical spreading depression is associated with changes in CBF characterized by hypoperfusion followed by hyperemia (15,21). Transient hypoperfusion in the cerebral hemisphere contralateral to the side of hemiplegia was previously shown in three children with HM (7,10,22,23). In our patient, the presence of hypoperfusion during motor weakness and headaches was shown to be associated with decreased CBF and increased MTT. The area with hypoperfusion matched the one with prominent, hypointense sulcal veins on SWI. Detailed evaluation of the regional changes in ADC values revealed an unexpected increase in the ADC values of the white matter similar to that of cortex in the hypoperfused area. This phenomenon reversed on follow-up imaging where the white matter regained lower ADC values. Similar increases in ADC values were recently reported in patients with migraine, and they were hypothesized to represent vasogenic edema (24).
In HM, the reduced CBF remains above the threshold for brain ischemia, as demonstrated by the absence of diffusion restriction. In fact, in our patient, subtle increases in ADC values were detected within the region of hypoperfusion. The dual threshold of relevant brain hypoperfusion was initially reported many decades ago. Below the first threshold, the neurons fail functionally with resultant neurological deficit but remain viable. Below the second lower threshold, irreversible ischemic injury with cell death is observed (21). The absence of restricted diffusion differentiates between HM and pediatric acute ischemia (PAIS) in acute onset hemiplegia, whereas PWI and SWI findings are similar in both diseases (25). The transient clinical and neuroimaging findings in our patient further argue for HM compared with PAIS.
Vasospasm during HM attacks may also contribute to hypoperfusion. Recently, Safier et al. showed transient vasospasm in MCA branches in seven children with HM using MRA (9,11). In our patient, a subtle smaller caliber of the left MCA ipsilateral to the hypoperfused left hemisphere was seen supporting the concept that vasospasm plays at least a partial role in HM. The presence of vasospasm and hypoperfusion further supports the importance of avoiding medications with vasoconstrictive properties such as triptans or dihydroergotamine as acute therapy of HM.
In summary, this case highlights the significance of utilizing multiple advanced MRI techniques, in particular the evolving role of SWI in combination with PWI and DWI/DTI in the setting of acute hemiplegia to (1) differentiate between HM and PAIS and (2) better understand the complex cerebral hemodynamics in HM. SWI is especially helpful in children because it gives valuable cerebral hemodynamic information without the necessity of injecting intravenous contrast agents.
Clinical implications
Multiple advanced MRI techniques in hemiplegic migraine help us differentiate it from a stroke and also to understand the underlying complex cerebral hemodynamics. Susceptibility-weighted imaging is especially helpful in children because it gives valuable cerebral hemodynamic information without the necessity of using an intravenous contrast agent.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
