Abstract
Deep brain stimulation (DBS) of the posterior hypothalamus was found to be effective in the treatment of drug-resistant chronic cluster headache. We report the results of a multicentre case series of six patients with chronic cluster headache in whom a DBS in the posterior hypothalamus was performed. Electrodes were implanted stereotactically in the ipsilateral posterior hypothalamus according to published coordinates 2 mm lateral, 3 mm posterior and 5 mm inferior referenced to the mid-AC-PC line. Microelectrode recordings at the target revealed single unit activity with a mean discharge rate of 17 Hz (range 13-35 Hz, n = 4). Out of six patients, four showed a profound decrease of their attack frequency and pain intensity on the visual analogue scale during the first 6 months. Of these, one patient was attack free for 6 months under neurostimulation before returning to the baseline which led to abortion of the DBS. Two patients had experienced only a marginal, non-significant decrease within the first weeks under neurostimulation before returning to their former attack frequency. After a mean follow-up of 17 months, three patients are almost completely attack free, whereas three patients can be considered as treatment failures. The stimulation was well tolerated and stimulation-related side-effects were not observed on long term. DBS of the posterior inferior hypothalamus is an effective therapeutic option in a subset of patients. Future controlled multi-centre trials will need to confirm this open-label experience and should help to better define predictive factors for non-responders.
Introduction
Cluster headache (CH) is a rare form of primary neurovascular headache, characterized by the sudden onset of excruciating unilateral periorbital pain. The pain is often accompanied by local signs of facial autonomic dysfunction such as lacrimation, rhinorrhoea, sweating and flushing. Attacks last up to 3 h and occur in clusters of a few months with multiple attacks per day, often followed by periods of remission (1, 2). In its chronic form, significant remission intervals are lacking as attacks appear on a daily basis (3).
Neuroimaging studies have revealed structural and functional abnormalities in the posterior hypothalamic area in CH patients, suggesting dysfunctional hypothalamic activity as being the pathophysiological correlate of CH attacks (4–6).
Thus, the posterior hypothalamic area was chosen first by Leone et al. as a target for stereotactic neurostimulation leading to a profound relief in the severity and frequency of attacks in chronic CH patients (7, 8). A further pilot study from a Belgian centre, however, found a beneficial effect of hypothalamic deep brain stimulation (DBS) only in a subgroup of operated patients.
To date, the outcome of 27 patients from four centres has been published (9–12). As the findings of these centres on hypothalamic DBS have to be confirmed and extended, we report a series of a further six cases who have been operated in a national multicentre setting. These patients were selected after the published criteria for DBS surgery in cluster patients and were operated on the basis of the published stereotactic coordinates (8, 13).
Methods and materials
Patients
All patients fulfilled the International Headache Society criteria for chronic CH (ICHD-II code 3.1.2) and the selection criteria of CH patients eligible for DBS as published by an international expert team (13). The patients' characteristics are listed in Table 1. One patient was diagnosed with familial CH. Patients were operated in four different centres in Germany experienced in DBS. Patients from each centre were selected in cooperation by a team of neurologists specialized in headache treatment and neurosurgeons experienced in functional neurosurgery. Written informed consent about DBS was obtained from each enrolled patient. Some aspects concerning the neurosurgical procedure of some of these patients have been previously reported elsewhere (14, 15). Surgery was indicated as an individual (orphan) treatment attempt that did not require a vote of the ethics committee according to German legislation.
Patients' characteristics, history of cluster headaches, medication prior to deep brain stimulation as well as surgical technique for electrode implantation

RBBB, Right bundle branch block; AC, anterior commissure; PC, posterior commissure; MCP, midcommissural point.
Clinical assessment
Patients eligible for hypothalamic DBS were asked to document attack frequency and pain intensity including autonomic characteristics 4 months preoperatively and from the DBS onward.
Preoperative assessment included a standard neurological work-up [cranial magnetic resonance imaging (MRI), blood tests including endocrine status] and an extended diagnostic work-up in two patients (cerebrospinal fluid analysis, Doppler ultrasound of the extra- and intracranial vessels, EEG, evoked potentials, neuropsychological testing). A detailed history of the cluster headaches including medication was taken. In three patients a nitrogylcerin test (1.2 mg sublingual) was performed prior to DBS to elicit a cluster attack.
The impact of headaches on daily life and activity was assessed with the Headache Impact test (HIT) and the Henry Ford Headache Disability Inventory (HDI). The Short Form (SF)-36 was used in four patients assessing the quality of life. The affective component was assessed using either the Beck Depression Inventory (BDI) or the Hamilton Depression Score (HDS). The preoperative assessment included a detailed history of autonomic functions: sleepiness, sleep quality and behaviour, body mass index, personality changes and eating behaviour. In two patients detailed sensory and autonomic testing was performed. The follow-up after initiating DBS was monitored using a pain diary. All patients were assessed by a neurologist experienced in CH treatment.
Neurosurgical procedure for electrode implantation in the ipsilateral posterior hypothalamus
A stereotactic head frame was applied prior to MRI or computed tomography (CT) to obtain high-definition imaging data. Gadolinium-enhanced T1 images or reformatted 3D CT scans were used to identify the anterior and posterior commissure (AC-PC). The target and trajectory planning was performed using a neuronavigation workstation (Framelink; Medtronic Stealth, Düsseldorf, Germany) according to standard neurosurgical procedures for DBS, as described previously (16–18). The revised stereotactic coordinates for the target published by the Italian group in 2003 were used (8): 2 mm lateral to the midline, 3 mm behind the midcommissural point and 5 mm below the intercommissural line (Table 2; Fig. 1). For target localization, stereotactic reference atlases (19, 20) as well as published criteria for hypothalamic stereotaxy were used (21) (Fig. 2). Stereotactic implantation of the electrodes was performed under local anaesthesia.
Data showing the effect of deep brain stimulation (DBS) on attack frequency and pain intensity as well as details about the follow-up period including current stimulation settings, medication and side-effects

Global rating was graded: 1, excellent; 2, good; 3, satisfactory; 4, sufficient; 5, insufficient; 6, failed.
VAS, Visual analogue scale.
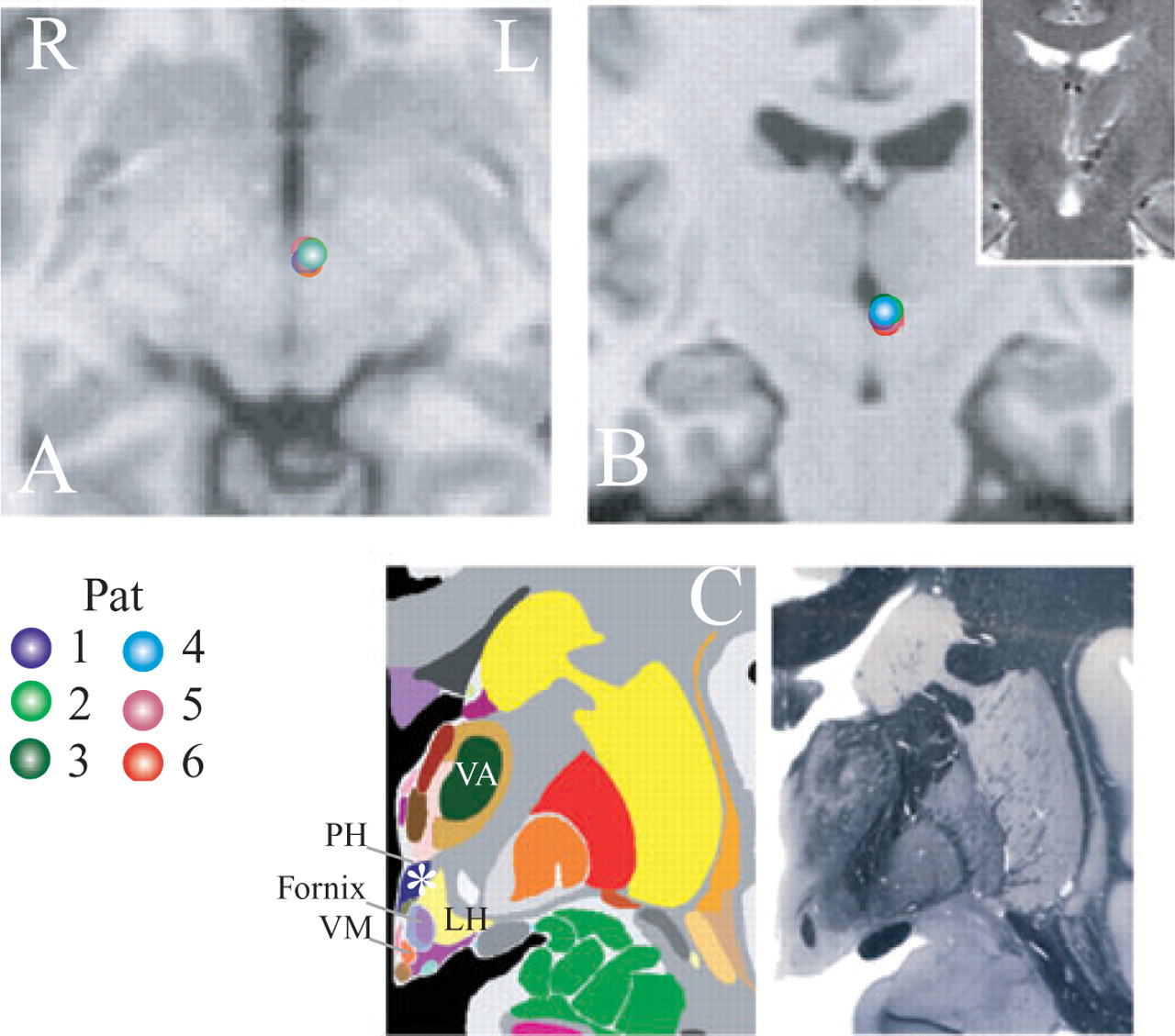
Synopsis of the electrode positions reconstructed from all three planes of the postoperative imaging of all deep brain stimulation (DBS) patients. Lead (target) electrode positions delineating the stereotactic coordinates (2 mm lateral, 3 mm posterior, 5 mm inferior) were plotted and superimposed on axial (A) and coronal (B) T1 magnetic resonance imaging reference images registered to the relative distance to the midcommissural point within the Talairach space [http://www.neurovia.umn.edu/cgi-bin/tal_atlas, used with permission by the International Neuroimaging Consortium (INC)]. Inset shows a representative electrode position in the coronar plane. (C) Corresponding schematic anatomical and histological plate from the reference brain atlas of Mai et al. (19) delineating the anatomy of the target region, used with permission. VA, Ventroanterior thalamic nucleus; PH, posterior hypothalamus; LH, lateral hypothalamic area; VM, ventromedial hypothalamic nucleus.

Postoperative imaging showing the position of the electrode tip on the axial plane on the level 5 mm below the midcommissural point within the posterior hypothalamic area. A–F corresponds to patients 1–6.
In four patients microelectrode recordings were performed at ± 1 mm around the target. To assess the functional nature of the recorded neurons, in three patients sensory stimulation in the trigeminal and spinal dermatomes was performed. Further testing in two patients included: motor stimulation (passive movement of elbow) and autonomic testing (counting backwards under stress, cold pack, and bladder filling). Further affective stimulation was performed showing pictures with emotional content to the patients.
Macrostimulation was performed on different levels from 10 mm above to the target level to check for intrinsic effects and side-effects (Table 2). In five patients the quadripolar electrode model 3387 (Medtronic, Minneapolis, MN, USA) was used, and in one patient model 3389. After removing the stereotactic ring the pulse generator (Medtronic Soletra®) was implanted under general anaesthesia in three patients, whereas in the other three patients the pulse generator was implanted after a test stimulation of 5–8 days with externalized electrodes.
Postoperative MRI or CT controls to document regular electrode positions were done either directly after wound closure (n = 2) or on the first day after the operation (n = 4).
Results
Clinical effects of neurostimulation
All patients reported a decrease of their attack frequency after initiating hypothalamic DBS (Fig. 3). In four patients the reduction of attack frequency amounted to 90–100% of the preop level within the first weeks. Two patients responded only a moderate transient decrease of < 30% of their attack frequency. In those patients (n = 4) who responded with a profound decrease of attack frequency, the pain intensity of the remaining residual attacks was rated significantly lower on the visual analogue scale (VAS) (VAS preop 10/10 vs. VAS postop 1–4/10). Also, the autonomic signs were rated less intense. In those two patients with only a transient initial effect the attack frequency and pain intensity returned to its preop level within 4 weeks after DBS. The latter patients showed an insertional effect lasting 2–4 days subsequent to implantation with either no or fewer attacks in the absence of neurostimulation. One patient initially showed ‘cold’ attacks with typical autonomic signs in the absence of headaches. In two patients (patients 1 and 5) a typical cluster attack was either provoked intraoperatively or by nitroglycerin.
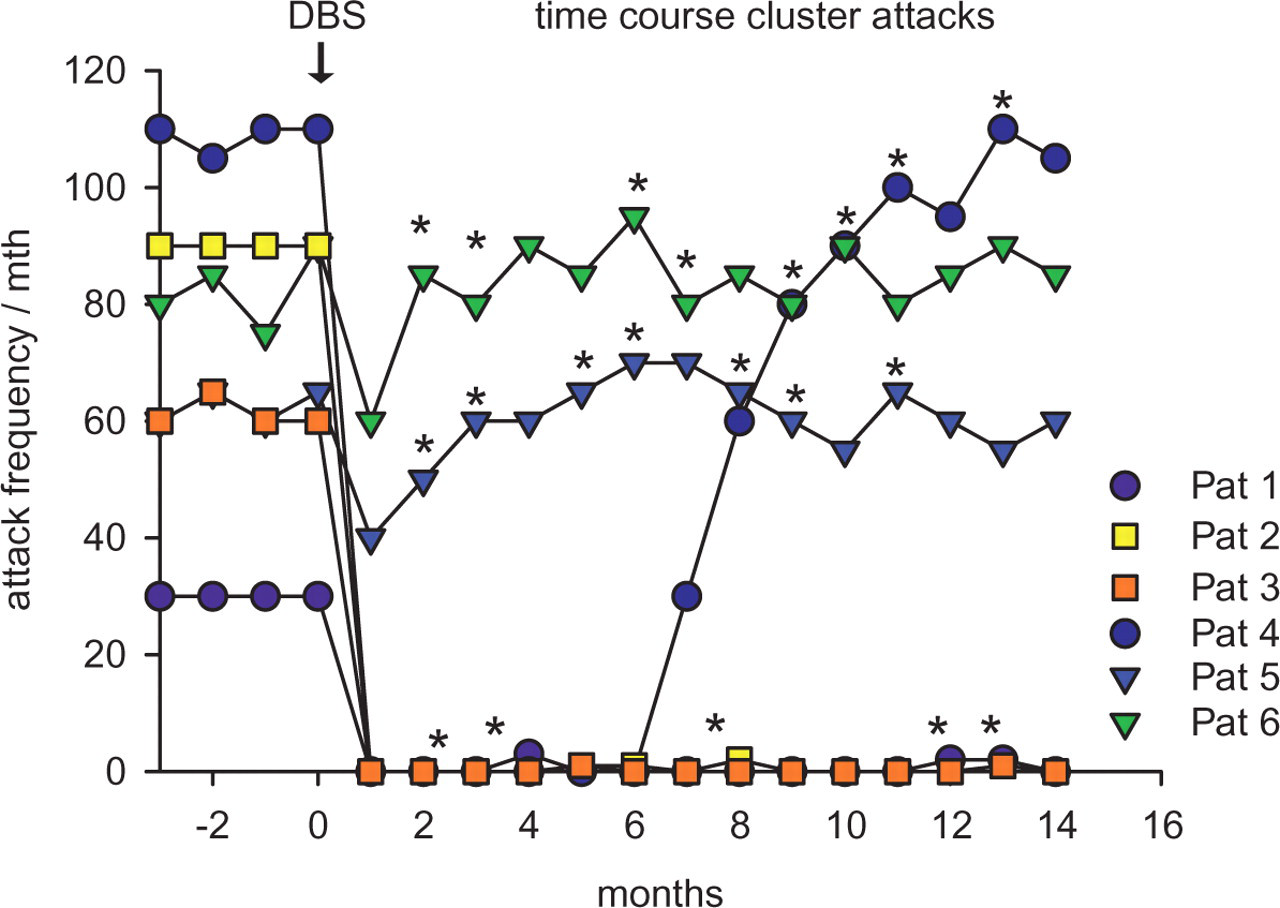
Time course of attack frequency before and after hypothalamic deep brain stimulation (DBS) in all six patients. All patients showed a decrease of the attack frequency after initiating DBS, but two patients showed only a marginal decrease within the first weeks before returning to the former frequency. One patient was completely attack free for 6 months before returning to the preop level. Three patients remained at a significantly low level of attack frequency within the follow-up interval of 12 months postoperatively. ∗Time point of adjusting the stimulation parameters.
Long-term follow-up
In two out of four patients with a profound stimulation effect repetitive adjustments of the amplitude and pulse width of the stimulation were required to maintain a prolonged stimulation effect. Reprogramming consisted of changing amplitude and pulse width and, if necessary, changing the active electrode. Three patients remained almost completely attack free in the follow-up phase 9–15 months after DBS (Fig. 3, Table 2). The total number of remaining attacks at the last follow-up was seven (patient 1), four (patient 2) and two (patient 3). One patient showed a beneficial effect for 6 months, but then over 2–3 months returned to its former frequency and intensity level, despite repeated adjustments of the stimulation parameters. In the two patients who had only a marginal transient effect, repeated adjustments over 17 months including controlled stimulation trials through each of the four electrode contacts did not show a longer-lasting effect that could be attributed to the DBS (Fig. 3). During reprogramming of the stimulation parameters, as the stimulation device was switched off twice in these two patients, both patients reported a marginal short-lasting worsening of the pain. The three patients with a long-lasting effect refused to switch off the device; due to ethical reasons this was not performed in a blind fashion in this pilot study.
Preoperative testing did show a considerable impact of headaches on daily life and activity in two patients (patients 5 and 6) as assessed with the HIT-6 and HDI (HIT-6 values 70 and 71/78, HDI values 72 and 67/100), as well as an affective component (BDI values 4 and 22, pain scores SF-36 10 and 11/11). In patients 2 and 3, postoperative scores of the HDS ranged within normal values (6 and 4 p); scores from the SF-36 indicated a tendency for an improvement of quality of life pre- vs. postoperatively. Due to small and inhomogeneous sample size, a formal statistical assessment was not possible. Qualitative pre- and postoperative assessment of autonomic functions did not show changes of sleep, body weight, personality or eating behaviour. Endocrine testing did not show evidence for changes of the hypothalamic–pituitary axis.
Effects of neurostimulation and side-effects
The stimulation was usually well tolerated. Only transient and mild side-effects were noted. The most frequent reported side-effects when initiating neuromodulation or reprogramming were short-lasting vertigo and transient double vision. The current stimulation settings are listed in Table 2. Patient 2 had an intraoperative cluster attack elicited by test stimulation at the target (1 V, 130 Hz, 60 µs) that was aborted by high-flow oxygen.
Neurophysiology and functional anatomy
Postoperative imaging confirmed the electrode location in the target area according to the published coordinates (21). Microelectrode recordings from the level of the target in four patients showed a tonic firing pattern with no discernible rhythmic pattern. The average discharge rate of these four neurons was 17 Hz (range 13–35 Hz, n = 4; Fig. 4). Intraoperative testing using sensory, motor, autonomic and emotional stimulation (two patients each) did not reveal obvious neuronal responses that could be attributed to the modality-specific afferent stimulation.

Representative microelectrode recordings from the target level illustrating the discharge pattern and frequency of the recorded neurons. Mean discharge was 17 Hz (range 13–35 Hz) with a tonic firing pattern. No responses to sensory, motor, autonomic or emotional stimulation could be elicited.
Discussion
This study has shown that hypothalamic DBS can be an effective treatment in medically refractory chronic CH. After a mean follow-up of 17 months, three out of six patients remain almost attack-free, whereas one patient did show a limited beneficial effect over 6 months. Two patients did not show a relevant early or late treatment effect using the generally accepted definition of a treatment response of > 50% reduction of attack frequency or severity. The response rate in our sample was thus 50%.
Hypothalamic DBS was first shown by Leone et al. to have an effect on the frequency on cluster attacks (7). Results were later confirmed by Schoenen et al., who reported that three out of six patients in their series had a favourable outcome (10). The Belgian as well as the Oxford group had used the coordinates initially published by the Italian group 2 years earlier (7, 11), but revised them after the first two operated patients to optimize stimulation. The original coordinates were 2 mm lateral, 6 mm posterior and 8 mm below the midcommissural point. In our patients the modified functional coordinates published by Franzini et al. in 2003 (in relation to the midcommissural point: 2 mm lateral, 3 mm posterior, 5 mm inferior) were chosen (21). Recently, the results from a further five patients have been published (11, 12), including a patient with episodic CH (11). Hypothalamic DBS using the same coordinates has also been shown to be effective in another type of trigeminovascular headache classified as drug-resistant short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing, since in this disorder activation of the posterior inferior hypothalamus was also observed (22, 23).
The time course of the neurostimulation effect, the magnitude of the effect and the stimulation settings in our patients are comparable to the data of the Italian and Belgian groups, showing a rapid effect in some patients, whereas the effect in other patients was achieved only due to repeated adjustments of the stimulation settings (9, 10). The side-effects of the stimulation, such as arterial hypertension, diplopia, vertigo and, in some patients, feelings of panic have been previously described for the stimulation of the posterior hypothalamus in humans (10, 21, 24, 25). The long-term results from the Italian group with a mean follow-up of 23 months show that 13 of 16 patients remain pain-free or almost pain-free and three patients improved, whereas no treatment failures were reported (9). Starr et al. (12) have reported an improvement after 1 year in two out of four patients after DBS, thus confirming our response rate of 50%. However, there are also possible complications with this procedure, as Schoenen et al. have reported one fatal intracerebral haemorrhage in a series of six patients, putatively due to a haemorrhage along the electrode pathway. In another patient the intraoperative procedure had to be aborted due to a panic attack with autonomic disturbances (10).
The efficacy of hypothalamic DBS in CH is probably comparable to the results of the less invasive occipital nerve stimulation (ONS). In two recent pilot studies, a total of 16 patients underwent occipital nerve stimulation; nine out of 16 patients reported a considerable benefit in these trials in terms of tolerability of stimulation, reduction of severity and frequency of cluster attacks (26, 27). However, in comparison with our DBS data, there seems to be a marked difference in the time course and magnitude of the analgesic effect, as the latency to effective treatment after ONS took several weeks and months, suggesting different mechanisms of action (26). Further data are needed for a definite evaluation of both methods in terms of a sequential treatment recommendation for invasive procedures in CH.
The reason for the three long-term treatment failures in our series remains unclear, but responsiveness to DBS may be associated with the individual anatomical variability of the target region. Unlike in DBS for Parkinson's disease, there are no characteristic clinical responses for test stimulation that would help to delineate the optimal functional target. On the other hand, considering our series, the short-term response and the magnitude of the effect after implantation seem to be predictors of long-term responsivity. We could not identify typical response characteristics of posterior hypothalamic neurons that would help to guide implantation. Stereotactic targeting for hypothalamic DBS therefore has to rely on an indirect (landmark-based) target definition, because MRI does not allow visualization of the target directly.
Our electrode positions correspond to the stereotactic target sites published as well as matching the hypothalamic activity in positron emission tomography (PET) during CH that was used for identifying the target region for DBS in the first hypothalamic DBS series by Leone et al. (5, 6, 21). However, as the PET studies that were used for delineating the target site were statistical group analyses, this method obviously does not account for the individual anatomical and functional variability of the posterior hypothalamus in cluster patients (6). Thus, as the exact functional role of the posterior hypothalamus in CH pathophysiology has still to be determined, in contrast to the role of the subthalamic nucleus in Parkinson's disease, functional variability may affect the variable outcome of hypothalamic DBS (10). Furthermore, habituation similar to DBS in the periaqueductal grey/periventricular grey (PAG/PVG) region may contribute to the time course of the DBS effect.
Intraoperative test stimulation does not show immediate analgesic effects, which is in contrast, for example, to DBS in the PAG/PVG region, where an immediate analgesic effect can be used for functionally determining the target site. A certain functional mapping of the posterior hypothalamus has been attempted by Sano et al. (25, 28), although the spatial resolution does not allow exact determination of the target region. Our microelectrode recording in the posterior hypothalamus corresponds to the older literature as well as to findings that neuronal activity in the posterior hypothalamus does not show a specific rhythmic pattern and only rarely and inconsistently shows responses to sensory stimulation (10, 25, 29). The neuronal discharge may thus reflect neuronal density within the posterior hypothalamus. This is in contrast to neuronal activity in the human PAG, that shows rather a burst-like pattern (30). The lack of neuronal responses to modality-specific afferent stimulation reflects the functional anatomy of the posterior hypothalamus in terms of not being a primary afferent and nociceptive relay centre in contrast to the PAG, which is critically involved in stimulus-induced analgesia (31–33).
On the other hand, direct hypothalamic involvement in CH is suggested by the finding in one of our patients, in whom a cluster attack could be provoked by intraoperative test stimulation. Furthermore, in two patients an insertional effect similar to that described for DBS in the PVG/PAG, and which is generally more clearly seen in DBS for movement disorders, was observed. The time course of the analgesic effect appears to be prolonged, as in that seen in PAG/PVG stimulation that frequently shows a habituation after some months (34). Interestingly, in one patient ‘cold’ attacks with solely typical autonomic signs could be observed in the first days after the operation, suggesting a dissociation between autonomic and nociceptive mechanisms in the hypothalamic relay circuits involved in CH (35–38).
The effect of hypothalamic neurostimulation is probably relayed via descending antinociceptive projections from the posterior hypothalamic area onto second-order neurons in the caudal trigeminal nucleus (39, 40). However, this stimulation-induced hypothalamic inhibition of nociceptive trigeminal neurons might involve further relay centres including the PAG (33, 41, 42). Additionally, investigations of cluster DBS patients studied with PET as well as the clinical observation that acute stimulation is not effective in aborting attacks suggest that the effect of DBS involves neuroplastic mechanisms (43, 44). In a recent PET study, neurostimulation showed activation of the ipsilateral hypothalamic grey, the ipsilateral thalamus, somatosensory cortex and praecuneus, the anterior cingulum, the ipsilateral trigeminal nucleus and ganglion (44). These findings underline the hypothesis that hypothalamic neurostimulation exerts its effects by neuroplastic mechanisms modulating hypothalamic and midbrain neuronal circuits.
In this context, animal data of hypothalamic-induced antinociception show that peptidergic neurotransmission in the posterior hypothalamus might be involved in the modulation of CH attacks. It has been demonstrated that activation of orexin A and B receptors as well as blockade of somatostatin receptors in the posterior hypothalamus selectively modulates trigeminovascular nociception (39, 40). Experimental studies have shown that the regulation of autonomic and neuroendocrine functions as well as nociceptive processing are closely coupled in the hypothalamus (45), and recent data strongly suggest an involvement of orexinergic mechanisms in the regulation of these functions (46). Orexins are selectively synthesized in the posterior hypothalamus and, together with genetic findings of an orexin receptor polymorphism in cluster patients, this suggests that orexinergic mechanisms in the posterior hypothalamus are involved in the pathophysiology of primary CH; however, more data are needed to corroborate this hypothesis (39, 47–51).
The study has shown that DBS of the posterior hypothalamus is an effective therapeutic option in a subset of patients with therapy-refractory chronic CH. Further studies are needed to define the predictors of an optimal treatment response, to elucidate the pathophysiological mechanisms of hypothalamic DBS and to describe the long-term outcome. A controlled multicentre study is now mandatory to evaluate the efficacy and safety of hypothalamic neurostimulation in a larger patient group. This therapy should be restricted to patients with a chronic form of this disorder.
Footnotes
Acknowledgements
We wish to thank Dr K. Knudsen, Dr F. Steigerwald and Dr J. Herzog for helpful input and technical assistance.
