Abstract
Background
Occipital nerve stimulation (ONS) has been shown to be effective for selected patients with intractable headache disorders. We performed a prospective critical evaluation of complications and direct treatment costs.
Methods
Twenty-seven patients with chronic cluster headache (CCH, n = 24) or chronic migraine (CM, n = 3) underwent a trial phase with bilateral ONS and subsequent implantation of a permanent generator (IPG), if responsive to treatment according to predefined criteria. Procedural and long-term complications as well as direct treatment costs of neuromodulation therapy of ONS were recorded over a mean follow-up period of 20 months (range 5–47 months).
Results
Twenty-five of 27 patients (93%) responded to treatment. Twenty-one complications in 14 patients were identified, necessitating reoperation in 13 cases. Overall treatment costs were €761,043, including hardware-related costs of €506,019, costs for primary hospital care of €210,496, and complications related to hospitalization costs of €44,528. This results in a per case-based cost of €9445 for hospitalization and €18,741 for hardware costs, totaling €28,186.
Conclusion
ONS for treatment of refractory CCH and CM is a cost-intensive treatment option with a significant complication rate. Nevertheless, patients with refractory primary headache disorders may experience substantial relief of pain attacks, and headache days, respectively.
Keywords
Introduction
Migraine and cluster headache are the most common disabling primary headache disorders (1). Chronic cluster headache (CCH) and chronic migraine (CM) have a substantial impact on health-related quality of life (HRQOL), as well as on social and economic costs (SEC) (2,3). Patients with CCH or CM report social deprivation in family relations and friendships, and of career drawbacks due to their inability to take part in social activities or work during attacks (4–6). Guidelines for the treatment of CM are based on multimodal therapeutic approaches including behavioral therapy, psychological support, physical therapy, and medical treatment for attack abortion (e.g. nonsteroidal anti-inflammatory drugs (NSAIDs) or triptans), and prophylaxis (e.g. topiramate or botulinum toxin) (7–10).
Therapy for CCH is based on permanent prophylactic medication (verapamil, topiramate or lithium), and on acute medication aborting the attacks (nasally or percutaneously administered triptans), and oxygen (11). Cost burden of the illness is mainly caused by subcutaneous or endonasal triptans and consumption of oxygen. In CCH monthly expenses for medical treatment will reach or even exceed €1500 per patient (2). For CM costs are estimated to be tenfold lower. However, socioeconomic burden of CM is approximately three times higher than for episodic migraine (12). Beside expenses for medical treatment, the socioeconomic costs caused in the wake of unemployability cumulate (13).
Despite multimodal approaches and prophylactic and acute medical therapy, there will be 5%–15% of patients with CCH and CM who will continue to suffer attacks and headache days refractory to treatment (11,14). For those patients neuromodulative surgical approaches were developed over the last decade. Deep brain stimulation (DBS) was effective but resulted in major complications in some patients. Therefore, less invasive procedures were introduced to treat refractory headache disorders (15,16). Among them, occipital nerve stimulation (ONS) has been favored for an assumed low rate of major complications, and substantial effectiveness in the response to treatment (17–21). Yet, data focusing on procedure-related complications are sparse, and literature dealing with direct treatment costs concerning procedure-related hardware as well as hospital costs for the health insurance system (based on the German Diagnosis Related Groups system (G-DRG)) are lacking (22). This prospective observational study reports treatment results of a single-center experience with 27 patients undergoing ONS for CCH or CM. Special attention was given to long-term complication rates and costs directly related to therapy. Study purposes and the treatment protocol were approved by the local ethics committee of the Essen Medical School (09-4143).
Material and methods
Patient selection and evaluation
Between December 2008 and September 2012, a total of 27 patients with refractory CCH (24) or CM (three) underwent ONS. Patient selection was performed at the Headache Outpatient Clinic of the Department of Neurology by experienced headache specialists. Headache refractory to treatment was diagnosed for CCH if the patient continued to suffer daily attacks without attack-free periods for more than four weeks per year despite prophylactic medication either alone or in combination (verapamil > 450 mg/day (d), topiramate >150 mg/d, lithium according to blood serum levels), or if side effects of the prophylactic or acute medication impeded sufficient control of attacks. CM was diagnosed according to International Headache Society (IHS) 2004 criteria in patients suffering 15 or more headache days per months with at least eight attacks fulfilling IHS criteria for migraine (23). CM was defined as refractory according to the recommendations of the German Migraine and Headache Society in patients who suffered persistent attacks despite adequate prophylactic medication and a multimodal treatment concept for more than 12 months (24). All patients were asked to keep a headache diary for at least three months to define an individual baseline of headache days and/or daily attacks. Patients were informed about the observational study and consent was given for participation. Prior to operation patients were assessed with a custom-made, nonstandardized questionnaire. A magnetic resonance imaging scan (MRI) of the neurocranium including contrast-enhanced series was performed to rule out secondary headache disorders.
Implantation
All patients underwent a preliminary implantation of bilateral electrodes, under general anesthesia with the patient in the prone position in a standardized fashion described previously, that were externalized at the flank (21,25). In all patients ONS was performed bilaterally for CCH and CM via a midline incision 3 cm beneath the external occipital protuberance. For stimulation either eight-pole (e.g. Octrode®, St. Jude Medical, St. Paul, MN, USA), or four-pole (e.g. Pisces Quad Plus®, Medtronic Inc., Minneapolis, MN, USA) electrodes were employed. Electrodes were placed strictly subcutaneously, targeting from the midline to the mastoid. Placement of the leads parallel to the atlas was intraoperatively ensured by fluoroscopy. A restrained loop was placed in a subcutaneous pocket suprascapular before the leads were connected to extension wires at the midthoracic level. Extension leads were externalized at the flank and secured with atraumatic suture (e.g. Prolene 2.0®, Johnson & Johnson GmbH, Neuss, Germany) to the skin. Intraoperatively, only adequate resistances were verified (200–500 Ohm), as from our experience this operation technique warrants a highly reproducible stimulation of the main trunk of the greater occipital nerve. Therefore, the intraoperative testing of the achieved paresthesia appears to be superfluous.
Stimulation settings
Leads were programmed in a way that paresthesia covered the occipital and nuchal region in the course of the greater occipital nerve. Stimulation parameters were only partially prefixed (impulse width of 390 us, at 40 Hz), which was convenient for all patients. All patients underwent a test stimulation period of 30 days (CCH) and 45 days (CM), respectively. During the test phase patients continued the headache diary. Success of stimulation was determined by a 50% reduction of headache days or attacks, and/or relief of pain intensity during the attacks as rated on a numeric rating scale (NRS) from zero (pain free) to ten (maximum pain intensity). Evaluation of the diaries was performed interdisciplinarily by a neurologist and a neurosurgeon. Patients responding to treatment received an implantable permanent generator (IPG; e.g. Synergy®, Medtronic Inc; EonC®, EonMini®, St. Jude Medical). Implantation site of the IPG was to the patient’s preference: either abdominal or gluteal.
Complications and costs
During the follow-up (FU) period, all complications were recorded and classified as procedure related or coincidental. To evaluate treatment costs directly linked to the procedure, we prospectively assessed all expenses for hardware on the basis of each current proposal for electrodes and IPGs at that time. Additionally, costs for hospitalization were included based on the G-DRG for CCH (G44.0) and complex migraine (G43.8), respectively. International Classification of Diseases—10 German Modification (ICD-10-GM) and the Operation- and Procedure-Classification (OPS) for 2012 were used to calculate G-DRG reimbursement including cost weight, base rate and adjustment factors. The OPS for peripheral nerve stimulation (PNS) were 5-059.81 and 5-059.c1, respectively, triggering DRG B17B with a reimbursement of €4,084 per hospitalization. In case of readmission of the patient for arbitrary reasons within 30 days, cases were conflated according to current regulation of the German health care system. Procedure surcharges were calculated only on the basis of cost of the hardware, leaving individual hospital negotiations with health insurance companies out of consideration.
Results
Diagnosis, gender, length of follow-up (months) and response to treatment for each patient.
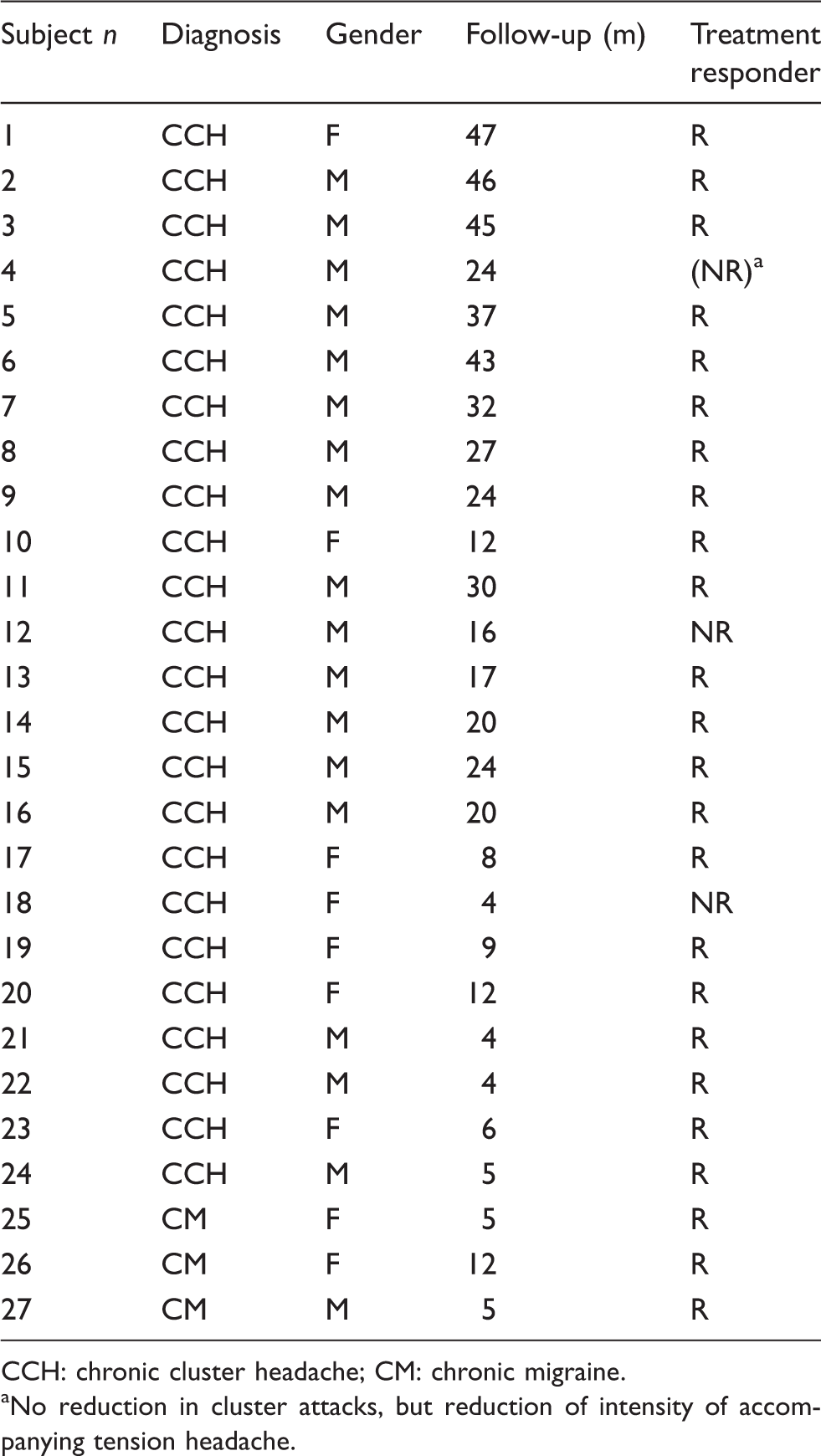
CCH: chronic cluster headache; CM: chronic migraine. aNo reduction in cluster attacks, but reduction of intensity of accompanying tension headache.
Clinical FU
Two patients did not respond to treatment, both suffering from CCH. One CCH patient did not respond with a reduction of the cluster attacks, but felt substantial relief of the accompanying migraine. Overall response rate in this single-center series was 89% at three months short-term FU. At last FU, 21 of 27 patients remained responders to ONS therapy (78%). All patients followed for more than six months reported intercurrent episodes with a significant increase of cluster attacks or headache days with aggravated migraine at latest FU, requiring medical intervention by increased doses of prophylactic drugs or acute medication.
CM patients suffered on average 27 headache days (20–30 days) preoperatively. At last FU mean headache days dropped down to mean 20 days (10–30 days). Two of three patients needed fewer triptans during migraine attacks. All CM patients stated that the duration of migraine attacks was reduced by 50%.
Patients with refractory CCH experienced five attacks per day on average (one to 14 attacks/day) with a mean pain score of 8 on the NRS preoperatively. When seen for latest FU, the number of attacks declined on average to three (zero to eight attacks/day). Additionally, the NRS score was lower at an average of 5 (0–9 NRS). Patients consumed 1.5 triptans (range zero to six doses) per day before stimulation. At latest FU a mean of 0.9 doses/day was used. Six patients were able to terminate the use of triptans for acute medication during the attacks. Thirteen patients repeatedly experienced pain-free days without CCH attacks for at least five days in a row.
Accordingly, 21 of 27 patients reported satisfaction with the operative result compared to their preoperative status in our questionnaire, but furthermore 22 of 27 patients would recommend the procedure to other patients.
Nonhardware-related complications
Five nonhardware-related complications were observed in 27 patients (19%). Two patients reported worsening of the cluster attacks by the stimulation after six months of initially successful trials. Therefore, one patient discontinued stimulation for four months before another trial was initiated. Afterward, the stimulation did not provoke cluster attacks anymore, but the initially good response returned to a level at which the stimulation only supported acute medication to interrupt the attacks more quickly. The other patient declined another trial and was lost to FU after eight months.
One patient lost effectiveness of the stimulation after six months, despite every conceivable reprogramming of the stimulation with regards to pulse width, amplitude, frequency, and continuous and discontinuous stimulation. The patient finally received intravenous dihydroergotamine (DHE; 10 mg total dose) as an off-label medication (DHE is not approved for CM and CH in Germany), which controlled CCH for an FU period of eight months, so far (26).
ONS provoked uncontrollable nausea in one patient. Nausea was directly related to the stimulation and was refractory to antiemetic medication. When the stimulation was turned off, nausea disappeared immediately. Even sub-threshold stimulation provoked this phenomenon. With all antiemetic medication failing, we even tried transcutaneous stimulation of the vagal nerve, ipsi- and contralaterally to control nausea (27). However, it did not show any influence on the complaints. The problem remains unsolved to date. So far, we have not found this complication reported in recent literature.
List of hardware-related or non-hardware-related complications and the subsequent necessity of re-operation.

Iatrogenic; maintained stimulation by second lead.
Referring to chronic cluster headache (CCH) patients only.
Hardware-related complications
In 14 of 27 (52%) patients, therapy-associated complications were noted. The majority of complications were local infections, leading to explantation of the hardware in four patients (15%). Of these patients, two experienced recurrent infections requiring several surgical interventions.
In two patients the gluteal IPGs had to be replaced for sciatic irritations. In one of these patients the extension lead presumably broke during the procedure and had to be exchanged in another operation. Two patients complained of local pain at the connector site; one of them had to be revised (21). Two generators had to be replaced because the battery ran down (after 46 and 48 months, respectively).
One stimulation electrode developed malfunction after 46 months and had to be revised. Another lead wire broke after four months, but stimulation could be performed for both sides with the contralateral electrode. Thus, the broken lead did not have to be replaced. This might be explained by the fact that in the same patient an iatrogenic lead displacement of an electrode occurred. When the extension wires were exchanged during IPG implantation, the stimulation lead was presumably pulled for too long, displacing it for about 3 cm compared to preoperative X-rays. Still, the lead was in place to establish a sufficient stimulation over the occiput in the patient, even more covering the contralateral site with the broken lead.
One extension lead had to be exchanged four weeks after implantation. Technical investigation of the electrode suggested a damage of the isolation, suggesting an iatrogenic complication. Yet, peri- and postoperative testing of the system in this patient showed normal resistances. The extension lead had to be replaced, and the system has worked ever since.
Hospitalization costs
Breakdown of in-hospital costs for the test period, secondary and subsequent operations.

Interventions at the peripheral nerve system.
Hardware costs
Standardized implantation procedures included bilateral stimulation leads (Octrode, St. Jude Medical; Pisces Quad (Plus), Medtronic Inc) and extension leads that were at first externalized, and after successful trial phase exchanged for another pair of wires. Individual treatment costs ranged between €2345 for the trial phase only, and €18,200 for successful stimulation including IPG implantation.
Hardware materials employed in all patients are listed.

IPGs: implantable permanent generators.
Overall treatment costs amounted to €761,043 for 27 patients in a 47-month period. Thus, mean treatment costs were calculated at €28,186 per case. This calculation neglects the continuing visits at the headache center, neurosurgical outpatient department or neurologist, as we deemed the effect of the consecutive costs of minor importance for overall treatment costs.
Discussion
Neuromodulative procedures have been shown to be effective as an additional treatment option for refractory headaches for more than a decade. For CCH, randomized trials with sham stimulation were not yet performed. Weiner and Reed were the first to report on ONS for intractable occipital neuralgia (18). Subsequently, ONS was adopted for other primary headache disorders in refractory conditions (19,28). Case series with short- up to long-term FUs were reported that showed good to excellent response of CCH and CM to ONS (19,20,29). Effectiveness of the stimulation was comparable to published data of DBS procedures but harbored a lower morbidity (15,30). Yet, multicenter studies conducted to analyze the value of ONS for the treatment of refractory CM lacked evidence of significant impact on CM (31,32). Nevertheless, CM and CCH patients seem to respond to treatment in about 80% of the cases with a reduction of headache days, headache attacks and/or reduction of pain intensity between 30% and 100% (20,21,29,32–34). Individual reduction of attacks and headache days vary significantly between studies (17,20,29,31–33,35,36).
Predictive factors for response to ONS could neither be established from the current literature nor from our own experience (37). Assuming a success rate of 80%, it is debatable whether lead electrodes and IPGs should be implanted all-in-one or whether a trial phase is justified to selected responders and nonresponders. From our personal experience, we suggest performing a test phase rather than implanting an all-in-one procedure (21). One may assume that one or two out of ten IPGs will be wasted in patients who do not benefit from ONS. This will spare treatment costs for IPGs of at least of €9939 before taxes per case (calculated for Synergy®, Medtronic Inc, cheapest multichannel IPG required with use of bilateral electrodes). Still, costs of the second operations rise and cumulate for all cases.
Complications have been reported in nearly every study, but morbidity rates can be taken only from systematic reviews (22,33). In the beginning of ONS, lead dislocation was a major issue (19,35). Using paddle electrodes reduced the rate of dislocated electrodes almost to zero. Paddles require a more invasive surgical approach, but may be associated with less scar formation around the leads (33). With improvement of implantation techniques, it became clear that creation of stress loops leads to stable placement of electrodes (38). Interestingly, the use of externalized extension leads did not cause more dislocations, nor is the location of the IPG (abdominal or gluteal) associated with increased lead migration in our series. Strict standard operation procedures (SOP) may help implanters to further reduce the rate of lead dislocations as well as to reduce the infection rate.
Infection is a hazardous complication for any implanted device as it necessitates explantation of the whole system in most cases (33). Perioperative administration of intravenous antibiotics (preferably third-generation cephalosporines) have lowered infection rate to a minimum (39). Even though we externalized the extension leads for 30 and more days, we did not find an increase of infections at the exit site or in conjunction with the leads themselves. Of the four patients suffering this specific complication in our series, infection site was distant to the extension leads. In two cases infections developed at the IPG site, far from the externalization and after removal of the extension leads. One of these patients suffered a localized infection around the connector of the stimulation lead prior to the IPG infection, due to a fistula that was caused by the laced thread securing the connector. In the remaining two patients repetitive generalized infections occurred despite extremely careful handling, strict aseptic proceedings and, after first relapse, performing an all-in-one implantation afterward. Both patients suffered from defilement of the skin and acne with history of recurrent pustules. After suffering infections for the second time implantation was abandoned in both patients. Even though we consider the complication rate high in the present series, the rate of re-operations is similar or even lower comparatively to previously published studies (20,31).
CCH and CM both carry a severe burden for the patients in regards to social isolation, but also economic distress. Direct treatment cost in CCH patients were calculated up to €9073 for a six-month period in a tertiary headache center (2). Stokes et al. calculated health care costs in CM patients to be substantially lower, amounting to approximately €1535 (United States (US)), and €588 (Canada) within one year, respectively (12). In a recent analysis, annual treatment costs for CM in Germany were estimated to be €1495 (13). In our series, individual average treatment costs for ONS were calculated to be €28,186.63 over a 48-month period (mean individual FU 20 months). So far, two generators had to be exchanged because of battery discharge. From our data, we suppose that nonrechargeable IPGs in ONS will have a durability of about four to six years. Lifespan for rechargeable IPGs is indicated for about 10 years (e.g. Eon Mini™ IPG, St. Jude Medical, technical information), producing even higher individual therapy costs.
Treatment costs of ONS have to be balanced against conservative pain treatment costs. Unfortunately, to date there is virtually no report on savings in therapy costs based on the calculation of ONS-related costs and the reduction of medical treatment costs. A major drawback of the present study is that we can offer only sparse information on the reduction of conservative treatment costs. Assuming that a daily intake of 1.5 triptan doses preoperatively costs €49,31 (calculated for Imigran®-Inject, GlaxoSmithKline GmbH & Co KG, Munich, Germany; single-dose equivalent €36.98), conservative medical treatment costs were reduced at latest FU to an average of €33,28 (0.9 doses/day) for CCH patients. However, a part of the patients used triptans nasal spray, which reduced the costs and most migraine patients are treated with oral triptans. CM patients rather use oral triptans or nasally administered triptans in severe attacks. This dilutes the economic aspect for ONS in CM further.
Furthermore, it is important to compare ONS treatment costs with treatment expenses managing other chronic neurological diseases. For this, we gathered data on therapy costs of Parkinson’s disease (PD) treatment with DBS and disease-modifying therapy for relapsing–remitting multiple sclerosis (MS). Managing PD with DBS is a widely accepted treatment option and numerous reports on long-term FUs have been published thus far. With respect to analysis of treatment costs, it is of great importance to outweigh procedure- and hardware-related costs against substantial decrease in medical costs. In a recent analysis patients receiving DBS versus best medical treatment (BMT) were followed prospectively for six months, identifying a decrease in levodopa equivalents (LEDD) by 13% in the treatment group. Still, medical costs did not decrease significantly in this study as a raised consumption for dopamine agonists and catechol-O-methyltransferase (COMT) inhibitors was found (40). However, in another study a substantial reduction in medical treatment costs for patients undergoing DBS for stimulation of subthalamic nuclei was found after one year. Hardware- and procedure-related costs for the initial implantation (€18,456) were comparable to our data (41). The study period, again, is too short to analyze subsequent costs of battery replacement.
Cost-of-illness studies in MS revealed that disease-modifying therapies account for 69% of the total annual treatment costs. Estimated mean annual MS-related health care costs in the US amount to $23,434 (42). A recent prospective study of annual treatment cost in Germany for a patient with relapsing–remitting MS totaled €25,270 (indirect costs: €4457, direct costs: €17,792); costs of secondary progressive MS are 1.7-fold higher (43). MS has an estimated prevalence comparable to CCH, therefore treatment costs for disease-modifying therapies can be balanced against treatment costs of ONS in CCH. Bearing this in mind, one might conclude from our data that ONS is an expensive treatment option initially, but concerning direct subsequent costs of ONS, therapy expenses may further decrease with length of observation period and ongoing success of therapy.
In our series we did not find a single patient who remained completely pain free after ONS, even though patients reported attack-free intervals during three months. Thus, remaining conservative medical treatment costs have to be taken into account when counting costs for ONS at all.
Nevertheless, for the clinical results in our series more than 80% of the patients were responders to ONS and gained substantial relief of frequency of attacks, intensity of attacks, and duration of attacks, respectively. This is in accordance with previously reported data (18,20,29,35). It is also reflected in the questionnaires, in which 78% reported that they were satisfied with their physical and psychological conditions compared to preoperative status. Furthermore, 81.5% would recommend ONS to other patients with refractory headaches. Success of treatment equally accounted for CCH and CM. Yet, mid- and long-term FU data for CM in our series cannot be provided. A longer FU for our CM patients has to be awaited. Additionally, it has to be kept in mind that any study on ONS is subjected to a negative selection bias as so far only the most refractory patients are subjected to ONS. This in turn justifies using even an expensive treatment as every other therapy failed in these patients.
Conclusion
ONS is an expensive procedure that should be reserved for patients with refractory headaches. The method carries a substantial risk of complications; among them infection seems to be of critical importance. In patients who respond to treatment attack-free periods of several weeks can be accomplished. The majority of patients will still suffer attacks, but they decrease in frequency and intensity as compared to an individually ascertained baseline. In comparison to other progressive neurological disorders, ONS for CCH and CM has a relatively high initial cost factor that may favorably decrease over a long observation period concerning annual individual treatment costs.
Clinical implications
ONS is a valuable but still cost-intensive therapy for treating refractory headache disorders. ONS harbors a significant complication rate even in experienced hands. Patients who are responsive to treatment may experience a substantial decrease of frequency of cluster attacks and/or intensity of their headaches days.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
O. Mueller has received honoraria from St. Jude Medical, Autonomic Technologies, and a restricted grant from Medtronic. C. Gaul has received honoraria for contributing to advisory boards or oral presentations from St. Jude Medical, Electrocore, Autonomic Technologies, and a restricted grant from Medtronic. H. Diener has received honoraria for contributing to advisory boards from St. Jude Medical and Medtronic. P. Dammann, K. Rabe, V. Hagel and U. Sure have nothing to declare.
