Abstract
Introduction
Individuals with tension-type headache (TTH), in addition to headache pain, typically suffer from pericranial muscle tenderness and increased cervical muscle tone. Physical and physiological outcomes related to musculoskeletal function, however, are not commonly assessed in clinical studies and not systematically proposed as outcome measures in headache-related practice guidelines.
Objectives
To review which musculoskeletal outcomes are used in the clinical assessment of patients with TTH and which are associated with headache pain and related dysfunction.
Methods: Literature searches were performed in MEDLINE, PubMed, the Cochrane databases and EMBASE using terms relating to musculoskeletal physical outcomes in TTH.
Results
Twenty-six studies met selection criteria. Physiological outcomes typically reported in laboratory studies were trigger points, pressure pain threshold, range of motion and tenderness. A greater number of trigger points and lower pressure pain threshold were reported in patients with episodic TTH in comparison with healthy subjects. Individuals with chronic TTH, when compared with non-headache controls, consistently showed a greater number of trigger points, a lower value of pressure pain threshold and a more severe forward head posture.
Conclusion
Musculoskeletal outcomes, such as trigger points, pressure pain threshold and forward head posture should inform TTH pathophysiology, diagnosis and interdisciplinary patient care.
Introduction
Tension-type headache (TTH) is characterized by a bilateral, pressing, tightening pain of mild to moderate intensity, occurring in short episodes of variable duration (30 minutes to 7 days) (1,2). TTH is the most prevalent type of headache across all age groups worldwide, with an occurrence reaching over 70% in some populations and it has the greatest socioeconomic impact of any primary headache type (3,4).
Individuals with TTH, in addition to headache pain, typically suffer from pericranial muscle tenderness and increased cervical muscle tone. Criteria of the 2nd edition of The International Classification of Headache Disorders (ICHD-II) (2) suggest that pericranial muscle tenderness on manual palpation, preferably as pressure-controlled palpation, may serve as a reliable physical measure, together with other symptom-based criteria, to diagnose TTH.
The purpose of this review is to address two fundamental questions for this headache phenotype:
Which musculoskeletal physical outcomes are commonly used in the clinical assessment of patients with TTH? Are any of the physical outcomes sufficient to be considered as TTH pain and related dysfunction biomarkers that could be applied objectively in clinical practice and/or future TTH research?
The importance of these questions and related research is driven by the need for effective treatment(s), which stems from patient burden and the economic impact of TTH.
A neurobiological basis for TTH is widely accepted (1). Pain detection thresholds are often decreased in patients with frequent episodic TTH (ETTH) (5,6), whereas both pain detection and tolerance thresholds decrease in patients with chronic TTH (CTTH) (6–10). This nociceptive hypersensitivity is consistently found in response to different stimulus modalities in various tissues at cephalic and extracephalic locations in patients with CTTH, strongly suggesting that synaptic transmission of nociceptive input within the central nervous system is increased in this population (1). Peripheral sensitization of myofascial nociceptors may play a role in increased pain sensitivity, but firm evidence for a peripheral abnormality is lacking (11). Therefore, it is hypothesized that the increased myofascial pain sensitivity in patients with TTH is attributed to central factors, including sensitization of second-order neurons at the level of the spinal dorsal horn/trigeminal nucleus, sensitization of supraspinal neurons and decreased antinociceptive activity from supraspinal structures (7).
ETTH (between 12 and 180 headache days/year) or CTTH (≥ 180 days/year) clinical studies usually focus on the efficacy of innovative treatment approaches. On the other hand, laboratory or fundamental TTH research explores physical and physiological outcomes related to musculoskeletal function and physiology. These physical and physiological outcomes, however, are not commonly assessed in clinical studies and not systematically proposed as outcomes measures in headache-related practice guidelines (2,12–14). Nevertheless, physical outcomes may provide relevant information regarding the clinical evolution as well as the physiological processes underlying TTH.
Methods
Scoping studies are used as a methodology to inform future research directions. This secondary research extracts breadth of scientific evidence from the literature rather than depth, and characterizes meaning for interdisciplinary knowledge transfer. Aims of this paper include enhancing evidence exchange between headache specialists, neuroscientists and biomechanics researchers.
Literature search strategy
The following keywords (MESH and non-MESH terms) and search terms were used alone and in combination:
Tension type headache [MeSH] AND (electromyography OR force OR stiffness OR endurance OR pressure pain threshold OR range of motion OR cross sectional area OR tenderness OR forward head posture);
Tension type headache [MeSH] AND (electromyography OR muscle strength OR physical endurance OR trigger points OR range of motion OR anatomy cross sectional OR tenderness OR forward head posture);
Tension-Type Headache [MeSH] AND (Electromyography [MeSH] OR Muscle Strength [MeSH] OR Physical Endurance [MeSH] OR Trigger Points [MeSH] OR Range of Motion, Articular [MeSH]).
Search terms were applied to identify peer reviewed scholarly publications from databases including, but not limited to, MEDLINE, PubMed, Cochrane databases and EMBASE. Additional data sources included the authors’ pre-existing knowledge of the literature and manual review of reference lists of laboratory studies. Studies published between 2004 (second ICHD classification (2)) and 2011 were retrieved. The search strategy was restricted to English and French publications.
Evidence selection criteria
Search results were electronically screened in two stages: title/abstract and full text for relevance. Only laboratory studies and interventional clinical trials involving subjects with TTH as diagnosed in accordance with the second version of the ICHD classification criteria (2) were selected as evidence. Physical and physiological outcomes of interest included tenderness, electromyography data, maximum voluntary contraction, muscle endurance, ranges of motion, presence of trigger points, cross sectional area, pressure pain threshold and forward head posture. Studies looking at variables such as muscle hardness, postural control and proprioception were excluded because of a limited amount of information. Review articles using the ICHD-1 were not assessed, as none focused on physiological outcomes. Oral communications, as well as studies involving animals or including only patient-reported outcomes (e.g. Visual Analogue Scale (VAS) or similar subjective scales) were excluded. Studies with unspecified headache types or mixed types of headache in the same treatment group were also excluded.
Data synthesis and analysis
Study data were extracted by two of the authors (AAM and JA) and entered into summary tables according to objectives, methods (laboratory or clinical study) and outcome measures. Table 1 (see Appendix) presents key features for each study included: headache population, sample size, objective outcomes and scientific/clinical significance of the reported results. Key features for each study included headache population, sample size, objective outcomes and scientific/clinical significance of the reported results. Data were gathered and when possible, normalized changes in outcome measures (% of change) were calculated. Relationships between physical/physiological outcomes and subjective clinical outcomes (e.g. headache intensity, frequency, duration) were documented as findings of interest when reported in the research articles.
Results
A total of 228 articles were identified from the literature search and 26 articles fulfilled selection criteria (Figure 1).
Flowchart of the study selection process.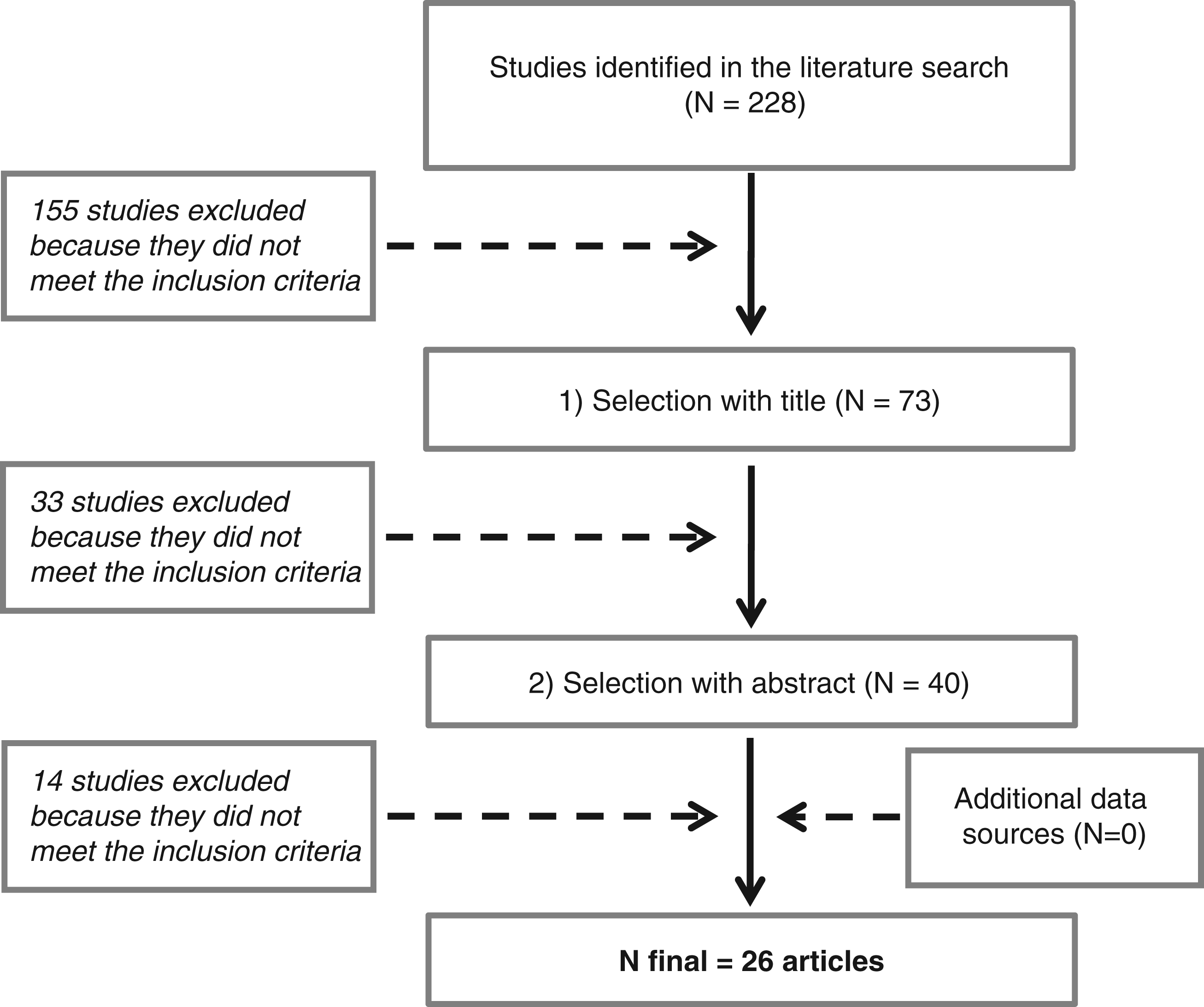
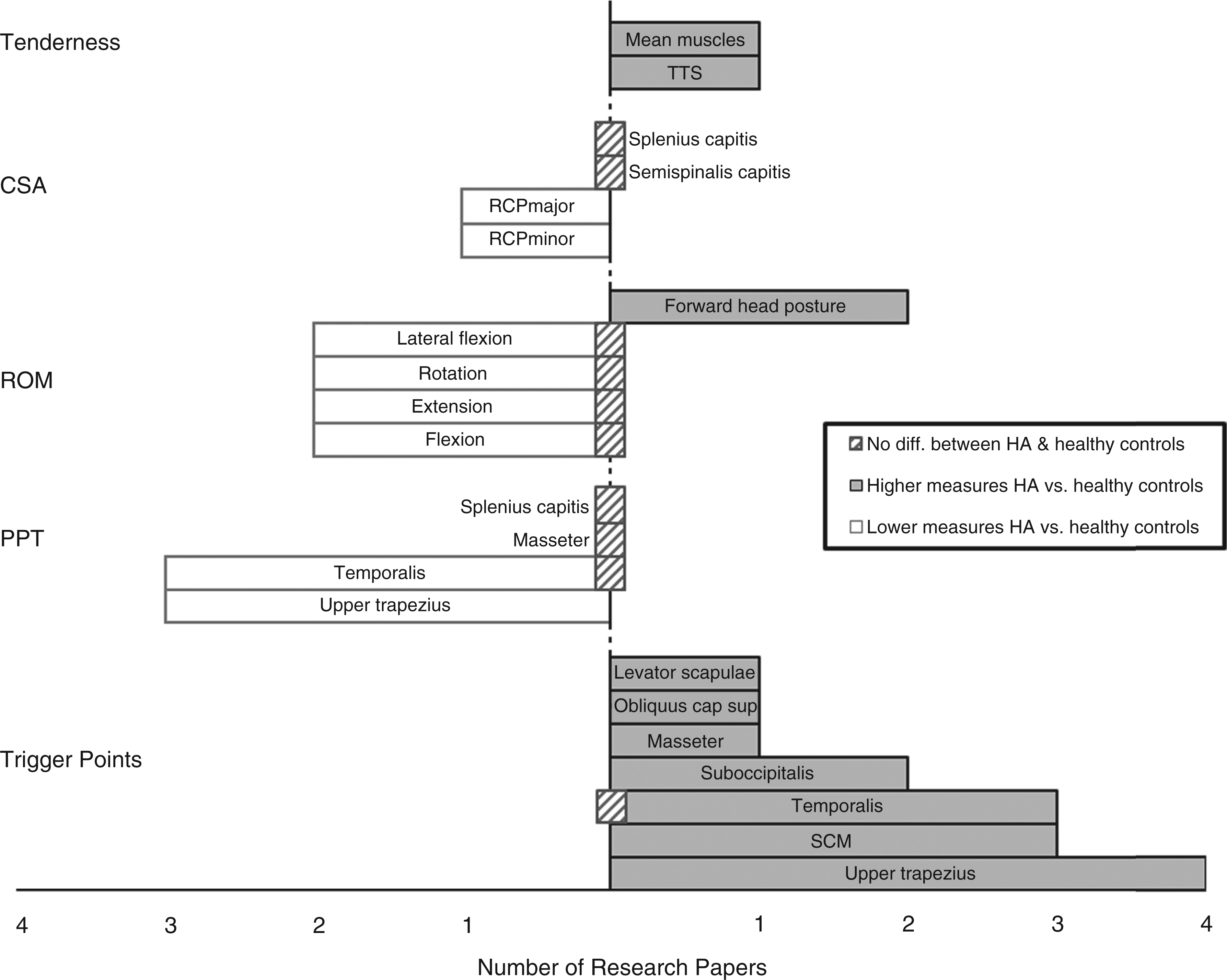
Of the 26 studies, 22 were laboratory studies (6,8,15–34) and four were clinical studies (35–38). Physiological outcomes typically reported in the literature included pressure pain threshold (PPT) and range of motion (ROM) in clinical studies and trigger points (TrPs), PPT, ROM and muscle tenderness in laboratory studies (please refer to Table 1). Outcomes, as assessed within the various research paper, are schematically represented in Figures 2 and 3.
Musculoskeletal outcome in patients with episodic tension-type headache versus healthy controls expressed in number of research papers. HA: headache; CSA: cross sectional area; ROM: range of motion; PPT: pressure pain threshold; SCM: sternocleidomastoids; *control areas. Musculoskeletal outcome in patients with chronic tension-type headache versus healthy controls expressed in number of research papers. TTS: Total tenderness score of masseter, temporalis, frontalis, sternocleidomastoids (SCM), trapezius, suboccipital muscles, coronoid and mastoid processes; mean muscles: mean of frontalis, masseter, temporalis, pterygoid, sternocleidomastoids, trapezius and neck muscle insertions; RCP: rectus capitis posterior; HA: headache; CSA: cross sectional area; ROM: range of motion; PPT: pressure pain threshold.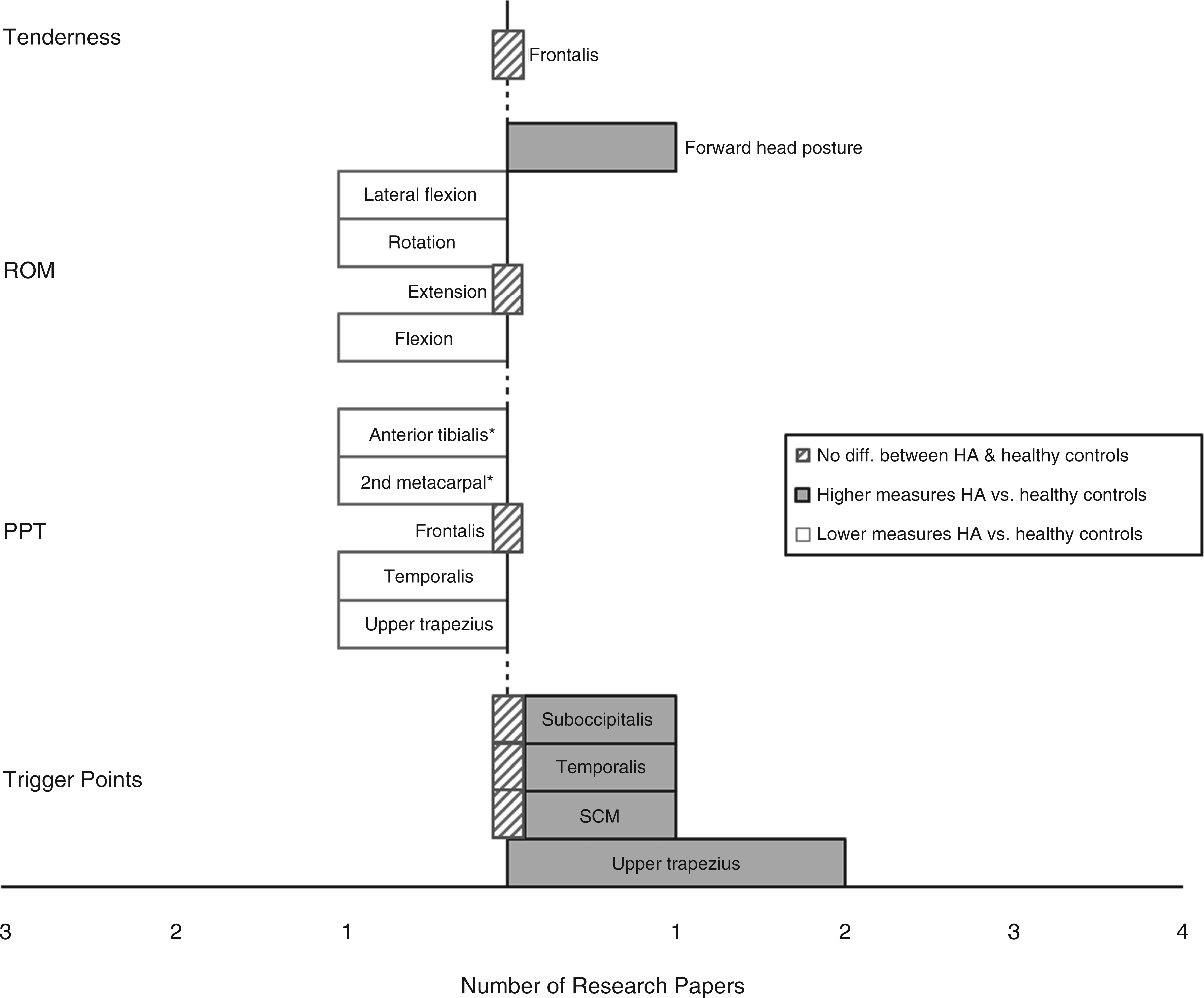
Trigger points
TrPs are defined as hyperirritable spots within a taut band of skeletal muscle (39). All experimental studies measuring TrPs reported a greater number of active or latent TrPs (trapezius, temporalis, sternocleidomastoids and suboccipital muscles) in patients with TTH than in healthy subjects (18,20,21,23,25–28,33). Among these, three studies did not report the average number of TrPs but used the number of participants who had active TrPs as outcomes (21,23,25). An increased number of TrPs (active and latents) has been reported to be 43–92% in individuals with CTTH and 59–86% in individuals with ETTH compared with healthy subjects (18,20,26–28,33). One study comparing the number of TrPs between patients with CTTH and ETTH revealed more TrPs in patients with CTTH (4.1 on average in ETTH versus 6.2 on average in CTTH) (33). Interestingly, five CTTH studies reported a positive correlation between the number of active TrPs and the following clinical outcomes: headache duration, frequency or intensity (20,26–28,33). However in ETTH, two studies reported no correlation between TrPs and clinical outcomes (18,23). Lastly, active TrPs in the temporalis muscle were associated with significantly lower PPT values (25).
Results of the clinical studies showed no difference in the presence of TrPs in patients with CTTH patients when comparing injection of Botulinum toxin A to a placebo, and the presence of TrPs within the sternocleidomastoid, suboccipital and superior oblique muscles has been acknowledged as a positive predictor of TTH in women (48,49).
Pressure pain threshold
Lower PPT was reported in trapezius, frontalis and temporalis muscles for CTTH and ETTH patients in experimental studies (8,19,20,24,27,29). PPT values had reduced by 25% to 52% in patients with CTTH and by 35% to 43% in patients with ETTH compared with control subjects when combining all muscles (8,20,24,27). Two experimental studies showed no significant differences in PPT between TTH groups (episodic or chronic) and control groups for temporalis, frontalis, masseter and splenius capitis muscles (15,32). Two studies compared PPT before and after pain-inducing stimuli in patients with TTH. Drummond and Knudsen (2011) reported PPT reduction after a pain stimulus (cold water) in patients with ETTH (15), whereas Schmidt-Hansen et al. (2007) reported lower PPT values in patients with ETTH and CTTH before and after pain stimuli (injection of hypertonic saline) versus controls (6). Lastly, two studies found that PPT, over the upper trapezius and center of temporalis muscle, was negatively correlated to clinical parameters such as frequency and duration of headache episodes in patients with CTTH (24,29). One study showed the same correlations for patients with ETTH over the upper trapezius (19). Conversely, two studies failed to report similar findings (8,25).
Three clinical treatment studies measuring PPT in patients with TTH were retrieved. One study found no difference in PPT values over the temporalis muscle after massage therapy versus placebo in patients with CTTH (38). However, a reduction of pain intensity, which lasted 24 hours after the massage therapy, was reported. A separate study reported reduced pain (total pain score: 28/80 to 14/80 after 8 weeks) observed through PPT value with manual therapy (35). PPT does not appear to be a good predictor for identifying patients with TTH (36).
Tenderness
Tenderness is considered a relevant criterion for differential diagnosis of individuals with TTH by ICDH-2 classification (2). Two out of four experimental studies assessed muscle tenderness using a palpation technique according to Langemark and Olesen criteria (40) or Bendtsen (41). Each zone was scored according to a four point numerical rating scale from 0 to 3, where 0 is no pain and 3 is considerable pain. In these same studies, higher tenderness scores were reported in patients with CTTH in comparison with healthy controls (8,34). The other two studies used modified versions of Langemark and Olesen criteria (31) or a sharpness measure to quantify muscle tenderness (15). Mongini et al. compared muscle tenderness in patients with CTTH versus patients with migraine and found no difference (31). Reduction of tenderness score after administration of a pain stimulus (cold water) was observed only in the control group; and no difference at baseline between patients with ETTH and controls (15). With regard to the relationship between tenderness and clinical parameters of headache, none of the studies reported any correlations involving intensity (8,15), frequency (8,34) or duration (8,34). However, a negative correlation was found between PPT and tenderness score (8). In one of the clinical studies, tenderness score appeared to be a strong positive predictor for identification of patients with TTH (36).
Electromyography and muscle functions
Decreased neck muscular strength was reported for patients with CTTH versus controls in both flexion and extension movements (16), as measured by a neck strength system in a seated position. Increased amplitude of EMG signal in patients with CTTH was also reported (16). Furthermore, patients with CTTH showed greater co-activation of an antagonist muscle during cervical extension and flexion contraction in comparison with healthy subjects (16). Finally an increased amplitude of the jaw-stretch reflex in patients with CTTH was reported in comparison with healthy controls (32).
A clinical study showed augmentation of the neck flexor endurance time (32.1 to 45.1 seconds) for patients with CTTH with manual therapy versus placebo treatment (28.8 to 31.7 seconds) (35).
Muscle atrophy has been suggested as a perpetuating factor for chronic pain, as it could affect the proprioceptive output from the affected muscles (7). Smaller neck muscle cross-sectional area (CSA) was reported in patients with CTTH in comparison with healthy controls (17), as well as an inverse relationship between CSA and intensity, frequency or duration of headache attack(s) (17). Moreover an association was found between higher number of active TrPs and smaller CSA (size) of suboccipital muscles in patients with CTTH versus those with latent TrPs (21).
Range of motion and forward head posture
Results showed lower flexion and lateral flexion ROM in patients with CTTH and ETTH (adults and young) when compared with healthy individuals (18,22,30). However, one study, assessing patients with ETTH, failed to identify neck movement differences in comparison with healthy controls (33). As for neck rotation ROM, several authors describe a decrease for patients with ETTH and CTTH when compared with healthy controls (18,22,33). Studies reporting neck extension ROM in patients with CTTH or ETTH were highly variable (see Figures 2 and 3) (18,30,33,46). When all movements were considered, patients with CTTH showed decreased ROM ranging from 7 to 15 degrees when compared with healthy subjects (22,30,33), whereas patients with ETTH showed a reduction between 7 and 21 degrees (18). Finally, in both CTTH and ETTH patients, no correlation between ROM and intensity (18,33), frequency or duration of headache attack (18,30,33) could be identified. Only rotational movement ROM was found to be negatively correlated with duration and frequency of headache episode (22).
Forward head posture is measured by calculating the angle between the horizontal line passing through C7 and a line extending from the tragus of the ear to C7. Results showed more severe forward head posture in patients with ETTH (18) and CTTH (22,33) in sitting and standing position versus healthy subjects. However, one study found no difference between patients with ETTH and healthy subjects (33). Difference in degrees between patients with CTTH and healthy controls ranged between 4.4 and 8.8, whereas difference between patients with ETTH and healthy controls was 5 degrees. Positive correlations were found between number of TrPs and forward head posture, indicating that forward head posture becomes more serious as the number of TrPs increase, especially active TrPs (upper trapezius, sternocleidomastoids, suboccipital and temporalis) (33). The same results were observed only with active TrPs in the sternocleidomastoids (18,26) and temporalis (18) muscles. Another positive correlation was found between forward head posture and ROM in all cervical motions, which means the greater the forward head posture, the lesser the neck ROM (22).
ROM outcomes were also identified in three clinical studies (35–37). One study showed ROM increases in all directions in patients with CTTH who receive manual therapy (a combination of mobilizations of the cervical and thoracic spine, exercises and postural correction) when compared with CTTH patients who did not receive therapy (35). Conversely, no difference in ROM was observed between patients with CTTH who received Botulinum toxin A injections and those who received placebo injections (37). Finally, ROM (rotation only) was shown to be a useful variable for characterizing patients with TTH in one clinical study (36).
Discussion
TTH is a common condition, in which clinical experience and the research literature confirm the contribution of neuromuscular factors. Physiological outcome measures are commonly studied in individuals with low back or cervical pain (47,48) and have been used effectively to discriminate healthy subjects from symptomatic patients with these conditions (49–51). Such outcomes may provide relevant information regarding the clinical evolution as well as the physiological processes underlying painful neuromuscular/skeletal conditions.
PPT and TrPs were the most frequently reported outcomes, followed by ROM and muscle tenderness (see Figures 2 and 3). The clinical treatment studies included PPT and ROM, which was unexpected given that pain threshold assessments are usually less common than strength or endurance assessments in clinical practice settings.
The literature synthesis revealed mixed results regarding physical measures in patients with ETTH. A greater number of TrPs in trapezius muscles and lower PPT were reported in patients with ETTH in comparison with healthy subjects. The literature was more robust for patients with CTTH (Figure 3). Individuals with CTTH, when compared with non-headache controls, consistently showed a greater number of TrPs (trapezius, temporalis, sternocleidomastoids and suboccipital muscles), a lower value of PPT (trapezius and temporalis muscles) and a more severe forward head posture. These results suggest that the development of chronic symptoms (pain frequency, intensity and duration) is associated with concomitant changes in musculoskeletal physical outcomes. Chronic TTH is a key contributor to reduced functionality and economic burden, and reinforces the need for early, effective treatment(s).
Although ICHD-2 clearly highlights the association between TTH and pericranial tenderness, only a few studies explored this outcome. Our results, however, are comparable to those found in previous studies using the first version of the ICHD (52).
The physical manifestation of chronic pain and the precise composite of disrupted physiological measures may, in time, serve as markers of pathogenesis in descending modulatory efferents secondary to central sensitization.
It is clinically relevant to ask whether there are links between physical/physiological outcomes and subjective clinical outcomes. Five studies showed a positive correlation between the number of active TrPs and the duration, frequency or intensity of headache episodes in patients with CTTH. Referred pain from neck muscles in the presence of active TrPs is also likely to be associated with head pain. Links between either PPT or ROM and headache clinical parameters in patients with TTH remain unclear. Finally, one study identified an inverse relation between CSA and duration, frequency or intensity of headache in patients with CTTH (17). These findings for CSA should be interpreted with caution in the absence of corroborating evidence.
Although not a primary objective of this literature review, it was of interest to assess whether certain physical measures typify the transition from acute to chronic TTH. Two studies compared physical measures directly in patients with ETTH versus CTTH (6,33) and showed no difference between them for PPT, forward head posture or even ROM. However, Sohn et al. found a higher number of active TrPs in patients with CTTH (33). Comparisons between patients with TTH and headache-free individuals indicate that changes in physical and physiological outcomes are similar in ETTH and CTTH (Figures 2 and 3). The number of studies supporting these changes is higher for patients with CTTH.
Limitations
As a result of the heterogeneity of studies, the findings of this review should be interpreted with caution. Conflicting study results (Figures 2 and 3) may be a result of different measurement tools used across studies and, in some cases, variable methods of assessment (e.g. differences exist regarding levels of reliability and validity between tools measuring ROM (60)). Variations in inclusion and exclusion criteria will also impact experimental findings. The physical outcomes included in this review were deemed to be closely related to head and neck muscular and articular function. Other physical outcomes such as joint position sense (61,62) and needle EMG (63,64) may also be clinically relevant, but were not included in this review because they involve instrumentation rarely available in clinical practice settings.
Research recommendations
TTH warrants ongoing experimental attention and further research investment. While designing future laboratory studies, researchers should consider including outcomes such as TrPs, PPT or forward head posture that will most likely be included in clinical trials. Including these outcomes in clinical trials may generate relevant information regarding the pathophysiology of TTH.
Given the various outcomes studied and the overall heterogeneity of the studies included in this review, determining whether physical and physiological outcomes inform clinical assessment and treatment of TTH should remain an active domain of research.
Conclusion
The review revealed that musculoskeletal physical outcome measures, such as TrPs, PPT and ROM are commonly studied in patients with TTH. An increased number of TrPs, a reduction of PPT and more severe forward head posture could help in the diagnosis and follow-up of patients with TTH. Whether these outcomes can further inform the prognosis of patients with TTH or other forms of headaches remains to be determined.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Clinical implications
This study reviews the musculoskeletal outcome changes observed in patients with tension-type headache.
Trigger points, pressure pain threshold and forward head posture can be considered relevant to the pathophysiology, diagnosis and care of patients with tension-type headache.
Appendix
Tension-type headache results summary table. CTTH: chronic tension-type headache; ETTH: episodic tension-type headache; M: men; W: women; SCM: sternocleidomastoid; MVC: maximal voluntary contraction; EMG: electromyography; VAS: visual analogue scale; TrPs: trigger points; PPT: pressure pain threshold; ROM: range of motion; CSA: cross-sectional area; MRI: magnetic resonance imaging; FHP: forward head posture.
References
Headache type
Study subjects
Physical and physiological outcomes
Clinical outcomes
Key findings
1. Drummond and Knudsen, 2011 (15)
Episodic tension- type headache (ETTH)
Group 1: N = 34 (23 women (W), 11 men (M)) With ETTH Age: 22 ± 3 Group 2: N = 32 (12 W, 20 M) With less than one episode of TTH per month (control group Age: 26 ± 10
Pressure pain threshold (PPT) (before and after pain stimuli): Fontralis Tenderness(before and after pain stimuli): Fontralis Pain stimuli: Participants immersed one hand in cold water for 1 minute
PPT and tenderness (before stimuli): No difference between groups Tenderness (after stimuli): Reduction only in control group PPT (after stimuli): Reduction in ETTH group (631 g to 579 g) No correlation between tenderness and headache intensity
2. Fernández-de-las-Peñas, 2007 (18)
ETTH
Group 1: N = 15 (3 M, 12 W) With ETTH Age: 39 ± 17 Group 2 : N = 15 controls (4 M, 11 W) Age: 37 ± 12
Trigger points (TrPs): upper trapezius, sternocleidomastoid (SCM), temporalis Forward head posture (FHP) Range of motion (ROM) (neck): flexion/extension, lateral flexion, rotation
Visual analogue scale (VAS)
TrPs (mean number): ETTH = 3.7 and control = 1.5 (only latent TrPs) Severe forward head posture in ETTH Lower flexion (47°), lateral flexion (34°) and rotation (58°) in ETTH than in control group (68°, 41°, 72°) Both ROM and FHP did not correlate with headache parameters
3 Fernández-de-las-Peñas, 2010 (19)
ETTH
Group 1: N = 25 (6 M, 19 W) With ETTH Age: 8.9 ± 1.8 Group 2: N = 50 controls (12 M, 38 W) Age: 8.8 ± 1.7
PPT: temporalis, upper trapezius, anterior tibialis, 2nd metacarpe
Pain scale (0–10) Beck depression inventory (BDI-II)
Positive relation (r = 0.525) between BDI-II score and headache frequency: the greater the frequency of headache attacks, the greater score of BDI-II Lower PPT for all the regions in ETTH Negative correlations: between years of headache and PPT over the trapezius (r = − 0.415) between the intensity of headache and PPT over the upper trapezius (r =−0.47)
4. Fernández-de-las-Peñas, 2006 (23)
ETTH
Group 1: N = 10 (2 M, 8 W) With ETTH Age: 35 ± 15 Group 2: N = 10 controls (3 M, 7 W) Age: 34 ± 13
TrPs: suboccipital muscles (rectus capitis right, minor and major)
VAS
Group 1: Pain = 4.3 6/10 ETTH had active TrPs No correlation between presence of TrPs and frequency, duration or intensity of headache
5. Castien, 2011 (35)
Chronic tension- type headache (CTTH)
Group 1: N = 41 (32 W, 9 M) With manual therapy (MT) Age : 40.2 ± 10.2 Group 2: N = 41 (32 W, 9 M) With usual care Age: 40.6 ± 11.3
Primary outcomes: frequency duration intensity Secondary outcomes: Cervical active ROM PPT pain perceived (trapezius and suboccipital muscles) Neck flexor muscles endurance
Pain scale
Primary outcomes at 8-week follow-up: Lower headache frequency (−6.4 days; 95% CI − 8.3 to −4.6), duration of headache (−5.3 hours, 95% CI −9.5 to −1.2) and headache intensity (−1.8, 95% CI −3.1 to −0.7) with MT Primary outcomes at 26-week follow-up: Lower headache frequency (−4.9 days, 95% CI −6.95 to −2.98) and pain intensity (−1.4, 95% CI −2.7 to −0.2) with MT Secondary outcomes at 8-week follow-up: Significantly greater ROM with MT (18.8 vs 2.0 p =0.023) Significant reduction of pain perceived over PPT values with MT (−9.2 vs 1.0 p = 0.001) Significantly greater endurance (13.0 vs 2.9 p = 0.011) with MT Differences in secondary outcomes at 26-week follow-up were no longer significant
6. Fernández-de-las-Peñas, 2008 (16)
CTTH
Group 1: N = 9 W with CTTH Age: 40.1 ± 7.2 Group 2: N = 10 controls (W) Age: 39.8 ± 6.6
EMG: Sternocleidomastoid (SCM) and splenius capitis Maximum voluntary contractions (MVC): cervical flexion and extension
An 11 point numerical pain rating scale
Group 1: Pain = 4.5 Significantly less force for both cervical flexion (32 ± 10%) and extension (24 ± 15%) maximum contractions EMG splenius capitis during extension: No difference between groups Higher EMG SCM during extension Higher EMG SCM and splenius capitis during flexion
7. Fernández-de-las-Peñas, 2007 (17)
CTTH
Group 1: N = 15 W with CTTH Age: 43 ± 12 Group 2: N = 15 controls (W) Age: 40 ± 10
Cross-sectional area (CSA) measured by MRI: extensor muscles; rectus capitis posterior minor/major, semispinalis capitis, splenius capitis
VAS
Smaller rectus capitis posterior minor and major CSA for CTTH Positive correlation between CSA of rectus capitis posterior and intensity (r = 0.72), frequency (r = 0.6) and duration (r = 0.65) of headache in CTTH
8. Fernández-de-las-Peñas, 2007 (20)
CTTH
Group 1: N = 20 (11 M, 9 W) with CTTH Age: 36 ± 11 Group 2: N = 20 controls (13 M, 7 W) Age: 35 ± 9
TrPs: upper trapezius PPT: Upper trapezius
VAS MPQ (McGill pain questions)
Group 1: Pain = 5.1 (mean) TrPs: Higher number of active TrPs for CTTH Mean number in CTTH = 1.4 Mean number in control = 0.8 (only latent) Larger referred pain area (cm2)in dominant side (4.95 ± 0.41) than the non-dominant side (2.54 ± 0.21) for CTTH CTTH with active TrPs showed greater headache intensity and frequency, and longer headache duration than those with latent TrPs PPT: CTTH showed decreased PPT (1.5 kg/cm2) than controls (2.5 kg/cm2)
9. Fernández-de-las-Peñas, 2008 (21)
CTTH
Group 1: N = 11 (W) with CTTH Age: 40 ± 10
CSA: rectus capitis minor/major TrPs: rectus capitis minor/major
Pain scale (0–10)
Pain = 4.6 (mean) 6/11 subjects had active TrPs Smaller rectus capitis minor CSA in CTTH with active trigger points than latent
10. Fernández-de-las-Peñas, 2006 (22)
CTTH
Group 1: N = 25 (11 M, 14 W) with CTTH Age: 42 ± 18 Group 2: N = 25 controls (12 M, 13 W) Age: 40 ± 12
FHP: cranio vertebral angle ROM (neck): flexion/extension, lateral flexion, rotation
VAS
Smaller cranio-vertebral angle in headaches (45.3°) than controls (54.1°) Smaller range of flexion/extension (103.9°), lateral flexion (67.1°), rotation (116°) in patients than controls (128.3°, 78.8°, 145.8°) Positive correlation between cranio vertebral angle and ROM: the greater the FHP, the lesser the neck ROM
11. Fernández-de-las-Peñas, 2010 (24)
CTTH Migraine
Group 1: N = 20 W with CTTH Age: 39 ± 8 Group 2: N = 20 W with migraine Age: 37 ± 9 Group 3: N = 20 healthy W Age: 37 ± 8
PPT (map): 11 points on trapezius
VAS
Group 1: Pain = 5.4 Lower PPT in CTTH than controls or migraine Lower PPT in upper trapezius in all groups Lower PPT in the center of trapezius than inferior part Higher PPT in cervico-thoracic part of the trapezius than in the upper trapezius Lower PPT in upper trapezius in CTTH than migraine or controls Negative correlation between frequency and duration of headache with PPT level
12. Fernández-de-las-Peñas, 2009 (25)
CTTH
Group 1: N = 40 W with CTTH Age: 40 ± 10
PPT: 9 points on temporalis TrPs: temporalis
Decreased PPT scores from the posterior to the anterior column Higher tenderness in the anterior column points Higher TrPs presence at anterior part and center of the muscle bilaterally Significantly lower PPT areas in temporalis muscle correlated with presence of active TrPs PPT levels were not correlated with headache clinical parameters
13. Fernández-de-las-Peñas, 2006 (26)
CTTH
Group 1: N = 25 (8 M, 17 W) with CTTH Age: 40 ± 16 Group 2: N = 25 controls (9 M, 16 W) Age: 38 ± 9
TrPs: upper trapezius, SCM, temporalis FHP: cranio-vertebral angle
VAS
Group 1 = 3.9 active TrPs (mean) Group2 = 1.4 latent TrPs (mean) Greater headache intensity and duration in CTTH with active TrPs in the right upper trapezius, than those with latent TrPs (p < .05) Greater headache intensity and duration in CTTH with active TrPs in the left SCM, than those with latent TrPs (p < .05) Longer headache duration with active TrPs in right temporalis muscle (p < .01) Greater headache intensity with active TrPs in left temporalis muscle (p < .05) Smaller cranio-vertebral angle in CTTH with active TrPs in the SCM muscles
14. Fernández-de-las-Peñas, 2007 (27)
CTTH
Group1: N = 30 (21 W, 9 M) with CTTH Age: 39 ± 16 N = 30 controls (21 W, 9 M) Age : 39 ± 12
PPT: Temporalis TrPs: Temporalis
Significantly lower PPT in CTTH (1.1 ± 0.2 right side, 1.2 ± 0.3 left side) vs controls (2.5 ± 0.5 right side, 2.6 ± 0.4 left side) (p < 0.001) Group 1 = 1.5 active and latent TrPs (mean) Group 2 = 0.3 latent TrPs (mean) Positive correlation between number of TrPs and duration and intensity of headache
15. Fernández-de-las-Peñas, 2007 (8)
CTTH
Group 1: N = 25 (12 W, 13 M) with TTH Age: 41 ± 14 Group 2: N = 25 controls (13 W, 12 M) Age: 39 ± 13
PPT: Temporalis Trapezius Tenderness: Masseter, temporal, frontal, SCM, trapezius, suboccipital muscles, coronoid and mastoid processes
VAS Medical outcomes study 36-Item short form (SF-36)
VAS = 5.1 PPT: Lower PPT in cephalic in CTTH (1.3 ± 0.4 for CTTH vs 2.1 ± 0.4 for controls) and neck points (1.5 ± 0.5 for CTTH vs 2.5 ± 0.4 for controls) (p < 0.001) No correlation between PPT and headache intensity, frequency and duration Tenderness: CTTH showed increased tenderness compared with controls Positive correlation between cephalic tenderness and neck tenderness (r = 0.66) Negative correlations between PPT and tenderness score None of the tenderness scores were related to headache intensity, frequency, and duration
16. Fernández-de-las-Peñas, 2011 (28)
CTTH
Group 1: N = 50 (36 W, 14 M) With CTTH Age : 8 ± 2 Group 2: N = 50 (36 W, 14 M) Without headache Age: 8 ± 1.6
TrPs: Upper trapezius, temporalis, masseter, SCM, superior oblique, levator scapulae and suboccipital muscles
TrPs: Mean active TrPs in CTTH = 4 Mean latent TrPs in controls = 0.3 The distribution of myofascial TrPs between patients and controls was significantly different for all regions Positive association between the number of active TrPs and headache duration: the greater the number of active TrPs, the longer the duration of headache attack
17. Fernández-de-las-Peñas, 2008 (29)
CTTH
Group 1: N = 15 (15 W) With CTTH Age 40 ± 7 Group 2: N = 10 (10 W) Without headache Age 40 ± 6
PPT: 9 points over the temporalis muscle
11 point numerical pain rate scale
Pain score = 5 PPT: Lower PPT values in patients as compared with controls Lower PPT levels on the non-dominant side as compared with the dominant side Lower PPT values in the anterior column of the temporalis muscle compared with the posterior part Negative correlation between headache intensity and duration and PPT at only one point (center of the muscle) over the temporalis muscle
18. Fernández-Mayoralas, 2010 (30)
CTTH
Group 1: N = 50 (13 M, 37 W) With CTTH Age: 8.5 ± 1.6 Group 2: N = 50 controls (13 M, 37 W) Age: 8.5 ± 1.8
Tenderness: Masseter, temporalis, frontalis, trapezius, SCM, suboccipital muscles, coronoid and mastoid processes ROM (total): neck flexion/extension, lateral flexion, rotation ROM (half-cycle): flexion, extension, lateral flexion, rotation
Pain scale (0–10) BDI-II
Group 1: BDI-II score = 3.7 Group 1: Pain = 5.1 Tenderness: 20 ± 5 in CTTH Lower range of flexion (55.4°), extension (69.9°), left lateral flexion (40.8°), right lateral flexion (39.2°), total ROM flexion-extension (125.1°) and total lateral flexion (80.0°) in CTTH than controls (64.6°, 78.2°, 48.8°, 48.4°, 142.8°, 97.3°) No correlation between ROM and intensity, frequency or duration of headache
19. Harden, 2009 (37)
CTTH
Group 1: N = 12 (5 W, 7 M) With CTTH Age: 49.6 ± 11.1 With the Botulinum toxin A (BT-A) injection Group 2: N = 11 (7 W, 4 M) With CTTH Age: 40.8 ± 10.3 With placebo injection
On the initial visit: TrPs: Upper trapezius, SCM, splenius capitis (involved cervical muscle groups) ROM: Flexion, extension, lateral rotation, left and right lateral flexion After 2 weeks: MPQ and PDI Participants received BT-A or placebo injection into the four most sensitive TrPs After the injections: MPQ TrPs and ROM assessment Follow-up at 2 weeks, 1, 2 and 3 months post injection: MPQ and PDI TrPs and ROM assessment New follow-up at 3 months: All participants received BT-A injections and follow-up assessment were the same
MPQ short form BDI Headache specific self-efficacy scale (HSES) State trait anxiety inventory (STAI) Pain disability index (PDI)
Headache frequency: Group 1 reported greater reductions in headache frequency than group 2 (week 1 to week 8) No difference at weeks 11-12 Headache intensity: No difference between groups ROM: No difference for each ROM planes between groups TrPs: No difference between groups BT-A injections don’t affect ratings of depressive or anxiety symptoms or self-efficacy
20. Mongini, 2005 (31)
CTTH migraine
Group 1: N = 41 W With migraine Age: 40.0 ± 11.7 Group 2: N = 34 W With CTTH Age: 37.3 ± 14.5
Tenderness (pericranial): Masseter, pterygoid (lateral and medial), temporal mandibular, temporal, cranial insertion, Tenderness (cervical): SCM, trapezius and nuke muscles
VAS STAI
STAI: No difference between groups Tenderness: No difference between groups VAS: Higher in migraine (67.90) than CTTH (52.35)
21. Peddireddy, 2008 (32)
CTTH
Group 1: N = 30 (15 M, 15 W) with CTTH Age M: 48.4 ± 7.9 Age W: 44.8 ± 3.2 Group 2: N = 30 controls (15 M, 15 W) Age M: 50.8 ± 11.4 Age W: 44.8 ± 2.2
Stretch reflex recording: EMG activity of masseter, temporalis during MVC PPT: masseter, temporalis, splenius capitis
Pain scale (0–10)
Group 1: Pain = 5.7 Lower EMG amplitude in masseter and temporalis during pre-test No difference in onset, offset, latencies and duration of the reflex response Significantly higher normalized peak-to-peak amplitude in CTTH (temporalis and masseter) No PPT difference between groups
22. Tommaso, 2006 (34)
CTTH
Group 1: N = 18 (10 W, 8 M) With CTTH Age 39.1 ± 11.5 Group 2: N = 12 (6 W, 6 M) Without headache Age 33.6 ± 12.8
Tenderness: Frontalis, masseter, temporalis, pterygoid, SCM, trapezius and neck muscle insertions CO2 laser stimulation: Electroencephalogram (EEG) recording through 19 electrodes Cutaneous heat stimuli (two series of 20 stimuli): Dorsum of the hand, frontalis, masseter, temporalis, SCM, trapezius and neck muscle insertions
Self-evaluating anxiety scale (SAS) Self-evaluating depression scale (SDS)
Tenderness (mean): Higher in patients (5.2) than controls (0.2) Tenderness was not correlated with frequency and duration of headache SAS and SDS: Difference between two groups SAS: Correlation with tenderness
23. Toro-Velasco, 2009 (38)
CTTH
N = 11 (8 W, 3 M) Age: 51 ± 15 yrs With massage therapy (head-neck) or placebo ultrasound
PPT (before, right after treatment and 24 hours after): temporalis muscles
NPRS (10-point)
Before intervention: no difference in PPT No significant group × time interaction for PPT levels in either right or left temporalis muscles. Significant group × time interaction for pain (p = 0.04) Sustained lower pain level at 24 hours post manual therapy for massage therapy (p < .05) (placebo p = 0.9)
24. Fernández-de-las-Peñas, 2011 (36)
ETTH CTTH
Group 1: N = 76 (72 W) With TTH Age: 46 ± 11.9 With multimodal physical therapy including both manual joint mobilizations and TrP therapy program
ROM: Flexion, extension, lateral flexion and rotation PPT: Temporal, upper trapezius, supra-orbital nerve (V1) Tenderness: Masseter, temporal, frontal, trapezius, suboccipital, SCM, coronoid and mastoid processes TrPs: Upper trapezius, SCM, temporal, levator scapulae, suboccipital and superior oblique
SF-36 NDI BDI-II Headache disability inventory (HDI) Global rating of change (GROC) 15-point scale
Patients category: 36 patients were categorized as having achieved a successful outcome (five or more of GROC) and 40 as non-successful outcome (four or less of GROC) TrPs: Baseline = 5.1 / Success = 5.7 / Non-success = 4.6 ROM (extension): Baseline = 55.3° / Success = 61.3° / Non success = 50.0° ROM (right rotation): Baseline = 70.1° / Success = 73.7° / Non-success = 66.9° ROM (left rotation): Baseline = 69.2° / Success = 72.8° / Non-success = 65.9° Tenderness (total score):v Baseline = 19.3 / Success = 17.3 / Non-success = 21.0° No other significant difference Strongest predictor of headache: -TrPs of SCM, suboccipital, superior oblique -ROM rotation -Tenderness -NDI -Headache duration These variables were used to form a combination of predictors for identifying patients with TTH likely to benefit from a treatment session including joint mobilization and TrP therapy
25. Schvmidt-Hansen, 2007 (6)
ETTH CTTH
Group 1: N = 24 (18 W, 6 M) With ETTH Age 38.9 ± 11.7 Group 2: N = 22 (15 W, 7 M) With CTTH Age 39.7 ± 15.1 Group 3: N = 26 (15 W, 11 M) Without headache Age 37.2 ± 10.6
Headache pain and PPT: Before and after infusion of hypertonic saline and VAS Same sequence was repeated in the next muscle Muscles examined: Temporalis, masseter and anterior tibial
Intensity of headache pain (VAS)
VAS: No difference between ETTH and CTTH PPT (baseline): Higher in control than CTTH and ETTH at all sites No difference between ETTH and CTTH Lower in women than men PPT (post injection): Higher in control than CTTH and ETTH at all sites No difference between ETTH and CTTH
26. Sohn, 2010 (33)
ETTH CTTH
Group 1: N = 36 (7 M, 29 W) with ETTH Age: 51.1 ± 14.4 Group 2: N = 23 (2 M, 21 W) with CTTH Age: 53.4 ± 16.9 Group 3: N = 42 controls (8 M, 34 W) Age: 51.7 ± 16.2
Trigger points (TrPs): upper trapezius, SCM, temporalis, suboccipital muscles Flexor head posture: Measurement of postural alignment of the head and shoulders in sagittal plan ROM: neck flexion, extension, lateral flexion, rotation
VAS
Mean number of all active TrPs: Group 1 = 4.11; Group 2 = 6.17; Group 3 = 0.57 Flexor head posture more severe in CTTH versus controls Lower range of neck rotation in CTTH (56°) than controls (68°) Positive correlation between number of TrPs and frequency (r = 0.662) and duration (r = 0.618)of headache for TTH Positive correlation between number of TrPs and degree of flexor head posture

