Abstract
Background
Supraspinal activity-dependent neuroplasticity may be important in the transition from acute to chronic pain. We examined neuroplasticity in a cortical region not considered to be a primary component of the central pain matrix in chronic tension-type headache (CTTH) patients. We hypothesised that neuroplasticity would be exaggerated in CTTH patients compared to healthy controls, which might explain (in part) the development of chronic pain in these individuals.
Methods
Neuroplasticity was examined following a ballistic motor training task in CTTH patients and control subjects (CS). Changes in peak acceleration (motor learning) and motor-evoked potential (MEP) amplitude evoked by single-pulse transcranial magnetic stimulation were compared.
Results
CTTH patients showed significantly less motor learning on the training task than CS (mean acceleration increase 87% CTTH, 204% CS, p < .05), and CS but not CTTH patients showed a significant increased MEP amplitude following training (CS: F = 2.9, p < .05; CTTH: F = 1.6, p > .05).
Conclusions
These findings suggest a deficit in use-dependent neuroplasticity within networks responsible for task performance in CTTH patients which might reflect reciprocal influences between primary motor cortex and interconnected pain processing networks. These findings may help explain the positive effects of facilitatory non-invasive brain stimulation targeting motor areas on chronic pain and help elucidate the mechanisms mediating chronic pain.
Introduction
Chronic pain conditions impose significant social and economic burdens on society, with reports that up to one fifth of the general population are affected by chronic pain (1). At present, the mechanisms responsible for the development of chronic pain are poorly understood. Increased excitability and synaptic efficacy of neurons in central nociceptive pathways triggered by nociceptive inputs (central sensitisation) are thought to play a role in the development of chronic pain (2), however, it is unlikely that central sensitisation at the level of the spinal cord alone can explain the transition from acute pain to chronic pain. More likely, is the involvement of activity-dependent neuroplastic changes at the supraspinal level. In fact, recent evidence shows unique patterns of grey matter changes distinguish different chronic pain conditions, such as back pain and complex regional pain syndrome, from each other and from healthy adults (3), suggesting an important role of supraspinal changes in chronic pain conditions.
Recent neuroimaging studies have shown substantial neuroplastic changes in higher brain regions, including sensorimotor cortices, in patients with chronic pain (4–8). The neuroplastic changes observed in patients with chronic pain affect the cortical representations of areas from which pain arises and, importantly, the extent of these neuroplastic changes has been shown to correlate with pain severity (8–12). While good evidence exists to show that intense or prolonged nociceptive input results in plastic changes within both spinal and cortical regions, why some patients develop chronic pain conditions and others do not when the initial injury and recovery is similar remains unknown. The importance of this issue has been noted in a recent, and influential, review (2).
It is plausible that some individuals have a higher propensity for developing central sensitisation and neuroplastic changes at the supraspinal level. Here, we have sought to examine this issue by investigating whether there was evidence of altered neuroplasticity in a cortical region not considered to be a primary component of the central pain matrix. We examined neuroplasticity in the primary motor cortex (M1) because both direct and non-invasive stimulation of M1 have proved useful in treating a variety of central pain syndromes including post-stroke pain, spinal cord pain, thalamic pain, trigeminal neuralgia, trigeminal neuropathic pain and complex regional pain syndrome (13). We examined whether motor cortical neuroplasticity induced by a ballistic training task was different in a patient population with a chronic pain condition, chronic tension-type headache (CTTH) – a well-characterised common chronic pain condition that is thought to be due to central sensitisation – and a population of control subjects (CS) with no history of chronic pain conditions. This ballistic training task has been shown to result in learning and changes in M1 that are consistent with neuroplastic changes mediated by a long-term potentiation-like mechanism (14). We hypothesised that there would be an exaggerated neuroplastic response in CTTH patients compared to healthy controls, which might explain (in part) the development of chronic pain in these individuals.
Materials and methods
Subjects
Eighteen right-handed healthy adults (seven males; mean age 28 ± 8 years (CS)) and 11 CTTH patients (five males; mean age 35 ± 13.2 years (CTTH)) participated in this study. Inclusion criteria for CTTH were: diagnosis of CTTH according to the International Classification of Headache Disorders, 2nd edition (ICHD-II) criteria (cod. 2.3) (15); diagnosis of CTTH for more than one year prior to commencing the trial; CTTH episodes of greater than four hours (if untreated); and CTTH onset before 50 years of age. Exclusion criteria for all subjects were: contraindications to transcranial magnetic stimulation (TMS) (16); neurological conditions; use of prophylactic migraine drugs (beta-blockers, calcium channel blockers, antidepressants, or antiepileptic drugs), or any drugs known to interfere in excitability to TMS (16) within eight weeks prior to commencing the trial. Participants in the trial were recruited through general advertising for persons with problematic headache. Screening was conducted by a single physician (PER) with extensive experience in headache diagnosis and treatment. In addition to detailed assessment of the current symptomatology, a lifetime headache history was obtained to distinguish episodic migraine evolving to daily or near daily headache from episodic tension headache evolving to daily or near daily headache. Care was also taken to exclude medication-overuse headache, concomitant psychiatric disease and other clinically significant pain conditions including fibromyalgia. All subjects were instructed to refrain from taking analgesics on the day of the experimental session. The protocol was in accordance with the Declaration of Helsinki and was approved by the University of Adelaide human ethics committee. All subjects gave written informed consent prior to testing.
Motor training
Subjects were seated in a comfortable chair with their head and neck supported throughout the session. The right arm was placed in a plastic cast secured to a board positioned across the chair. The right elbow was flexed at approximately 90° with the forearm in a semipronated position and all fingers fixed within the cast. Movement of the right thumb was not restricted in any direction. An accelerometer was secured onto the distal phalanx of the right thumb to record the acceleration of the thumb movements (in both abduction-adduction and flexion-extension axes). Abduction and flexion signals were filtered (DC-100 Hz, CED 1902) and digitised at a sampling rate of 5 kHz (CED 1401 interface, CED, UK).
The motor training task consisted of two blocks of 225 thumb abduction movements, with five minutes rest between the two training blocks (total 450 trials) (17). Movements were paced by a metronome at a rate of 0.25 Hz (total time of motor training, including five-minute break, 35 minutes). Subjects were instructed to abduct their thumb as quickly as possible after each tone and then return their thumb to the neutral rest position. Throughout the training task, peak acceleration of each abduction movement was presented on a computer screen in front of the participant and verbal feedback was provided to the subject to encourage optimal performance.
TMS
For all TMS measurements, the arm was removed from the plastic cast, supported by cushions, and placed in a semipronated position with the right elbow was flexed at approximately 90°. Electromyographic (EMG) activity was recorded from the relaxed right abductor pollicis brevis (APB) using surface electrodes placed in a belly-tendon configuration. The EMG signal was amplified (×1000; CED 1902 amplifier, CED, UK), band pass filtered (20–1000 Hz) and digitised at a sampling rate of 2 kHz (CED 1401 interface, CED, UK). A Magstim 200 stimulator (Magstim Co., Whitland, UK) generated single-pulse stimuli, delivered through a figure-of-eight coil (90 mm diameter) placed tangentially to the scalp with the handle pointing backward and at a 45° angle away from the midline to produce a posterior-anterior current in the cortex. Suprathreshold pulses were delivered over the left M1 at a number of sites in order to identify the optimal site for consistently evoking motor-evoked potentials (MEPs) in the relaxed right APB and marked on the scalp with a water-soluble pen.
Resting motor threshold (RMT) was determined before and two minutes after the motor training protocol; RMT was defined as the minimum intensity (as a percentage of maximal stimulator output (MSO)) required to elicit MEPs in the relaxed APB of at least 50 µV in at least five out of 10 consecutive trials. The TMS intensity that elicited MEPs of approximately 1 mV (SI1mV) in the relaxed APB was determined at baseline and was used to examine changes in MEP amplitude after the motor training protocol. Blocks of 15 single-pulse TMS trials (SI1mV), with an inter-trial interval of seven seconds (±10%), were delivered at baseline and 0, 5, 10, 20 and 30 minutes after the end of the motor training protocol. Testing sessions were conducted in the afternoon (after 12 p.m.) to minimise time of day influences (18).
Data analysis
Peak acceleration of the initial abduction movement after the response tone was calculated for each trial (m/s2). Trials were grouped into blocks of 25 trials (i.e. 18 blocks). A repeated-measures analysis of variance (ANOVA) was performed to test for changes in mean peak acceleration with motor training (18 blocks) between the two groups (GROUP: CTTH, CS).
Individual MEP data trials were excluded if EMG activity was present in the 100 ms immediately prior to the TMS pulse. The peak-to-peak MEP amplitude (in mV) was measured for each trial. Repeated-measures ANOVAs were used to test for differences in mean MEP amplitude across time (TIME: baseline, 0, 5, 10, 20, 30 minutes post-intervention) between the two groups (GROUP: CTTH, CS). SEM is presented in all figures.
Results
Motor learning
There was no significant difference in mean age of the CTTH and CS samples (t(27) = 1.8, p > .05). Figure 1 shows mean peak acceleration of thumb abduction movements with motor training for CTTH and CS. A repeated-measures ANOVA showed a main effect of MOTOR TRAINING (F(17,27) = 16.8, p < .05) and a significant interaction between MOTOR TRAINING and GROUP (F(17,459) = 4.2, p < .05). The significant interaction shows greater increase in acceleration with motor training in CS than CTTH. In the first major training block, before the five-minute break, CS showed significantly greater mean peak acceleration than CTTH in sub-blocks three through nine (all t > 2.4, all p < .05). In the second major training block, after the five-minute break, CS showed significantly greater mean peak acceleration than CTTH in sub-blocks 12–18 (all t > 2.4, all p < .05) (Table 1). A repeated-measures ANOVA performed on the coefficient of variation of mean peak acceleration in each sub-block showed a significant main effect of MOTOR TRAINING (F(17,27) = 4.1, p < .05) but no interaction between MOTOR TRAINING and GROUP (F(17,459) = 0.6, p > .05), showing that mean peak acceleration variability decreased with motor training but was not different between the two groups.
Mean peak acceleration across the motor training protocol for CTTH (filled symbols) and CS (open symbols). CS showed a significantly greater increase in mean peak acceleration with motor training than CTTH. Error bars show SEM. CTTH: chronic tension-type headache; CS: control subjects. Mean peak acceleration and standard deviation (m/s2) for each sub-block (25 trials) of the motor training protocol for CTTH and CS. CS showed a significantly greater increase in mean peak acceleration with motor training than CTTH (significant differences in mean peak acceleration between the groups shown in bold italics). (Note: Five-minute break in motor training protocol between Block 9 and Block 10.) CTTH: chronic tension-type headache; CS: control subjects; MEP: motor-evoked potential.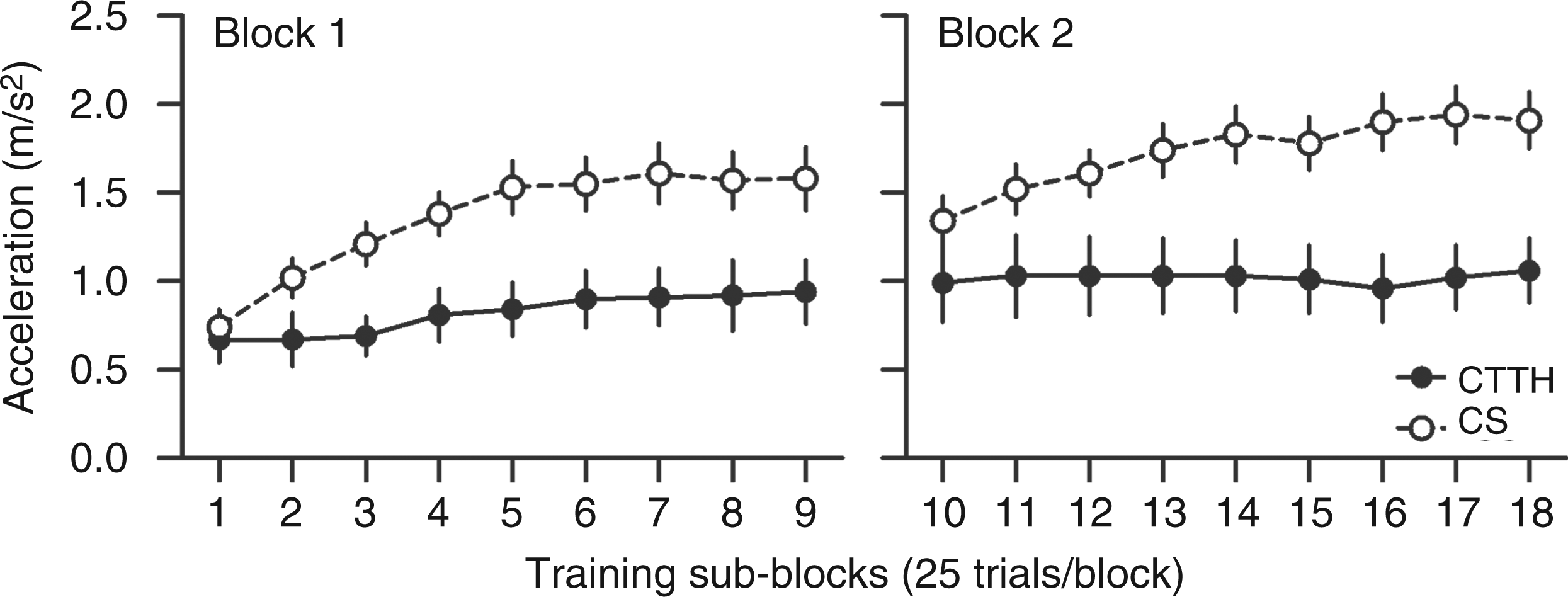

Neurophysiological measures
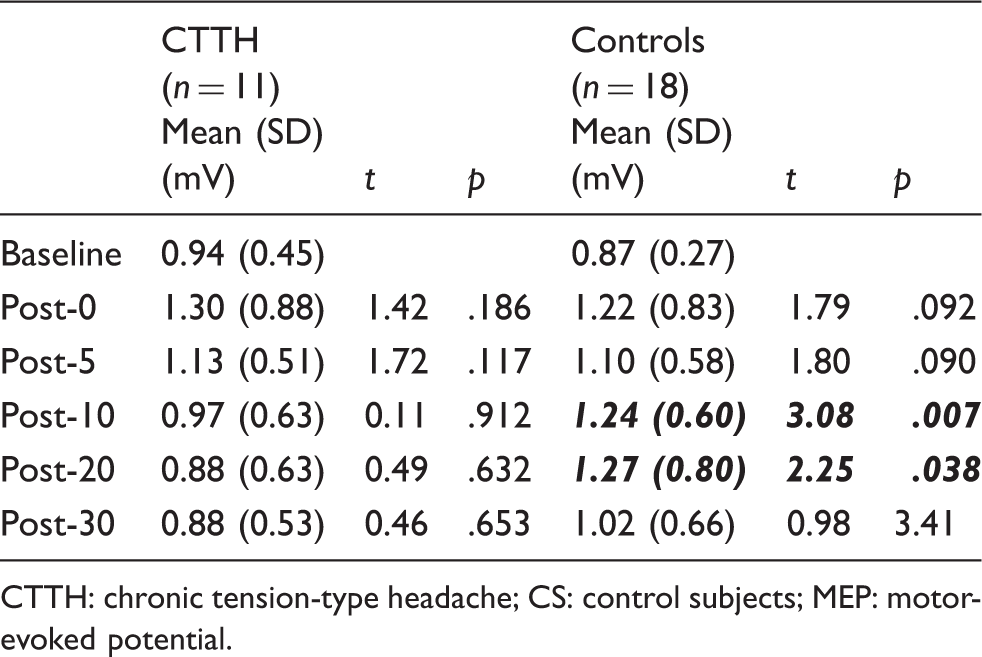
The mean intensity required to evoke an MEP of approximately 1 mV (SI1mV) was 55% and 57% MSO for CTTH and CS, respectively (t(27) = 0.9, p > .05) and the mean MEP amplitude evoked by SI1mV at baseline was 0.9 (±0.45) and 0.9 (±0.27) mV for CTTH and CS respectively (t(27) = 0.6, p > .05). The mean RMT at baseline was 45% and 46% MSO for CTTH and CS respectively (t(27) = 0.2, p > .05). Following motor training, there was no significant change in RMT compared to baseline for either CTTH (t(10) < 0.1, p > .05) or CS (t(17) = 1.2, p > .05).
Figure 2 shows mean MEP amplitude following motor training for CTTH and CS. A repeated-measures ANOVA showed no significant main effect of TIME (F(1,5) = 2.3, p > .05) but a significant interaction between TIME and GROUP (F(5,135) = 4.9, p < .05). Separate one-way repeated-measures ANOVAs showed a significant main effect of TIME for CS (F(5,85) = 2.9, p < .05) but not CTTH (F(5,50) = 1.6, p > .05), with CS showing a significant increase in MEP amplitude after motor training. Paired-sample t tests performed on CS data showed MEP amplitude was significantly increased 10 and 20 minutes post-motor training (both t(17) > 2.5, p < .05), and the increase in MEP amplitude approached statistical significance at 0 and 5 minutes post-motor training (both t(17) = 1.8, p = .09). At the 30-minute post-motor training measurement block, MEP amplitude had returned to baseline levels (t(17) = 1.0, p > 0.5) (Table 2).
Mean MEP amplitude at baseline and following motor training for CTTH (filled symbols) and CS (open symbols). CS but not CTTH showed a significant increase in MEP amplitude following motor training. Error bars show 95% CI. *p < .05. CTTH: chronic tension-type headache; CS: control subjects; MEP: motor-evoked potential; CI: confidence interval. Mean MEP amplitude (and standard deviation) in mV at baseline and all time points following the motor training protocol for CTTH and CS. CS, but not CTTH, showed a significant increase in MEP amplitude after the motor training protocol (significant increase in MEP amplitude from baseline shown in bold italics). CTTH: chronic tension-type headache; CS: control subjects; MEP: motor-evoked potential.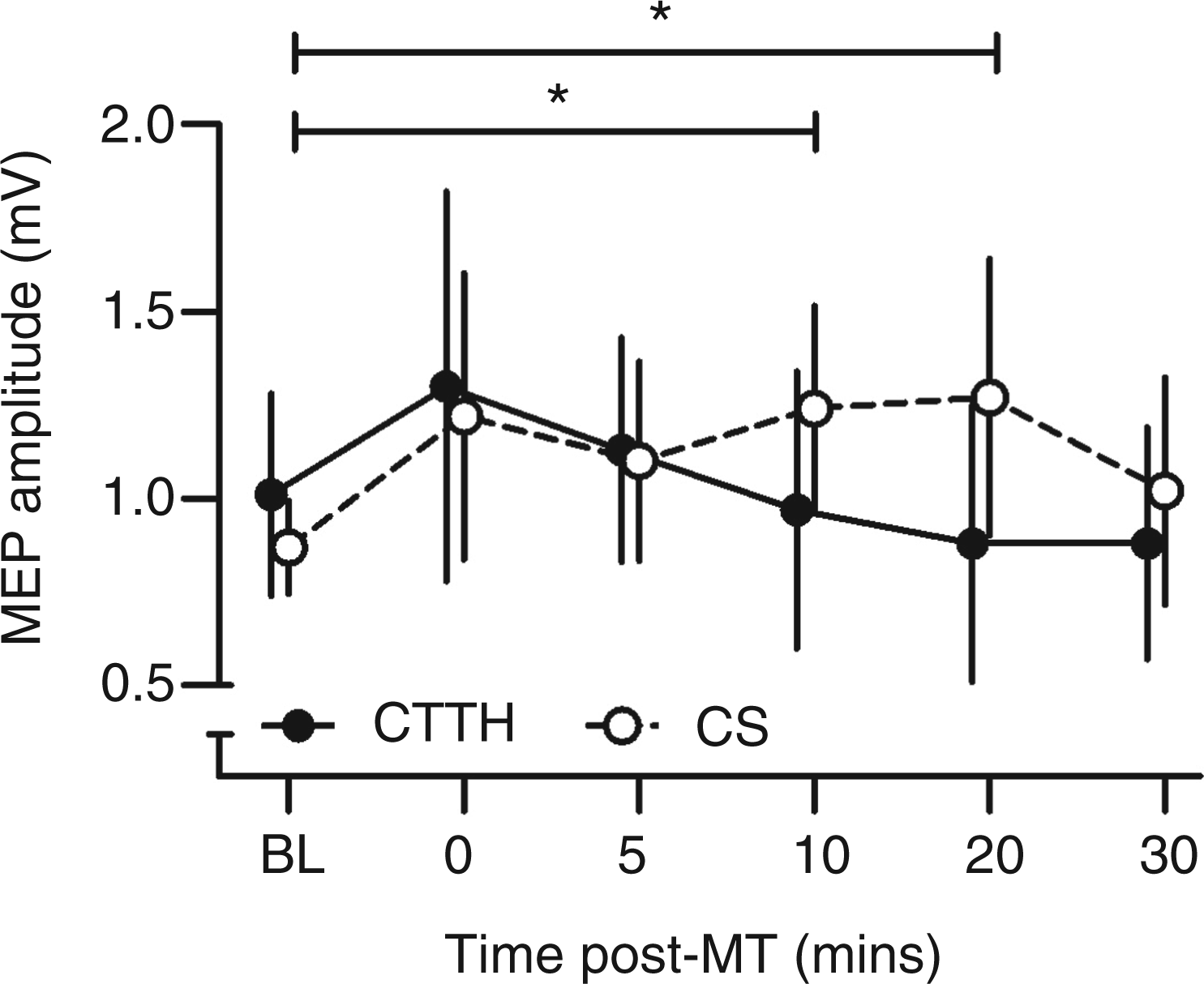
Headache intensity during experimental session
Immediately prior to the commencement of the experimental session, self-reported headache intensity in the CTTH subjects ranged from 1 out of 10 to 6 out of 10 on a visual analogue scale (mean 3.5 ± 1.7). Correlational analyses were performed to examine whether headache severity at time of testing was related to neuroplastic responses (Figure 3). There was no relationship between headache severity at the time of testing and either the increase in peak acceleration with motor training (r = 0.17; 95% confidence limits: −0.52, 0.72) or the change in MEP amplitude following motor training (r = 0.20; 95% confidence limits: −0.50, 0.73).
Scatter diagrams showing relationships between headache severity at time of testing and change in mean peak acceleration with motor training (top) and mean MEP change following motor training (bottom) for CTTH patients. MEP: motor-evoked potential; CTTH: chronic tension-type headache.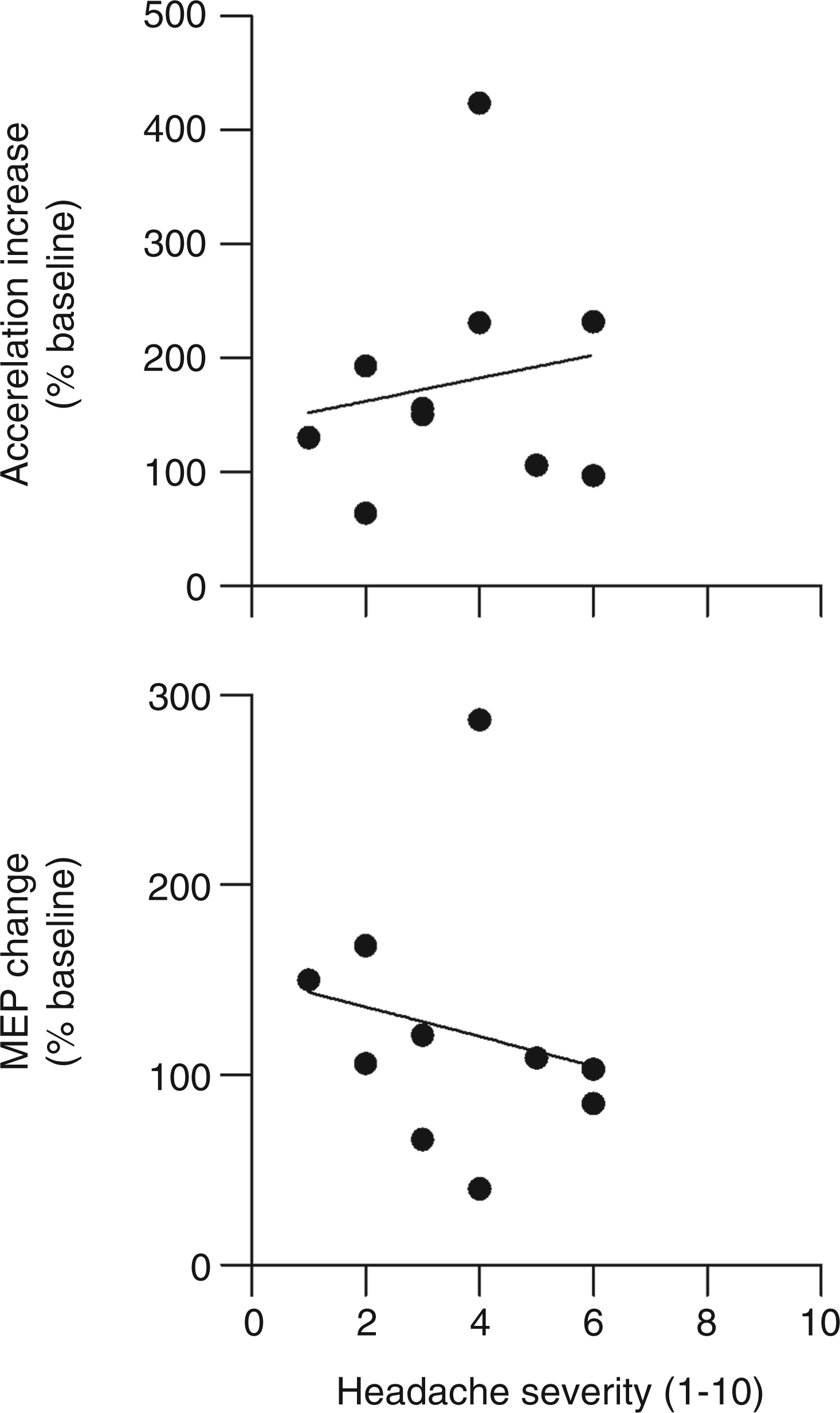
Discussion
Here we compared neuroplastic responses between CTTH and controls. We selected the population of CTTH as it is a common pain syndrome and current theory and evidence suggest that the dominant mechanism is acquired central sensitisation with minimal or no role for altered afferent nociceptive activity (19,20). The novel finding reported here is that individuals with CTTH showed significantly less motor learning on the training task than healthy CSs, suggesting a deficit in use-dependent neuroplasticity within networks responsible for task performance in patients with CTTH.
The motor training task used in the present study has been employed in numerous other studies and induces robust motor learning in which M1 has an important role (14,21). In healthy CS, we found significant motor learning with training which was accompanied by an increase in corticospinal excitability that lasted at least 20 minutes following the end of training, consistent with previous studies (21,22). CTTH patients showed significantly poorer motor learning than controls and showed only a brief (non-significant) increase in corticospinal excitability immediately following the end of training, suggesting an impairment of use-dependent neuroplasticity in the cortical networks important for this task. While this finding contrasts with our original hypothesis, it does not rule out the suggestion that chronic pain conditions are characterised by excessive generalised neuroplasticity. Indeed, it is plausible that the reduced neuroplasticity evident in M1 in CTTH might reflect reciprocal influences acting on M1 from (hyperexcitable) interconnected pain processing regions. Interregional reciprocal influences on cortical excitability (changes which act as a neurophysiological marker of neuroplasticity) are known to exist between various brain regions including, for example, the motor cortices of the two hemispheres (23) as well as cortical areas within the same hemisphere (24). Furthermore, M1 has extensive projections to thalamic nuclei. Thus, it is plausible that reciprocal modulatory influences exist between M1 and the cortical networks involved in the generation of chronic pain. However, due to the cross-sectional nature of this study, whether reduced motor plasticity is a predisposing factor in development of CTTH or an adaptive response to the condition cannot be determined.
At present, the influence of activity within M1 on pain is not clear. It is known, however, that epidural cortical stimulation of M1 is an effective treatment option for drug-resistant chronic pain, especially of central origin (for a review, see Cruccu et al. (25)). It has been suggested that top-down activation of descending pain control systems from the motor cortex to the thalamus, and perhaps to motor brainstem nuclei, might mediate motor cortex stimulation-induced pain relief (26). More recently, non-invasive brain stimulation techniques such as repetitive TMS (rTMS) and transcranial direct current stimulation have been trialled as non-invasive alternatives to epidural stimulation. In the small number of studies investigating the use of rTMS and other non-invasive brain stimulation techniques to treat chronic pain, the most positive results have been observed following administration of facilitatory paradigms, that is, paradigms that act to increase excitability of M1 (for a review, see O'Connell et al. (27)). As with epidural motor cortex stimulation, it is thought that non-invasive stimulation of M1 modulates the excitability of projections to remote brain regions involved in pain processing (25). The beneficial effect of facilitatory M1 stimulation on pain might be due to the normalisation of a hypoexcitable M1 in chronic pain patients. Although we do not have direct evidence for reduced M1 excitability in the current study, we did find that the change in corticospinal excitability following motor training in CTTH patients was significantly smaller than their healthy counterparts. This is consistent with the finding that reduced M1 excitability impairs motor performance, learning and consolidation (22,28,29). It is tempting to speculate that the beneficial effect of motor cortex stimulation on pain is due to reciprocal interactions between M1 and pain processing networks in which the excitability of M1 is up-regulated and the excitability of pain pathways are down-regulated.
It is worth noting here that performance on the motor training task is influenced by attention. Thus, it might be argued that the differences in task performance were due to the CTTH patients having a poorer attentional focus than the CS. However, we think this explanation is unlikely for a number of reasons. First, the fact that the CTTH patients volunteered for the study is indicative of their high motivational drive. Second, poor attentional focus during the task would result in much more variable performance. However, the coefficient of variation of the acceleration data was no different between groups, indicating similar levels of performance variability during the task. Finally, it might be argued that the differences in task performance were due to CTTH patients experiencing pain during the experimental session. However, here there were no correlations between headache severity at the time of testing and task performance or neuroplastic MEP change, showing that pain during the experimental session did not affect the results.
In summary, there is evidence to suggest a role of both central sensitisation at the spinal level and neuroplastic changes in higher brain regions in the development of chronic pain conditions (2,30). The underlying mechanisms of CTTH, and indeed many other chronic pain conditions, are still not well understood. We have demonstrated impaired learning on a simple motor training task in CTTH patients. This finding is consistent with impairments in M1 neuroplasticity which we suggest might reflect reciprocal influences between M1 and interconnected pain processing networks. These findings may help explain the positive effects on chronic pain of facilitatory non-invasive brain stimulation paradigms targeted to M1 and help elucidate the mechanisms mediating chronic pain. Insights as to whether these changes are a predisposing feature or a response to the condition will come from longitudinal studies of response to treatment.
Clinical implications
Chronic tension-type headache (CTTH) patients showed impaired learning on a simple motor training task compared to healthy controls. Control subjects (CS) but not CTTH patients showed a significant increased motor-evoked potential (MEP) amplitude following the motor training task. These findings suggest a deficit in use-dependent neuroplasticity within networks responsible for task performance in CTTH patients. This deficit in neuroplasticity within primary motor cortex in CTTH patients might reflect reciprocal influences between primary motor cortex and interconnected pain processing networks.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgements
MCR is a senior research fellow (ID 519313) of the National Health and Medical Research Council of Australia. The authors would like to thank James Swift for assistance with patient recruitment.
Conflict of interest
None declared.
