Abstract
Background
Sleep disorders, nightmares and visual hallucinations have been reported in migraine patients, which may suggest the involvement of rapid eye movement (REM) sleep regulation in migraine. However, the relationship between migraine and REM sleep behaviour disorder (RBD) remains unclear.
Methods
To investigate the clinical correlates of dream-enacting behaviours (DEB) in migraine patients, we assessed episodic migraine patients (
Results
A significantly increased frequency of DEB was observed in migraine patients compared to controls (24.2% vs. 14.3%). Migraine patients with DEB presented higher scores on the Migraine Disability Assessment and Pittsburgh Sleep Quality Index and an increased rate of smoking compared to those without DEB. Duration of migraine and headache frequency and intensity were not different between migraine patients with or without DEB.
Conclusion
DEB was associated with impaired sleep and severe headache-related disability in migraine patients and may reflect brainstem dysfunction and increased brain excitability in migraine patients.
Introduction
Migraine is a disabling neurological disorder that involves several neurotransmitters, including serotonin, dopamine and calcitonin gene-related peptides. Sleep disturbances and daytime sleepiness are more common in migraine sufferers than in headache-free individuals. Compromised sleep conditions can worsen headaches, and severe forms of headache are more frequently associated with sleep disturbances (1). Notably, migraine attacks can be triggered by sleep deprivation or excessive sleep, and healthy sleep is associated with relief from migraine attacks. Increased migraine frequency has been described in patients with somnambulism and narcolepsy (1). Moreover, a significant relationship between restless legs syndrome and migraine has been confirmed by several studies (2). Thus, a strong link between migraine and sleep disturbances suggests that sleep and pain perception involve similar anatomic structures, such as the brainstem, hypothalamus and thalamus (1).
Rapid eye movement (REM) sleep behaviour disorder (RBD) is a REM parasomnia characterised by a loss of normal muscle atonia and by dream-enacting behaviours (DEB) during REM sleep, often causing injury to the patient or their bed partner (3). RBD predominantly affects males over 50 years of age in association with neurodegenerative diseases. In contrast, early-onset cases (affecting individuals under 50 years old) are associated with medications, such as tricyclic antidepressants, selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs). Furthermore, narcolepsy and brain lesions affect REM sleep regulation (3). Ju et al. (4) proposed that three separate subgroups are affected by RBD: older men with neurodegenerative diseases, young patients with narcolepsy and middle-aged women with autoimmune diseases. However, the detailed clinical features of early-onset RBD remain ill defined.
Disturbed dreaming is found in a variety of neurologic, psychiatric and sleep-disordered conditions. Lippman (5) described three different recurrent dream patterns (nightmare, nostalgic Technicolor dream and waking dream) that often occur in patients with migraine, and these may be used as a diagnostic aid for migraine. The frequent occurrence of migraine attacks during REM sleep has been reported (6). Hallucinations in patients with migraine have also been described (7,8). Dysfunctions in migraine of neural structures involved in the regulation of REM sleep are suggested by a lower index of high-frequency electroencephalographic arousals during REM sleep (9), and increased REM sleep and prolonged REM latency are found in migraine patients (10). These observations suggest that REM sleep is impaired in migraine, leading us to investigate whether DEB is frequently observed in migraine patients.
To our knowledge, an association between RBD and migraine has not yet been evaluated. In this study, we evaluated the frequency of DEB in migraine patients under 50 years of age with the Japanese version of the RBD screening questionnaire (11), which is a recently developed and validated screening scale for RBD assessment (12), and we aimed to determine the factors associated with DEB in migraine patients.
Methods
To assess the frequency and characteristics of DEB in migraine patients, 262 migraine patients (38.2 ± 13.0 years; range, 14–72 years; 47 men and 215 women) and 163 healthy control subjects (37.3 ± 12.1 years; range, 22–74 years; 25 men and 138 women) were selected from a previously conducted case-control survey on “sleep-related disorders in migraine patients” performed between June and November 2010 at the Department of Neurology, Dokkyo Medical University Hospital (2). In Japan, unlike in Europe and America, there is no formal system of medical referral. A referral is recommended but not necessary; in fact, most patients who visited our headache outpatient clinic were not referral cases. Patients living around the university hospital can decide whether to go to local clinics or our outpatient clinic. Patients with chronic migraine (
All patients and control subjects were instructed to complete all questions on sleep, habits (smoking, alcohol and caffeine intake), depressive state and medications. The participants were informed of the approximate time required to finish the questionnaires (30 minutes). The dose-dependent effects of smoking were indicated by the Brinkman index (daily number of cigarettes × years) (14). The RBD screening questionnaire (RBDSQ) is a recently developed self-rating instrument consisting of the following 10 items on the most prominent clinical features of RBD (12): items 1 to 4, the frequency and content of dreams and their relationship to nocturnal movements and behaviour; item 5, self-injuries and injuries to the bed partner; item 6, four sub-items specifically assessing nocturnal motor behaviour, e.g. questions about nocturnal vocalisation (6.1), sudden limb movements (6.2), complex movements (6.3) or bedside items that fell down (6.4); items 7 and 8, nocturnal awakenings; item 9, disturbed sleep in general; and item 10, the presence of any neurological disorder. Each item could be answered as “yes” or “no”. The maximum total score of the RBDSQ is 13 points. The clinical usefulness of the RBDSQ has been demonstrated by assessing RBDSQ scores in polysomnography (PSG)-confirmed RBD patients (12). A Japanese version of this questionnaire was assessed in 52 idiopathic RBD patients, 55 controls with obstructive sleep apnoea on successful continuous positive airway pressure therapy and 62 age-matched healthy subjects (without PSG confirmation). Sensitivity was 88.5% and specificity was 90.9% in sleep apnoea patients (vs. 88.5% sensitivity and 96.9% specificity in healthy subjects) (11). According to the RBDSQ validation study, an RBDSQ score of 5 points as a positive test result showed a sensitivity of 0.96 and a specificity of 0.56, where specificity was highest for item 6.3 (complex movements during sleep; 91.1%), item 6.4 (things that fell down around the bed; 88.4%) and item 5 (injury of the patient or bed partner; 85.3%). In most subjects of the control group (
The Migraine Disability Assessment (MIDAS) questionnaire, which ranged from 0 (no disability) to 270 (worst disability), was used to measure headache-related disability during the previous three months (15). Headache frequency was assessed by MIDAS question A. Headache intensity was assessed by a MIDAS question B with an 11-point pain scale (0–10), with 0 indicating no pain at all and 10 indicating pain as bad as possible. The Beck Depression Inventory (BDI)-II was used to evaluate depressive symptoms (16). Sleep disturbance was evaluated using the Pittsburgh Sleep Quality Index (PSQI), and patients with global scores of 6 or greater were considered “poor sleepers” (17). The following seven component scores were also estimated (range of subscale scores, 0–3): C1, sleep quality; C2, sleep latency; C3, sleep duration; C4, habitual sleep efficiency; C5, sleep disturbances; C6, use of sleeping medications; and C7, daytime dysfunctions. The risk of sleep-disordered breathing was assessed using the average of the sum of the PSQI sub-items C5d “Cannot breathe comfortably” and C5e “Cough or snore loudly”. We measured daytime sleepiness using the Japanese version of the Epworth sleepiness scale (ESS) (18). An ESS score of 10 or higher was considered indicative of excessive daytime sleepiness (EDS). The migraine patients were divided into migraine with DEB and migraine without DEB groups to compare the clinical and demographic data and detailed medication use information.
The study was approved by the Dokkyo Medical University institutional review board, and written informed consent was obtained from all patients enrolled in the study.
Statistical analysis
Mann-Whitney
Results
Clinical profiles of migraine patients and control subjects.
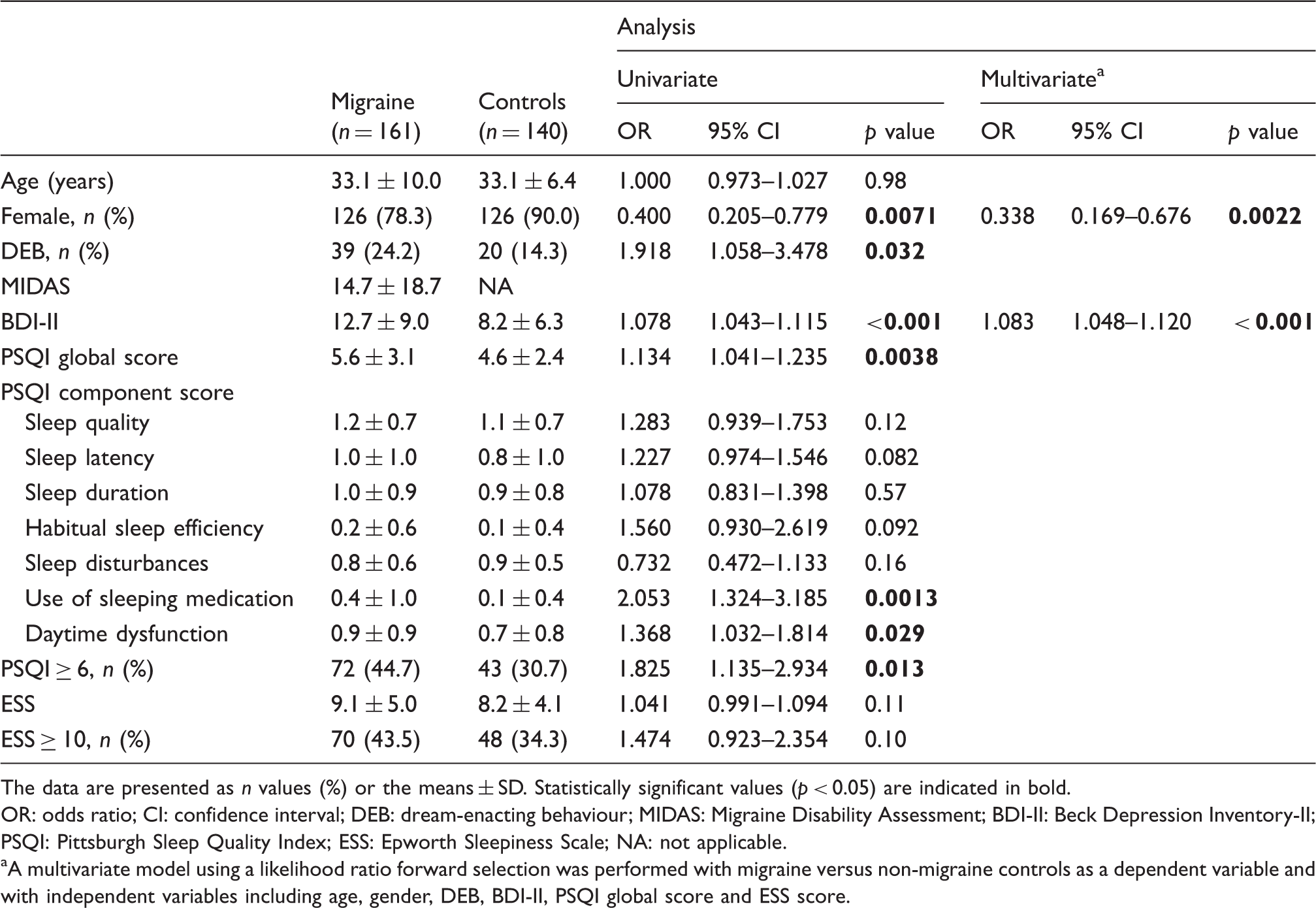
The data are presented as
OR: odds ratio; CI: confidence interval; DEB: dream-enacting behaviour; MIDAS: Migraine Disability Assessment; BDI-II: Beck Depression Inventory-II; PSQI: Pittsburgh Sleep Quality Index; ESS: Epworth Sleepiness Scale; NA: not applicable.
A multivariate model using a likelihood ratio forward selection was performed with migraine versus non-migraine controls as a dependent variable and with independent variables including age, gender, DEB, BDI-II, PSQI global score and ESS score.
Clinical characteristics of migraine patients with and without DEB.

The data are presented as the
DEB: dream-enacting behaviour; RBDSQ-J: REM sleep behaviour disorder (RBD) screening questionnaire, Japanese version; MIDAS: Migraine Disability Assessment; BDI-II: Beck Depression Inventory-II; PSQI: Pittsburgh Sleep Quality Index; ESS: Epworth Sleepiness Scale.
The rate of positive results of RBDSQ-J sub-item 5, 6.3 or 6.4
Comorbid diseases and medications of migraine patients with and without DEB.
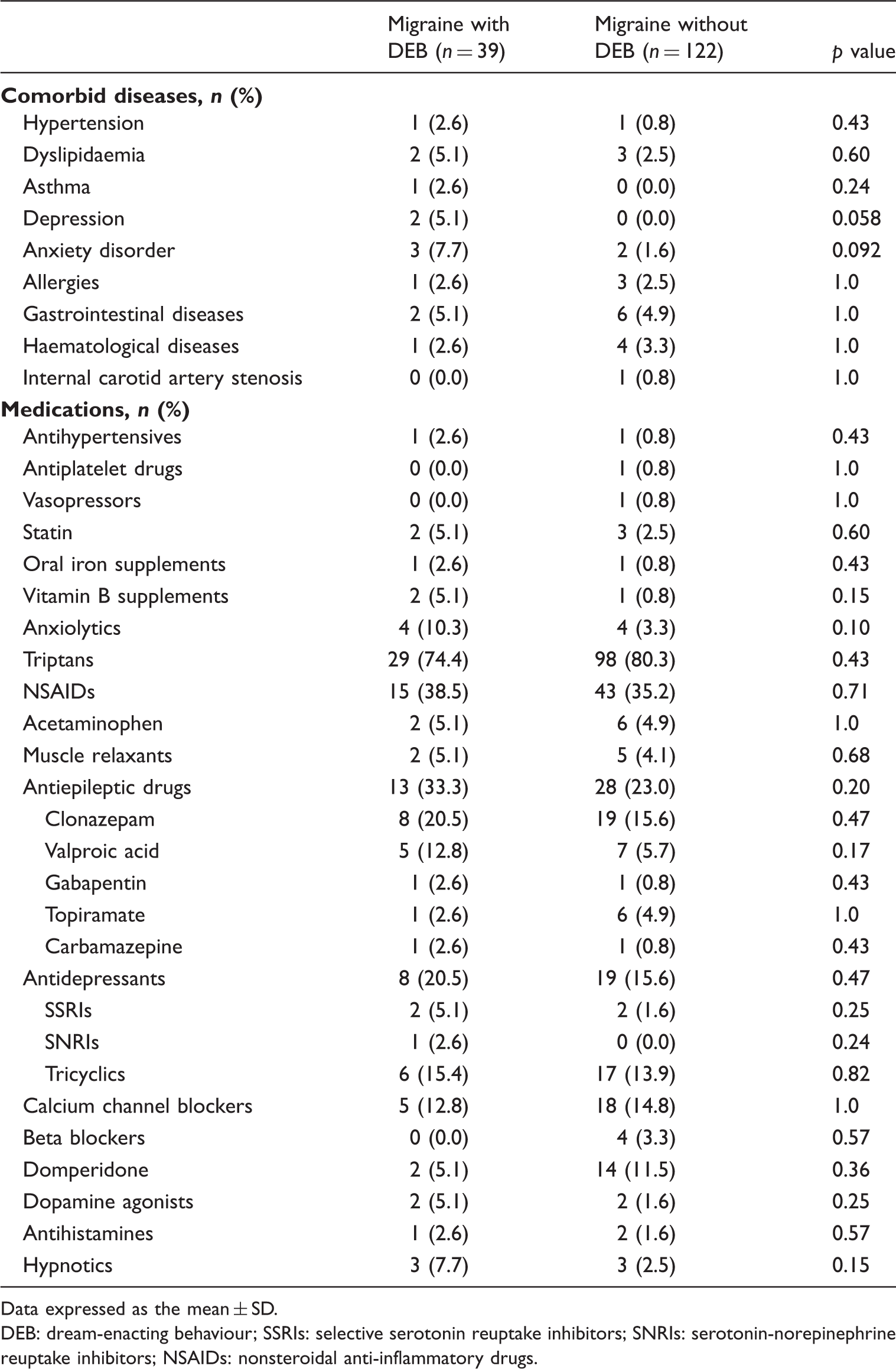
Data expressed as the mean ± SD.
DEB: dream-enacting behaviour; SSRIs: selective serotonin reuptake inhibitors; SNRIs: serotonin-norepinephrine reuptake inhibitors; NSAIDs: nonsteroidal anti-inflammatory drugs.
Cronbach’s alpha of the RBDSQ-J was 0.71 in migraine patients and 0.68 in controls. The item-total correlation showed significant coefficients ( Answer patterns of RBDSQ-J sub-items for migraine patients and controls. Item-total correlation and Cronbach’s alpha of RBDSQ-J in migraine patients. RBDSQ-J: REM sleep behaviour disorder (RBD) screening questionnaire, Japanese version.

In the migraine patients, the logistic regression analysis revealed that age (OR, 0.933; 95% CI, 0.890–0.979;
Discussion
We observed a significantly increased frequency of DEB in migraine patients compared with controls (24.2% vs. 14.3%). Idiopathic RBD frequency is reported to be less than 1% in the general population aged 15 to 100 years (3), which is significantly lower than the DEB frequency for the controls in our study. However, the RBD frequency may have been overestimated because we diagnosed DEB with a questionnaire. A similar RBD frequency (13%) was reported in controls in a recent study that employed an RBD questionnaire (19). Moreover, as suggested by Ju et al. (4), sleep disorders have recently received greater attention, which may result in a change of the initially estimated prevalence and characteristics of idiopathic RBD.
However, the current diagnostic criteria of RBD based on the International Classification of Sleep Disorders-2 require PSG to detect the presence of REM sleep without atonia and to rule out several medical conditions that can mimic RBD. With regard to the cut-off points of RBDSQ-J, we used a cut-off of 5 in our study. An RBDSQ score cut-off of 5 has been considered useful for differentiating idiopathic RBD patients from healthy controls and patients with sleep apnoea syndromes (11,12). We therefore applied the term DEB rather than RBD in the setting of this study. In our study, migraine patients had a Cronbach’s alpha of 0.71 and significant item-total correlation for all RBDSQ-J sub-items, supporting the internal consistency of the RBDSQ-J as satisfactory. In addition, we performed a linear stepwise regression analysis of the RBDSQ-J score that had no cut-off points and revealed identical results as with the logistic regression models.
The present study is the first to assess the factors contributing to DEB in migraine patients. We found that migraine patients with DEB manifested severe headache-related disability and insomnia, particularly sleep-onset insomnia and impaired sleep efficiency, in comparison to those without DEB. Logistic and stepwise linear regression analyses demonstrated that headache-related disability was one of the significant determinants of DEB in migraine patients. In our study, migraine patients exhibited a higher frequency (albeit non-significant) of EDS than controls, which is similar to previous studies (20,21). EDS prevalence in the general population varies widely between studies. A global cross-sectional survey revealed between-country differences in EDS ranging from 6.2% to 24.5% (22). The increased frequency of EDS in control subjects in our study might have been related to the relatively young age of the sample. Breslau et al. (23) reported that approximately one-third of 997 young adults showed high levels of EDS. The mean sleep latency of young subjects has been reported to be shorter than that of older subjects (24).
Increased frequency of smoking in migraine patients with DEB compared to those without DEB was observed in this study. Nicotine may prolong the onset of REM sleep latency and decrease REM and slow-wave sleep time, due to a consequence of an increase in wakefulness (25). A recent multicentre case-control study assessing environmental and lifestyle risk factors for idiopathic RBD revealed that smoking, head injury, pesticide exposure and farming are potential risk factors for idiopathic RBD (26).
The proposed anatomic substrate for REM sleep control in humans includes a “REM-off” region, consisting of the ventrolateral portion of the periaqueductal grey matter (vl-PAG) and the lateral pontine tegmentum, and a “REM-on” region, consisting of the precoeruleus and sublaterodorsal nucleus, the extended portion of the ventrolateral preoptic nucleus, the locus coeruleus (LC), the laterodorsal tegmental nucleus, the pedunculopontine nucleus and the raphe nucleus (RN) (27). The interaction between these brainstem nuclei is crucial for maintaining muscle atonia during REM sleep. Lesions in the vl-PAG and RN cause migraine symptoms (28), and the RN and LC in the brainstem are activated during and after migraine attacks (29). The involvement of the vl-PAG has been reported in migraine and RBD, possibly implying that shared structural changes between the two diseases represent common pathophysiological substrates. In a study employing diffusion tensor imaging (DTI), patients with migraine with aura exhibited lower fractional anisotropy, which reflects the cellular and subcellular tissue components and their preferable orientation, in the ventral trigeminothalamic tract. Patients with migraine without aura manifested lower fractional anisotropy in the vl-PAG (30). The authors suggested that these findings likely reflect axon enlargement as a response to overactivity rather than to brain lesions and indicate the interictal alteration of the trigeminal somatosensory pathway and PAG in migraine patients. A study by Scherfler et al. (31) using DTI demonstrated significant decreases in fractional anisotropy in the tegmentum of the midbrain and rostral pons and increases in the mean diffusivity within the pontine reticular formation in idiopathic RBD patients. In contrast, a voxel-based magnetic resonance imaging study in patients with idiopathic RBD identified grey matter volume loss in the tegmental portion of the pons, including the sublaterodorsal nucleus (32).
In migraine patients, recurrent vivid dreams (5) with negative affect (6) and persistent and frequent migraine attacks that disrupt REM sleep (6) suggest dysfunction of the brainstem, which regulates REM sleep, and limbic regions, which are implicated in motivation and emotional control. Heather-Greener et al. (33) studied the relationship between migraine attacks and dream contents and found that pre-migraine dreams contained a significantly higher occurrence of aggressive interactions, misfortune, anger and apprehension than non-migraine dreams. This finding supported the hypothesis that the presence of persistent negativity in dreams precipitated the migraine attacks (5,6). Dream disturbances characterised by stimulation augmentation, such as dream stereotypy or the recurrence of particular dream contents, occur in migraine (34). In REM sleep, there is enhanced activation of limbic and paralimbic regions and relatively reduced activity of the cortical areas involved in higher-level cognition compared to waking. There is also a prominent increase in the neural activity in the pons, midbrain, thalamus, basal ganglia and limbic subcortex, including the amygdala, hypothalamus and ventral striatum, during REM sleep compared with non-REM sleep (35). Observations from animal experiments have also demonstrated a significant influence of brainstem nuclei, such as the LC and dorsal RN, on sensory cortex neuronal excitability by way of the thalamus (36). Reports of RBD following acute aseptic limbic encephalitis and voltage-gated potassium channel antibody-associated limbic encephalitis suggest the involvement of the limbic system in RBD and imply that autoimmune mechanisms may play a role in RBD pathogenesis (37). Moreover, Stankewitz and May (38) studied the neuronal response to olfactory stimulation in migraine patients using event-related functional magnetic resonance imaging. The results revealed that during spontaneous and untreated migraine attacks, migraine patients exhibited significantly higher blood oxygen level-dependent signal intensities in limbic structures such as the amygdala, the insular cortex and the rostral pons. This finding suggests the possible role of the rostral pons in olfactory and trigemino-nociceptive pathways. In migraine patients, the vulnerability of the limbic system may amplify pain after years of sensitisation (39).
We only analysed migraine patients and controls under 50 years old in this study because the characteristics of early-onset RBD are still unclear, and the enrolled patients with migraine were relatively young. In early-onset RBD patients, the male-to-female ratio of 1.25:1 and the high prevalence of autoimmune disease among women suggest that there may be a new sub-group of RBD patients with a distinct pathophysiological process compared to late-onset RBD, which is associated with neurodegenerative diseases (4). Recent studies also correlated early-onset RBD with antidepressant use and current and past psychiatric diagnoses. Because several medications such as SSRIs, SNRIs, tricyclic antidepressants and beta-blockers are associated with RBD (3), we closely examined medication use in this study. However, there was no significant difference in the medication use rate between patients with and without DEB. In our study, we excluded patients with chronic headache or meditation-overuse headache, and we focused on patients with episodic migraine, as patients with chronic headache are associated with an increased frequency of sleep disturbances (40) and several factors known to cause secondary RBD such as antidepressant use and caffeine overuse (3).
A major limitation of this study is that a PSG examination was not performed. The current diagnostic criteria for RBD require PSG findings, including a loss of REM sleep-related muscle atonia with excessive sustained or intermittent elevation of the submental electromyographic tone or excessive phasic submental or limb electromyographic twitching. We therefore could not evaluate whether DEB arose from REM or non-REM sleep owing to a lack of PSG recordings. Arousal disorders, such as somnambulism, pseudo-RBD caused by sleep apnoea syndrome or nocturnal epilepsy, cannot be fully ruled out. Patients with mild DEB who were unaware of or could not remember abnormal movements during REM sleep may have been missed. In this study, we used a validated questionnaire for screening RBD, the RBDSQ-J, and obtained a satisfactory internal consistency in migraine patients; however, its usefulness has not been confirmed in migraine patients, who have more sleep problems than healthy subjects. In addition, this study included a large number of female patients because of the study setting. Lastly, in our study, the severity of DEB could not be assessed. Revision of the RBDSQ would have enabled us to evaluate DEB severity.
In conclusion, our study is the first to demonstrate an increased frequency of DEB in migraine patients, and this was associated with headache-related disability and impaired sleep. DEB may be associated with negative emotions experienced during sleep or wakefulness and may reflect increased brain excitability in migraine patients due to brainstem involvement. Further study is required to evaluate clinical characteristics of migraine patients with PSG-confirmed RBD.
Clinical implications
The presence of sleep disorders, nightmares and visual hallucinations in migraine suggests impaired rapid eye movement sleep. A significantly increased frequency of dream-enacting behaviours was observed in migraine patients compared to controls. Dream-enacting behaviours were associated with impaired sleep and severe headache-related disability in migraine patients. Dream-enacting behaviours may reflect brainstem dysfunction and increased brain excitability in migraine patients
Footnotes
Acknowledgements
We are very grateful to Ayaka Numao, MD, from the Department of Neurology, Dokkyo Medical University, and to the medical staff of the outpatient clinic for their assistance with this study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
