Abstract
There is still a controversy regarding the relationship between sleep apnoea syndrome and headaches, especially morning headaches. Our objectives were: (i) to compare the prevalence and the clinical data of headaches in sleep apnoea syndrome (SAS) and control (snorers) groups defined by polysomnographic recording; (ii) to analyse the clinical improvement of headaches with appropriate treatment; and (iii) to correlate headaches with mood disorders, and nocturnal respiratory and architectural sleep parameters in order to understand the underlying pathophysiological mechanisms. This is a prospective study of 324 consecutive patients referred to our sleep centre for snoring. Of these, 312 patients who underwent sleep polysomnography were finally included. Patients and controls were interviewed about their medical past, headache history and clinical characteristics, their daytime sleepiness (Epworth's sleepiness scale) and their mood disorders (Zerssen's scale). Follow-up of patients with headaches (SAS and control groups), treated or not, was also assessed. According to our definition of SAS, patients were dissociated in SAS (n = 164) and snorers (n = 148). Fifty-three SAS patients had headaches, of whom 58.5% (n = 30) suffered from morning headaches. However, there was no statistical difference between the two groups concerning the prevalence and the clinical characteristics of headaches. In addition, headaches and morning headaches were not correlated with nocturnal respiratory and architectural sleep parameters, nor with excessive daytime sleepiness, but were strongly correlated with mood disorders. In 36 SAS patients, headaches improved under treatment, but this was not statistically different from what was found among untreated snorers. Headaches and morning headaches are common in patients with SAS but may be considered as a non-specific symptom. The underlying mechanisms are not fully elucidated but depression could play an important role. Despite this absence of specificity, the treatment of SAS, especially nasal continuous positive airway pressure, leads to an improvement in headaches in several cases.
Introduction
Sleep apnoea syndrome (SAS) is a common disorder with a prevalence estimated to be 4% among males and 2% among females (1) and receives increasing interest. The characteristic clinical pattern associates obesity, excessive daytime sleepiness, heavy habitual snoring and neuropsychological disturbances. Headache, especially recurrent morning headache, is also described as an important part of these clinical features, as Guilleminault et al. (2) reported that 18 of 50 SAS patients suffered from frontal or diffuse morning headache. Several other reports supported an important relationship between SAS and headaches (3–9), although others did not (10–12). However, the mechanisms by which SAS could lead to morning headaches were not fully explained.
Therefore, we decided in a prospective study to evaluate the relationship between headaches and sleep apnoea or snoring defined by polysomnographic data. The headache response to continuous positive airway pressure (CPAP) or uvulopalatopharyngoplasty (UPPP) in SAS patients was also studied. In order to understand the pathophysiological mechanisms of headaches, we investigated the relationship between headaches, polysomnographic data and a standardized measure of depression, which is a frequent comorbid condition in patients with chronic headache.
Materials and methods
Patients
All patients referred to our sleep laboratory at Poitiers Hospital, France, because of snoring were included in this prospective study between April 1996 and July 1999. However, patients with known neurological diseases (i.e. strokes, cerebral tumours) were excluded. All included patients underwent a standardized questionnaire about their medical past, sleeping habits, mood and headaches. The first two questions of the headache questionnaire (HQ) were ‘Do you suffer from headache?’ and, if the answer was ‘yes’, ‘How often do you have headache?’. The patient was classified as suffering from headache if the answer was ‘daily, more than once a week, once a week, and more than once a month’ and not suffering from headache if the answer was, of course, ‘no’ for the first question and ‘once a month or less’ for the second question. If the patient was classified as ‘headache’, he/she was then asked to respond to other questions about: (i) the circadian timing of the headaches (usual onset during night (yes or no), usual onset during day (yes or no), ‘Do you wake up with a morning headache?’ (yes or no)); (ii) the side (unilateral vs. bilateral); (iii) the localization (frontal vs. non frontal); (iv) the characteristic (pulsating vs. non-pulsating quality); (v) the intensity (annoying or not); (vi) the presence or absence of phonophobia, photophobia, nausea, vomiting, or aggravation by routine physical activity; and (vii) requiring any anodyne medication.
For each patient age, sex, occurrence of hypertension (blood pressure >160 mmHg systolic or >95 mmHg diastolic on at least two occasions, or previous hypertension with treatment), smoking habits (smoker, ex-smoker and non-smoker) and body mass index (BMI) in kilograms per square meter were recorded.
All patients underwent a full nocturnal polysomnography, including electroencephalogram, electro-oculogram, electrocardiogram, chin electromyogram, recording of respiratory thoraco-abdominal efforts, naso-buccal airflow, and measurement of arterial blood oxygen saturation (SaO2) with a finger oxymeter (Ohmeda). Sleep parameters were determined by a visual analysis according to Rechtschaffen and Kales criteria. The apnoea/hypopnoea index (AHI) representing the number of apnoeas and hypopnoeas per hour of sleep was calculated. Apnoea was defined as 10 s of interrupted naso-buccal airflow with or without thoraco-abdominal movements. Thus central and obstructive apnoeas were counted together. Hypopnoea was defined as a 50% decrease of the naso-buccal airflow, measured with a thermic captor, inducing at least a 4% decrease of the SaO2 in comparison with the level just before the respiratory event. An AHI≥15 per hour of sleep defined the SAS group (group I) and an AHI<15 per hour of sleep was considered as normal and defined the group of snorers (group II). Among these patients (group II), those who had an AHI<5 were considered as controls (group IIa), and those with an AHI=5 but <15 were considered as an intermediate group (group IIb) and were excluded from the study in a secondary analysis.
A subjective sleepiness score was performed using the Epworth Sleepiness Scale (13).
Emotional state was quantified using the Zerssen scale (14). The patient was considered as depressed if the score was ≥27 and/or if he/she had current antidepressant treatment.
Follow-up
Follow-up information was obtained by a phone interview in June or July 2000. Patients with headaches (SAS and snorers) were interviewed by one of us (MB) who was unaware of the treatment (CPAP, surgical treatment or not). The BMI was re-evaluated. Headaches were also re-evaluated in terms of presence or absence (including, if possible, the delay of recovery). The CPAP compliance was estimated on the basis of the mean duration of time that the apparatus was used each day (determined from the CPAP time built-in counter) and by the patients' self-assessment.
Statistical analysis
Data analysis was performed on a PC microcomputer with a Statistical Analysis System package (15). The different variables of the two groups for snoring (group II) and SAS (group I) were compared by using Fisher's exact test, Mann–Whitney's test or stratified Mantel-Haenszel's test. In a secondary analysis using the same tests, the variables of group IIa (AHI<5) and group I (AHI≥15) were compared. Two-tailed P-values below 0.05 were considered significant. Univariate analysis was performed to detect factors associated with headaches in both groups (P<0.10), using the χ2 test. These factors were incorporated as independent variables and headache as the dependent variable in a multivariate analysis by forward stepwise logistic regression (16).
Results
Patients
We studied 324 consecutive patients referred to our department for snoring from April 1996 to July 1999. Twelve patients were secondary excluded (3.7%), as they refused the polysomnographic study. There were eight men (54.6 ± 10.0 years) and four women (48.3 ± 3.0 years), with a mean age of 52.5 ± 8.7 years. Three of them (25.0%) suffered from headache.
A total of 312 patients were definitely included in our study. There were 256 men (51.7 ± 11.1 years) and 56 women (51.2 ± 8.6 years), with a mean age of 51.6 ± 10.7 years.
According to our SAS definition, there were 164 patients in group I (54.0 ± 10.7 years) and 148 patients in group II (48.9 ± 10.0 years). Group I was significantly older (P<0.001), heavier (P<0.001), with a majority of male gender (P<0.01) and a higher percentage suffering from hypertension (P<0.0005). However, depression and Zerssen's score were not statistically different (respectively P=0.24 and P=0.16) between the two groups (Table 1).
Clinical characteristics of SAS (group I) and snorers (group II)

The prevalence of depression was very high in both groups, as more than 25% of the patients had a score ≥27 at the Zerssen's scale or took an antidepressant treatment daily.
Polyomnographic study
Polysomnographic data are shown in Table 2, demonstrating that patients with SAS had significant differences in respiratory parameters and nocturnal sleep architecture, with increased percentages of stages 1–2 and decreased percentages of stages 3–4 and REM sleep. The wide range of the AHI indicated that patients with mild to severe SAS were included in the study.
Polysomnographic parameters of SAS (group I) and snorers (group II)

Furthermore, subjective daytime sleepiness among patients with SAS was only slightly higher than that observed among snorers (P<0.05).
Headache
The prevalence of headache was high in both groups of patients but not significantly different. Furthermore, the different characteristics (Table 3) of headaches were quite similar in both groups, especially morning headaches, which were frequent and reported in 18.9% of the patients in the SAS group and 16.2% of the snorers group (P=0.15). If the International Headache Society (IHS) criteria were applied (17), 14.9% of the total number of snorers and 9.1% of the SAS patients had migraine without aura (P=0.12), which was more frequent among women (P<0.001) and younger patients (P=0.05). If we excluded these patients with migraine from the study, the prevalence of morning headaches would be significantly higher (P<0.05) in SAS patients than in snorers.
Clinical characteristics of headache among SAS and snorers
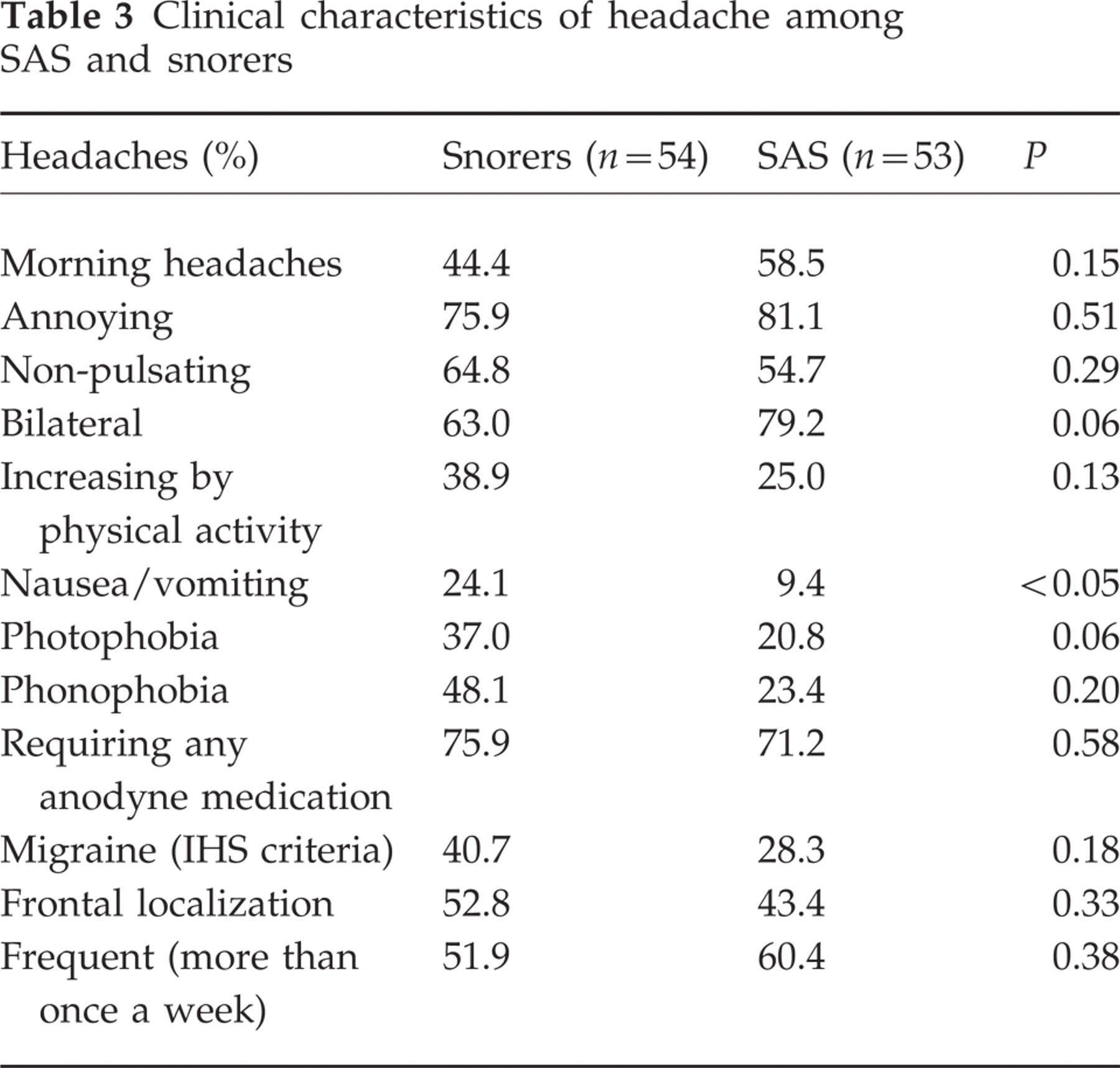
In a secondary analysis, omitting the intermediate group (group IIb), the prevalence of headaches, the characteristics of headaches and especially morning headaches were not statistically different between group I and group IIa (AHI<5).
For the whole population (groups I and II), univariate analysis demonstrated the following factors associated with headaches: higher depression score (Zerssen's score), lower AHI, lower percentage of sleep stage 1, higher percentage of sleep stages 3–4, and lower awakening SaO2 (Table 4). After multivariate analysis, headaches among snorers and SAS were not associated with nocturnal respiratory or architectural disturbances, nor with daytime sleepiness, age, sex and hypertension. However, headaches were strongly associated with the presence of depression (defined be a Zerssen's scale≥27) and with the Zerssen's score (continuous variable).
Clinical and polysomnographic parameters among patients with and without headache

The different patterns of nocturnal respiratory parameters, sleep architecture, and the Zerssen's and Epworth's scores were not significantly different among cephalalgic SAS patients with or without morning headaches.
Follow-up
Among 53 SAS patients with headache, two were lost to follow-up (3.8%). Twenty-eight patients underwent nasal CPAP treatment, four uvulopalatopharyngoplasty and 19 no treatment except for dietetic advice. Marked improvement of headaches was observed in 36 patients (70.6%) after a few weeks (3.9 ± 3.2 weeks), but was not correlated with any treatment (P=0.80) nor with CPAP (P=0.19) and initial AHI (P=0.77). However, complete relief of headache was observed in 20 of these 36 patients and was significantly correlated with treatment (P<0.04) and with the initial SAS severity (P<0.01).
Among 54 snorers with headache, 18 were lost to follow-up (33.3%). Five patients (13.8%) underwent uvulopalatopharyngoplasty (UPPP) and 31 (86.2%) had no treatment except for dietetic advice. Marked improvement of headaches was observed in 17 patients without any correlation with surgical treatment (P=0.54) nor significant loss of weight (P=0.78).
Although, there was no statistical difference between improvement of headaches among snorers and SAS patients (P=0.10), complete relief of headache was significantly more frequent among SAS patients (P<0.05) and among patients (SAS patients and snorers) who had a treatment (CPAP or UPPP) or a significant loss of weight (P<0.02).
Discussion
In 1978, Guilleminault et al. reported that 36% of patients with SAS complained of diffuse or morning headaches (2). Since this first report, the high prevalence of headaches in SAS has been confirmed in the literature, at variable levels of importance from 15% (5, 10) to 25% (11) and up to more than 50% (3, 4, 9). On the other hand, between 12% (6) and 41.7% of patients with severe and recurrent nocturnal or morning headaches had a SAS on polysomnography (18).
This apparent discrepancy for frequency of headaches is essentially due to: (i) different types of study (i.e. retrospective [3, 9, 10] or prospective [5]); (ii) different definitions of headaches, essentially depending on their frequency (i.e. ‘experienced headaches in the year prior to their study’[3] or ‘frequent headaches’ defined as those occurring often or almost always [10] or ‘headache at least once a week’[5]) or on their intensity (headaches or ‘markedly intense headaches’[4]) and the time of onset for headaches or solely morning headaches (7, 11). In our study, we confirm the high frequency of headache (32.3%) and solely morning headache (18.9%) in our SAS population.
However, despite this high prevalence, there is no statistical difference for frequency of headaches between our two groups, SAS and snorers. Other reports comparing SAS patients, patients with periodic leg movements of sleep and psychophysiological insomnia (11) or sleep-disordered breathing (10) were also unable to find any significant differences between groups of patients. In contrast, frequent morning headaches were found to be more frequent in patients with other sleep-disordered breathing than in patients with sleep apnoea (10). Indeed, morning headaches can also be encountered in many other diseases other than SAS, including intracranial and arterial hypertension, depression, tension-type headache, and cluster, hypnic and migraine headaches. Nevertheless, two recent prospective studies demonstrated a strong relationship between headache and two types of sleep-disordered breathing: heavy snoring and SAS (5, 7). In the Swedish study (7), morning headache was at least three times more frequent among male and female, heavy snorers (448 patients) and SAS (324 patients) than among the general population (583 patients). Furthermore, headache in the control group was significantly more common among snorers than non-snorers (7). As in our study, 18% of the heavy snoring and SAS groups reported experiencing headache often or very often upon awakening (7). In the Danish study (5), self-reported snoring was also significantly associated with headache among 3323 men, aged 54–74 years, independently of potential confounders such as lifestyle factors, age and body mass index (5).
In one recent retrospective study concerning 80 consecutive SAS, 60% of them had headaches in the year prior to the study, of whom 41% reported morning headaches. These awakening headaches were significantly more common in the SAS group compared with the control group of 22 patients with periodic limb movement disorder, but the proportion of common type headaches was similar in both groups (9). These awakening headaches were brief, lasting no more than 30 min, and their occurrence and severity were correlated with SAS severity (9).
If, as in other studies, there was no significant difference in the prevalence of headache among snorers and SAS patients, our study also failed to demonstrate any statistical difference between the two groups concerning the different clinical characteristics of headaches. However, if patients with migraine according to the IHS criteria were excluded from the study, morning headaches were significantly more common among SAS patients as previously reported (9), but without correlation with the SAS severity. One possible bias explaining this absence of difference of headache among controls and SAS patients could be that there is no truly normal control population (snorers). In fact, it can easily be argued that the snorers had at least qualitatively similar pathophysiological changes during sleep as the sleep apnoea patients. Furthermore, oesophageal manometry was not performed, and it can also be argued that some patients with upper airway resistance syndrome were included in the control group.
The mechanisms by which headache could be associated with heavy snoring or SAS remain unknown. Hypoxia, hypercapnia (12), hypercapnic vasodilatation, disturbances of cerebral blood flow autoregulation during the early morning hours, excessive neck movements (19), bruxism (20), transient increases in intracranial pressure (21, 22) and sleep disturbances were evoked (23). Hypoxia, which could explain headache in high altitude headache (24), has also been implicated in cluster headache (25–27). In cluster headaches, oxygen desaturation between 65% and 89% for 1–33 min preceded 8 out of 14 attacks of cluster headache among 10 patients (25). However, as in Aldrich and Chauncey's study (10), we were unable to find any significant correlation with headache, morning headache and nocturnal respiratory parameters (except for awakening arterial blood oxygen saturation) among our patients. Although, morning migraine was associated with prolonged episodes of sleep stages 3–4 and REM (28), our study does not support this hypothesis, as there was no correlation between headache, morning headache and sleep parameters in the SAS group. Another potential mechanism, which was earlier emphasized by Aldrich and Chauncey (10), could be anxiety and depression. Indeed, in our study, depression (qualitative as well as quantitative variable) was the only parameter remaining significantly correlated with headache after multivariate analysis. Depression, which is frequently reported among SAS patients, at between 20% and 56% (29, 30), was unfortunately never taken into account in previous studies (5, 7, 9, 10). Furthermore, it is known that nasal CPAP treatment could lead to a significant improvement in depression (30, 31).
Our study emphasizes that improvement, and especially complete relief of headache, can be observed in more than half of the SAS patients. This complete relief is correlated with treatment (CPAP, UPPP or diet) and with the initial SAS severity. This improvement could also be observed among snorers, but was uncorrelated with treatment (UPPP or diet). No other specific prospective study was devoted to the effect of treatment on headache, except for anecdotal case reports demonstrating improvement in morning headache after surgical (32) or CPAP treatment of SAS (6, 26). One small series showed marked (28.6%) or moderate (21.4%) improvement in half of 14 SAS patients without any data regarding the delay of recovery. There was no significant difference in age or sex of the responders, but these latter had significantly more severe SAS than the non-responders (11). This improvement was essentially observed in patients with awakening headaches compared with minimal improvement of migraine, tension and cervicogenic headaches (9, 33).
The main conclusion to be drawn from this study is that headaches are frequent among SAS patients but the relationship between headache, morning headache and SAS remains ambiguous. Our results clearly indicate that depression could be one of the most important mechanisms associated with headache among snorers and SAS patients and that the role of hypoxia and sleep disturbances was probably overestimated. As our results suggest that more than two-thirds of patients with headache and SAS will be helped with nasal CPAP, we believe that inquiring about clinical signs of SAS should become a routine practice among patients with chronic headaches.
Footnotes
Acknowledgements
The authors are grateful to Ronald Prendergast for helpful comments and English revision.
