Abstract
Background
It has been suggested that migraine attacks strike according to circadian patterns and that this might be related to individual chronotype. Here we evaluated and correlated individual chronotypes, stability of the circadian rhythm, and circadian attack timing in a large and well-characterised migraine population.
Methods
In 2875 migraine patients and 200 non-headache controls we assessed differences in: (i) distribution of chronotypes (Münich Chronotype Questionnaire); (ii) the circadian rhythm’s amplitude and stability (Circadian Type Inventory); and (iii) circadian timing of migraine attacks. Data were analysed using multinomial and linear regression models adjusted for age, gender, sleep quality and depression.
Results
Migraineurs more often showed an early chronotype compared with controls (48.9% versus 38.6%; adjusted odds ratio [OR] = 2.42; 95% confidence interval [CI] = 1.58–3.69;
Conclusion
Migraine patients are less prone to be of a normal chronotype than controls. They are more languid and more rigid when changes in circadian rhythm occur. Most migraine attacks begin in the early morning. These data suggest that chronobiological mechanisms play a role in migraine pathophysiology.
Introduction
Several studies have suggested that migraine attacks show seasonal and circadian periodicity with attacks more likely to occur in the early morning, implicating chronobiological mechanisms in attack triggering and initiation (1–3). Candidate mechanisms include those involved in: (i) the later stages of the sleep cycle and/or the sleep/wake transition; (ii) various intrinsic circadian cycles; and (iii) mechanisms functioning as
Chronotype refers to an individual’s endogenous circadian clock rhythm and how it synchronises (entrains) to the 24 h day. Chronotypes depend on sex, age, genetic and environmental factors (4), and are distributed normally in a given population. Some are very late (evening) people (‘owls’) while others are very early (morning) people (‘larks’) (5). Early and late chronotypes have been associated with several diseases. Early chronotypes have been associated with depression (6) and epilepsy (7), paroxysmal brain disorders with strong bidirectional co-morbidity with migraine (8). Late chronotypes have been associated with suicide attempts (9) and bipolar disorder (10), psychiatric disorders showing unidirectional co-morbidity with migraine (9,11). Only a few small studies have investigated chronotype and migraine, with inconsistent results (2,12). Whether early or late chronotypes are associated with specific circadian timings of the onset of migraine attacks is unknown. Besides circadian phase (i.e. chronotype), low amplitude and high flexibility of circadian rhythm enable better coping with changes in sleep/wake pattern (13).
The present study has three aims. First, to analyse chronotypes in a large and well-defined migraine population. Second, to assess circadian rhythm amplitude and stability in relation to migraine. Finally, to study whether chronotype and circadian timing of migraine attacks are associated.
Material and methods
Participants
Our study was conducted as a part of the Leiden University Medical Center Migraine Neuro Analysis (LUMINA) programme (14). Participants were Dutch adults aged 18–74 years, both migraine patients and healthy controls. Patients with migraine with and without aura fulfilled the International Classification of Headache Disorders (ICHD-3 b) criteria (15). Controls did not suffer from migraine, cluster headache, chronic tension type headache or medication overuse headache. Both migraine patients and controls were recruited via public announcement, advertising in lay press and via the research website, and were considered eligible after a two-step inclusion process using validated questionnaires (see Supplementary Text 1 for details).
Respondents and non-respondents in this study
Eligible participants (both migraine patients and non-headache controls) within the LUMINA study were sent an invitation to participate in this study into chronotype by e-mail. A reminder was sent twice. Those not having participated after two reminders were considered non-respondents. Baseline and demographic data of the non-respondents were available in the LUMINA study.
Standard protocol approvals, registrations and patient consents
The study had been approved by the local medical ethics committee. All participants provided written informed consent prior to the procedure.
Design
In this observational and cross-sectional study, eligible participants were sent an invitation to a digital questionnaire on sleep habits and sleeping problems. This included questions on phase, rhythm and stability of circadian chronotype as well as items on circadian timing of migraine attack onset. Questionnaires were filled out between September 2010 and September 2011. Non-responders were reminded twice by email and once by telephone.
Chronotype assessment
Circadian chronotype phase
Chronotype was assessed using a Dutch translation of the Munich Chronotype Questionnaire (MCTQ) (16,17). The MCTQ obtains one’s subjective self-reported chronotype (early, normal or late). From the MCTQ, the ‘timing of mid sleep on free days’ is calculated. This timing is an objective measure of chronotype derived from the timing of mid-sleep on free days (MSF), the point of time exactly in the middle of the total sleep time on free days (individual sleep timing and duration are independent traits). For participants who indicated they were on shift-work at the moment of filling the questionnaire, additional questions on timing of sleep, going to bed, etc. – separately for each of the different shifts – were visible and obliged to fill out. At the moment of our study, the specific MCTQ that is validated for shift-work (18) was not yet available.
Chronotype, sleep duration, age and gender
Both sleep duration and sleep timing on free days are influenced by the sleep-debt accumulated over the workweek (17). These parameters were therefore corrected for the confounding effect of sleep-debt during the workweek, which were used in the analyses (17). Analyses between migraineurs and controls were adjusted for gender and age, given its age- and gender-dependency (17).
Circadian rhythm amplitude and stability
Amplitude and stability were assessed using the Circadian Type Inventory, a scale measuring individual capabilities and preferences regarding changes in sleep pattern (19). The scale consists of two subscales. The languid-vigour scale reflects the individual capability to recover from a change in sleep pattern and is linked to the amplitude of the circadian rhythm. The flexible-rigid scale reflects preferences regarding sleep pattern and is linked to the rhythm’s stability. Each subscale contains 15 items, with five answer options (in the range of 1: ‘practically never’ to 5: ‘practically always’), and total scores were in the range of 15–75 per subscale. A higher score on languid-vigour indicates that an individual is more tired after changes in circadian rhythm (i.e. more languid: difficulty to overcome drowsiness and lethargy after reduced sleep). A lower score on flexible-rigid indicates that an individual is coping less with being active or sleep at unusual hours (i.e. less flexible, more rigid).
Circadian timing of attack onset
Migraineurs were asked to indicate on what time of the day attacks usually started, in 6-h intervals (00:00–06:00; 06:00–12:00; 12:00–18:00; 18:00–00:00; or ‘cannot indicate’). If 6-h intervals were indicated, patients were asked to be more precise in 2-h intervals, if possible.
Migraine characteristics, demographics, data on sleep quality and depression
Within the LUMINA cohort, data on migraine characteristics were available. Of both migraineurs and controls, demographic data on intoxications, sleep medication and sleep data (Pittsburgh Sleep Quality Index; range 0–21, with score >5 indicative for poor sleep quality (20)) were collected. For depression, data from the HADS questionnaire (21) (Hospital Anxiety and Depression Scale with Anxiety and Depression subscales; total range 0–42, with HADS-D score ≥8 indicative for depression), CES-D (the Center for Epidemiologic Studies Depression scale; total range 0–60, CES-D > 16 indicative for depression (22)) and a combined life-time depression algorithm (8) (HADS-D ≥ 8 or CES-D > 16 or physician-made diagnosis of depression or use of antidepressants with indication of depression) were used (23).
Statistics
General characteristics were compared between migraineurs and controls using Student’s t-tests for continuous variables and Chi-square tests for categorical data. To assess differences in chronotypes between patients and controls Chi-square tests and multinomial regression analyses were performed with chronotype as dependent variable (levels: early, normal and late chronotype), adjusted for age and gender, and additionally for sleep quality and HADS depression score and shift-work. Mid-sleep on free days corrected for sleep debt (MSFsc) was compared between patients and controls using a linear regression model, adjusted for age and gender. Continuous data on circadian rhythm’s stability and amplitude were analysed using linear regression models (to identify determinants). The relationship between circadian timing of attack onset and chronotype was also assessed using Chi-square tests and multinomial regression analyses. All data analyses were performed using SPSS 17.0 (SPSS Inc., IBM, USA), with the statistical threshold at
Results
Study population
Baseline characteristics of migraineurs (n = 2389) and non-headache controls (n = 189).

BMI, body mass index; F, female; HADS-D, Hospital Anxiety and Depression Scale, Depression subscale; PSQI, Pittsburgh Sleep Quality; SD, standard deviation.
Confounders
Compared with controls, migraineurs more frequently were female, had a lower educational level and had a slightly higher body mass index (BMI). They consumed fewer units of alcohol per week, had higher total scores on the Pittsburgh Sleep Quality Index and Hospital Anxiety and Depression Scale -Depression subscale (HADS-D), as well as a higher prevalence of lifetime depression.
Non-respondent data
Non-responder analysis in controls showed higher HADS-D score compared with responders due to one non-responder control who was an outlier with severe depression (
Chronotypes in migraineurs and controls
Self-reported chronotypes
Chronotypes were distributed differently between groups (unadjusted proportions;
Self-reported chronotypes in migraine subtypes
Earlier chronotypes were over-represented in both migraine with aura and migraine without aura (Chi-square test;
Timing of mid-sleep on free days corrected for sleep-debt (MSFc)
MSFsc was not different between migraineurs and controls (mean ± SD): 3:39 ± 0:58 versus 3:43 ± 0:59, adjusted for age and gender
Amplitude and stability of the circadian rhythm
Amplitude of the circadian rhythm
Predictors of higher scores on languid-vigour (LV) and flexible-rigid (FR) subscales in migraineurs and non-headache controls.
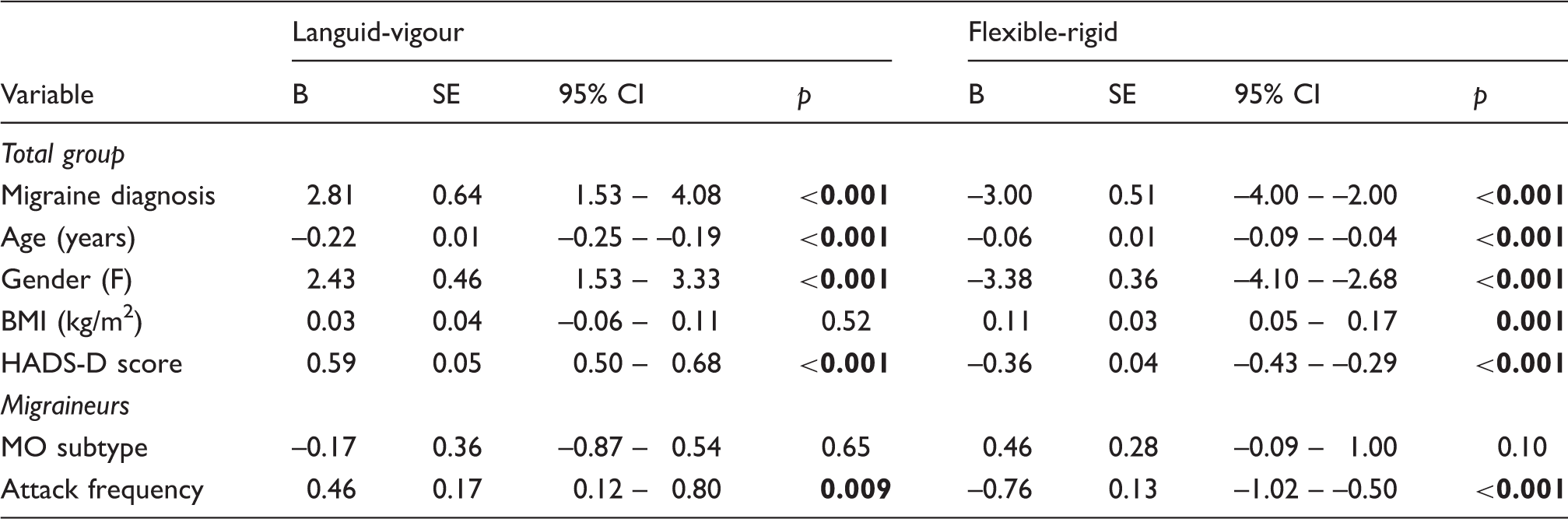
Higher score on LV scale reflects more languidness. On the FR scale, higher scores reflect more flexibility while lower scores reflect more rigidity.
B, regression coefficient; CI, confidence interval; F, female gender; HADS-D, Hospital Anxiety and Depression Scale, Depression subscale; MO, migraine without aura; SE, standard error.
Stability of the circadian rhythm
Migraineurs were more rigid (less able to cope with changes in sleep/ wake pattern) than controls (reflected by lower scores): 48.0 ± 0.6 versus 51.0 ± 1.0;
Circadian timing of attack onset in migraineurs
In total, 1462/2389 (61.0%) of migraineurs indicated a specific circadian timing for their migraine attacks, most often between 00:00–06:00 (505/1462; 34.5%), and between 06:00–12:00 (463/1456; (31.7%). Out of these, 1050/1462 (71.2%) were able to indicate the usual timing of the onset of their attacks in 2-h segments: 02:00–04:00 and 04:00–08:00 were reported most frequently (together: 399/1050; 38.0%) (Figure 1).
Distribution of circadian periodicity of migraine attack onset in migraine patients. The upper panel depicts the timing of clinical onset of migraine attacks in 1456/2389 (61.0%) migraineurs who were able to specify the circadian timing of their attacks in 6-h intervals. Attack most often began between 04:00 and 06:00 (15.4% of total) or between 06:00 and 08:00 (11.8% of total). In the lower panel, specifications into 2-h intervals are depicted, with the bars accented in grey showing patients who could not further specify in 2-h intervals. Percentages in the lower panel add up to 100%.
Patient chronotype linked to clinical migraine characteristics
Chronotypes were associated with attack time (Chi-square test; overall model Chronotype in relation to migraine attack onset. Early chronotypes are over-represented among migraine patients with early attack onset. The proportion of migraine patients with early chronotype declines with advancing circadian attack onset time, while the proportion of late chronotypes increases. Normal chronotypes are evenly prevalent among subgroups with different attack onset times.
Discussion
We found that migraineurs are less prone to be of a normal chronotype compared with healthy controls and that they are less flexible in adapting to changes in the sleep/wake cycle. Migraine attack onset peaks in the early morning and is related to early chronotype. These findings suggest a different setting of the circadian clock in migraineurs and that mechanisms which are involved in the initiation of migraine attacks are linked to chronobiological pathways.
In contrast to two other, smaller studies which reported contrasting results, our study found that both migraine with and without aura patients are less prone to be of a normal chronotype. Gori et al. also reported over-representation of both morning and evening type participants in 100 migraine without aura patients versus 30 healthy controls (2). In 93 patients with menstrual migraine, Cevoli et al. found no differences in chronotype distribution compared with 85 controls (12). We found that over 60% of patients reported circadian periodicity of their attacks, and that those migraine attacks showed a predilection for the early morning, mostly in patients with early chronotype. This is in line with earlier smaller studies (1–3). Fox et al. reported that migraine attacks started most frequently between 04:00–08:00, based on 3598 migraine attacks in 1698 patients (1). In an 11-month prospective study with 58 female patients, Alstadhaug et al. found that migraine attacks tended to peak around the middle of the day (3). Amplitude (languid-vigour) and stability (flexible-rigid) of the circadian rhythm have not been studied before in relation to migraine. Our data show that migraine patients are more languid, indicating that they have more difficulty to overcome the effects of reduced sleep. They also have a more rigid circadian rhythm, i.e. they prefer to sleep and be active at set hours. Both effects are most pronounced among patients with high attack frequency. The effect sizes, however, are small and the exact clinical relevance needs to be further studied.
Our study has several strengths. The study sample is very large and the patients are well characterised. Non-headache controls and patients were recruited in exactly the same way, minimising the risk of inclusion bias. The use of validated instruments for migraine diagnosis and chronotype (5,14) assured large populations of well characterised migraine patients and healthy participants and detailed evaluation of circadian rhythmicity. Third, the web-based questionnaire was easy to fill out and send in, resulting in high response rates in both groups.
Some limitations of the study can be addressed. There were some differences between the migraine and control groups. Migraine patients were more often female, had lower education levels and used less alcohol. They showed lower sleep quality and higher depression scores. Ideally, the differences between the migraine and the control groups would have been smaller. To minimise potential bias, the primary analyses were adjusted for the effects of age and gender. As an additional check, additional corrections were performed for the effects of sleep quality and depression. These, however, did not affect the differences between migraineurs and controls. Furthermore, we were not able to include the specific MCTQ shift-work version (18) in this study since it was published after our data collection period. However, in participants who indicated that they were doing shift-work at the time of filling out the questionnaire, additional questions for each of the possible shifts separately were included. Unfortunately, as the number of participants with recent shift-work (last week) was very small, no useful separate analysis could be made.
Although the number of control participants in our study was considerably smaller than the number of migraine patients in our study, and smaller than numbers from population based studies, the distribution of chronotypes in the control group is similar to the general population. We believe the smaller size of the control group has hardly affected the statistical power of the study. The number of cases (n = 2389) was high and the number of controls (n = 189) was still considerable, resulting in post-hoc power of 0.93 to detect the 12% difference in proportion of early chronotypes between both study groups at alpha 0.05. With regard to circadian rhythm amplitude and stability scales, post-hoc power to detect the differences we found was 1.0 (see Supplementary text). Increasing the number of controls would have involved disproportionately large and, in fact, unnecessary efforts leading to only moderate increase in study power.
The participants within the LUMINA study have partly been self-selected, since registration and participation via the study website was obligatory. The population we invited for participation was highly motivated as was reflected by the high response rates (over 80%). Although we cannot rule out a self-selection bias, since both patients and controls have been recruited similarly, we feel this potential bias has affected both groups in a similar way. Since, in the LUMINA cohort, only 4% of participants were included from our dedicated headache outpatient clinic and 87% of participants have been diagnosed with migraine previously by a physician, we feel these patients are representative of the migraine population in our country.
Over-representation of early chronotypes, early circadian attack onset and high circadian rigidity suggest that migraineurs have a different setting of the endogenous pacemaker in the suprachiasmatic nucleus, the main circadian rhythm initiator. This nucleus has extensive projections to the hypothalamus and the pineal gland and is pivotal in regulating wakefulness, the sleep/wake cycle (24) and various other body rhythms. The suprachiasmatic nucleus has been suggested to play an important role in the pathophysiology of episodic brain disorders such as cluster headache and migraine. It is unknown where, how and why migraine attacks begin and the hypothalamus might be the site of initiation. Several observations and arguments support this hypothesis. Anatomically, the hypothalamic A11 dopaminergic nucleus facilitates and modulates trigeminovascular nociception (25), the underlying mechanism for headache in migraine. Clinically, hypothalamic involvement is suggested by the nature of the premonitory symptoms which frequently occur several hours to even days before the headache and other features of the migraine attack begin: the circadian rhythmicity of migraine attacks (1,2); the temporal relationship between fluctuations in female hormone levels (menarche, menstruation, pregnancy and menopause) and onset, recurrence and disappearance of migraine attacks in women; and changes in several other hormones (26). Altered hypothalamic activation during spontaneous migraine attacks has also been detected in a positron emission tomography study (27). More recently, a functional imaging study covering three untreated migraine attacks in one patient showed, in addition to hypothalamic activation, altered functional coupling to the trigeminal spinal nuclei prior to an attack. Functional changes in the hypothalamo-brainstem coupling might be an important driver for attack initiation (28).
Early chronotypes are also over-represented in depression and epilepsy (29). These paroxysmal brain disorders show strong bidirectional co-morbidity with migraine, suggesting overlapping pathophysiologic mechanism, possibly shared genetic factors (8). These might predispose to early and late chronotypes and circadian rigidity. This hypothesis is further supported by observations in two rare genetic conditions, in which migraine is associated with marked changes in sleep pattern or biorhythm. First, in familial advanced sleep phase syndrome, a very rare, highly penetrant autosomal disorder caused by a mutation in the casein kinase 1-delta (
In conclusion, most migraineurs are early birds and have difficulties in coping with (acute) changes in the sleep/wake cycle. Attacks preferentially strike in the early morning. These observations underscore an important role for chronobiological mechanisms in migraine attack initiation.
Clinical implications
Migraine patients are less prone to be of a normal chronotype and they are more languid and more rigid when changes in circadian rhythm occur. 60% of migraine patients report diurnal periodicity of headache attacks, of which one-third reports attack beginning between midnight and 06:00, and one-third between 06:00 and noon. Taken together, these data suggest that chronobiological mechanisms play a role in migraine pathophysiology.
Supplemental Material
Supplementary Text - Supplemental material for Chronotypes and circadian timing in migraine
Supplemental material, Supplementary Text for Chronotypes and circadian timing in migraine by WPJ van Oosterhout, EJW van Someren, GG Schoonman, MA Louter, GJ Lammers, MD Ferrari and GM Terwindt in Cephalalgia
Supplemental Material
Supplementary Tables - Supplemental material for Chronotypes and circadian timing in migraine
Supplemental material, Supplementary Tables for Chronotypes and circadian timing in migraine by WPJ van Oosterhout, EJW van Someren, GG Schoonman, MA Louter, GJ Lammers, MD Ferrari and GM Terwindt in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: W.P.J. van Oosterhout reports support for conference visits from Menarini and Allergan. G.G. Schoonman reports support for conference visit from Pfizer. M.A. Louter reports support for conference visits from Menarini and Allergan. G.J. Lammers is member of the international advisory board on Narcolepsy of UCB Pharma. M.D. Ferrari reports grants from Medtronic, and independent support from the Netherlands Organization for Scientific Research (NOW),National Institute of Halth (NIH), grant from the European Community, and the Dutch Heart Foundation. G.M. Terwindt reports support for conference visits from Menarini and independent support from NWO, ZonMW, the Dutch Heart Foundation and the Dutch Brain foundation. The other authors report no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants of the Netherlands Organization for Scientific Research (NWO) (VIDI 917.11.319 to G.M.T.) and the European Commission (EC) (FP7-EUROHEADPAIN - no. 602633). They had no role in the design or conduct of the study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
