Abstract
Background
Low β endorphin level in serum and cerebrospinal fluid (CSF) has been reported in migraine. The basis of pain relief in migraine by repetitive transcranial magnetic stimulation (rTMS) may be related to β endorphin (BE), which has not been evaluated. It is proposed to measure plasma β endorphin level in migraine patients and the change in β endorphin level following rTMS, and to correlate these changes with migraine relief.
Methods
Twenty-five patients with migraine diagnosed as per International Headache Society criteria and 20 gender- and age-matched controls were included. Their clinical characteristics including duration of migraine, its frequency, severity and functional disability, triggers, allodynia and number of analgesic used were noted. Plasma β endorphin level was estimated before and after the third rTMS session. rTMS was delivered on the hot spot of right abductor digiti minimi on alternate days for 3 days and each session consisted of 600 pulses at 10 Hz. The clinical response was noted weekly for 1 month and correlated with β endorphin level.
Results
The median age of the patients was 35 (20–50) years and 19 were females. Eight patients had episodic and 17 chronic migraine. β endorphin level was significantly lower in migraine (4.35 ± 2.29 ng/ml) compared to controls (6.68 ± 2.93 ng/ml). β endorphin level was lower in chronic compared to episodic migraine (3.74 ± 2.20 versus 5.65 ± 2.02 ng/ml). Following rTMS, the headache frequency, severity, functional disability and analgesic intake significantly reduced on the seventh day of rTMS and remained significant until the fourth week compared to the baseline. The clinical improvement was associated with increase in β endorphin level (4.35 ± 2.29 versus 6.58 ± 3.33 ng/ml).
Conclusion
It can be concluded from this study that the basal plasma β endorphin level was low in migraine patients, especially in chronic migraine. The improvement in migraine after rTMS was associated with increase in β endorphin level.
Introduction
Migraine is a common condition affecting 18% of women and 6% of men (1). The exact etiology of migraine is not known but is attributed to diverse genetic and environmental factors. The pathophysiology of migraine is complex, ranging from extracranial vascular dilatation, cortical spreading depression (CSD) to trigeminovascular system. Recently hypothalamic dysfunction, channelopathies and imbalance of neurotransmitters have been postulated as the underlying cause (2). CSD has been shown to activate the trigeminovascular system in rodents and has been postulated to activate the trigeminovascular systems in humans as well (3,4). CSD may also be triggered by increase in K+ ion and glutamate as a result of intense cortical excitation. Migraine also has a strong and complex genetic basis and is susceptible to various endogenous (estrogen, 5HT, insulin, endorphin) and exogenous (stress, sleep deprivation, sunshine, cold) factors. High frequencies of various endogenous and exogenous triggers have been reported in migraine patients (5–7). β endorphin, as with other endogenous opioids, reduces neurophysiologic response to acute pain (8,9). β endorphin levels have been used as a marker of acute pain and efficacy of opioid analgesics after surgery, malignancy and burn (10,11). There are isolated reports describing low β endorphin level in patients with migraine and cluster headache (12–15). β endorphin regulation is dependent on the hypothalamopituitary adrenal axis and the hypothalamus has been implicated in the pathophysiology of migraine.
Transcranial magnetic and electrical stimulation are upcoming therapies in migraine (16–21). Low rate repetitive transcranial magnetic stimulation (rTMS) results in excitation of cortical neurons, whereas high rate rTMS results in inhibition (18). Single pulse TMS has been shown to disrupt CSD, thereby aborting the acute migraine attack (18). The role of rTMS in migraine prophylaxis has been evaluated in only few studies with contradictory results. This difference in the results may be because of the variation in stimulation parameters, site of stimulation and number of sessions (20,22). Brighina et al. found a beneficial effect of 10 Hz rTMS on left dorsolateral prefrontal cortex compared to placebo (22). Left dorsolateral prefrontal cortex has specific nociceptive effect as it relieved both right- and left-hand capsaicin-induced pain, whereas right homologous stimulation was ineffective at relieving pain (16). High rate rTMS consisting of at least 1000 pulses over the primary motor cortex (M1) has also been associated with pain relief in patients with chronic neuropathic pain (23,24). In an earlier study, we used 10 Hz rTMS of 600 pulses on left frontal region corresponding to the hot spot of abductor digiti minimi. Three sessions of rTMS on alternate days resulted in migraine relief in 80.4% patients at 4 weeks (17). The measurement of β endorphin levels before and after rTMS in migraine patients may provide insight about the mechanism of migraine relief following rTMS. The present study has been undertaken to measure the plasma β endorphin level before and after rTMS and correlate the changes with migraine relief.
Methods
Migraine patients between the ages of 20 and 65 years having more than four attacks per month were included in this study. The study was approved by the Institute Ethics Committee SGPGIMS, Lucknow, and the patients gave informed consent. The diagnosis of migraine was based on International Headache Society criteria (25). Patients with severe hypertension, coronary artery disease, seizure, pregnancy, neurological deficit, metal implants, systemic and psychiatric disease, malignancy, glaucoma, intracranial lesions on imaging and chronic pain resulting from other causes were excluded.
Clinical evaluation
The patients were evaluated for the duration of migraine, frequency of headache, triggering factors, family history of headache and associated medical illness. The frequency and type of migraine were noted and patients having more than 15 attacks per month were considered as chronic migraine and less than that as episodic migraine (26). The severity of headache was evaluated on a 0–3 scale (0 = none, 1 = mild, 2 = moderate, 3 = severe). The functional disability was also graded on a 0–4 scale in which 0 = none, 1 = mild, 2 = moderate, 3 = severe impairment of daily living, 4 = inability to perform daily activities requiring bed rest. The severity of associated symptoms such as nausea, vomiting, phonophobia and photophobia were recorded on a 0–3 scale (0 = none, 1 = mild, 2 = moderate, 3 = severe). All the patients maintained a headache diary.
Investigations
Blood counts, hemoglobin, blood sugar, serum creatinine, electrolytes and serum calcium were measured. Electrocardiogram and cranial computed tomography (CT) scan or magnetic resonance imaging (MRI) were carried out.
β endorphin assay
Five milliliters of venous blood from the antecubital vein were collected in an ethylenediaminetetraacetic acid (EDTA) vial between 10 am and 12 noon before the rTMS and 5 days later. The plasma was separated and kept at −80°C until analysis. β endorphin level was estimated by enzyme linked immunosorbent assay using Endorphin, beta (Human) - EIA Kit (Phoenix Pharmaceuticals, Burlingame, CA, USA (EK-022-14)). β endorphin was also estimated in 20 healthy controls without history of headache.
Repetitive transcranial magnetic stimulation
rTMS was performed using a Magstim Rapid (Whitland, South West Wales, UK) magnetic stimulator with an air cooled figure of eight coil, each loop 4.5 cm in diameter. The coil was angled 5 cm lateral and 1 cm anterior to the line drawn from the vertex to the left tragus corresponding to the hot spot of abductor digiti minimi. Each session of rTMS consisted of 600 pulses in 412.4 seconds. The impulses were given in 10 trains, each consisting of 60 pulses at 10 Hz with inter-train interval of 45 seconds. rTMS was delivered at 70% of motor threshold. The motor threshold was determined at the hot spot of right abductor digiti minimi as the minimum stimulus intensity able to elicit five or more motor evoked potentials of 50 µV amplitude out of 10 consecutive stimuli. Three sessions of stimulation were delivered on alternate days between 10 am and 12 noon. This protocol of rTMS was used because of our earlier experience (17).
Outcome measures
The frequency, severity of headache, functional disability, number of rescue medication and Visual Analogue Scale (VAS) which is a 0–100 scale were noted 1 month before rTMS and repeated at weekly intervals for 4 weeks following rTMS.
Statistical analysis
The baseline β endorphin level was compared between migraine patients and controls as well as episodic and chronic migraine using the Mann-Whitney U test. The change in β endorphin level on the fifth day of rTMS in patients with migraine was compared with the baseline value using paired t test. β endorphin level was also compared between episodic and chronic migraine using the Mann-Whitney U test. The clinical response to rTMS was compared by one-way analysis of variance using Tukey’s multiple comparison test. The correlation of β endorphin level with the clinical response was done by Pearson correlation test. The two-tailed p value of <0.05 was considered significant and was presented without adjustment for multiplicity. All the statistical analysis was done using SPSS 15 version or GraphPad Prism 5 software.
Results
Twenty-five migraine patients and 20 controls were subjected to plasma β endorphin measurement. The median age of the migraine patients was 35 (20–65) years and 19 (76%) were females. The median age of the control group was 37 (18–55) years and 15 were females. The control group was matched for gender (p = 1.00) and age in 5-year intervals (p = 0.64) with the study group. The median duration of migraine was 9 years (1.5–30) and all except one patient had migraine without aura. Eight patients had episodic migraine and 17 chronic migraine. The frequency of attack ranged between six and 30 per month, lasting for a median duration of 0.9 (0.4–3) days. All the patients had severe headache resulting in significant functional disability. The majority of patients had multiple triggers (median 12, range 3–14). Allodynia was present in 18 patients; cranial in six and extended to extracranial region in 12.
β endorphin levels
Plasma β endorphin level was significantly lower in the migraine patients (4.35 ± 2.29) ng/ml compared to the controls (6.68 ± 2.93) ng/ml (p = 0.005). β endorphin level was significantly lower in the patients with chronic migraine 3.74 ± 2.20 ng/ml compared to episodic migraine 5.65 ± 2.02 ng/ml (p = 0.04) (Figure 1), but it was not significantly lower in episodic migraine compared to controls (5.65 ± 2.02 ng/ml vs. 6.68 ± 2.93 ng/ml; p = 0.41). In our study, only one patient had migraine with aura whose β endorphin level was 5.25 ng/ml. In 18 patients, β endorphin level was estimated during headache (4.37 ± 2.67 ng/ml) which was not significantly different compared to those without headache (4.31 ± 0.90 ng/ml). β endorphin levels correlated with a number of migraine triggers such as fasting (r = 0.40, p < 0.05) and dry hair (r = 0.43, p = 0.03).
Error bar diagram shows significantly lower level of plasma β endorphin level in patients with episodic migraine and chronic migraine (CM) compared to controls.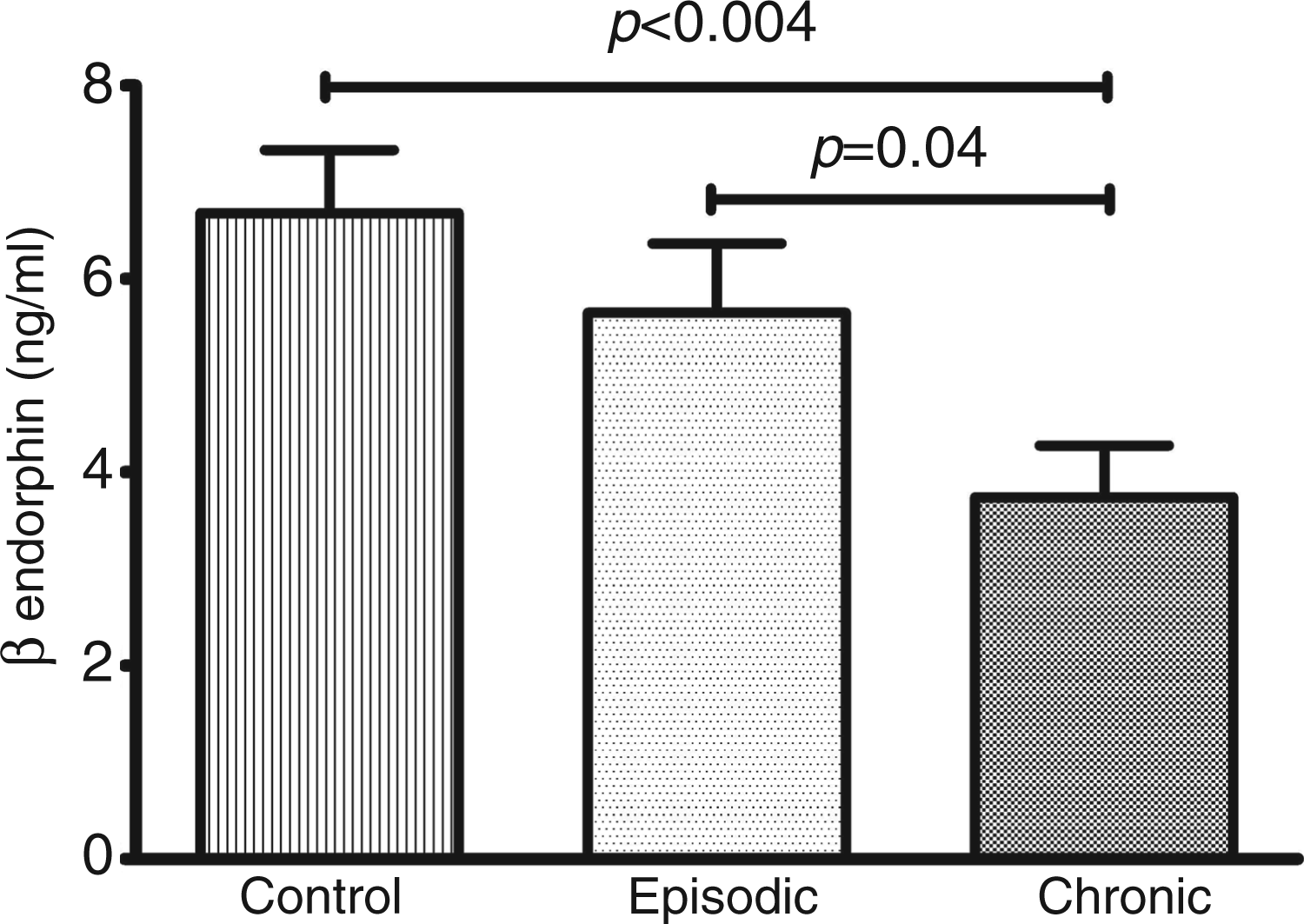
β endorphin in response to rTMS
β endorphin levels significantly increased on fifth day of rTMS (6.58 ± 3.33 ng/ml) compared to basal level (4.35 ± 2.29 ng/ml; p = 0.007). β endorphin level on the fifth day of rTMS in migraine patients reached the control value (6.58 ± 3.33 versus 6.68 ± 2.93 ng/ml, p = 0.92). In the chronic migraine group, β endorphin level significantly increased after rTMS compared to baseline (6.59 ± 3.20 ng/ml versus 3.74 ± 2.20 ng/ml; p = 0.01) but in episodic migraine the increase in β endorphin was not significant (6.58 ± 3.82 ng/ml versus 5.65 ± 2.02 ng/ml; p = 0.49) (Figure 2). The change in β endorphin after rTMS was more in chronic migraine (2.84 ± 3.78 ng/ml) compared to episodic migraine (0.93 ± 3.60 ng/ml), but the difference was not significant (p = 0.24).
Error bar diagram shows β endorphin level at baseline and at fifth day of rTMS in patients with episodic migraine and chronic migraine (CM).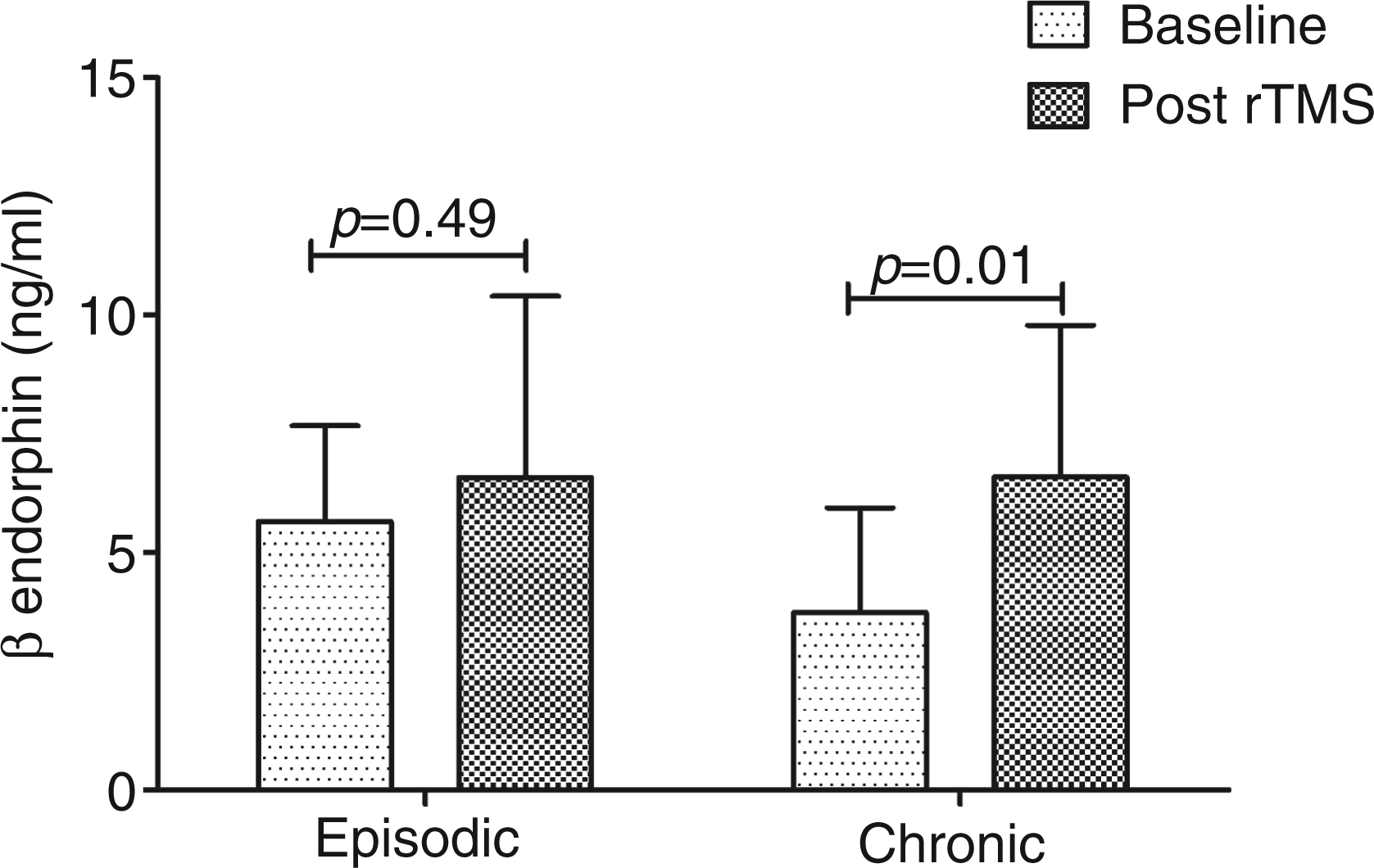
The improvement in migraine parameters at different time points following repetitive transcranial magnetic stimulation (rTMS).
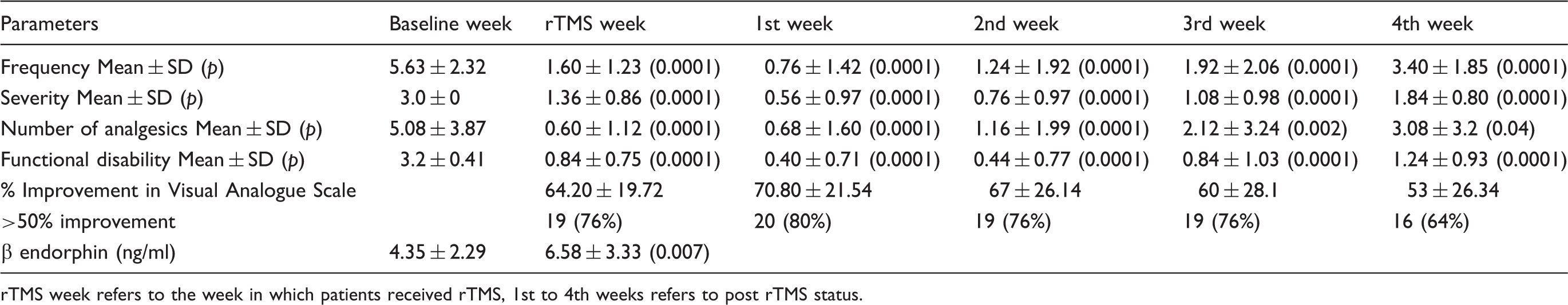
rTMS week refers to the week in which patients received rTMS, 1st to 4th weeks refers to post rTMS status.
Discussion
Our study reveals that migraine patients had lower β endorphin level compared to controls, and that the level was lower in chronic migraine compared to episodic migraine. rTMS resulted in significant migraine relief which was associated with an increase in β endorphin level. Endogenous opioid cooperates with 5-hydroxy tryptamine (5HT) in modulating spontaneous pain (8). Morphine like factors have been found to be lower in the CSF of migraine patients, suggesting failure of the pain suppressing system and associated neurotransmitters such as 5HT and β endorphin (27–29). Opioid receptors are located presynaptically in the neuronal terminals. Stimulation of β endorphin receptors by exogenous or endogenous opioids inhibits release of neurotransmitters such as noradrenaline, acetylcholine and 5HT from the presynaptic neurons to the synaptic cleft (30).
In a study on 11 patients with common migraine and continuous migraine with inter paroxysmal headache, CSF β endorphin and ACTH were measured. CSF β endorphin levels were significantly reduced in chronic migraine compared to those with episodic migraine and controls. The β endorphin level correlated with the severity of migraine (31). Plasma β endorphin level negatively correlated with total duration of migraine attack in the previous month (32). In tension type headache, β endorphin level was not significantly reduced suggesting a different pathophysiology of migraine and chronic tension headache (33,34). In a study on 50 patients with chronic tension headache, there was no difference in plasma and CSF β endorphin levels compared to controls. Moreover plasma and CSF β endorphin levels did not correlate (34). Circulating β endorphin level is considered to be of anterior pituitary origin, whereas β endorphin in CSF is released from cells of the central nervous system (31). β endorphin in CSF and blood may therefore reflect activity of two separate opioid systems. We have measured plasma β endorphin level which is a surrogate marker of hypothalamopituitary β endorphin level.
In our study, chronic migraine patients had significantly lower basal level of β endorphin compared to episodic migraine. This relationship was also confirmed in another study (31). In our study, β endorphin level during the ictal and interictal periods did not differ significantly. Low β endorphin level has been reported during migraine attacks in one study (35) but was not confirmed in another (36). During headache-free periods, plasma β endorphin level has been reported to be normal. The lack of relationship between ictal and interictal levels of β endorphin in our study may be due to small sample size and the majority of our patients having a high frequency of migraine attacks.
In our study, β endorphin level correlated with a number of migraine triggers such as fasting and dry hair. The prodromal migraine symptoms are attributed to a temporary dysfunction in homeostatic functioning controlling hunger, thirst, body temperature, awareness and mood; these symptoms are regulated by the hypothalamus and midbrain. It is therefore possible that the origin of migraine may be from these areas of brain. These areas are also related to hypoendorphinemia resulting in neuronal excitability (2). Hypothalamic dysfunction in migraine has been reported on the basis of PET MRI studies. A study on seven migraine patients having headache for less than 6 hours revealed activation of hypothalamus, periaqueductal grey matter, red nuclei and locus coeruleus contralateral to the pain and posterior reticular formation. Following sumatriptan treatment, headache subsided but the activation of midbrain and hypothalamus persisted (37).
Various non-pharmacological treatments have been used for the management of migraine, although the basis of only a few modalities has been systematically evaluated. A large amount of encephalin and endorphin are released during exercise and have been suggested to be responsible for relieving pain, anxiety and depression (38). In a study on 40 migraine patients, exercise resulted in beneficial effects in all migraine parameters which were related to the β endorphin level (32). In our study also, rTMS resulted in significant improvement in all headache parameters as well as a significant rise in β endorphin level on the fifth day compared to baseline. The post rTMS β endorphin level correlated with improvement in headache frequency in the first week after rTMS. The change in β endorphin level was much higher in patients who had low basal β endorphin levels. Low basal level of β endorphin and its significant rise following rTMS suggest a state of hypoendorphinemia in chronic migraine in our patients. Low β endorphin may be responsible for chronification of migraine. Low β endorphin level has also been reported in other chronic painful conditions such as trigeminal neuralgia and rheumatoid arthritis (39,40). There is no study showing the efficacy of rTMS in migraine in reference to β endorphin level, although both low and high frequency rTMS have been evaluated for abortive therapy in migraine (18,41,42). The benefit of rTMS in migraine may be somewhat similar to that in pain relief in acupuncture in which β endorphin level has also been reported to rise (43). β endorphin levels are elevated following seizures and vagal nerve stimulation. In a study, following intracranial stimulation, there was rise in CSF and plasma serotonin, β endorphin, gamma-aminobutyric acid (GABA) and dehydroepiandrosterone (DHEA); and a drop in plasma cortisol and tryptophan. These changes were associated with improvement in pain, headache, insomnia, depression and spasticity (44). In our study, the increase in β endorphin following high rate rTMS on the left frontal region might have been mediated by the insular hypothalamopituitary axis. Transcranial magnetic stimulation is more widespread and less restricted than electrical stimulation (45) and might have stimulated the insular cortex. Motor cortex stimulation results in an increase in cerebral blood flow in the ventrolateral thalamus, medial thalamus, insular cortex, anterior cingulate gyrus and brainstem (24). Motor cortex stimulation therefore may enhance the release of opioids from various pain processing brain structures. A placebo controlled study using functional brain imaging is needed to confirm this observation.
Clinical implications
β endorphin is reduced in migraine which is more marked in chronic migraine compared to episodic migraine. β endorphin level is increased following high rate repetitive transmagnetic stimulation (rTMS). Rise in β endorphin level following rTMS is associated with migraine relief.
Footnotes
Acknowledgement
We thank Mr S P Singh for technical, Mr Rakesh Kumar Nigam and Ms Rupali Mishra for secretarial help.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
