Abstract
Background
Evoked potentials are widely used to investigate sensory and nociceptive processing abnormalities in migraine. However, electrophysiological distinctions between migraine subtypes remain insufficiently characterized in the literature. The aim was to systematically review and summarize neurophysiological abnormalities in evoked potential studies (visual, auditory, brainstem, somatosensory and laser) in migraine patients, with a particular focus on latency, amplitude, habituation and clinical correlations across subtypes and healthy controls.
Methods
Following PRISMA guidelines, we searched PubMed, EMBASE and Web of Science for studies, terms included “Migraine Disorders,” “Migraine,” “Vestibular Diseases” and “Evoked Potentials”, which were published from 2000 to 2024 were included. Risk of bias was assessed using a modified Newcastle–Ottawa Scale.
Results
In total, 813 studies were screened, resulting in 55 studies meeting the inclusion criteria. Patients with migraine with aura demonstrated higher amplitudes and asymmetry of visual evoked potentials compared to those with migraine without aura. Habituation deficits were particularly evident across all types of evoked potentials. A few studies compared chronic and episodic migraine, reporting higher brainstem and somatosensory evoked potential amplitudes in chronic migraine.
Conclusions
Migraine patients have a consistent habituation deficit on all evoked potential parameters. Migraine with aura and chronic migraine may have higher cortical excitability. Further research with larger sample sizes, standardized methodologies and an accurate comparison of migraine phases will enlighten our understanding of the migraine subtypes.
Trial Registration
PROSPERO ID: CRD42024502803.
This is a visual representation of the abstract.
Introduction
Migraine is a chronic neurological disorder marked by altered sensory processing and hypersensitivity to light, sound, smell and touch (1–3). Understanding the migraine brain and its effects on the nervous system has been the focus of neurophysiological and functional imaging studies (4,5).
Evoked potentials (EPs) constitute a core neurophysiological approach for investigating sensory processing abnormalities in migraine. They offer reproducible and quantifiable insights into the sensory and nociceptive pathway function, cortical excitability and pain-modulating networks, capturing both peripheral input and central integration processes that underlie underlying migraine pathophysiology. The pioneering work of Golla and Winter in 1959 (6) first identified alterations in the hyperexcitable migraine brain. Since then, EPs have played a pivotal role in advancing our understanding of this complex disorder (7). Electrophysiological measures assess brain activity in response to specific stimuli, revealing changes in cortical activation, excitability and overall brain function in patients with migraine (8). These potentials are beneficial due to their non-invasive nature, ease of performance and applicability in human studies (9).
We aimed to comprehensively review and document the variability among studies, categorize and analyse their findings, and focus particularly on migraine subtypes in comparison with healthy controls (HCs). By examining the differences between patients with migraine and HCs across various modalities, we explored potential correlations between specific EP patterns, latency, amplitude, habituation patterns and clinical relationships. Our objective was to clarify whether migraine exhibits distinct EP patterns and to document the differences between different subtypes, as migraine with aura (MA), migraine without aura (MO), chronic migraine (CM) and episodic migraine (EM).
Methods
Search strategy
A systematic review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) consortium (10). The protocol was prospectively registered in the International Prospective Register of Systematic Reviews (PROSPERO) platform (Registration ID: [CRD42024502803; 26 January 2024]). The search was conducted on 9 February 2024, across three databases: MEDLINE-PubMed, Web of Science-Web of Science Core Collection (Clarivate) and EMBASE (Elsevier). Furthermore, manual checking of the references of all included studies and relevant reviews identified in PubMed and Cochrane Library was performed to supplement the electronic search, thereby enhancing the rigour and completeness of our search strategy. A literature search using medical subject heading terms and synonyms combined with the Booleans operators “AND” and “OR” was applied. The search groups and main terms were: (1) “Migraine Disorders”, “Migraine”, “Vestibular Diseases” and (2) “Evoked Potentials”. The filters applied were temporal (2000–2024), language (English) and publication type (clinical trials, randomized controlled trials, case–control and cohort studies). The comprehensive search strategy used is detailed in the supplementary data section (see supplementary material, Text S1)
Selection criteria
The search aimed to select studies assessing EPs in migraine patients, comparing these findings with HCs and among patients with different migraine subtypes. Inclusion and exclusion criteria are summarized in Table 1
Papers eligibility criteria

Abbreviations: BAEPs = brainstem auditory evoked potentials; cVEMP = cervical vestibular evoked myogenic potentials; EP = evoked potentials; FDG PET-scan = fludeoxyglucose-18 positron emission tomography scan; IDAPs = intensity dependence of auditory evoked potentials; HFO = high-frequency oscillations; LEPs = laser evoked potentials; LFO = low-frequency oscillations; oVEMP = ocular vestibular evoked myogenic potentials; RCT = randomized controlled trials; SSEPs = somatosensory evoked potentials; SSVEPs = steady-state visual evoked potentials; VEMP = vestibular evoked myogenic potential; PRVEPs = pattern reversal visual evoked potentials; VEFR = visually evoked cerebral blood flow velocity responses; VEPs = visual evoked potentials.
Article selection
The search was conducted by a librarian specialist at the Vall d’Hebron Barcelona Hospital Campus in conjunction with FB and LG. The reviewers FB, LG and PY were paired off (FB/LG, FB/PY, LG/PY) to independently evaluate all articles identified in the initial search for potential inclusion based on their titles and abstracts, adhering to the eligibility criteria (Table 1). Subsequently, reviewers independently screened the full text of records initially identified by their titles and abstracts. In case of disagreement, the third reviewer made the final determination. Any persisting discrepancies or uncertainties were resolved through discussions.
Additionally, if any article initially identified did not meet the inclusion/exclusion criteria upon examination for data extraction by SU/EU/NT, it was also excluded. Furthermore, to ensure comprehensive coverage, all reviewers manually searched for references in relevant primary articles, aiming to identify any additional eligible papers that might have been overlooked during the initial search process.
Data extraction
Three reviewers (SU, EU, NT) performed data extraction for all the selected studies, with information recorded in an Excel electronic spreadsheet. The selected studies were divided among pairs of reviewers, each responsible for extracting essential information from each paper: title, author, year of publication, recording condition including visual evoked potentials (VEP) as well as pattern reversal visual evoked potentials (PR-VEP), and steady-state visually evoked potentials (SS-VEP), auditory evoked potentials (AEP), somatosensory evoked potentials (SSEP), brainstem auditory evoked potential (BAEP), LEP, intensity dependence of auditory evoked cortical potentials (IDAP), sample size and demographics (age, gender distribution), migraine subtypes (MA, MO, EM, CM,), subject note (interictal or ictal phases, use of preventive therapy or any other treatment, concomitant diseases, otological or neurological examination, exclusion criteria), and main findings (EPs’ amplitudes, peak to peak amplitudes, latency, habituation, potentiation, long-term effects).
Primary outcomes focused on the main EP findings, including alterations in response amplitudes, latencies and habituation patterns between patients with migraine and HCs. Secondary outcomes involved comparing main EP findings between patients with different migraine subtypes (EM, CM, MA, MO), comparing migraine patients with HCs to evaluate clinical endpoints and exploring potential correlations of EP patterns and migraine profiles.
Risk of bias
The quality of included articles was independently assessed by five reviewers (SU, EU, NT, PY, LG) following the Cochrane Collaboration's Risk of Bias tool for randomized controlled trials and assessed and rated poor, fair or high quality using a modified Newcastle–Ottawa Scale for Observational Studies (mNOS). In cases of discrepancies, discussions were conducted to achieve consensus.
Albatross plot
For studies not directly reporting effect sizes, we estimated Cohen's d from published p-values and group sample sizes. Two-tailed t-values were derived from p-values and degrees of freedom (df = n1 + n2 − 2) and effect sizes were computed as: d = t · √(1/n1 + 1/n2)
Approximate values were used for threshold p-values (e.g. p < 0.05) and non-specific results (e.g. p > 0.05) were excluded. These standardized estimates were then plotted for the sensory habituation parameters in the albatross plot to visualize study findings across sample sizes. All analyses and plots were performed using Python, version 3.11.9 (https://www.python.org).
Results
Study characteristics
Our search in reliable databases identified a total of 1018 papers and reviews, which were reduced to 813 after removing the duplicates and filtering by title and abstract. Out of these 813 articles, 241 were full-text screened for eligibility. Additionally, one article was included in the review after being identified through searching of the cited references (Figure 1). All articles were strictly screened following the inclusion and exclusion criteria (Table 1). Thus, we finally included 55 articles for the analysis. Regarding the risk of bias judgments for the included studies, Table 2 details the risk of bias for each study. In summary, 54 case–control/cross-sectional studies and one randomized controlled trial were included, 53 of them scored above 7 points on the mNOS scale, indicating high-quality data. Specifically, two scored 6 points, 42 studies scored 7 points and 11 studies scored 8 points (Figure 2).

PRISMA flowchart of the studies in the systematic review.

Sensory stimulation modalities and corresponding evoked potential types.
Modified Newcastle–Ottawa Scale (mNOS) assessment of included studies

Abbreviation: RCT = randomized controlled trial.
Findings of visual evoked potential studies (n = 34)
Visual stimuli were applied in 34 studies; of the VEP studies, five used SS-VEP, one used hemifield visual evoked potential (HVEP) and the others used PR-VEP
Amplitude variability and latency findings
In total, 17 studies have examined VEP latency, with a subset of these studies reporting no significant differences (n = 11) (11–21) while six studies found differences, with shorter VEP latencies (n = 2), others reported longer latencies (n = 4) (17,22–26).
Compared to HCs, half studies reported abnormal waves in the migraine group (n = 16/34) (see supplementary material, Table S1). When the pathological amplitudes (n = 12) were analysed, nine studies reported predominantly increased VEP amplitudes in any migraine subtypes compared to HCs amplitudes (12,17,22,27–32). When comparing migraine subtypes, MA amplitude exceeded MO in five out of seven instances, while MO exceeded MA in two out of seven instances (15,17,22,27,30,33,34). Significantly higher interhemispheric asymmetry in VEP responses was also observed in all studies compared migraine to HCs (n = 3).13,20,21 Fong et al. (29) reported N2 amplitude was elevated in migraine patients only at high spatial frequency (SF) compared to HCs. The duration of disease was correlated with various parameters, including P100 amplitude, P1 latency, P1N2 amplitude, P100-N145 habituation slope and N1-P1 habituation slope (n = 4) (16,22,27,33).
Habituation traits
The majority of the studies (n = 11/17), migraine group exhibited habituation deficits or potentiation compared to HCs (11,12,14,35–42). Additionally, four out of 17 studies also reported habituation deficits varied depending on techniques and stimuli (16,30,33,41). Sharp et al. (41) observed VEP amplitude potentiation at higher frequencies (9 Hz) in the migraine group. Bednář et al. (43) reported habituation deficits in migraine at both low and high contrasts. Taken together, 14 out of 17 studies identified habituation abnormalities either independently or modulated by stimulus techniques (see supplementary material, Table S1). Studies analysing the ictal phase showed that habituation normalizes during migraine attacks (35,40).
Findings of somatosensory evoked potential studies (n = 7)
Amplitude variability and latency findings
In five out seven SSEP studies, no significant differences in latency, and, in four out of seven studies, no differences in amplitude were observed between migraine patients and HCs (see supplementary material, Table S2) (44–50). Regarding amplitudes, two studies revealed interictally lower SSEP amplitudes and one of these studies reported higher ictal amplitudes in migraine group (44,48,50).
Habituation traits
All SSEP studies that analysed habituation traits (n = 5) showed habituation deficit interictally, particularly in N20 responses (44,47–50).
Clinical correlations with SSEP responses
An habituation deficit was found to be negatively correlated with monthly headache days or attack frequency (n = 3) and with headache severity (n = 2) (35,49,50). In studies of Abanoz et al. (45) and Kalita et al. (49), N19/N1-P1 amplitudes were found to be positively correlated with disease duration. Restuccia et al. (47) and Kalita et al. (49) reported age, sex, allodynia, analgesic intake, functional disability, triggers, headache duration and disease progression were found to be correlated with SSEP habituation deficit or amplitude. Restuccia et al. (47) reported that N20 amplitude showed potentiation in patients whose migraine frequency worsened (n = 7), whereas it remained stable in those who experienced spontaneous improvement (n = 10).
Findings of auditory evoked potential studies (n = 10)
Amplitude variability and latency findings
Prolonged BAEP peak latencies and/or interpeak latencies (IPLs) were detected in migraine patients compared to HCs (n = 3/4), while AEP studies (n = 3) showed no significant latency differences (see supplementary material, Table S3) (11,51–55).
Most of the studies reported no significant difference in the amplitudes of BAEP (n = 3/4) and AEP P50/P300 waves (n = 2/3) between migraine and HCs (42,51,52,55,56). Increased IDAP slopes were observed in migraine patients compared to HCs (11,36).
Habituation traits
All AEP studies that examined habituation (n = 5) reported habituation deficits in migraine patients (11,36,42,51,52).
Findings of laser evoked potentials (n = 7)
Habituation traits
Habituation deficit was reported in most studies in the migraine group compared with HCs (n = 5/6) (57–61). Uglem et al. (62) reported no difference in habituation between EM and HCs, except in ictal EM (see supplementary material, Table S4).
Pain perception and other findings
de Tommaso et al. (57) found that, despite similar pain thresholds between groups, migraine patients reported higher pain perception compared to HCs when exposed to stimuli of the same pain intensity. In another study, de Tommaso et al. (63) reported during P2 dipole analysis, EM (MO) patients showed significantly greater x-axis displacement compared to HCs. In CM patients, both x and z coordinate shifts were significantly larger than in HCs and EM patients, indicating a broader alteration in dipole localization with migraine chronicity (63) (see supplementary material, Table S4).
Comparisons between MO, MA and HCs (n = 23)
VEP (n = 18)
The majority of VEP latency studies comparing MO, MA and HCs reported non-significant differences (n = 15/18) (see supplementary material, Table S5). While only half of the VEP amplitude studies detected significant differences (n = 8/17), subtype analyses consistently demonstrated higher amplitudes in MA compared to MO (n = 5/7) with both migraine subtypes elevated amplitudes relative to HCs (15,17,22,27,29,30,33,34).
Habituation deficit or potentiation was detected majority of the VEP habituation studies (n = 7/9) compared to HCs with no consistent differences observed between migraine subtypes. One exception was Omland et al. (15), who reported higher habituation deficit MO compared to MA, specifically in larger checker sizes.
While Shibata et al. (30) reported that both MA and MO lacked habituation at high contrast and SF, it emerged with decreasing contrast and lower SF. Notably, potentiation at 0.5 cycles per degree was observed exclusively in MA (30). In phase based analysis, pre-ictal and ictal migraine groups demonstrated habituation patterns similar to HCs, whereas both interictal migraine groups showed habituation deficit (35,42).
BAEP and AEP (n = 3)
No differences were observed between MO and MA in terms of amplitude and latency. In the study by Hamed et al. (54), analysis of interside differences revealed that BAEP III latencies were longer on the right side than on the left in MO patients. All studies demonstrated habituation deficits in migraine subtypes, with one study reporting a greater deficit in MO compared to MA at 40 dB (11,42).
SSEP (n = 3)
Two out of three studies in amplitudes and all studies found no difference in the latency and habituation between subtypes (44,46,47).
LEP (n = 1)
No significant LEP amplitude and habituation difference was found between MO, MA and HCs in one study (62).
Comparisons between MO and HC (n = 12)
VEP (n = 3)
There were no differences in VEP latencies (n = 2) and amplitudes (n = 1) between MO and HCs (14,19) (see supplementary material, Table S6). Lisicki et al. (14) found lower VEP amplitudes and significant habituation deficit compared to HCs with a first-degree relatives of migraine patients, with the first block N1-P1 amplitude was negatively correlated with the habituation slope only in those with family history.
AEP (n = 2) and SSEP (n = 1)
While two studies found no latency differences in AEP, one reported higher amplitudes in MOs compared to HCs (51,52). Both studies (n = 2) reported habituation deficits in P50 and P300 waves in the MO group, not in HCs (51,52). Abanoz et al. (45) found no differences in trigeminal SSEPs’ ipsilateral latencies, N1-P1/N2-P1 amplitudes and sensory thresholds.
LEP (n = 5)
All studies analysed habituation parameters, consistently reported habituation deficit in MO and normal habituation in HCs (n = 5) (57–61). No significant differences were found in latency (n = 5) (57–61) and amplitude parameters (n = 4) (58–61). Higher pain perception was observed in MO compared with HCs, whereas pain thresholds did not differ between groups (57).
Comparisons between MA and HCs (n = 5)
In two studies, no significant VEP amplitude differences were reported between MA and HCs (see supplementary material, Table S7) (21,23). However, four studies reported significant differences, including decreased P100 amplitude in MA with visual aura, lateral HVEP-P100 asymmetry aligning with the visual aura side, lower amplitudes during aura compared to MA and HCs, and greater interhemispheric asymmetry (N75, P100) with larger checker sizes (13,21,23,24).
No significant interictal VEP latency differences were observed between HCs and MA (n = 4/5). However, subgroup analyses revealed shorter VEP latencies in MA during the interictal phase, prolonged P100 latencies in MA during aura compared to both HCs and interictal MA, and greater interhemispheric asymmetry in VEP latencies (N75, P100) in MA when larger checker sizes were used (13,21,23).
Coppola et al. (12) found habituation deficits in with both pure visual auras and complex auras. VEP amplitude was higher in complex aura group than pure visual aura and HCs.
Comparisons between EM, CM and HCs (n = 13)
VEP studies (n = 8)
Only Lisicki et al. (38) directly compared amplitudes between EM, CM and HCs, finding no significant amplitude differences (see supplementary material, Table S8). Most studies compared EM and HC amplitudes reported no differences, however; Lisicki et al.(28) was the only study observed increased VEP amplitude in EM (37,38,43,64).
Habituation analyses consistently showed interictal deficits in EM, with normal habituation in HCs (36,37,39,40,43). Some studies noted ictal–interictal differences: Kalita et al. (39) reported deficits only ictally, Bednar et al. (43) in both phases while Ambrosini et al. (36) found deficits only interictally.
SSEP (n = 2) and BAEP studies (n = 2)
Coppola et al. (48) found significantly higher SSEP amplitudes (N20-P25) in CM and ictal EM, with CM exhibiting greater amplitudes than EM. By contrast, interictal EM showed reduced amplitudes compared to HCs. Kalita et al. (49) did not find differences in amplitude (N19-P25) among EM, CM and HCs, but the CM group insignificantly had higher amplitudes than EM. Coppola et al. (48) showed a habituation deficit in interictal EM but normal habituation in CM and ictal EM. Conversely, Kalita et al. (49) found habituation deficits in CM and ictal EM groups but not in interictal EM. In both studies, there were no latency differences between the groups (48,49).
Kalita et al. (65) reported that, BAEP amplitudes (waves II, III, IV) were higher in migraine patients (EM and CM) compared to HCs, with CM showing greater amplitudes than EM.
In summary of sensory parameters, four studies compared EM and CM amplitudes across all EPs. Half of these studies reported significantly higher amplitudes in CM than in EM (SSEP & BAEP), while one found higher amplitudes with no statical significant difference, the SSEP study showed non-significant higher amplitudes in CM than EM and HCs (VEP and SSEP) (38,48,49,65). Coppola et al. (48) reported that interictal EM exhibits habituation deficits, whereas CM and ictal EM may demonstrate normal habituation. Conversely, Kalita et al. (49) found habituation deficits in CM and ictal EM but not in interictal EM.
LEP (n = 3)
Studies directly compared LEP amplitudes and latencies among EM, CM, and HCs and did not find significant differences (58,63). de Tommaso et al. (63) reported greater displacement of the P2 dipole source along the x and z coordinates in CM compared with EM (MO) and HCs, indicating altered cortical source localization. Regarding to habituation parameters Uglem et al. (62) compared LEP habituation traits in ictal, preictal, postictal, interictal phases EM vs HCs, and found normal habituation in interictal EM and HCs and habituation deficits only in ictal EM in post-hoc analysis.
The only study compared habituation traits between CM and EM (MO) reported higher N2a–P2 amplitude habituation deficit in CM, following the pattern CM > EM (MO) > HCs (58).
Discussion
The present review set out EP abnormalities in migraine, with a particular attention to migraine subtypes in comparison to HCs. Notably, VEP studies offered the most internal consistent evidence: while VEP latencies were similar between migraine patients and HCs, a consistent habituation deficit was observed in the migraine group not only in VEPs but also across all EP modalities (Figure 3).
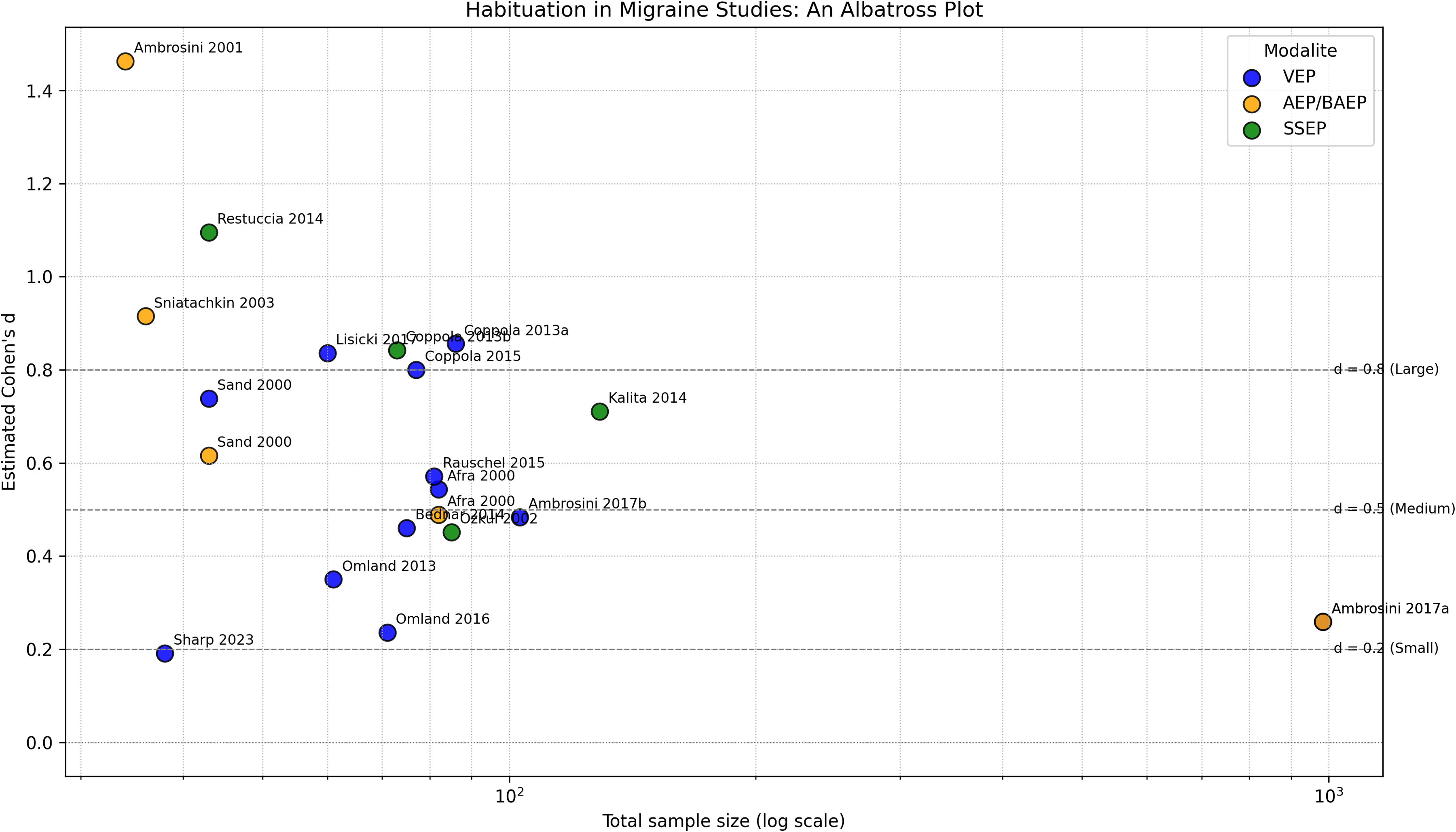
Albatross plot of habituation differences between migraine patients and healthy controls. AEP = auditory evoked potential; BAEP = brainstem auditory evoked potential; VEP = visual evoked potential; SSEP = somatosensory evoked potential.
By contrast, findings related to VEP amplitude were markedly heterogeneous. Interestingly, when migraine subtypes were compared, MA more consistently exhibited higher VEP amplitudes than MO during the interictal phase. This differential pattern, while modest in isolated comparisons with HCs, becomes more salient when dissected by migraine subtype (17,26,27,30,33). Notably, hyperresponsivity in MA appeared to include a lateralized component, as suggested by interhemispheric asymmetries in VEP and BAEP responses (13,20,21,54). Despite limited number of studies, the present findings align with converging evidence from electrophysiology, neuroimaging and metabolic studies supporting the notion that MA may exhibit distinct neurobiological signatures.
Although cortical spreading depression (CSD) remains the most widely accepted mechanism underlying migraine aura, the precise pathophysiological cascade in MA is still not fully elucidated. Perfusion imaging studies supported the CSD model by demonstrating transient, retinotopically organized hypoperfusion that can occur not only during aura, but also interictally spreading across the occipital cortex in a pattern consistent with the spatiotemporal dynamics of CSD (66–69). In line with this, interictal studies using visual stimulus–evoked BOLD (i.e. blood oxygenation level dependent) functional magnetic resonance imaging (fMRI) revealed amplified occipital and extended visual network responses, indicative of trait-like cortical hyperresponsivity distinguishing MA from MO (70–72). fMRI findings extend this interictal vascular signature; as compared to MO, MA is characterized by a distinct network configuration. This includes stronger within-visual connectivity and altered coupling between intra visual cortical areas, visual cortex-extrastriata regions (including salience network hubs) and higher-order control systems, as well as thalamic and brainstem recruitment during visual stimulation between the attacks (66,72–75). Structural neuroimaging implicates reduced cortical thickness and more extensive white-matter alterations, while magnetic resonance spectroscopy studies showed decreased cortical N-acetyl aspartate, increased occipital lactate particularly in MA group (72,76–80). A prevailing hypothesis is that these findings are consistent with mitochondrial metabolic inefficiency that may lower the threshold for cortical hyperexcitability and sensory hyperreactivity may offering a plausible mechanism for the heightened occipital excitability consistently observed in MA (66,76,77,81).
Electrophysiological studies revealed a more consistent pattern in MA patients during the interictal phase, characterized by increased posterior alpha power, interhemispheric asymmetries, increased delta and theta wave occurrence (82–88). Accompanied by disrupted alpha–beta synchronization during visual stimulation, findings were pointing to reduced intracortical inhibition and altered long-range connectivity in MA group (82,84,86–88).
TMS data further support these distinctions, with MA patients showing shortened cortical silent period suggesting GABAergic hypofunction and lower thresholds for evoking magnetophosphenes indicating cortical hypersensitivity (85,89–91). Furthermore, paradoxical responses to excitatory/inhibitory protocols and impaired habituation of nociception-specific blink reflexes have been reported (82,89–93). By contrast, these abnormalities were less consistently observed in MO, suggesting that MA is associated with more pronounced alterations in cortical excitability, hypersynchronization and sensory adaptation (91,94). Collectively, these findings align with the hypothesis that MA represents a more functionally disrupted phenotype, underpinned by disrupted inhibitory control and altered sensory integration mechanisms.
Diving on the aura subtypes, Coppola et al. (12) opened a new discussion point, as they reported a sustained increase in VEP amplitude specifically in MA with complex aura than visual aura, indicating a genuine excitability shift. Supporting this, Mastria et al. (95) reported that complex aura phenomena, such as those seen in Alice in Wonderland syndrome, may be associated with more pronounced alterations in cortical excitability and functional connectivity compared to typical aura presentations. This was further supported by functional and structural MRI findings which demonstrated that complex aura is associated with widespread network dysfunctions and reduced cortical thickness extending beyond the visual cortex (78).
Although EP studies exploring distinctions between EM and CM remain limited, we believe preliminary findings are suggestive. Our data revealed increased amplitudes in CM patients specifically within the SSEP and BAEP modalities, whereas the single VEP study in our dataset showed no significant differences between CM and EM groups (38,48,49,65). These findings suggest a sensory modality-specific alteration in CM, consistent with a pattern of enhanced cortical responsivity and sensory gating, particularly in pathways related to somatosensory and brainstem processing (96–98). Although Chen et al. (99) has reported abnormalities in VEPs – especially using magnetic paradigms, our results do not support an alteration in VEP components between CM and EM. Regarding habituation notably, some variability persists across studies: while Kalita et al. (49) reported SSEP reduced habituation in CM, Coppola et al. (48) reported a late and normal habituation in CM may resemble ictal-like patterns. Medication overuse headache may partially account for such variability, as it is known to modulate cortical excitability (100–102). Based on the results of our review, de Tommaso et al. (63) identified that, compared to EM, CM patients exhibited a broader spatial shift in P2 dipole localization, involving significant displacements along both x and z axes. While EM showed only lateral displacement, CM sources extended into more ventromedial cortical regions, including the rostral ACC and insula. In the broader literature, studies employing nociceptive paradigms -such as LEPs, contact heat-EPs- have highlighted abnormal amplitude–intensity scaling; moreover fMRI studies have demonstrated activation of within these same key pain-processing cortical networks in CM relative to EM (63,103–105). This distinction may suggest that CM is associated with deeper and more widespread reorganization within pain-related cortical networks, possibly reflecting cumulative effects of repeated migraine attacks and chronification mechanisms (63,97,103,104). The inferior displacement of cortical sources in CM may also reflect modulatory input from deeper subcortical structures such as the hypothalamus, which has been shown to exhibit increased functional connectivity with cortical networks in CM (105,106). These findings support the view of CM as a condition characterized by widespread cortical hyperexcitability and enhanced responsivity across both exteroceptive and nociceptive systems, reflecting increased involvement of the pain-related cortical matrix (97,99,102,103).
The main contribution of EPs to migraine pathophysiology is with interictal abnormalities which support migraine is a brain disorder not solely an ictal disease (107). These findings have broadened our understanding and opened new discussions on subcortical-cortical interactions, inhibitory control and cortical plasticity (108). EPs are inexpensive, scalable and mechanistically informative. Nonetheless, clinical translation remains limited at present due to substantial heterogeneity across studies in stimulation protocols, recording montages, analysis pipelines, and critical variables such as migraine phase, medication use and comorbidities, all of which reduce effect sizes and complicate meta-analytic synthesis. Neurophysiological findings support that migraine is a dynamic, phase-dependent disorder involving widespread network dysfunction across cortical, subcortical, and brainstem structures. Future studies should aim to dissect these mechanisms across migraine subtypes with stratification by clinical, genetic and neurophysiological phenotypes. Particular attention should be paid to hypothalamic and brainstem modulation their role in aura phase and chronification of the disease.
The review has limitations. We exclusively included studies that demonstrated reproducible results with a minimum of 20 patients to ensure basic clinical representativeness. While this pragmatic threshold helped stabilize data, it also led to the exclusion of studies with smaller sample sizes. Our review is further limited by small patient cohorts, heterogeneous methodologies, variability across migraine phases, and differences among migraine subtypes in the studies.
Conclusions
EP studies have consistently revealed abnormal sensory processing in migraine, most notably interictal habituation deficits. Although the findings on amplitudes remain heterogeneous, subtype comparisons indicate distinct electrophysiological patterns, with MA and CM showing alterations in cortical excitability. Future research should prioritize standardized methodologies, subtype-compared analyses and studies with larger cohorts.
Clinical implications
Habituation deficits are the most consistent abnormality differentiating migraine disease from HCs. Highlighted increased cortical excitability in MA from MO and CM from EM, which may indicate subtype-specific alterations in cortical responsivity, may inform tailored therapeutic strategies. Revealed consistent interictal EP abnormalities, supporting migraine as a disorder of cortical sensory processing and network dysfunction, beyond its characterization as a transient pain condition. This highlights the need for further research into the interictal migraine brain. Underscored the need for standardized EP protocols to improve diagnostic reliability and cross-study comparisons.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251398792 - Supplemental material for Evoked potential studies in migraine: A systematic review of neurophysiological patterns across migraine subtypes
Supplemental material, sj-docx-1-cep-10.1177_03331024251398792 for Evoked potential studies in migraine: A systematic review of neurophysiological patterns across migraine subtypes by Samiye Ulutas, Emel Ur Özçelik, Laura Gómez Dabó, Fleury Bolla, Nermin Tepe, Paul Yambao, Yu-Hsiang Ling, Li-Ling Hope Pan, Shuu-Jiun Wang and in Cephalalgia
Footnotes
Acknowledgments
We thank the International Headache Society (IHS) board for organizing the International Headache Academy of the International Headache Society (IHS-iHEAD) working group. L. L. H. Pan and S. J. Wang are supported by the Brain Research Center, National Yang Ming Chiao Tung University, from the Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education of Taiwan.
Author contributions
Laura Gómez Dabó, Fleury Bolla, Nermin Tepe and Paul Yambao contributed equally to the preparation of this manuscript.
Data availability
Search strategies, inclusion/exclusion criteria and screening records are available upon reasonable request from the corresponding author.
Declaration of conflicting interests
S. J. Wang has received honoraria as a moderator from AbbVie, Biogen, Eli-Lilly, Hava Biopharma and Pfizer, has received consulting fees from AbbVie, Eli-Lilly Taiwan, Percept Co. and Pfizer Taiwan, and has been the PI in trials sponsored by Eli-Lilly, Lundbeck and Novartis. He has received research grants from Taiwan branches of Eli Lilly, Novartis and Orient Europharma. These funders had no role in the design and conduct of the review; in the collection, management, analysis and interpretation of the data; in the preparation, review or approval of the manuscript; and in the decision to submit the manuscript for publication. LG-D salary has been partially funded by Río Hortega grant Acción Estratégica en Salud 2021–2023 from Instituto de Salud Carlos III (CM24/00072). This funding source had no role in the design of this study and had no role during its execution, analyses, interpretation of the data or decision to submit results.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
