Abstract
Aim:
The aim of this article is to generate hypotheses for the mechanism of pituitary adenoma headache.
Patients and methods:
Fifty-eight patients with pituitary adenoma were prospectively analysed for prevalence and manifestation of tumour headache. Intrapersonal and neoplasm-associated risk factors were assessed.
Results:
Twenty-four patients (41%) had tumour-attributed headache, seven had migraine-like, 11 tension-type headache-like headache, and three both. Cluster headache-like headache was found once, and two headaches remained unclassified. Tumour-attributed headache was associated with a positive history of headache (p = 0.03; OR 3.4), nicotine abuse (p < 0.01; OR 4.7), intake of acute headache medication (p = 0.04; OR 3.3), and a higher tumour proliferation indicated by a Ki67-labelling index (LI) >3% (p = 0.02; OR 11.0). For patients with migraine-like tumour-attributed headache, risk factors were younger age (p = 0.02), nicotine abuse (p < 0.01; OR 10.9), acute headache treatment (p < 0.01; OR 9.0), and Ki67-LI >3% (p = 0.03; OR 14.1). For tension-type headache-like headache, the main risk factor was a positive history of tension-type headache (p = 0.045; OR 5.6).
Conclusion:
Headache predisposition and local tumour effects might be important for the pathophysiology of pituitary adenoma headache and tumour headache in general.
Introduction
Headache can be a symptom of intracranial neoplasms. Although its pathophysiology is still unknown, the International Classification of Headache Disorders (ICHD-II) (1) suggests that two mechanisms might be responsible for the development of tumour headache, namely an ‘elevation of intracranial pressure’ (ICP, coded as IHS 7.4.1) or a ‘direct tumour influence’ (IHS 7.4.2). ‘Direct tumour influence’ was defined by its clinical manifestation by aggravation during horizontal posture (i.e. worsening in the morning) or when bending forward or coughing. Since these aggravating factors are associated with an increase of ICP, the differentiation between IHS 7.4.1 and IHS 7.4.2 seems to be obsolete because of the identical mechanism ‘elevated ICP’.
In the past, multiple studies demonstrated that tumour headache resembles primary headaches, such as tension-type headache (TTH) or migraine, and is rarely associated with typical symptoms of increased ICP (2–4). In general, the activation of trigeminal meningeal afferents is thought to be important for the pathophysiology of headache (5). Different thresholds for such an activation might be based on an individual predisposition and may be the reason why the history of primary headaches is a risk factor for the occurrence of headache in association with brain tumours in general (2,3) or pituitary adenoma in particular (6,7).
In this prospective study, we aimed to identify additional pathophysiological mechanisms for tumour headache by analysing the tumour subgroup pituitary adenoma. We addressed which factors are associated with the occurrence of tumour headache. In a first step, we identified patients with tumour headache according to the criteria for secondary headaches listed in the ICHD-II (presence of post-operative amelioration). Second, we assessed which clinical or tumour-associated parameters were correlated with tumour headache.
Patients and methods
Study population
All patients with the clinical and radiological diagnosis of a pituitary adenoma who were seen for surgery at the Neurosurgery Department of the University of Munich, a tertiary-care centre for pituitary adenoma surgery, between June 2007 and February 2009, were asked to participate in this study. The study was approved by the Ethics Committee at the University of Munich (project no. 233-07). All patients gave their written informed consent. The decision to surgically treat the tumour had been made prior to admission based on the judgement of the surgeon and an endocrinologist. All parameters for data collection were pre-defined at the onset of the study. After admission and prior to surgery, we recorded personal details (age, sex, family history of headache or cardio-/cerebrovascular events) from each patient. They were asked to fill out a standardised questionnaire, which included information on pre- and post-operative symptoms with a focus on headache. We were interested in medical history, general medication, cardiovascular risk factors (arterial hypertension, hypercholesterinaemia, diabetes mellitus and nicotine abuse) and alcohol abuse. Each patient underwent a standardised neurological examination on the day of admission.
Patients were excluded post-operatively if the histological diagnosis was incompatible with a pituitary adenoma.
History of headache, pre-operative and pituitary adenoma-associated headache
History of headache (defined as a headache history of longer than five years before the occurrence of first symptoms of the adenoma) and pre-operative headache (significant headache in the last three months) were characterised using a self-administered headache questionnaire validated on a population basis for migraine, TTH and, to a limited extent, for trigeminal autonomic cephalgia (TAC) (8). Patients were instructed to distinguish between the history of headache and the pre-operative headache when answering each question. As recommended by the IHS, we separately coded each headache syndrome in a patient (1). For statistical purposes, headache was coded as either present or absent.
The patients had to state their pain treatment habits (pain medication and prophylaxis: substance, frequency). The diagnosis of tumour-associated headache was made in accordance with the ICHD-II criteria for ‘secondary headaches’. These cover the criteria for ‘headache attributed to intracranial neoplasm’ (IHS 7.4) except for criterion A and the time course of criterion D. The phenotype of the headache was left out since we also aimed to assess the clinical presentation of the headache. Further, we allowed a longer time for the post-operative amelioration since the exact time-course of pituitary adenoma headache improvement after surgery is not known. The presence of all three of the following criteria was therefore required:
The presence of pre-operative headache. The exclusion of medication overuse (MOH), which was defined according to the ICHD-II (1) as: headache on more than 15 days per month and intake of pain medication on more than 10 (triptans, opioids, combination analgesics) or 15 (other analgesics) days per month for more than three months. Presence of post-operative amelioration assessed three to 18 months after surgery by telephone interview and defined as relief of headache by at least 50% in respect of frequency or severity.
Endocrinological assessment
The endocrine activity of the tumour was characterised clinically according to the pre-operative laboratory values as non-secreting adenoma, prolactinoma, acromegaly, Cushing’s syndrome, TSHoma, LH/FSHoma, or hybrid forms.
Radiological assessment
All tumours were categorised as microadenoma (diameter <1 cm), macroadenoma (1–4 cm) or giant adenoma (>4 cm). If clinically necessary, patients underwent magnetic resonance imaging (MRI) either on a 1.5 Tesla (Symphony, Siemens Medical Solutions, Erlangen, Germany) or on a 3 Tesla MR scanner (Signa HDx, GE Healthcare, Solingen, Germany). On the 1.5 Tesla MR the following sequences were performed: pre-contrast axial and coronal T1-weighted images (slice thickness: 3 mm), and a coronal T2-weighted sequence (slice thickness: 3 mm). The protocol performed on the 3 Tesla scanner included in addition a contrast-enhanced 3D-FIESTA-C (slice thickness: 0.6 mm), and a contrast-enhanced MR angiography (slice thickness: 0.4 mm). Analysis of adenoma volume was done using the open-source DICOM viewer OsiriX Imaging software (9). First, the area of the tumour was determined by manually encircling the adenoma on each T1-weighted contrast-enhanced axial image. To calculate tumour volume, the sum of the tumour area of the slices was then multiplied by the respective slice thickness.
Cavernous sinus invasion was assessed as follows: purely intrasellar, at least invasion of the cavernous sinus, at least contact with the internal carotid artery (ICA), at least partial or complete embracing of the ICA.
Some patients presented prior to surgery with outpatient non-electronic brain imaging. In these patients, cavernous sinus invasion and tumour volume could not be assessed.
Histological assessment of endocrine activity, proliferation status and invasiveness
The neuropathological diagnosis was obtained by the Center for Prion Disease and Neuropathology of the University of Munich during daily clinical routine. Depending on the immunohistological staining, the diagnosis was specified as adenoma without abnormal hormonal expression (no over-expression), expression of prolactin (PRL), growth hormone (hGH), corticotropin (ACTH), thyrotropin (TSH), gonadotropin (LH/FSH), multiple hormones, or normal pituitary tissue (no adenoma tissue). We determined proliferation status by using Ki67-labelling index (LI, low: <3%, or high: >3%) (10) and assessed p53 immunoreactivity (low: <1%, or high: >1%) as a marker for aggressive growth behaviour (11). The antibody Ki67 recognises proliferating cells during nearly all phases of the cell cycle, but not during the G0-phase (12). The Ki67-LI represents the percentage of positive nuclei and thus is elevated in highly proliferative tumour tissues.
Statistical analysis
Statistical analysis of clinical data was performed using SPSS 17.0 for Windows (SPSS Inc., Chicago, IL, USA). Categorical variables were analysed using Chi square or Fisher’s exact test (in case of expected values <5). We used the two-sample t test for continuous variables to identify significant group differences. Data are presented as means ± standard deviation (SD), and, if appropriate, as odds ratio (OR) and as 95% confidence interval (CI).
Results
Study population and characteristics of the pituitary adenomas
Characteristics of the study population (N = 58) including tumour-attributed headache (n = 24).

SD: standard deviation; NA: not applicable.
Characteristics of the pituitary adenomas (N = 58). Tumour volume could be assessed in 44 patients.
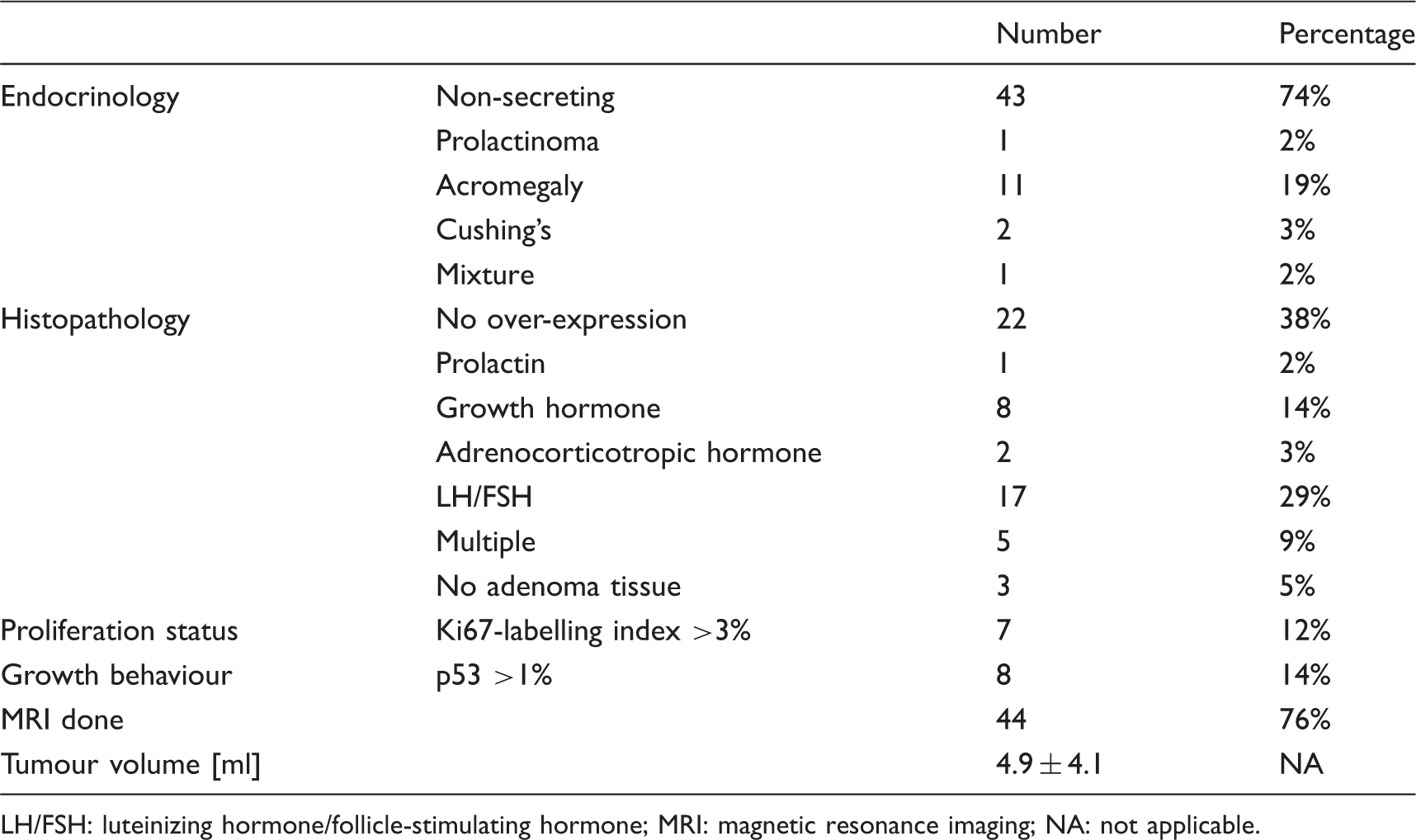
LH/FSH: luteinizing hormone/follicle-stimulating hormone; MRI: magnetic resonance imaging; NA: not applicable.
Prevalence of pituitary adenoma-attributed headache
Table 1 lists 33 patients with pre-operative headache. Of these, three had MOH and six had no post-operative amelioration, leaving 24 patients with pituitary adenoma-attributed headache (prevalence 41%).
In the 24 patients with pituitary adenoma-attributed headache, the self-administered questionnaire assigned the diagnosis of migraine-like headache to seven (29%), TTH-like headache to 11 (46%) and both migraine-like and TTH-like headache to three (13%). Ten of the 24 patients with tumour headache did not have a history of headache. Of the remaining 14 patients, comparison with the history of headache revealed a change in phenotype in four (either from migraine to TTH or vice versa), whereas the phenotype was unchanged in 10 patients (data not shown). Among the patients with either migraine-like headache or migraine-like and TTH-like headache, migraine with aura was found in five patients (data not shown). The headache of one patient was classified as trigeminal autonomic cephalgia (cluster headache-like; this patient had a history of TTH), and two remained unclassified. Of the unclassified headaches, one was a purely unilateral headache, but without any features of migraine or trigeminal autonomic cephalgia. This patient has never tried indomethacin to rule out hemicrania continua. The other unclassified headache was bilateral, dull and featureless, but interfered with routine daily activity and thus did not fit with TTH. The patient with the history of trigeminal autonomic cephalgia had tumour-attributed migraine with aura prior to surgery.
Statistical analysis
Univariate analysis for tumour-attributed headache assessed by Chi square, Fisher’s exact test, or two sample t test (ap < 0.05).
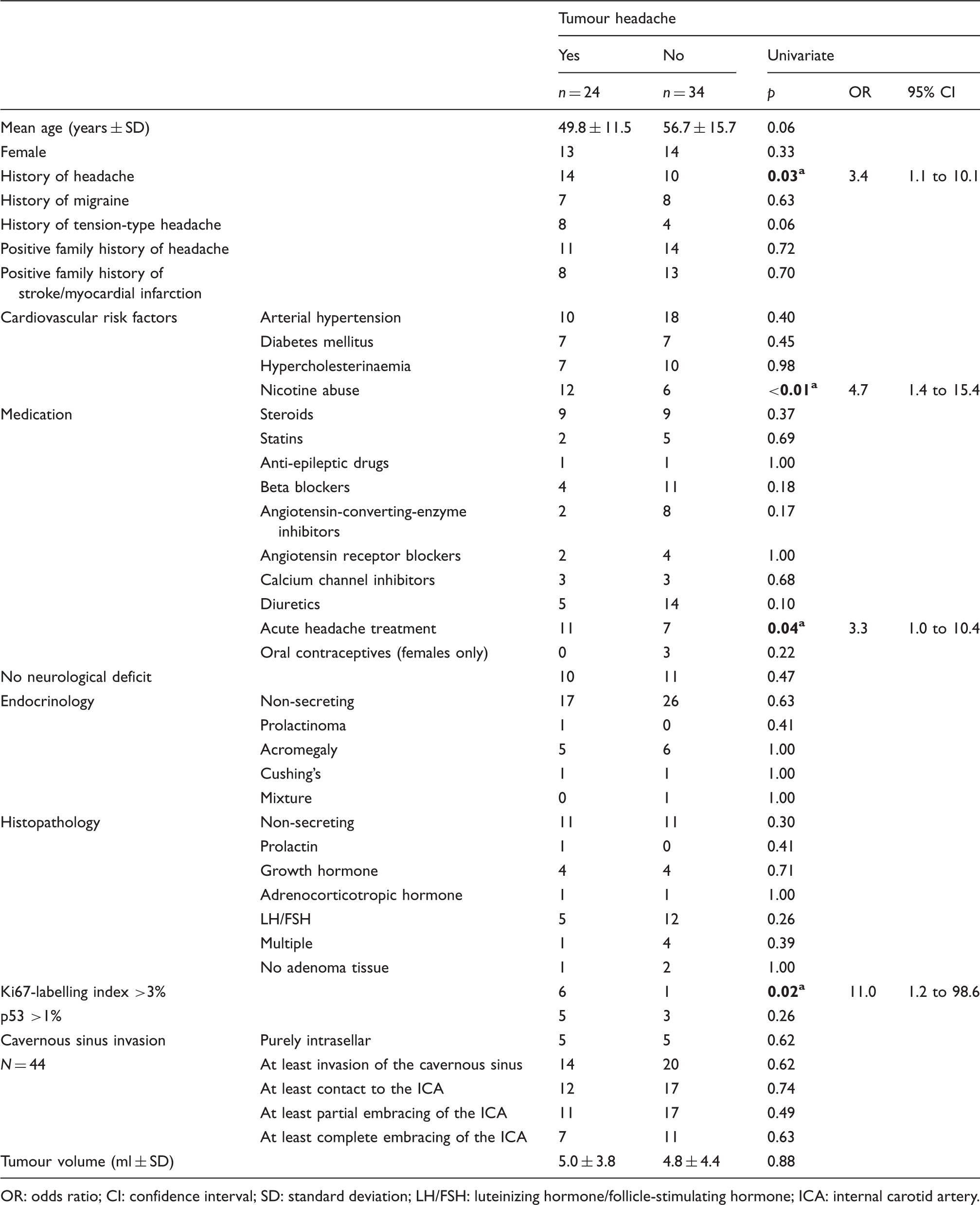
OR: odds ratio; CI: confidence interval; SD: standard deviation; LH/FSH: luteinizing hormone/follicle-stimulating hormone; ICA: internal carotid artery.
Univariate analysis of the intrapersonal and tumour-associated risk factors for the subgroups migraine-like and tension-type headache-like tumour headache assessed by Chi square, Fisher’s exact test, or two sample t test (ap < 0.05, only relevant data shown).
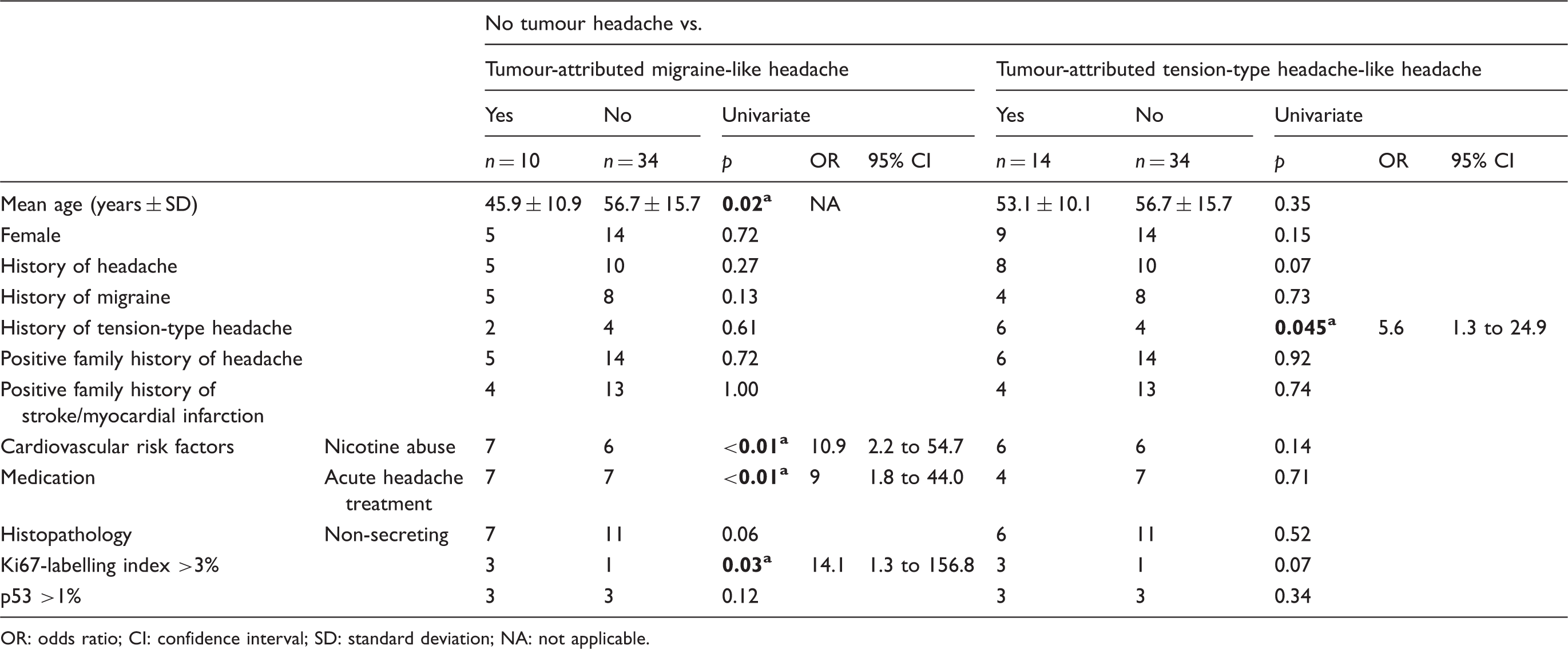
OR: odds ratio; CI: confidence interval; SD: standard deviation; NA: not applicable.
No association was found for all other parameters listed in Table 3. These values were not included in Table 4 for the purpose of clarity.
Discussion
The prevalence of migraine-like tumour headache (17% or 10 of 58 patients) in our patients with pituitary adenoma was slightly higher than the reported prevalence for migraine in the general population (10% in (13), 6.8% in (14)), whereas the prevalence of TTH-like tumour headache (24% or 14 of 58 patients in our study) was similar to the prevalence of TTH in the general population (19.9% in (14)). According to the questionnaire, one patient had a history of migraine and TAC, another had TAC-like tumour headache. Due to the low specificity of the questionnaire for TACs (8), this has to be interpreted with caution and will not be discussed here further.
In our study 33 patients complained about headache prior to surgery; 24 (73%) of those had tumour headache. In other words, 73% of patients with headache and pituitary adenoma improved after surgery, since tumour headache was defined by post-operative amelioration of at least 50%. A limitation of our study is that the amelioration was not objectively documented by a headache diary pre- and post-operatively. However, it could be speculated that patients with severe headache and pituitary adenoma might profit from surgery when medical headache treatment fails. This is supported by a recent retrospective study of 41 patients with small pituitary lesions. Fifteen had nonfunctioning pituitary microadenoma with intractable chronic headaches. Post-operative improvement of headache was found in 12 (i.e. 80%) (15) patients. A placebo effect from surgery has to be kept in mind, which is particularly high when more invasive treatments are performed (16). However, it has to be noted that the placebo effect also largely depends on a high expectation (17). Our patients were admitted for surgical treatment of a pituitary adenoma, and they were not told that this surgical procedure would improve the headache. Thus it is more likely that patients had a high expectation in respect to treatment of their pituitary adenoma and not of their headache. The long time frame of up to 18 months for the assessment of post-operative amelioration has to be kept in mind since it might have resulted in a higher prevalence of tumour headache because of improvement due to natural time course or regression to the mean instead of true improvement by surgery.
A positive history of headache was found to be a risk factor for the occurrence of headache in the presence of a pituitary adenoma. Tumour headache was more likely to be migraine-like if there was a history of migraine (tendency), but more likely TTH like in patients with TTH in the past. The definition of headache history requiring duration of longer than five years was chosen pragmatically since it is retrospectively not possible to assess how long a patient had a pituitary adenoma. As about 50% of macroadenomas show significant growth leading to surgery within five years (18), one would expect that these tumours would have been smaller five years prior to surgery with fewer symptoms and presumably less tumour-attributed headache. Our finding is consistent with studies on headache after craniotomy (19), and in the presence of a meningioma (20) or of brain tumours in general (2–4). Together with the finding of Levy et al. (6), who have shown a correlation of a positive family history of headache with pituitary adenoma-associated headache, this points to a shared pathophysiological mechanism of secondary and primary headaches. Possible connections between primary and secondary headaches might be the mechanisms by which trigeminal fibres of the meninges are activated. In other words, the tumour might act as a headache trigger in predisposed patients with primary headache in the past. In addition, the tumour itself might be responsible for both susceptibility to headache and trigger.
According to our data, mechanisms, which predispose toward or trigger tumour-attributed headache, might include local tumour effects or lifestyle factors, such as smoking. In contrast, systemic endocrine processes (hormonal activity or inactivity, acromegaly, prolactinoma, Cushing’s syndrome), or cavernous sinus invasion might be of less relevance.
Mechanical factors
As already shown by others (6), we were not able to demonstrate that the occurrence of headache itself depends on the size of the neoplasm or the presence of cavernous sinus invasion. Although this contradicts the recent findings by Gondim et al. (21), we could speculate that cavernous sinus invasion is less important for headache generation than previously thought, and that alterations within the sella could be more relevant. Wang et al. (22) have shown that the side of cranial autonomic symptoms in patients with pituitary adenoma and headache is concordant with both the side of the tumour and the headache in case of unilateral headaches. Therefore, cavernous sinus invasion might be more important for the laterality of the symptoms and less important for the presence or absence of headache.
Histologically, Ki67-LI >3% was an additional risk factor. The antibody Ki67 recognises proliferating cells (12). In our study, headache is therefore associated with highly proliferative tumour tissue. Thus, a lack of adaptation to a growing mass could elevate intrasellar pressure, which might account for headache. This is consistent with a study demonstrating higher intrasellar pressure in macroadenomas confined to the sella than in cases of extrasellar extension (23). In another study, intrasellar pressure was shown to correlate with headache (24). Thus, even small intrasellar tumours may lead to increased intrasellar pressure, which might result in headache. Contrasting migraine-like with TTH-like pituitary adenoma-associated headache, Ki67-LI as a marker for the velocity of growth was found to be a risk factor only for migraine-like headache. The main clinical features differentiating migraine and TTH are severity of the pain and interference with daily routine activity. Higher intrasellar pressure therefore might be responsible for a more severe pain level. Since we did not measure intrasellar pressure, confirmation of this hypothesis requires further prospective studies with invasive measurement of intrasellar pressure.
Another risk factor for migraine-like headache was younger age, whereas this was not relevant for TTH-like headache. Similarly, Valentinis et al. (4) found that headache was more likely to be the presenting symptom of a brain tumour in younger patients than in the elderly, probably because of generalised brain atrophy in the latter, which might also affect the pituitary gland. Alternatively, the predisposition to migraine-like tumour headache might decrease with age as migraine prevalence does (25), resulting in a reduced susceptibility to a triggering pituitary adenoma.
Smoking
In our study, nicotine abuse was significantly more frequent in patients with tumour headache and its subgroup with migraine-like headache than in the respective non-headache group. Similarly, patients with headache in general (26), with cluster headache (27) and with migraine (28) are more often smokers compared to controls without headache. The relationship between smoking and headache is controversial (28). A possible mechanism of headache in smokers with pituitary adenoma might be based on the central effects of nicotine: Acute exposure to nicotine results in an increase of ACTH, PRL (29) and hGH (30) and in an increase of sleep-onset latency (31). In rats, this influence on the hypothalamus-pituitary adrenal axis and on the control of endogenous rhythms and sleep architecture is mediated by brainstem catecholaminergic neurons with a subsequent noradrenaline release in the hypothalamic paraventricular nucleus (32). Functional imaging studies demonstrated a specific activation of these regions in headache (e.g. dorsal pons in migraine (33,34)). An altered influence of nicotine on the hypothalamus or the brainstem nuclei due to the presence of a pituitary adenoma might provide a pathophysiological link between pituitary adenoma-associated headache and smoking.
Systemic endocrine factors
In our prospective study, we could not demonstrate a correlation between endocrinological state and tumour headache. Although numerous case reports exist on headache in patients with a prolactinoma (35,36) or in patients with acromegaly (37,38), no prospective clinical study has demonstrated a correlation between tumour headache and endocrine function of the adenoma.
Limitation
One major limitation of the study is the limited size of the cohort, which reduces the statistical strength of our findings. The prevalence of headache attributed to a pituitary adenoma (41%) was lower than in the studies by Levy et al. (6) and Gondim et al. (21), who demonstrated headache in 70% and 60% of patients, respectively. This might be because our patients were pre-selected for surgical therapy, whereas the patients in Levy et al. were seen at a hospital unit offering both surgical and medical treatment options. The patients in Gondim et al. were included from an endocrinological setting. This is consistent with the data from Abe et al. (39), who also assessed patients prior to surgery and found a similar prevalence to ours (37%). The frequency of prolactinomas was markedly lower in our study (2%) than in the populations presented by Levy et al. (27%) and Gondim et al. (18.8%), whereas we had more non-secreting adenomas (74% vs. 32% and 47%). Similarly, the tumours from our patients (only 9% microadenomas) were larger than those from Gondim et al. (19%). This relatively low frequency of prolactinomas with essential larger tumours has to be kept in mind for the discussion of our findings. Similarly, a cross-sectional study from Belgium identified 68 patients with clinically relevant pituitary adenomas out of a population of 71,972. Of these, 55.9% underwent surgery. Thus, our findings correspond primarily to these two thirds of patients with surgical treatment, and not to the patients with conservative medical treatment or observant therapy (40).
Further, this was a cross-sectional study asking for history of headache and pre-operative headache by using the same questions. This might bear the risk that patients tended to fill in similar answers for both history of headache and pre-operative headache. Such pre-occupation might result in a high frequency of migraine-like or TTH-like pre-operative headache in patients with history of migraine or TTH.
Conclusion
In the majority of cases, pituitary adenoma-attributed headache was migraine-like or TTH-like. This and the elevated risk for patients with history of headache point to a shared pathophysiological mechanism of primary and tumour headaches. The tumour might be a trigger of headache in predisposed patients or might be both a predisposing and a trigger factor. Mechanisms of tumour headache might include the following: (i) Mechanically, a high proliferation rate of intrasellar adenomas might be important owing to increased intrasellar pressure. In contrast, no evidence was found for a significant role of tumour size or cavernous sinus invasion. (ii) Currently, the relevance of a systemic endocrine effect of the tumour remains unclear. Smoking, however, is a risk factor for tumour headache, probably due to an endocrine effect or an effect on endogenous rhythm generators.
Footnotes
Clinical implications
Headache in patients with pituitary adenoma is common. Main risk factors for the occurrence of headache in patients with pituitary adenoma are: history of headache, nicotine abuse, younger age and high tumour proliferation rate. In patients with pre-existing primary headaches, pituitary adenoma headache resembles phenotypically the previous headache. This points to some pathophysiological overlap of primary headaches and tumour headaches.
Funding
The study was supported by a grant from the BMBF (Chronic headache, project D1), and the Förderprogramm für Forschung und Lehre (FöFoLe, reg.-no. 561 to C.J.S.) of the University of Munich.
Acknowledgements
We thank Ms. K. Ogston for her help in preparing the manuscript.
Conflict of interest
No conflicts of interest were declared.
