Abstract
Background: The rate of illicit drug use in cluster headache (CH) patients is unknown.
Methods: Two hundred and ten CH patients (162 males and 48 females) attending two headache clinics provided information about their lifetime use (once or more in their lifetime, LTU), recent use (once or more in the past year, RU), and current use (once or more in the past 30 days, CU) of illicit drugs. General population data (IPSAD®Italia2007–2008) served as the control group.
Results: LTU of each illicit drug but hallucinogens, RU of cannabis, cocaine, amphetamines and ecstasy, and CU of cannabis and cocaine were significantly higher in the male CH patients than in the general population, whereas no difference was found between the CH women and the controls. In the CH group, 28.5% of patients reported having used illicit drugs for the first time after CH onset and 71.5% before CH onset. Compared with the controls, the male CH group showed a greater prevalence both of lifetime sustained intensive use of any illicit drug and of current intensive use of cannabis.
Conclusion: The results of this study indicate that male CH patients are prone to overindulge in illicit drug use. This finding possibly reflects a common biological susceptibility that predisposes these subjects to CH and to addictive behaviour.
Introduction
Investigation of psychiatric and psychological comorbidities in headache has focused far more on migraine and tension-type headache than on cluster headache (CH). High-quality studies in this field (that is, studies including adequate CH patient and control groups and using specific psychometric instruments or indicators) are lacking (1). Most of the hypotheses on personality traits, psychological features and coping style in CH patients, such as those which suggest that these individuals are characterised by impulsiveness, aggressive behaviour, emotional ability, anxiety and poor social contacts, have not been validated by solid observational data (1–6).
To date, the abuse of tobacco in most CH patients is the most consistent finding in this population. This observation was documented by Kudrow in the early 1970s (7) in a small group of CH patients and subsequently confirmed by other groups (8,9). More recently, Manzoni investigated a large cohort – 340 subjects – of male CH patients and confirmed a very high percentage of smokers among people with this type of headache: 78.9% vs 35% in the general population (POP), with chronic CH patients appearing more inclined to smoke than episodic CH sufferers (10). The high prevalence of chronic use of nicotine in CH has also been confirmed in more recent studies, including non-clinic-based populations of CH patients in different countries (11–13). The significance of this association as regards the pathophysiology of CH is unclear, with some authors considering nicotine a potential trigger factor and others hypothesising the existence of a common genetic link predisposing people to both CH and nicotine addiction (12,14).
Data on CH patients’ use of other non-illicit drugs are more controversial than those relating to their smoking. In Manzoni’s study, CH patients significantly overused coffee and alcohol, too, which suggests that they have a tendency to overindulge in certain lifestyle behaviours (10). However, in a recent study by Schürks et al., conducted in Germany in a large clinic- and non-clinic-based CH population, the CH patients proved less likely than the general population to show hazardous drinking behaviour (15). Since alcohol is a well-known trigger of CH attacks during the active periods of the disease, these findings may indicate the presence of alcohol-avoidance behaviour in CH patients with frequent attacks.
In a clinical descriptive study of chronic CH patients, Donnet et al. reported that 26% of these patients were regular cannabis consumers (16). However, the use of illicit drugs in CH patients has never been investigated systematically. Acquisition of data on the use of illicit drugs in CH patients may be important in order to ascertain whether these patients are prone to overindulge in addictive behaviours.
The aim of this study was to examine the rate of illicit drug use in a clinic-based sample of CH patients compared, in the same period of time, with POP data.
Patients and methods
This cross-sectional study included a primary well-characterised study sample formed by CH patients and POP data serving as comparison data.
Patient study sample
Consecutive patients aged between 18 and 65 years, with CH diagnosed according to the International Classification of Headache Disorders and attending two headache centres (INI Grottaferrata and IRCCS C. Mondino, Pavia) for a first or follow-up visit were evaluated over an 18-month period (January 2007–June 2008). Non-Italian patients were included only if they had lived in Italy since before the age of 10 years. No other specific inclusion criteria relating to either clinical picture or socio-demographic profile were applied, and we thus recruited all patients able to provide reliable information about their headache history and undergo other assessments relevant to the protocol. A total of 210 CH patients were recruited; the group included 162 males and 48 females.
All the patients gave their written informed consent to their participation in the study, which had been approved by the local ethics committee.
The POP comparison data: the Italian Population Survey on Alcohol and Drugs (IPSAD)
The POP data used in this study were obtained from the IPSAD®Italia 2007–2008. IPSAD is a yearly survey monitoring alcohol, tobacco and drug consumption in the general population and is consistent, in terms of methods, with that carried out by the European Monitoring Centre for Drugs and Drug Addiction – EMCDDA – in several EU countries (17). The IPSAD®Italia 2007–2008 sample was formed by random selection of census data held by local councils involved in the survey (18). The councils were selected to ensure that all Italian regions and provinces were represented. The youngest age-classes were oversampled (15–19 years, 5 × 1000, 20–25 years, 4.5 × 1000, 25–29 years, 3 × 1000, 30–34 years, 2.5 × 1000, 35–39 years and 40–44 years 2 × 1000, 45–49 years and 50–54 years, 1.5 × 1000, 55–59 years and 60–64 years, 1 × 1000). Participation in the survey was voluntary, anonymous and at no cost to the interviewee (the questionnaires investigating illicit drug use and other variables were returned in pre-paid envelopes). The study had a responder rate of 35.1% with the final sample numbering 10,940 subjects, including the 4757 males and 6183 females comprising the POP group in our study.
Demographic characteristics of cluster headache (CH) patients and the general population sample (POP).

§: Chi-square test with Yates’ correction; *: Fisher’s exact test.
The comparison data were obtained from open-access publications (18) and, partly, from the National Council of Research, Epidemiologic Unit, Pisa.
In Italy the use of cannabis, cocaine and opioids is higher than the European average, especially in young age classes, although it is similar to that observed in the EU countries that are comparable to Italy for number of inhabitants and other demographic and economic variables (i.e. Spain, France, Germany and UK (18). In contrast, the use of stimulants is lower than the European average (18).
Data collection and definitions
Comprehensive information regarding socio-demographic status and smoking was obtained directly from each patient. Information regarding headache characteristics was obtained from each patient’s medical history and included CH subtype diagnosis (episodic cluster headache (ECH) or chronic cluster headache (CCH)), disease duration, age at onset of CH, mean bout frequency, mean bout duration and mean number of attacks per day.
Information regarding illicit drug use was obtained by means of a short questionnaire filled in anonymously and voluntarily by the patients at the end of the visit.
The questionnaire included the EMCDDA core items for assessing period prevalence of drug use in the general population (17). In detail, it included questions about the patients’ use during the previous month (last-30-days’ prevalence, often called current use (CU)), during the previous year (last-12-months’ prevalence, often called recent use (RU)), and during their lifetime (that is, any use during their lifetime (LTU)) of several substances (cannabis, opioids, cocaine, ecstasy, amphetamines and hallucinogens). In accordance with the EMCDDA guidelines, for each drug two basic items (age at first use and frequency of use in the past 30 days) were included to investigate patterns of use, which provide valuable insight into the incidence and intensity of use and their correlates. Data regarding the frequency of use in the previous 30 days were used to identify current intensive users, defined as patients who, in the previous 30 days, had used cannabis for 20 days or more, or one or more of the other drugs for 10 days or more. Moreover, in order to have, for each drug, an additional measure of lifetime intensity of use, we asked responders also to specify how many years they had used the drug(s) intensively in their lifetime. Patients presenting with intensive use of any illicit drug for at least six months in their lifetime were defined as sustained intensive users (SIUs).
Statistical analysis
Descriptive statistics (means, standard deviations (SD) and proportions) were used to describe each variable. Bivariate comparisons of groups were performed using the Chi-square test and Fisher’s test (where applicable) for categorical variables, the t-test for continuous variables. The analyses were sex-matched for all comparisons.
In order to find possible predictors of illicit drug use in CH patients, a binary logistic regression analysis was adopted. Three independent analyses were performed by a backward conditional method to predict the LTU, the RU and the CU of at least one of the illicit drugs. In each model, as possible predictors we considered age, gender, educational level, occupational status (student/employed/unemployed), economic income, age at cluster onset, type of cluster (episodic/chronic) and a measure of CH severity.
As a measure of severity, we considered the mean number of headache attacks per year of disease when analysing LTU; the number of CH attacks in the last 12 months when analysing RU; and the number of attacks in the last month when analysing CU.
A level of p < 0.05 was considered significant. Analysis was performed using SPSS for Windows, version 11 (SPSS, Inc, Chicago, IL).
Results
Study population
Of the 224 CH patients recruited during the study period, 210 (93.7%) agreed to participate in the study (114 came from INI and 96 from the Pavia centre). All but two were Caucasians and 98.1% were Italian. Almost 83% of the patients had ECH, 13.4% had CCH (females 10.8%, males 19.6%, Fisher’s test p > 0.05) and 4% were newly diagnosed at the time of the study (follow-up visits subsequently established a diagnosis of ECH in all these cases). Ninety-one per cent of patients were recruited during an active phase, 82.7% at a follow-up visit and 17.3% at a first visit. The mean age at CH onset was 27.5 ± 9.1 years (males 27.8 ± 9.1, females 26.5 ± 7.4, p > 0.05) and the mean age at study enrolment was 41 years ± 10.8 (males 41.3 ± 11.2, females 40.8 ± 9.4, p > 0.05). The ECH patients recorded a mean bout frequency of 1.05 ± 0.7 per year (males 1.2 ± 0.58, females 0.92 ± 0.4, t = 11.3, p < .005); their mean number of attacks per day was 2.15 ± 1.3 (males 2.16 ± 1.49, females 1.96 ± 1.54, p > 0.05), and their mean bout duration was 51.3 ± 24 days (males 54.6 ± 39.7, females 41.3 ± 24, t = 2.1, p < 0.05).
Compared with the POP group, the male CH group included a significantly lower proportion of patients in the age range 15–24 years (12.3% vs 32.2%, Chi-square = 28.1, p < 0.0001) and a significantly higher proportion in the age group 35–44 years (30.8% vs 22.1%, Chi-square = 4.05, p < 0.05).
Compared with the POP group, the female CH group included a significantly lower proportion of patients in the age range 15–24 years (Fisher’s test, 13%, vs 33%, p < 0.05) and a significantly higher proportion in the age group 55–64 years (24% vs 6.2%, Chi-square = 18.1, p < 0.01). The CH patients included a significantly higher proportion of divorcees compared with the POP group (males 6.7% vs 1.4%, Chi-square = 19.9, p < 0.0001, females 15.3% vs 1.6%, Chi-square = 41.3, p < 0.001).
Prevalence of lifetime use (LTU) of illicit drugs in cluster headache (CH) patients and the sex-matched general population (POP).

Chi-square test with Yates’ correction; *: Fisher’s exact test.
CU of tobacco was significantly higher in the CH than in the POP subjects, males (80.2% vs 31.9%, Chi-square = 209, p < 0.0001) and females (54.3% vs 28.7%, Chi-square = 14.8, p < 0.001).
LTU of illicit drugs
LTU of each of the illicit drugs surveyed but hallucinogens was significantly higher in the CH males than in the POP group (Table 2, cannabis 55.5% vs 36.6%, Chi-square = 27.1, p < 0.0001; opioids 5.5% vs 2.3%, Chi-square = 5.67, p = 0.01; cocaine 24.1 vs 8.7%, Chi-square = 39.1, p < 0.001; amphetamines 12.9% vs 3.7%, Chi-square = 35, p = 0.0001; ecstasy 13.5% vs 4.2%, Chi-square = 32, p = 0.0001). Conversely, in the CH females, no significant differences in LTU of any illicit drug were found (Table 2).
RU of illicit drugs
Prevalence of recent use (RU, previous 12 months) of illicit drugs in cluster headache (CH) patients and in the sex-matched general population (POP).
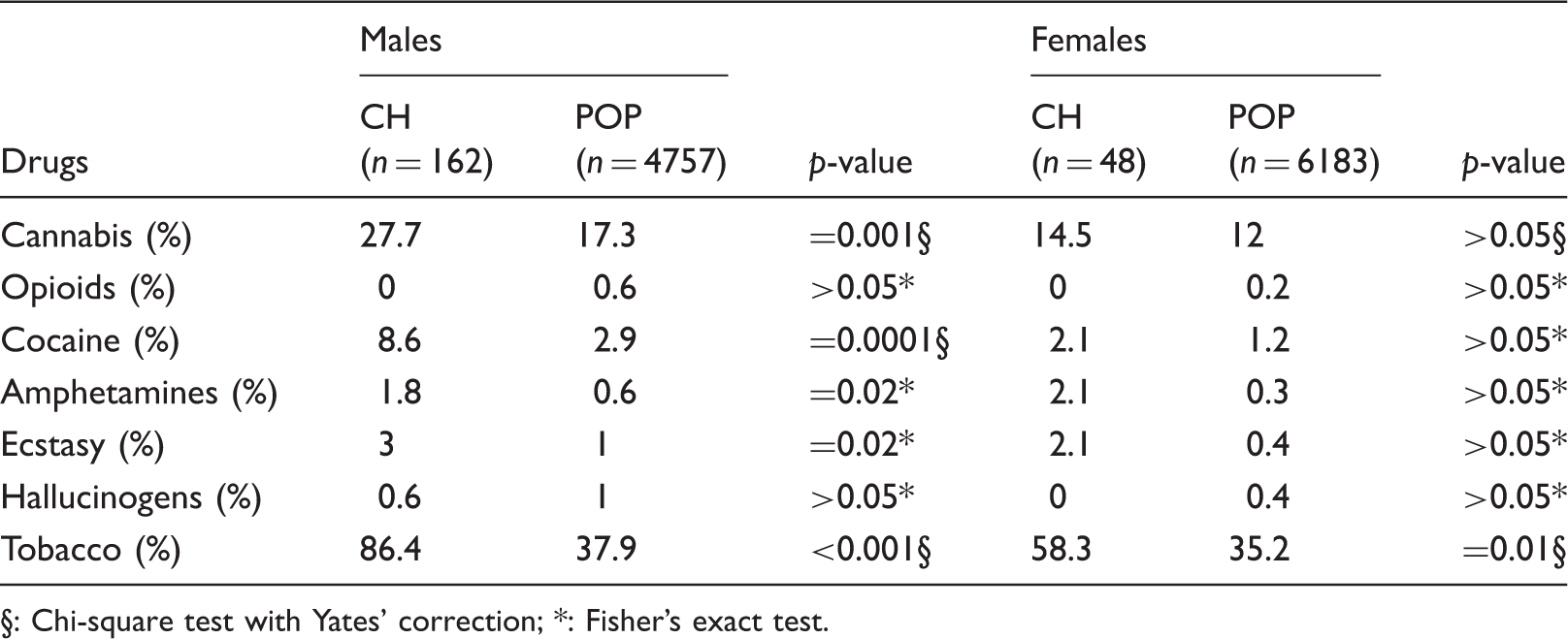
Chi-square test with Yates’ correction; *: Fisher’s exact test.
In the CH females, no significant differences in RU of any illicit drug were found (Table 3).
CU of illicit drugs
Prevalence of current use (CU, previous 30 days) of illicit drugs in cluster headache (CH) patients and in the sex-matched general population (POP).

Chi-square test with Yates’ correction; *: Fisher’s exact test.
Recent poly-drug use
Data regarding poly-drug use (that is, the use of more than one drug or type of drug by an individual) consumed at the same time or sequentially (17) from the POP were available only for recent use of cannabis, cocaine and heroin. Simultaneous recent use of cannabis and cocaine was significantly higher in CH males than in the POP (70% of recent users of cannabis used cocaine vs 12.7% in the control group, Fisher’s test p = 0.0002) whereas no difference was found for contemporary recent use of cannabis and heroin (0% vs 3.1%, Fisher’s test p > 0.05), heroin and cocaine (0% vs 11%, Fisher’s test p > 0.05) and in the CH female group (Fisher’s test, all p > 0.05).
Age at first use of illicit drugs
Age at first use of illicit drugs (data are presented as mean age in years and SD) in cluster headache (CH) patients and in the sex-matched general population (POP).

t-test.
No significant difference was found in the age at first use of amphetamines/ecstasy (data regarding hallucinogens were not available for comparison), or between CH females and POP subjects.
Prevalence of illicit drug use in cluster headache (CH) males and general population (POP) by age-classes.

: p < 0.01 Chi-square test; §: p < 0.05 Chi-square test.
Current intensive use of illicit drugs
The rate of current intensive use of cannabis was significantly higher in the male CH patients (11.2%) than in the POP group (2.19%, Chi-square = 53.2, p < 0.001). No significant difference was found in current intensive use of other illicit drug (cocaine, CH 1.6% vs POP 0.2%, Fisher’s test p > 0.05; opioids, CH 0% vs POP 0.13%, Fisher’s test p > 0.05; amphetamines/ecstasy, CH 0% vs POP 0.05%, Fisher’s test p > 0.05; hallucinogens, CH 0% vs POP 0.07%, Fisher’s test p > 0.05). In the CH females, no significant difference was found in the prevalence of current intensive use of any of the illicit drugs surveyed (cannabis, CH 2.1% vs POP 1.8%, Fisher’s test p > 0.05; cocaine, CH 0% vs POP 0.1%, Fisher’s test p > 0.05; opioids CH 0% vs POP 0.05%, Fisher’s test p > 0.05; amphetamines/ecstasy, CH 0% vs POP 0.03%, Fisher’s test p > 0.05; hallucinogens, CH 0% vs POP 0.03%, Fisher’s test p > 0.05).
Lifetime sustained intensive use
The prevalence of lifetime SIU of any illicit drug was significantly higher in the CH males (22.8%) than in the POP group (4.8%, Chi-square = 108, p < 0.0001). In the women with CH no significant difference was found in the prevalence of SIU (5.2% vs 1.7%, Fisher’s test p > 0.05).
Episodic vs chronic CH and patients in active phase vs patients in remission phase
No significant difference was found between the ECH and CCH patients in any of the illicit drug use parameters considered, or in current use of cigarettes (CCH 80.5% vs ECH 73.1%, p > 0.05).
In the same way no significant difference was found in any of the illicit drug use parameters between the 189 patients investigated in the active phase and the 21 patients in remission phase (Fisher’s test, all p > 0.05).
SIU versus non-intensive users with CH
The age at onset of CH in the male SIUs was significantly lower than in the non-intensive users (24.3 ± 9.1 vs 29 ± 11.6, t = 2.35, p < 0.05), while bout frequency in these subjects was higher than in the non-intensive users (1.4 ± 0.7 vs 1 ± 0.6, t = 3.5, p < 0.001). No significant difference was found in bout duration, attack frequency, attack duration or rates of CCH (t-test, all p > 0.05). In female SIUs with CH no difference was found versus non-intensive users in any of the clinical parameters considered (t-test all, p > 0.05).
Age-matched prevalence of illicit drug use in males
Because age is an important determinant of levels of illicit drug use, data regarding the prevalence of illicit drug use in males have been analysed by age-classes. In the age group 25–34 years, LTU of amphetamines (Chi-square = 5.87, p = 0.01) and ecstasy (Chi-square = 7.8, p < 0.001) and CU of cannabis (Chi-square = 3.9, p = 0.04) were significantly higher in the CH patients than in the POP group. In the age group 35–44 years, LTU of cannabis (Chi-square = 10.3, p < 0.001), opioids (Chi-square = 10.6, p < 0.001), cocaine (Chi-square = 17, p < 0.001), amphetamines and ecstasy (Chi-square = 21, p < 0.001), RU (Chi-square = 27, p < 0.001) and CU of cannabis (Chi-square = 39, p < 0.001), and RU (Chi-square = 21, p < 0.001) and CU (Chi-square = 38, p < 0.001) of cocaine were significantly higher in the CH patients than in the control group. In the age group 45–54 years, LTU use of cocaine (Chi-square = 5.43, p = 0.01) was significantly higher in CH patients than in the POP.
Women were not analysed by age-classes because their sample size was too small.
Predictors of use of illicit drugs
In the LTU and RU models, only gender and age resulted in being predictors of use of any illicit drug. For CU, predictors were gender, age and educational level.
For LTU, male gender was a factor that predicted illicit drug use (p = 0.004; odds ratio (OR) = 2.785; 95% confidence interval (CI) = 1.875–5.592), whereas age resulted in a protective factor (p = 0.002; OR = 0.960; 95% CI = 0.935–0.985). The model resulted in being significant (Chi-square = 18.961; degree of freedom (df) = 2, p < 0.0001) and it was able to predict 63.2% of patients.
For RU, male gender was a factor that predicted illicit drug use (p = 0.063; OR = 2.337; 95% CI = 1.01–5.718), whereas age was a protective factor (p = 0.002; OR = 0.950; 95% CI = 0.919–0.981). The model resulted in being significant (Chi-square = 14.516; df = 2 p = 0.001) and it was able to predict 73.7% of patients.
For CU, male gender was a factor that predicted illicit drug use (p = 0.022; OR = 4.665; 95% CI = 1.247–17.447), whereas age (p < 0.001; OR = 0.917; 95% CI = 0.874–0.962) and educational level (p = 0.05; OR = 0.555; 95% CI = 0.308–1) were protective factors. The model resulted in being significant (Chi-square = 25.485; df = 3, p < 0.0001) and it was able to predict 77.5% of patients.
Discussion
This study is, to our knowledge, the first to present data regarding illicit drug use in a clinic-based sample of CH patients compared with POP data. The main finding of the study is that men with a CH record had significantly increased rates of occasional, sustained and regular use of illicit drugs, mainly cannabis and cocaine, whereas CH women did not show similar addictive behaviour. As expected, tobacco use was significantly higher in CH males and females than in the POP sample.
The finding of gender-related differences in illicit drug use needs to be interpreted cautiously, since it possibly reflects methodological problems, i.e. the small sample size – that does not allow an age-matched comparison and limits the statistical power necessary to disclose significant differences – and the use of data obtained from very skewed samples. However, under these premises, it may be hypothesised that gender-related differences in illicit drug use is related to a more attenuated clinical expression of CH in women. Indeed, in our study bout frequency and bout duration were both significantly lower in the women than in the men, and isolated clinical descriptive studies from the literature have indicated that women with CH may not show exactly the same clinical and epidemiological characteristics as men with CH (19–21). Thus, in men, the phenotypic expression of CH may be accompanied by comorbid addictive behaviour leading to illicit drug use. Further studies including a greater number of female patients are necessary to better define the gender-related differences in CH clinical expression and comorbidity.
The cross-sectional design of this study precludes definite assumptions of causality between CH and addictive behaviour in males. For example, we cannot exclude the possibility that exposure to illicit drugs and/or nicotine is a factor triggering the development of CH (22). However, the marked differences in the neuropharmacological properties of the different drugs of addiction and the observation that almost 30% of CH patients started to use illicit drugs after CH onset make this hypothesis unlikely. Alternatively, the use of illicit drugs may constitute an attempt to manage CH-related discomfort. Indeed almost 30% of CH patients reported that they first started using illicit drugs after CH onset, and illicit drugs may have some effect on CH symptoms (23–25). It has been well established that CH is often misdiagnosed, undertreated and mismanaged (11,13,26). Web sites devoted to CH actively promote therapeutic use of illicit drugs, and, by doing so, they possibly create or reinforce a specific interest in and demand for these drugs for CH (27). However, it appears an unlikely explanation because the majority of CH patients started to use illicit drugs before CH onset, not all illicit drugs have the potential to improve CH symptoms and no difference in illicit drug use was found either between CCH and ECH, or between CH patients in active and remission phase.
The most likely hypothesis is that male CH patients are prone to overindulge in addictive behaviours (both tobacco and illicit drug use), possibly because of a common biological susceptibility that predisposes them to both CH and addiction. According to this line of reasoning, it is noteworthy that in our sample, the age at first use of cannabis, cocaine and opioids was significantly lower in the male CH patients than in the control group. Recent research on addiction suggests that age at first use of addictive substances is genetically influenced and linked to more severe addictive behaviour (28,29). Thus, our finding might indicate that male CH patients have a more marked genetic predisposition to try addictive substances. Consistent with this hypothesis is the finding that current intensive use of cannabis and sustained intensive use of any illicit drug were more prevalent in CH males than in controls.
The neuropeptide orexin (also known as hypocretin) has recently been implicated in both drug addiction (30) and CH susceptibility (31–33) and thus emerges as a potential candidate to explain the present findings.
Several possible limitations of this study preclude the drawing of definite conclusions regarding some aspects of illicit drug use in CH patients. First, as recommended by the EMCDDA, in order to overcome a birth cohort effect and differences in the age profile of the CH patients and general population, the measures of illicit drug use would have been better analysed by age groups. Unfortunately, on account of the low prevalence of CH and the difficulty in recruiting a large sample of CH patients within the study period, this analysis could not be performed in women. In men, the study lacked the statistical power necessary to disclose significant differences in the use of less prevalent drugs and in age groups including fewer subjects. Indeed, due to the combined effect of age at onset of CH (infrequent in the second decade), the time of specialist consultation and diagnosis (usually delayed by several years in relation to CH onset), and oversampling of the youngest age-classes in the POP, young subjects were under-represented in the male CH group, whereas those aged 35–44 years were over-represented. Similarly, due to the presence of two peaks of onset in CH in women (third and sixth decade) and oversampling of the youngest age-classes in the POP, young subjects were under-represented and older subjects over-represented in the female CH sample. Since illicit drug use decreases with age, the differences in the age profile of the CH and POP groups might have resulted in underestimation of the differences in illicit drug use parameters, especially in the female groups. Unfortunately, we could not adopt a regression-based approach using age as a covariate that would have been more appropriate for the unavailability of the database regarding the POP survey.
The second limitation is that especially in women the comparisons between samples was too skewed, so there is a much larger sample in the POP data versus CH. This calls into question the validity of the results with respect to gender differences.
The third limitation is that, since the patient sample was selected in a headache centre setting, our results cannot be applied to the CH population as a whole.
Fourth, the study used self-reported data on substance use without the possibility of verifying in an objective manner the answers to the survey questions. Considering the nature of the survey, it may have led to an under-reporting of illicit drug use even if literature data suggest that if anonymity and confidentiality are protected, as in our study, people’s self-reporting of illicit drug is a reliable method (34).
Fifth, since illicit drug use differs in different geographical areas, ideally the controls should have been selected from the same geographical region as the CH cases. However, the CH patients enrolled in the two participating centres came from 12 of Italy’s 20 administrative regions (and 39 of its 100 provinces). Thus, the use of data from a control group representative of the national population represented the best possible compromise.
Sixth, possibly due to the nature of the topic of the survey, as well as the method with which the data were gathered, the response rate obtained in the general population, 35.1%, may result in an information distortion in the collected data. In fact it may be hypothesised that the non-respondent group may be engaged in a much heavier use of drugs (in which case the data gathered would underestimate the issue), or that, on the other hand, drug users took advantage of the anonymous nature of the survey to participate in it and thus affirm their drug use (in which case, the data gathered would be inflated). The former hypothesis is the more likely one, even if there is no clear evidence to support it (18).
Finally, we did not investigate the role of genetic, psychological (e.g. victimization, psychiatric comorbidity), social (e.g. disadvantaged familial and social network), economic (e.g. low income) and specific situational factors potentially influencing drug use (35).
Further studies including larger non-clinic-based populations of CH patients and exploring socio-environmental and neurobiological factors are necessary to better understand the nature of the association between cluster headache and addictive behaviour.
Footnotes
Funding
Funded by the Ministry of Health to IRRCS Mondino Institute – Current Research for 2009-2011 triennium.
Conflict of interest statement
None declared.
