Abstract
Objective:
The objective of this article is to study the characteristics of headaches following an experimental cold stimulus and evaluate whether the ICHD-II criteria for headache attributed to ingestion or inhalation of a cold stimulus (HICS) are adequate for the diagnosis of the headache induced by ice on the palate area (ice-induced headache (IH)).
Results:
One hundred and fifty-three out of 414 (37.0%) individuals tested had IH, but only 77/153 (50.3%) satisfied the ICHD-II criteria. The frontal (60.8%) and temporal (48.4%) areas were the most affected ones, with bilateral (77.1%) predominance, often of the pulsatile type (41.2%). One hundred and forty-seven out of 379 (38.8%) individuals who presented with a previous history of primary headache presented with IH, while only six out of 35 (17.1%) who had no history of previous headache reported pain in the test (OR 3.063, 95% CI 1.241–7.557). The ice-induced headache test was positive in 115/240 (47.9%) of the migraine sufferers and in 32/139 (23%) of the tension-type headache sufferers (OR 3.076, 95% CI 1.924–4.918).
Conclusion:
IH is predominantly frontotemporal, bilateral and throbbing, most commonly affecting migraine sufferers, and the ICHD-II criteria are insufficient for classifying all individuals. We should, however, recommend caution regarding such a generalization from our findings with experimentally provoked cold stimulus headache to cold stimulus headache in general (i.e. HICS, ICHD-II).
Introduction
Headache attributed to ingestion or inhalation of a cold stimulus (HICS) is a common form of headache recognized by the International Headache Society since the first classification in 1988 (1,2). In spite of being of frequent occurrence, there are few studies on HICS (3–10) and none of them has ratified the more recent International Classification of Headache Disorders 2nd Edition (ICHD II-2004) diagnostic criteria (2). They have apparently never been validated. For instance they refer to HICS as non-throbbing and acute pain that develops immediately after the cold stimulus. Nevertheless, a few studies have shown that HICS can be throbbing (8,9). It is difficult to understand what ‘acute/immediately’ means, since many individuals take more than a minute to develop HICS. The ICHD-II also established that HICS disappears within five minutes after removal of the cold stimulus. It is also a fact that several individuals have had HICS for more than five minutes. This was reported by 9.9% of those interviewed by Fuh and colleagues (6).
Unlike other headaches, HICS is not disabling but because it is of such short duration (seconds to minutes) and avoidable by removing the provoking stimulus (e.g. frozen ice treat), it seldom interferes with an individual’s daily tasks. Another HICS characteristic is that only certain individuals are susceptible, with some evidence of greater susceptibility in migraine sufferers (3,8,9).
HICS prevalence varied from 7.6% to 93% (5–11). In adult populations it was observed that 8% of those interviewed reported having had HICS (8,10); other studies reported prevalences of 15% (11), 37% (7) and 60% (9). In teenage populations prevalence ranged from 41% (6) to 79% (5).
In a literature review we found only four studies in which HICS was provoked (4,5,8,9), through the ingestion of ice (9), ice cream (4,5) or even icy water (8). When HICS prevalence is evaluated in a given population by asking whether the use of icy foods has ever provoked HICS, rates cannot be compared to those obtained during experimental triggering using cold stimuli. The variation is great, ranging from a low frequency of 7.6% in a female population between 40 and 74 years old in Sweden (8) to 93% among migraineurs (of both genders) hospitalized for several causes, who reported a previous history of HICS in the United States (3).
As the diagnostic criteria for HICS of the ICHD-II (2) were still not evaluated, the aim of this study was to provoke HICS by an experimental design of ice on the palate area (headache following an experimental cold stimulus or ice-induced headache (IH)) and to verify whether all the patients that happened to develop headache would satisfy such criteria. In the absence of a good correspondence, new criteria will be suggested, based on the results of this study. Since some criteria are based on whether the individual is a migraine sufferer, we evaluated the HICS characteristics in the two groups – migrainous and non-migrainous (tension-type headache) patients.
Methodology
The final sample comprised 414 volunteers consisting of 266 (64.3%) females and 148 males (35.7%), from eight to 84 years of age, with a mean age of 31 ± 12 years. They were healthy subjects without any significant or pre-existing health problems, with the exception of previous history of primary form(s) of headaches. The study was carried out between November 2008 and May 2009 in the city of Caruaru, Pernambuco, Brazil. Healthy university students and their family members were recruited to voluntarily participate in the experiment at the Associação Caruaruense de Ensino Superior. There was no financial compensation. This was a cross-sectional analytic observational study.
Demographic characteristics of the 414 volunteers.
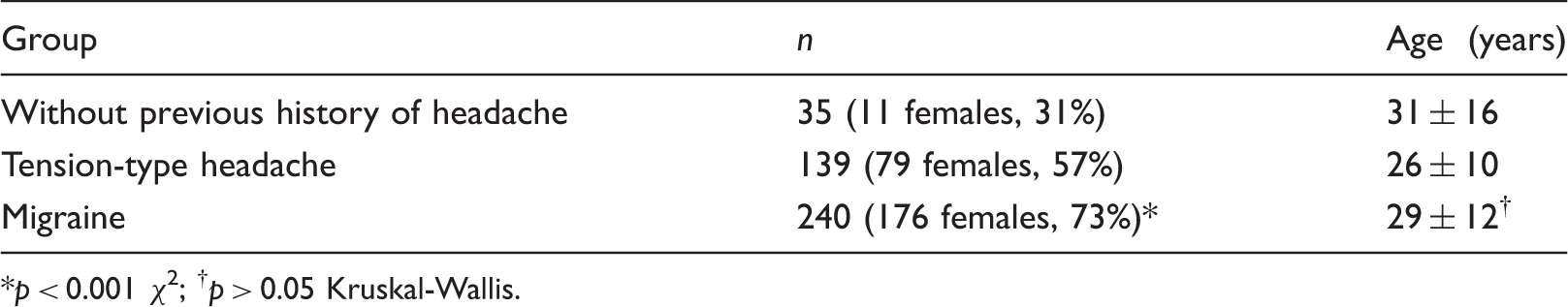
p < 0.001 χ2; †p > 0.05 Kruskal-Wallis.
Procedure of stimulation by using ice
In the attempt to provoke headache induced by ice in the volunteers, a previously described procedure (9) was used. To induce pain, ice cubes measuring approximately 20 × 20 × 35 mm (weighing 14.4 ± 0.5 g, n = 10) were placed between the hard palate area and the tongue (upper surface in contact with the palate, 15 × 20 mm, 300 mm2; lower surface in contact with the tongue, 20 × 35 mm, 700 mm2). Each volunteer was stimulated just once with a single ice cube. Before the procedure participants were told to remove any dental prosthesis that might be covering the palate. They thus had to place the ice cube on the tongue and press it against the hard palate, trying to keep it centralized. This precaution avoids teeth being stimulated by the ice. After placing the ice cube, the volunteers were instructed to close their mouth and keep their jaw motionless during the test. While the mouth was closed the volunteers were asked to indicate with the hand the onset and location of the pain. After the 90-second procedure of ice stimulation there was a 55% weight loss in the ice cube (ice cube weight after 90 seconds (s) stimulation 6.5 ± 0.7 g, n = 10).
The contact time of the ice cube on the palate was determined in 90 s. Counting began at the moment of closure of the mouth. Latency for the onset of the headache was the lapse of time from the moment the ice cube entered into contact with the palate up to the instant the subject felt the pain. Pain duration was timed from onset to relief, whether or not the ice cube was in contact with the palate. Information on the characteristics and intensity of pain were also obtained. The test was considered to be negative when no pain appeared until 10 minutes after the ice cube was removed. Of the 416 volunteers, two were excluded because they showed intolerance to ice and could not maintain the ice cube in contact with their palate for 90 s.
13.11.2 Headache attributed to ingestion or inhalation of a cold stimulus according to the International Classification of Headache Disorders 2nd Edition (ICHD-II) (2).

In relation to the nature of the headache, the volunteers classified it as throbbing or pulsatile, burning or blasted, weight or pressure or shock sensation. To evaluate headache intensity, a numerical visual scale was used showing figures from ‘0’ to ‘10,’ thus permitting the patients to identify their pain intensity among the indicated categories: 0, pain absence; 1 to 3, mild; 4 to 6, moderate; 7 to 9, high; and 10, unbearable. During the procedure no patient was allowed to use any prophylactic medicine for headache.
Ethical considerations
All the interviewees received detailed data concerning the purpose of the research. All doubts were clarified and each volunteer freely agreed to participate by giving his or her written consent. The research was approved by the Ethics in Research Committee of the Health Science Center of the Federal University in Pernambuco, Brazil, under protocol number 089/2008 in accordance with resolution 196/96 of the National Health Council.
Statistical analysis
Data are shown as mean ± standard deviation (SD). The Kolmogorov-Smirnov test was used to verify the type of distribution of variables to be studied. When the variables showed a normal distribution, the parametric test (Student t-test) was employed; when they did not, the Mann-Whitney non-parametric test was used. Based on the analysis of the categorical variables, we applied the chi-square test according to the expected frequency on the cells. The significance level considered was 5% (p < 0.05). For processing and analysis of data, the SPSS program version 13.0 for Windows was used.
Results
Field testing of ICHD-II criteria
Of the 414 individuals tested, 153 (37.0%) had IH (106/266 women (39.8%) vs. 47/148 men (31.8%), p = 0.126 χ2; Figure 1), but only 77 of these (50.3%) satisfied the ICHD-II criteria.
The bars show the percentage of individuals with ice-induced headache in the three groups: i. individuals with no previous history of primary headache (n = 35), ii. individuals with a previous history of tension-type headache (n = 139); and iii. individuals with a previous history of migraine headache (n = 240); (a) p = 0.0006 versus no headache group (odds ratio 4.447, 95% confidence interval 1.781–11.10); (b) p < 0.0001 versus tension-type headache group (odds ratio 3.076, 95% confidence interval 1.924–4.918) (χ2).
Considering the ICHD-II (2), the following examples would not receive the diagnosis of HICS: (a) non-migrainous patients with headache induced by a cold stimulus, located in a region other than the frontal one (34.2% in our series); (b) migrainous patients with headache located in a region other than frontal one, when it was not the usual site of migraine attacks (4.3% in our series); and (c) any pulsatile headache induced by a cold stimulus (41.2% in our series).
A previous history of HICS was reported by 200 (48.3%) of the individuals studied. The experimentally provoked IH was more frequent in those individuals who reported a previous history of HICS, when compared with the individuals without a previous history of HICS (128/200 (64%) vs. 25/214 (11.7%), odds ratio (OR) 13.44 (95% confidence interval (CI) 8.092–22.32), p < 0.001 χ2).
Pain location
Headache aspects observed among volunteers with positive ice test (n = 153).

Throbbing vs. non-throbbing nature of pain
Headache aspects observed among volunteers with migraine or tension-type headache.

2 Pearson test; †χ2 Yates test; ‡Mann-Whitney test.
Pain intensity
In 50 of 153 (32.7%) individuals who had IH, pain intensity varied from high to unbearable. The pain score was 5.5 ± 2.1 (SD) (Table 3). There were no statistical differences in the pain intensity scores between migraine and tension-type headache sufferers (Table 4).
Latency for pain onset induced by ice
Latency between ice placement in contact with the palate and resulting onset of pain was from 8 s to 8 min (65.5 ± 65.4 s). In 90/153 (58.8%) individuals with IH, latency was less than 60 s. In 150/153 (98.0%) individuals with IH it was ≤5 min. Only three volunteers presented latency longer than 5 min (330 s, 320 s and 480 s, respectively) (Table 3).
Pain duration
Pain duration varied from 90 s up to 26.6 min (207 ± 220 s). It was less than 2 min in 31/153 (20.3%) and less than 5 min in 136/153 (88.9%). Ten of the 153 (6.5%) had IH with a duration of between 5 and 10 min. In seven out of 153 (4.6%) the pain lasted for more than 10 min (26.6 min, 25.6 min, 20.5 min, 18.8 min, 12.0 min, 10.8 min and 10.0 min, respectively) (Table 3).
HICS in individuals with a previous history of migraine
In 13/38 (34.2%) patients without a previous history of headache or with tension-type headache with HICS, the pain was located in a region other than the frontal one, compared to 46/115 (40%) migraine patients; in only two out of 46 (4.3%) migrainous patients with headache located in a region other than the frontal one, it was not the usual site of migraine attacks (Table 4).
The ice-induced headache test was positive in 47.9% (115/240) of the migraine sufferers and in 23.0% (32/139) of the tension-type headache sufferers (χ2 p < 0.0001, OR 3.076, 95% CI 1.924–4.918) (Figure 1).
As for pain characteristics, 43.5% of migraine sufferers and 34.4% of the tension-type headache individuals, respectively, reported that their pain was throbbing or pulsatile (Table 4).
One hundred and forty-seven of the 379 (38.8%) who presented a previous history of primary headache (migraine or tension-type headache) presented IH, while only six out of 35 (17.1%) who had no history of previous headache reported pain in the test (χ2 p = 0.0111, OR 3.063; 95% CI 1.241–7.557).
Of the 147 with a previous history of primary headache whose ice test was positive, 71 (48.3%) reported that IH occurred at the habitual place of their previous primary headaches.
In migraineurs, the prevalence was similar between the genders (male 46.9% vs. female 48.3%).
Discussion
The present results using ice as an experimental way of provoking HICS reveal that the ICHD-II criteria are unsatisfactory for the diagnosis of this form of cold stimulus-induced headache, since only half the volunteers who developed headache after the application of ice to the palate would be thus diagnosed using such criteria.
Is all pain induced by a cold stimulus acute frontal non-pulsatile headache? Criterion ‘A’ states that the pain induced by a cold stimulus must be acute, frontal in location and of a non-pulsatile nature. However, an exception is admitted: In the case of migrainous patients, HICS may be referred to the usual site of migraine headaches.
We found that in about one third of tension-type headache subjects the IH was located in a region other than the frontal one, and in a small number of migrainous individuals the headache was also located in a region other than the frontal one, when it was not the usual site of migraine attacks. In addition, in a substantial number of subjects a pulsatile headache was induced by ice. HICS is a bilateral pain predominantly located at the frontal or temporal region (6,8).
Criterion ‘B’ clearly establishes the obvious presence of a cold stimulus to the palate and/or posterior pharyngeal wall due to the ingestion of cold food or drink or to the inhalation of cold air. Cold refers to the condition or unpleasant perception of having a low temperature (12–16).
A number of authors suggest a neurovascular mechanism in the genesis of the HICS (9,17). Migraine is a disease of the central nervous system with vascular participation, and the trigeminal nerve has an important role in migraine pathogenesis (18). Raskin and Knittle (3) suggested that HICS could represent a model for migraine because of the close relationship between HICS and migraine. The hyperexcitability of the trigeminal pathway that occurs in migraineurs and discharges of this pathway under certain circumstances (i.e. cold stimulus) could trigger a migraine-like attack. In this regard, the susceptibility of migraineurs to develop a headache induced by ice much more easily may be a form of cold allodynia or suballodinia (12), since the same stimulus does not trigger pain so readily in non-migraine sufferers.
The transient receptor potential (TRP) ionic channels are expressed in subpopulations of sensory neurons, and it has been argued that they mediate innocuous and noxious cold sensation (12–16).
An increase in blood flow and a visible flare in the area are seen after menthol (via TRP8 ionic channels) application, indicating a neurogenic vasodilatation. This may also explain a longer duration of the pain induced by ice and, perhaps, its pulsatile nature, since vascular arterial involvement and allodynia may both occur (19–22).
The use of ice in the present model of headache may activate low temperature nociceptors not only in the palate area, but also those present on the tongue. In addition, more than half of the ice cube thaws during the test, and the icy water and the cold air present inside the oral cavity, could as well decrease the local temperature. The icy water, once ingested, would also come into contact with the posterior pharyngeal wall.
In our volunteers the latency between the contact of the ice on the palate and the onset of headache ranged from 8 s to 8 min. The vast majority (98%) presented a latency of less than 5 min. Three individuals developed HICS after 5 min, indicating that the cold stimuli can trigger headache minutes later, probably through a mechanism other than the direct activation of nociceptors by cold. We believe that 5 min is a reasonable expected lapse of time for the occurrence of a headache provoked by a cold stimulus, even though it is possible, in a few cases, for an IH to occur after a longer latency.
Ninety-seven percent of the headaches induced by ice in the present study had a duration ≤10 min. Seven of the 143 patients with IH presented an episode of headache lasting longer than 10 min. Obviously, in the case of an HICS with a duration longer than 10 min it should be borne in mind that the HICS may trigger a primary headache attack superposed (without a headache-free interval) on the HICS. In the present study this did not seem to happen with the triggering of a migraine attack, since we would have expected a duration of hours in the typical example.
The present study demonstrated that individuals with a previous history of primary headache (lifetime prevalence) are twice as likely to suffer from IH.
Only a few studies have used icy substance to provoke pain (4,5,8), and only one used ice (9). Raskin and Knittle (3) conducted their research through an interview and found prevalences of 93% and 31% in the migrainous patients and control group, respectively. Mattsson (8) induced pain by ingestion of 150 ml of icy water (0–4°C) with a straw. He found a prevalence of 8% of HICS in his sample and suggested that females with active migraine had a greater predisposition to develop HICS. Using a questionnaire, Fuh et al. (6) found a prevalence of HICS in 55.2% of migrainous patients and of 39.6% in non-migrainous ones. By provoking palate stimulation with an ice cube for 90 s, Selekler et al. (9) found a prevalence of HICS in 74% of the migrainous patients and in 32% of those with headache of the tensional episodic type.
As stated by Selekler and colleagues (9), it seems that the prevalence of HICS is influenced by the duration of the exposure and the low temperature of the stimuli. We do not believe that the 90-second duration of the stimulus used was exceedingly long, as compared to a few intraoral stimuli experienced in real-life situations of consuming a cold substance. For example, it is common for ice cream to be continuously ingested for several minutes.
We should perhaps recommend caution regarding such a generalization from our findings with experimentally provoked cold stimulus headache to cold stimulus headache in general (i.e. HICS). The circumstances here are somewhat artificial – holding an ice cube against the palate – and do not entirely replicate what occurs when people take multiple gulps of cold drinks or swallow multiple boluses of ice cream. In 48% of the subjects evaluated in the present study, a previous history of HICS was reported. And we were able to induce an IH in 64% of them. Nevertheless, the IH was much more frequent among those who reported a previous history of HICS, suggesting that the artificial nature of our experimental stimulus in some way triggered a headache in those individuals susceptible to other forms of cold stimuli. Thus, our results suggest that the ICHD should take into account that, although artificial, the induced head pain in volunteers with an experimental cold stimulus and its characteristics (i.e. IH) may represent an HICS. An important question, however, remains to be answered: Would other kinds of cold stimuli trigger different forms of HICS?
Suggested modifications to 13.11.2 Headache attributed to ingestion or inhalation of a cold stimulus according to the International Classification of Headache Disorders 2nd Edition (ICHD II) (2).

Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
None declared.
