Abstract
Background: Sleep disturbances are consistently associated with chronic headaches, yet the mechanisms underlying this relationship remain unclear. One potential barrier to generating new hypotheses is the lack of synthesis between models of headache and models of sleep. The goal of this paper is to present a perspective on the chronification of migraine and tension-type headaches based upon conceptual models used in sleep research.
Methods: We provide a critical review of the literature on sleep and headache, highlighting the limitations in sleep methodology. Models of sleep physiology and insomnia are discussed, along with the potential implications for the chronification of migraine and tension-type headache. In addition, we propose a biobehavioral model that describes the interaction between behaviors related to coping with headache, the impact of these behaviors on insomnia and sleep physiology and the downstream propensity for future headache attacks.
Conclusions: We hope that this perspective will stimulate interdisciplinary activity toward uncovering the pathway for more effective interventions for chronic headache patients.
Introduction
Chronic headache (CH) is a debilitating disorder that affects 3–5% of the general population and 70–80% of patients presenting to headache clinics (1–3). Compared to episodic headaches, CH causes greater disability, greater impairment of quality of life and increased health care utilization (4,5). The cost of CH patients is estimated to be over 1000 U.S. dollars per person over three months, with hospitalizations accounting for over 60% of total direct costs (4,5). Out-of-pocket costs for medications are also significantly higher for CH compared to other forms of episodic headache (4,5). Classification of CH includes the presence of primary headaches that occur 15 days or more per month for at least three months, including migraine, tension-type headache (TTH), new daily persistent headache, and hemicrania continua (6,7). Chronic migraine (CM) is the most common type of CH, and there are often clinical features of TTH that develop during the transformation from episodic to daily or near-daily headaches (8). Therefore, this paper focuses on these two types of headaches.
Within a one-year period, up to 14% of patients can shift from episodic to CH (9). A growing literature has repeatedly found associations between sleep dysregulation and the chronification of headaches. However, the specific mechanisms or risk factors that could help uncover pathways for intervention remain unknown. One potential barrier to generating new hypotheses is the lack of synthesis between headache models and sleep models. Therefore, the goal of this paper is to present a perspective on the development and maintenance of CH based upon models of sleep physiology and the pathophysiology of insomnia, the most common form of sleep disturbance among headache sufferers (10). In particular, we focus on coping behaviors that appear to serve as a common link between headaches and insomnia and consider the downstream physiological impact of these behaviors. Other sleep disorders, such as obstructive sleep apnea and restless legs syndrome, have been found to be associated with headaches (11–17), but these disorders are more closely associated with sleep fragmentation, which is outside the scope of this review. As such, the aims of this paper are to provide a critical review of the literature on sleep and headache, outlining the key limitations from a sleep perspective, and to propose a biobehavioral model of CH and insomnia and discuss the research and clinical implications of this model. We hope that this perspective will catalyze multidisciplinary teams to work together in an effort to further clarify the relationship between CH and sleep, thereby improving patient care for this population.
What is known about the relationship between sleep and headaches?
Summary of findings evaluating sleep and headache.

HA: headache; EM: episodic migraine; CM: chronic migraine; TM: transformed migraine; MM: menstrual migraine; TTH: tension-type headache: REM: rapid eye movement; NREM: non-REM; SWS: slow-wave sleep; SDB: sleep-disordered breathing; MT: melatonin.
Self-report measures
Most studies have employed self-report measures, which can provide a convenient estimate of sleep quantity. Self-report measures of sleep are generally in the form of simple questions (e.g. How often do you have difficulty falling asleep?), and answer choices may be in the form of a yes/no format versus frequency determination (e.g. 1 = never, 2 = rarely, 3 = occasionally, 4 = frequently but not daily, 5 = daily). Duration of sleep is recorded as an estimate of sleep time or by utilizing sleep diaries. With these methods, self-report data in headache sufferers consistently show a high prevalence of insomnia, characterized by difficulty initiating sleep or maintaining sleep, or by non-restorative sleep that accompanies the short sleep time (19,20,29–31). Some studies report that short sleep can precede an acute headache attack (20,28) while others demonstrate an association between prolonged sleep time and headache episode (20,32). Interestingly, episodic migraine and TTH sufferers frequently report daytime naps, which may have an acute palliative effect on a headache episode (21,33,34). However, frequent daytime napping may shorten the duration of nighttime sleep, thereby contributing to worsening headache the following day. As discussed later, this might lead to greater variability in sleep patterns.
Polysomnography
Because self-report measures cannot assess sleep stages, polysomnography (PSG) is used as the “gold standard” objective measure of sleep physiology. PSG provides objective physiological data on electroencephalogram (EEG), electromyogram (EMG), electrocardiogram (EKG) and pulse oximetry data. PSG findings have generally supported the findings of reduced sleep time among migraine and TTH sufferers using self-report methods. In addition, studies have found that dysregulation of slow-wave sleep (SWS) and rapid eye movement (REM) sleep occurs in migraine, but the pattern is not consistent across studies (23,26,35). In one of the first studies using PSG, Dexter demonstrated an increase in SWS and REM prior to an acute migraine episode (35). Goder and colleagues found evidence of decreased REM density, fewer EEG arousals and less beta EEG activity (fast-frequency activity) in the nights prior to a migraine attack, suggesting decreased cortical activity prior to an attack (23,36). In CM, one study evaluating children also reported reduced SWS and REM (27), while another reported minimal objective sleep disturbance in episodic headache suffers when compared to CH sufferers who demonstrate chronically poor sleep (26). These findings suggest that sleep dysregulation may reflect the physiological changes that occur in the headache chronification process, rather than merely reflecting the differences between the ictal and interictal headache state.
Actigraphy
Another objective method used to assess sleep and sleep-related behaviors is actigraphy. Actigraphs are small devices that utilize a motion sensor known as an “accelerometer” to monitor the occurrence and degree of motion. This type of sensor integrates the degree and speed of motion and produces a small signal whose magnitude and duration depend on the amount of motion. The signal is amplified and digitized by the onboard circuit and stored in memory onboard the device as activity counts. Actigraphy is commonly used to assess sleep/wake patterns and has been well validated against PSG for the assessment of sleep (37,38). Unlike standard PSG, a key advantage of using actigraphy is the ability to assess sleep-wake patterns across a number of days or weeks. In a series of studies that used actigraphy monitoring in TTH sufferers over seven days, Kikuchi and colleagues found that longer sleep time and poor sleep quality were both associated with headache the next day (32) and that the occurrence of TTH attacks was associated with a simultaneous and subsequent decrease in physical activity (39). Unfortunately, it is unclear if the longer sleep time reflected more time in bed or more sleep time, given that sleep quality was rated as poor. Using actigraphy on migraine patients, Bruni et al. found decreased physical activity prior to a migraine attack, along with some difficulty initiating sleep (24). These limited findings provide evidence of decreased physical activity and potentially more time in bed when using actigraphy. However, this device has been underutilized in evaluating the sleep/wake patterns of CH patients.
Melatonin
An emerging area of interest is the relationship between headaches and the circadian rhythms that are involved in sleep-wake regulation. Circadian rhythms are influenced by melatonin, a hormone secreted by the pineal gland and regulated by the suprachiasmatic nucleus. Melatonin has been studied in headache patients for various reasons, relating to its use as a potential anti-inflammatory agent or toxic free-radical scavenger, its ability to modulate pro-inflammatory cytokines, its use as a nitric oxide or dopamine inhibitor, to help with membrane stabilization, to help with GABA/opioid potentiation or to modulate serotonin (40). Some studies have found that migraine attacks peak in the early morning or midafternoon (41,42), with evidence for a circadian phase delay in migraine sufferers (40,43,44). Studies have found that melatonin levels are lower in migraine sufferers (45–48) and that administration of melatonin has analgesic effects, improving headache symptoms (44,49). Claustrat et al. found that melatonin infusion provided headache relief in a small sample of patients with status migrainosis. Of the six patients, four patients had relief on the first night and the remaining two had relief by the third night, primarily described as decrease in the pulsatility of pain. Interestingly, two of these patients who were described as having delayed sleep phase demonstrated advancement of their sleep with treatment (43). Nagtegaal et al. administered melatonin by mouth for three months in patients with comorbid headache and delayed sleep phase, with improvement in headache frequency and intensity (44). These preliminary findings suggest that a deeper understanding of the chronobiology of migraine is warranted.
Limitations of the headache and sleep literature
There are several important limitations to the literature discussed above. First, the majority of findings are based upon self-report measures of sleep, with a relative dearth of studies employing objective measures. Self-report measures are subject to biased perception of sleep, as reflected by the consistent findings that those with insomnia overestimate the amount of time awake and underestimate the amount of time asleep when compared with normal sleepers (50–52). Also, self-report measures frequently do not differentiate the time spent in bed from the time spent sleeping. Many individuals will consider time spent in bed as sleeping, thereby conflating time in bed with time spent asleep. While this difference might be insignificant in normal sleepers, it is exaggerated in poor sleepers and could lead to overestimation of sleep time when using self-reported data, which may partially explain the mixed findings of sleep time (short and long) and headaches. Moreover, most studies reviewed did not employ measures to account for daytime naps. As a result, studies using self-report measures are not able to distinguish actual sleep time across a 24-hour period (i.e. nighttime sleep plus naps) from sleep-related behaviors (e.g. time going to bed, time getting out of bed and time laying down to rest without sleeping). As discussed later, these behaviors are particularly important for individuals with headaches and have downstream consequences on sleep physiology. Although PSG data provides key objective data, limitations of using PSG include a use of one-night study that may not necessarily be representative of a usual night of sleep and foreign environment. While actigraphy can provide objective data across several nights, few studies have used this method of sleep in differentiating between episodic and CH sleep patterns.
A second key limitation is the dearth of studies that have examined sleep physiology and circadian phase in the context of headache. Most of these studies were conducted on small samples and generally focused on sleep macroarchitecture, which includes sleep stages (percentage of night and latency to each stage), but not sleep microarchitecture (e.g. EEG power). Methods of examining sleep microarchitecture include spectral analyses of EEG activity during REM and non-REM (NREM) periods, from which more specific details about the mechanisms of sleep can be derived. These methods are becoming more commonly used in sleep research to examine patterns of cortical activity during sleep (53), yet have not been fully explored as a potential tool for examining sleep among headache sufferers. The use of spectral analyses could help connect the physiological mechanisms of sleep with the pathophysiology of CH.
In addition, we could not find any studies of headache patients that have assessed dim-light melatonin onset (DLMO), which has become the gold standard for measuring circadian phase in sleep research (54). Assessment of DLMO can yield important information on the potential role of circadian phase misalignment as a risk factor for CH without using invasive methods (i.e. plasma melatonin measures), and does not depend on menstrual stage (i.e. urinary melatonin measures). Assessment of DLMO could also help clarify the role of melatonin in headache chronification.
A final limitation is the lack of an integrated framework or model to explain the relationship between CH and sleep. Several review papers have focused exclusively on anatomical or physiological mechanisms (55–59). These have generally focused on the hypothalamus and the homeostatic functions that govern NREM sleep, regulated by the hypothalamus (58,59), along with hypotheses implicating dysregulation in REM sleep (55). Other hypotheses have included the role of neurotransmitter systems, including adenosine, orexin and cytokines (58,59). However, very little attention has been given to behavioral coping strategies (e.g. going to bed to relieve a headache), which could have a significant impact on sleep physiology. Thus, a model that integrates behavioral and biological factors could uncover biobehavioral interactions that have not yet been fully investigated.
Models of sleep and insomnia
In this section, we review the relevant literature on sleep physiology and pathophysiology. Because previous reviews have discussed anatomical and molecular pathways common in sleep and headache disorders (55,57–59), we focus on two sleep models that we consider to be the most relevant for the development and maintenance of CH. The first model is the two-factor model for sleep-wake regulation. This is considered the most widely recognized model for understanding sleep-wake regulation and sleep physiology. The second model is a biobehavioral model for chronic insomnia. This model provides a perspective of how sleep disturbance is developed and maintained over time, with a focus on the behavioral response to the initial sleep disturbance as a perpetuating factor. This model is widely accepted as a key etiological model for chronic insomnia, and many of the features appear to fit well with the chronification of headaches.
Two-factor model of sleep regulation
The most widely accepted model for understanding sleep-wake physiology is the two-factor model of sleep regulation (see Figure 1) (60,61). This model posits that sleep and wake mechanisms are primarily governed by the interaction between a homeostatic process (Process S) and a circadian process (Process C). Process S accumulates as a homeostatic drive or “sleep pressure” across the wake episode, reaching a maximum at bedtime. During sleep, Process S is indexed by dissipation of slow wave activity (SWA) throughout the sleep period. Process C is biphasic and independent of sleeping and waking states, assisting in the timing of alertness and sleep during the habitual wake and sleep periods, respectively, generating alerting signals during the habitual wake episode, and actively promoting sleep in the latter half of the sleep period (62). Process S and Process C interact to regulate sleep and wakefulness in humans.
Two-process model of sleep regulation. Process S (sleep homeostasis) reflects the accumulation of wake time, which dissipates during sleep. Process C (circadian process) is a biphasic process independent of wake time or sleep time, interacting with Process S to help with alertness and sleep onset. Figure adapted from Borbely AA (1982). A two process model of sleep regulation. Human Neurobiology 1(3): 195–204. Reproduced with kind permission from Springer Science+Business Media B.V.
94
.
It does not appear that this model has been specifically applied to the study of sleep disturbance in the context of headaches. However, there appear to be several important implications for how dysregulation in either the homeostatic or circadian process could impact headache pain. Because daytime naps are known to adversely affect the accumulation of Process S (61), this can lead to a delay in bedtime (potentially exacerbating a circadian phase delay), with increased difficulty initiating and maintaining sleep, and decreased SWA at night. Moreover, selective disruption of SWS (but not total sleep) has been associated with decreased pain thresholds and inflammatory response (63) and might lead to an increased vulnerability to a headache attack.
Several links have emerged to suggest that the circadian system is likely involved in headache mechanisms. In a review, Peres (40) proposes several possible links, including an anti-inflammatory pathway, toxic free-radical scavenger, modulation of pro-inflammatory cytokines, nitric oxide or dopamine inhibition, membrane stabilization, GABA/opioid potentiation or serotonin modulation. Studies have found lower levels of plasma melatonin in migraine sufferers (45–48). In migraine sufferers who have a delay in circadian phase (40,43,44), the administration of melatonin can prevent or ameliorate headaches (43,44). It is possible that melatonin is directly involved in the pathophysiology of migraine (i.e. role in photophobia) or that the circadian misalignment indirectly leads to increased sleep disturbances.
Biobehavioral model of chronic insomnia
The most widely accepted conceptual view of chronic insomnia is a biobehavioral model (see Figure 2) that includes predisposing, precipitating and perpetuating factors of sleep disturbance (64). This model proposes that certain predisposing factors (e.g. anxious personality traits, physiological predisposition for sympathetic activation) combine with precipitating factors (e.g. psychosocial stressors) that exceed a particular threshold to trigger acute sleep disturbance. These elements are congruent with a diathesis-stress model. Efforts to cope with the sleep disturbance, including compensatory daytime naps, use of sleep medications at night or use of caffeine during the day, then develop into perpetuating factors. Although these behaviors might seem to provide short-term relief, they are maladaptive, and instead serve to maintain the sleep disturbance, leading to chronic insomnia. This biobehavioral model of insomnia provides an explanation for the interaction between biological and behavioral factors over time in the context of insomnia.
Spielman biobehavioral model 3-P of insomnia. Conceptual model of the development of chronic insomnia and the changing factors that play a role over the course of the disorder. In this particular case, at the onset of the insomnia, precipitating factors predominate. When the insomnia becomes chronic, perpetuating factors become the main feature contributing to the sleep disturbance. This figure and caption was published in Principles and Practices of Sleep Medicine, 5th edition; Spielman, Yang, Glovinsky (Authors); “Assessment Techniques for Insomnia”; adapted from Spielman, Caruso, Glovinsky. Copyright Elsevier 2012.
95
.
An important consequence of the perpetuating factors is increased night-to-night (or intra-individual) variability of sleep. This variability or instability in sleep can have physiological consequences, leading to dysregulation in both Process S (increased sleep pressure during the work week) and Process C (delayed rise time leads to misalignment of the circadian rhythm). Recently, sleep researchers have begun to focus on variability as opposed to average sleep time. In a study in older adults with chronic insomnia, Buysse and colleagues found evidence for more night-to-night variability in sleep efficiency (calculated as total amount of sleep time divided by total amount of time in bed) and wake time after sleep onset (i.e. time awake in the middle of the night) for the chronic insomnia group compared to a healthy control group (65). Furthermore, there were few sleep parameters that correlated from one night to the next among those with insomnia. Another study found more night-to-night instability in total sleep time among those with comorbid insomnia compared to primary insomnia (66). Finally, one study found an association between stress and intra-individual variability in sleep parameters across nights (67). Examining the variability in sleep rather than the average sleep could provide insights into the dysregulation of sleep among CH sufferers.
Interestingly, there seems to be much overlap between the perpetuating factors in the biobehavioral model of insomnia and the coping behaviors used by headache sufferers. Both migraine and TTH sufferers are known to use naps or sleep as a coping strategy to manage headaches (20,21,34,68). Caffeine, an adenosine antagonist, has known palliative effects for headaches (58,59) and many headache sufferers report using caffeine and medications that have sedating properties (68,69) to cope with headaches. Use of these substances could lead to disruptions in sleep and wakefulness or difficulty establishing regular sleep/wake patterns. Thus, it would appear that coping strategies might provide a common link between the perpetuation of headaches and insomnia.
Ong and colleagues have proposed a behavioral hypothesis positing that the reinforcing effects of sleep as a palliative coping strategy for headache might lead to disruptions in normal sleep/wake regulation (21). Preliminary data for this hypothesis comes from research showing that TTH sufferers report sleep problems and stress as a trigger of headaches, while going to sleep is used as a coping strategy for pain (21). Furthermore, 81% of TTH sufferers endorsed “going to sleep” as a self-management strategy and rated this as the most effective strategy. These findings suggest that episodic headache sufferers face a unique dilemma whereby the efforts to manage headache pain by going to sleep might disturb the physiological processes involved in sleep regulation. This hypothesis connects the biobehavioral model of insomnia with the downstream physiological consequences based upon the two-process model of sleep-wake regulation (60).
A biobehavioral model of chronic headache and insomnia
Building upon the literature reviewed, we propose a biobehavioral model that describes the interaction between behaviors related to coping with CM and TTH, the impact of these behaviors on insomnia and sleep physiology, and the propensity for future headache attacks (see Figure 3). This model has three basic tenets: (1) coping behaviors for headaches can precipitate and perpetuate sleep disturbance; (2) disruption in sleep physiology increases the propensity for headaches; (3) over time, these cycles interact and serve to transform or perpetuate episodic headache into CH. We elaborate on each of these aspects in more detail below.
Proposed biobehavioral model of the mechanisms underlying chronic insomnia and chronic headaches.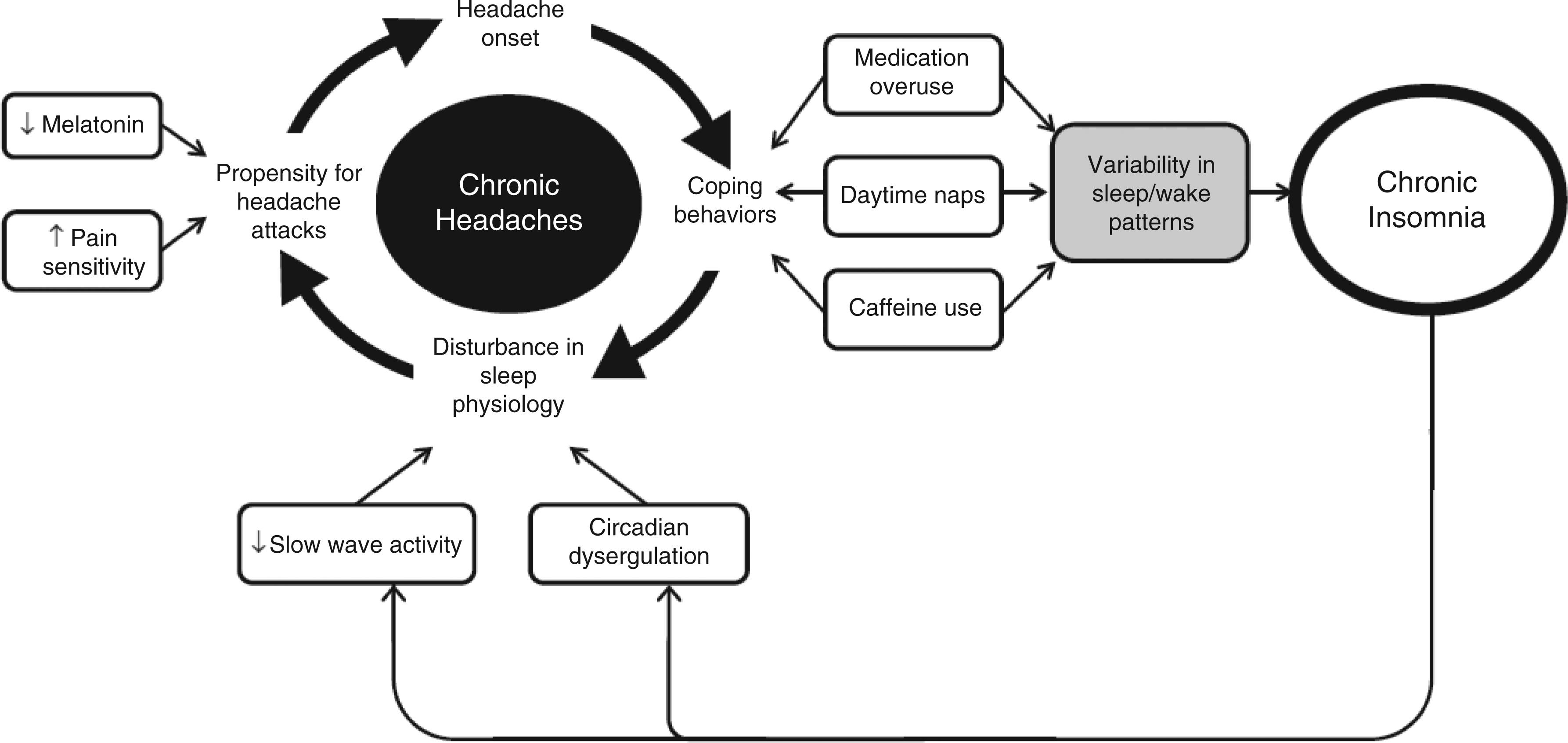
1. Coping behaviors for headache can precipitate and perpetuate sleep disturbance
As noted earlier, coping behaviors for both migraine and TTH include use of sleep or naps, caffeine, sleep medication or lying down in a dark room (68). Unfortunately, these coping behaviors for headache can lead to poor sleep hygiene, which might serve as a precipitating factor for a comorbid insomnia disorder according to the biobehavioral model of insomnia (64). These behaviors can result in variable sleep patterns, particularly with bedtimes, wake times and napping across days, creating a cycle of insomnia. Repetitive engagement in these coping behaviors can perpetuate the sleep disturbance to chronic insomnia and can result in “downstream” disturbances in sleep physiology. Specifically, frequent napping and variability in bedtimes can lead to decreased SWS. Also, caffeine, which is an adenosine antagonist (70), has been shown to suppress SWS (71,72), and some older hypnotic medications (e.g. benzodiazepines, barbiturates) are known to suppress SWS (73–76). These behaviors may also cause disturbances in the circadian system. Disturbances of circadian pathways have been demonstrated in other pain models, including fibromyalgia (77–79) and chronic fatigue syndrome (78).
2. Disruption in sleep physiology increases the propensity for headaches
Altered sleep physiology could increase the propensity for future headache episodes. Although not specific to headache, studies on pain and sleep have found that partial sleep deprivation can exacerbate pain (80,81) and impairs pain-inhibitory functioning (82). Hakki-Onen and colleagues found that recovery of SWS after total sleep deprivation has analgesic effects that surpass those found by level I analgesics (i.e. acetaminophen, aspirin, ibuprofen) (83). Given that adenosine is involved in SWS and that daytime naps and increased time in bed can decrease SWS during nocturnal sleep, this biobehavioral interaction might provide a multilevel explanation from behavior to neurotransmitter dysregulation. If the pain threshold is decreased, an individual might be more prone to having a headache or be more sensitive to headache attacks.
Disturbances in the circadian system could also have an impact on propensity for headaches. As noted earlier, melatonin appears to have analgesic effects (84), so reductions in the level of melatonin could be hyperalgesic. Also, phase delays or phase advances could lead to circadian misalignment, which has been associated with mood disorders and could also play a role in susceptibility to future headache episodes.
3. Over time these cycles interact and serve to transform or perpetuate episodic headache into chronic headache
From this cycle, a comorbid insomnia disorder might also develop that further perpetuates the cycle of CH. Or, it is possible that chronic insomnia develops first, leading to the transformation to CH. This process is not seen as being independent of other factors that have been suggested in the transformation of migraine (e.g. dietary factors, stress), which could also propagate headache episodes (85). It is possible that these other factors can enter at different points during the cycle or might have independent pathophysiological mechanisms. Much like two interlocking gears, the “co-perpetuation” leads to entanglement of the cycles and chronification of both headache and insomnia.
Research and clinical implications
This model is a proposed starting point for synthesizing models of sleep disturbance with models of headache. Research is now needed to validate the model and to test its specificity and generalizability. As noted earlier, this biobehavioral model is focused on insomnia and does not account for other types of sleep disorders. Studies are needed to test this model for conditions such as sleep apnea, where sleep fragmentation due to respiratory events could also lead to disturbances in sleep physiology in the current model. Also, this model was developed largely based upon the literature on CM and chronic TTH, both of which show strong associations with insomnia, and thus it is unclear if the model would generalize to other types of CH, such as new daily persistent headache or hemicrania continua. Furthermore, we felt there was insufficient evidence at this time to distinguish different sleep mechanisms for TTH and migraine. It is anticipated that future research might reveal core features that would be common across chronic headaches (e.g. coping behaviors that are maladaptive for sleep), while other elements would be unique to specific types of headache (e.g. unique disturbance in REM sleep). An agenda for future research should include using state-of-the-art methods for assessing sleep in the context of headaches. For example, evaluation of 24-hour sleep patterns, including night-to-night variability and daytime naps, would provide greater insight into sleep-wake patterns over time, which could help clarify certain paradoxical findings (e.g. the associations of headaches with both short and long sleep). Incorporating analyses of Process C with circadian phase angle assessments via DLMO measurements and Process S with power spectral analyses of sleep microarchitecture would allow comparisons on sleep physiology between CH with normal subjects or other populations. We encourage multidisciplinary collaborations between sleep specialists and headache specialists to help meet these research needs.
By employing sophisticated sleep measures, further research can also move forward to examine the impact of sleep interventions aimed at changing the coping behaviors. Effective behavioral treatments for sleep and circadian disturbances are available (86) and could be adapted to address the coping behaviors for headache that serve to perpetuate the sleep disturbance. Calhoun and Ford conducted a pilot study on CM patients using a brief behavioral sleep modification intervention consisting primarily of sleep hygiene instructions (87). Compared to a sham behavioral intervention, the authors found that the behavioral modification group reported significant reductions in headache frequency and intensity relative to the control group, with 48.5% of the treatment group reverting from chronic to episodic migraines. Given that sleep hygiene is only a component of most behavioral treatments for insomnia, it remains to be seen if a full course of cognitive-behavioral therapy for insomnia, typically eight weekly sessions, would have superior results. Nevertheless, this pilot study provides preliminary evidence that sleep-related behaviors are a modifiable risk factor in the transformation of migraine.
Although the clinical implications are not yet clear, the ultimate goal of the biobehavioral model would be to improve patient care. If further research supports this model, clinicians should carefully assess the coping behaviors in addition to other recommendations for sleep disorders (88–91). In addition to multidisciplinary research teams, this model would call for multidisciplinary clinical teams, with sleep specialists involved in the care of headache patients. Sleep specialists could help to implement assessment of sleep variability through actigraphy or help screen for sleep disorders including sleep apnea (92,93), given its common association with headaches. Clinical decisions regarding the sequence (i.e. target headache, then sleep or vice versa) or modality (e.g. behavioral or pharmacological) of treatments could then be made jointly with both specialists, thus providing more personalized patient care.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
The authors have indicated no financial conflicts of interest.
