Abstract
Background: The prevalence and characteristics of morning headaches (MH) in habitual snorers are not well known, with only one retrospective study reporting MH in 23.5% of snorers. The role of MH in bed partners of snorers has not yet been examined. Therefore, the objective of this study was to assess MH prospectively in habitual snorers and their bed partners.
Methods: We recruited habitual snorers and their bed partners via newspaper articles. The participants completed a semistructured interview, filled in questionnaires about sleep quality, daytime sleepiness, depression and anxiety and kept a 90-day headache and sleep diary.
Results: We included a total of 102 snorers and 63 bed partners. Seventy-six snorers (25 female) and 41 bed partners (31 female) completed the study, recording a total of 6690 and 3497 diary days, respectively. MH was present on at least 1 day in 57% of the snorers and in 61% of the bed partners and recorded on 7.2% and 3.6% of the diary days, respectively. Independent predictors of MH in snorers were pre-study headache frequency (p < 0.001), anxiety disorder (p < 0.001), waking up because of pain (p = 0.002) and waking up too early (p < 0.001); and independent predictors in bed partners were migraine (p = 0.02), difficulties breathing during sleep (p < 0.001), waking up because of pain (p = 0.01) and waking up too early (p < 0.001). The analysis of couples only (n = 41) revealed pre-study headache frequency (p < 0.001), waking up too early (p < 0.001) and nocturnal confusion (p < 0.001) as independent predictors of MH in snorers, and migraine (p = 0.02), difficulties breathing during sleep (p < 0.001), waking up because of pain (p = 0.01) and waking up too early (p < 0.001) as independent predictors in bed partners.
Discussion: MH shows a 90-day prevalence of around 60% in habitual snorers as well as in their bed partners. Predictors of MH are related to sleep, headache and psychiatric comorbidity.
Introduction
Snoring is the most common presenting symptom in the sleep clinic, being given by 95% of male and 90% of female patients (1). Snoring loudly every night increases the risk of obstructive sleep apnea syndrome (OSAS). OSAS occurs in 4% of men and 2% of women, and excessive daytime sleepiness and morning headaches (MH) are the most common clinical manifestations besides snoring (2).
The prevalence of MH ranges between 5% and 7% in the general population (3–5). Clinic-based studies in OSAS patients reported prevalence rates between 18% and 74% (6,7). On the other hand, OSAS has been diagnosed in 12–42% of patients with nocturnal headache or MH (8).
Comparing the prevalence of MH in OSAS patients and controls in two population-based studies yielded conflicting results (3,9). Ulfberg et al. (3) reported that MH occurred significantly more often in OSAS patients than in controls. In contrast, Olson et al. (10) did not find an association between OSAS and MH. In a clinic-based study by Göksan et al. (7), prevalence of MH was significantly higher in snorers with moderate-to-severe OSAS than in those with mild OSAS and healthy controls.
In habitual snorers MH was found to be independently predicted by OSAS as well as by a history of migraine, insomnia and psychological distress (10).
Our knowledge of the impact of habitual snoring on bed partners is limited to a single study showing that continuous positive airway pressure treatment of OSAS increased sleep efficiency and time spent in rapid eye movement sleep and decreased the number of arousals in bed partners of habitual snorers with OSAS (11).
All studies mentioned have exclusively relied on retrospective assessment of MH, and the role of MH in bed partners of habitual snorers is entirely unknown. To address this paucity of information we performed a prospective diary study including habitual snorers and their bed partners. The aim of this study was 1) to characterize the prevalence and clinical characteristics of MH, 2) to classify each single MH episode according to the second edition of the International Classification of Headache Disorders (ICHD-II) (12) and 3) to identify risk factors for the occurrence of MH both in habitual snorers and their bed partners.
Patients and methods
Between January 2009 and May 2011 habitual snorers were recruited via newspaper articles either alone or together with the non-snoring bed partner. Snorers and bed partners had to be between 18 and 75 years old, and they should be able to fill out the study questionnaires and headache and sleep diaries. Exclusion criteria for snorers and bed partners were shift work, regular intake of sleep-promoting substances, deafness or regular use of earplugs during sleep, substance addiction, severe respiratory disorders and other severe medical or psychiatric conditions. Habitual snoring was defined as snoring daily or near daily. MH was defined as present if the diary question ‘Do/Did you have headache this morning?’ was answered with ‘yes’. The study protocol was approved by the ethics committee of the Medical University of Vienna and written informed consent was obtained from all study participants.
At baseline all snorers and bed partners underwent a semistructured interview covering demographics, height, weight, general medical and psychiatric history, previous headache diagnoses and pre-study headache frequency (headache frequency during the last 3 months before study inclusion, given as 1–2 days a year, less than 1 day in 3 months, 1 day a month, 2–3 days a month, 1 day a week, 2–3 days a week and ≥4 days a week).
In addition, the participants were required to complete the Headache Impact Test (HIT-6), the Pittsburgh Sleep Quality Index (PSQI), the Epworth Sleepiness Scale (ESS), the Self-rating Depression Scale (SDS) and the Self-rating Anxiety Scale (SAS).
The HIT-6 is a short questionnaire that includes six questions relevant to headache severity, frequency and disability. Ratings of ‘never,’ ‘rarely,’ ‘sometimes,’ ‘very often’ and ‘always’ are weighted so that the total score lies between 36 and 78. A total score of 60 and more indicates that the headache is having a severe impact on the patient’s life (13,14).
The PSQI measures the quality and quantity of sleep in the 4 weeks preceding the investigation. It differentiates ‘poor’ from ‘good’ sleep by seven areas: subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleeping medication, and daytime dysfunction over the last 4 weeks. Scoring of answers is based on a 0 to 3 Likert scale, where 3 reflects the negative extreme. A total score of 5 or more indicates a ‘poor’ sleeper (15,16).
The ESS is also a self-rating instrument to evaluate the tendency for dozing off during daytime. It consists of eight items concerning everyday situations. Reponses to each item are ranked from 0 to 3 according to the probability of dozing off during a task (0 = never, 1 = low probability, 2 = moderate probability, 3 = high probability). A score >10 indicates excessive daytime sleepiness.
The SDS is a 20-item self-report questionnaire of the symptoms of depression. Subjects rate each item according to how they felt during the preceding 7 days. Item responses are ranked from 1 to 4. The sum of the 20 items produces a score ranging between 20 and 80, a value of >40 suggests clinically relevant depression.
The SAS measures affective and somatic symptoms of an anxiety disorder. The structure of the SAS is similar to that of the SDS. It also consists of 20 questions, which refer to the last seven days. The result of the SAS is obtained by summing up the scores. A value of >40 suggests the presence of a clinically relevant anxiety disorder.
In habitual snorers, a portable device (Stardust, Respironics/Philips Medcare/Habel Medizintechnik, Vienna, Austria) was used for monitoring blood oxygen saturation during sleep and recording the number and extent of nocturnal apnea/hypopnoea episodes and arterial oxygen desaturation during 1 night within the 90-day diary period. The Apnea/Hypopnea Index (AHI) was used for classifying the severity of OSAS. According to the American Academy of Sleep Medicine Task Force (17) an AHI of <5 h−1, 5–15 h−1, 16–30 h−1, and >30 h−1 indicate no, mild, moderate and severe OSAS, respectively.
Diagnostic criteria of sleep apnea headache proposed by the International Classification of Headache Disorders (ICHD-2, code 10.1.3).

Statistical analyses
Data are presented as total numbers and percentages. Continuous variables are given as mean and standard deviation (SD). Baseline demographic and baseline headache data were compared between snorers and their bed partners using t-tests, Chi2 tests or Fisher exact tests. To investigate differences between patients and bed partners during the 90-day diary period with regard to subjective sleep parameters, sleep complaints and clinical characteristics of MH, linear or various types of logistic regression models for repeated measures data were calculated.
Influence factors for MH used in the logistic regression models.

BMI: body mass index, AHI: Apnea/Hypopnea Index, SDS: Self-report Depression Scale, SAS: Self-report Anxiety Scale, PSQI: Pittsburgh Sleep Quality Index.
Results
Demographic data of snorers and bed partners.

f: female, m: male, BMI: body mass index, SD: standard deviation. p-values in bold are statistically significant.
Baseline headache characteristics
Patient-reported physician headache diagnoses as assessed in the semistructured interview comprised migraine in six (8%), tension-type headache in eight (11%), another type of headache in two (3%) and no headache in 60 (79%) snorers. Among bed partners seven (17%) gave migraine, seven (17%) tension-type headache, one (2%) another type of headache and 26 (63%) did not report a previous headache diagnosis. None of the snorers or bed partners gave a pre-study headache frequency of 15 days per month or more.
Baseline questionnaires
PSQI scores were significantly higher in bed partners than in snorers (6.3 ± 3.6 vs. 4.6 ± 2.3, p = 0.008). ESS scores were significantly higher in snorers than in bed partners (9.0 ± 3.5 vs. 7.4 ± 3.6, p = 0.03), whereas HIT-6 (46 ± 9 vs. 49 ± 9, p = 0.09), SDS (36 ± 11 vs. 34 ± 8, p = 0.3) and SAS scores (34 ± 11 vs. 36 ± 9, p = 0.6) did not differ between snorers and bed partners.
Apnea screening
In snorers, the mean AHI was 9.7 ± 13.5 (range 0–65.2). The mean nocturnal arterial oxygen saturation was 96 ± 1% and the mean lowest nocturnal arterial oxygen saturation was 85 ± 6%. Forty-four (58%) snorers did not suffer from OSAS (AHI <5), 20 (26%) suffered from mild OSAS, four (5%) from moderate OSAS and eight (11%) from severe OSAS.
Headache diary
In total, we analysed 6690 days of 76 snorers (25 female) and 3497 days of 41 bed partners (31 female). On average, snorers completed 88 ± 11 diary days and bed partners 85 ± 16 diary days.
MH characteristics
Headache of any time of onset was recorded on 760 diary days (7%), that is, on 529 days (8%) in snorers and on 231 days (7%) in bed partners. Headache occurring exclusively during daytime, at bedtime or during the night was recorded on 290 (4%), 17 (0.3%) and 17 (0.3%) vs. 164 (5%), 18 (0.5%) and 11 (0.3%) days in snorers and bed partners, respectively. MH was recorded on 572 days (5.6%), that is, on 454 days (7.2%) in snorers and on 118 days (3.6%) in bed partners. Analysis of whether MH was related to headache during the preceding 24 hours showed no headache, headache not persisting until bedtime and headache at bedtime on 205 (45%), 242 (53%) and 7 (2%) respectively, of all MH days in snorers, and on 38 (32%), 74 (63%) and 6 (5%), respectively, of all MH days in bed partners.
Forty-three (57%) snorers and 25 (61%) bed partners documented at least 1 day with MH. The mean number of days with MH was 11 ± 14 (range 1 to 83 days) in snorers and 5 ± 6 (range 1 to 28 days) in bed partners.
The onset of MH was recorded 85 ± 101 minutes (median 45) and 45 ± 60 minutes (median 26) after awakening in snorers and bed partners. The vast majority of MH was present on awakening or started within 1 hour after awakening (Figure 1). The onset of MH could not be identified owing to missing diary entries on 142 (31%) and 37 (32%) days in snorers and bed partners, respectively.
Onset of MH after awakening. Columns represent the percentages of MH present upon awakening, 1–15 minutes, 16–30 minutes, 31–45 minutes, 46–60 minutes and more than 60 minutes after awakening in snorers (black columns) and bed partners (grey columns).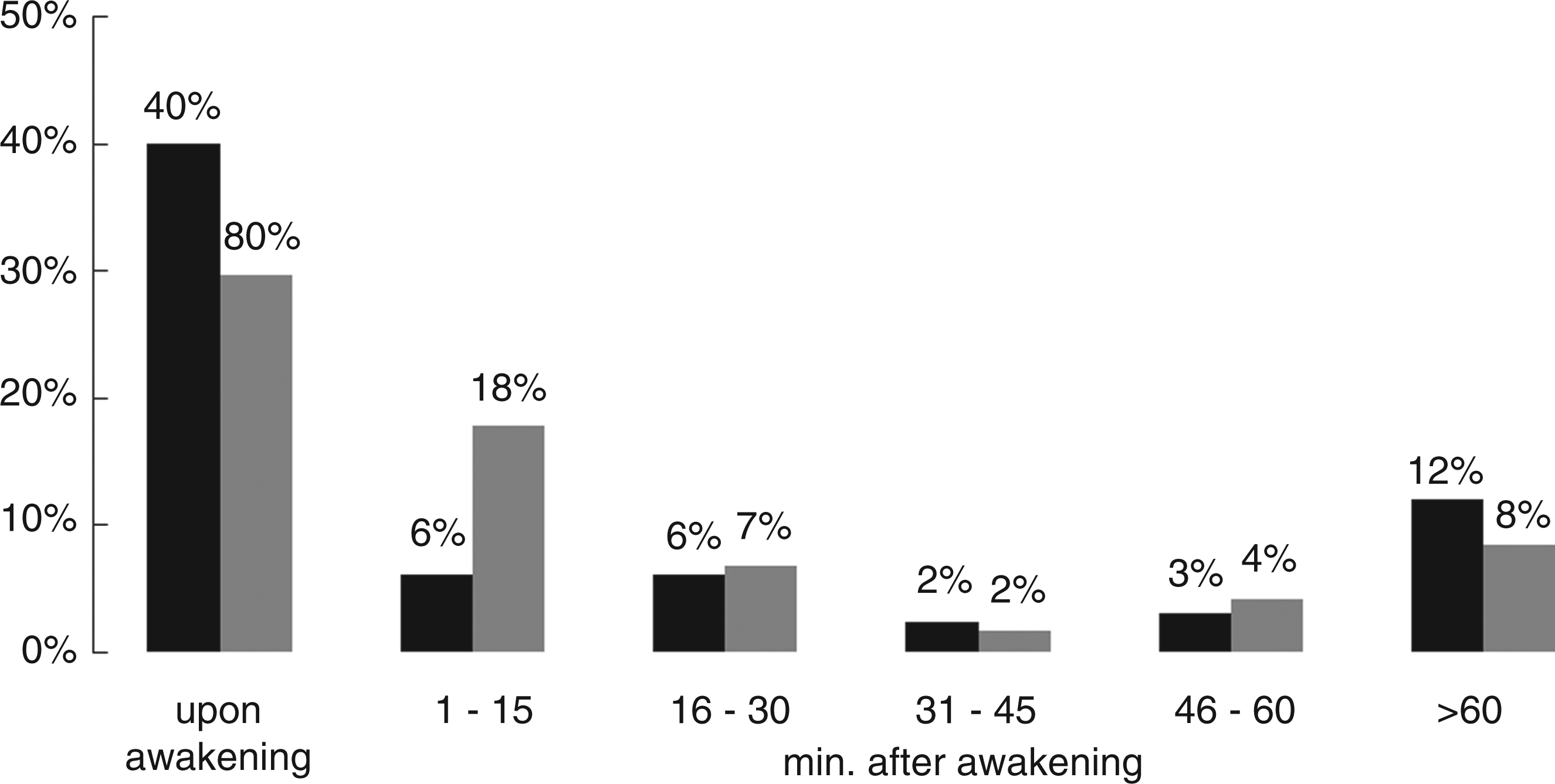
Clinical characteristics of morning headache (MH) recorded in the diary by snorers and bed partners.

TTH: tension-type headache, SD: standard deviation. p-values in bold are statistically significant.
When testing MH in snorers for the diagnostic criteria of sleep apnea headache (Table 1), MH fulfilled criteria A2, A3 and C only on one single day in one snorer without OSAS. All remaining MH in snorers did not fulfil even these criteria.
Subjective sleep parameters
Subjective sleep parameters and sleep complaints recorded in the diary by snorers and bed partners.

SD: standard deviation. p-values in bold are statistically significant.
Predictors for MH in habitual snorers and bed partners
Multivariate regression analyses showed that the self-reported pre-study headache frequency reported during the semistructured interview (p < 0.001), an anxiety disorder (p < 0.001), waking because of pain (p = 0.002) and waking up too early (p < 0.001) during the night before MH were independent predictors for MH in habitual snorers and migraine (p = 0.02), difficulties breathing during sleep (p < 0.001), waking up because of pain (p = 0.01) and waking up too early (p < 0.001) during the night before MH in bed partners.
The analysis of couples only (n = 41) revealed pre-study headache frequency (p < 0.001), waking up too early (p < 0.001) and nocturnal confusion (p < 0.001) as independent predictors of MH in snorers; and migraine (p = 0.02), difficulties breathing during sleep (p < 0.001), waking up because of pain (p = 0.01) and waking up too early (p < 0.001) in bed partners.
Discussion
This is the first study prospectively investigating the prevalence and characteristics of MH in habitual snorers and their bed partners. The 90-day MH prevalence was approximately 60% snorers and in bed partners. The vast majority of MH was not preceded by headache at bedtime, but 53% of the MH days in snorers and 63% of those in bed partners were preceded by headache during the previous day not persisting until bedtime. According to ICHD-II, MH most commonly resembled TTH and not one MH episode fulfilled the criteria of sleep apnea headache. Predictors of MH were related to sleep disturbance or insomnia, pre-study headache frequency and psychiatric comorbidity.
In a previous retrospective study, MH was reported by 23.5% of habitual snorers with and without OSAS (10). Göksan et al. found MH to be present in 33.6% of polysomnography-confirmed snorers with OSAS compared with 8.9% in controls (7). The prevalence of MH in our group of habitual snorers was 1.5–2 fold higher and the prevalence of MH in bed partners of habitual snorers was much higher than the prevalence reported in the general population (4). The prospective diary-based study design enabled us to also detect MH in subjects with rare MH which may not be recalled in a retrospective assessment.
Previously, MH has been reported predominantly as mild, pressing-tightening in quality, and lasting <30 minutes (6,7,18). In our study, MH more often showed pulsating quality and moderate intensity. Recent data suggested that MH in OSAS patients may last 1–4 hours (7). MH recorded by the habitual snorers in our study lasted even longer.
Due to our diary-based approach we were able to classify each single episode of MH according to ICHD-II (12). MH fulfilled the criteria of (probable) migraine and (probable) tension-type headache in 12% and 74% in snorers and in 14% and 68% in bed partners. All snorers and bed partners had at least one episode of MH classified as TTH and, surprisingly, more than 50% had at least one episode classified as (probable) migraine. In a previous study in habitual snorers MH fulfilled ICHD-2 criteria of migraine in 19% of the patients (10). Unfortunately, these authors (10) did not present data regarding the prevalence of probable migraine in habitual snorers.
In contrast to all previous studies (5,7,10,19), we did not focus only on MH but asked the participants to record the occurrence of headache at any time of the day. Accordingly, we could demonstrate that more than 50% of the MH episodes were preceded by headache on the previous day, albeit this headache did not persist until bedtime. Thus, we could show that MH frequently represents recurrence of diurnal headache on the day before and that ‘true’ MH (MH not preceded by headache during the previous 24 hours) was present in only 45% (snorers) and 32% (bed partners) of the MH episodes. It is commonly accepted that migraine and tension-type headache attacks have to be preceded by ≥24 hours without headache to be acknowledged as new onset headache. The majority of MH attacks were either classified as (probable) migraine or (probable) tension-type headache. Thus, we felt that it was important to differentiate between ‘true’ MH and MH most likely resembling a relapse of headache experienced during the previous day.
Previously, migraine, insomnia, psychological distress and OSAS have been identified as independent predictors of MH in habitual snorers (10). Two recent studies reported an association between MH and the severity of OSAS (7,19). In contrast, we did not find the severity of OSAS (the AHI) to be an independent predictor of MH in habitual snorers, which is in line with the data of a large population-based study (19). Our results emphasize the relationship between self-reported headache frequency, a pre-existing anxiety disorder, insomnia symptoms and MH in habitual snorers. In a large population-based study the prevalence of MH was 7.6% (4) and MH were significantly associated with age, anxiety and depressive disorders, insomnia and circadian rhythm disorder (4). In accordance with Ohayon et al. (4) difficulties maintaining sleep was a predictor for MH in bed partners. In addition, in our group of predominantly female bed partners, migraine was an independent predictor of MH. Most recently, a large study found migrainous and non-migrainous headaches to be associated with insomnia symptoms (20). We also analysed the snorer-induced sleep disturbances in bed partners and did not identify any of them as an independent predictor of MH in bed partners.
Our study is strengthened by its prospective design, the use of a combined headache and sleep diary, the classification of MH according to ICHD-II and the inclusion of bed partners of habitual snorers. The results of our study are limited by the lack of a control group, that is, a group of bed partners with non-snoring spouses. The sleep disturbance caused by the snoring partner may have induced a negative bias regarding the quality of sleep and thus the frequency of MH in bed partners. We also did not screen bed partners for OSAS. This might have induced a certain bias to the relationship between the nocturnal oxygen saturation and the occurrence of MH.
In conclusion, MH is more prevalent in both snorers and their bed partners than previously reported. The majority of MH can be classified as TTH and the mean frequency is once a week. Insomnia symptoms are the most prominent independent predictors of MH in snorers and bed partners.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interests
CW has received honoraria and travel funding from Allergan, AstraZeneca (Austria), Hermes Arzneimittel GmbH, Linde Gas (Austria), A. Menarini Pharma GmbH (Austria) and Pfizer (Austria). The other authors declare that they have no conflict of interest.
