Abstract
Objective
Ketorolac is a non-triptan, non-opioid, mixed cyclooxygenase (COX)1/2-inhibitor for short-term management of moderate-to-severe acute pain. This trial evaluated an intranasal formulation of ketorolac tromethamine (SPRIX®) containing 6% lidocaine (ROX-828) for the acute treatment of migraine with and without aura as defined by the International Headache Society.
Methods
Patients were randomly assigned 1:1 to self-treat with intranasal ROX-828 (31.5 mg ketorolac tromethamine/200 µL, containing 6% of lidocaine) or placebo (with 6% lidocaine) within four hours of a new migraine attack rated ≥ moderate in pain intensity. Assessments included headache intensity and associated migraine symptoms (nausea, vomiting, phonophobia, photophobia) measured at baseline and at regular intervals through 48 hours post-dosing, and global impression of efficacy (seven-point scale) measured at two hours.
Results
Randomized patients who had a migraine attack (N = 140) were evaluable (ROX-828, N = 68; placebo, N = 72). Patients receiving ROX-828 showed a significant (p < 0.05) improvement in pain relief at all time points except 0.5 and 24 hours compared with those who received placebo. More patients achieved pain-free status with ROX-828 than with placebo at 1.5, 3, 4, 24 and 48 hours (p < 0.05); significance at the two-hour time point, which was the primary endpoint, was not met. Patients’ global impression of efficacy showed statistically significantly better results for patients receiving ROX-828 than for those receiving placebo. Associated migraine symptoms were significantly improved (p < 0.05) with ROX-828 relative to placebo at several time points throughout the observation period. The most frequently reported adverse events in both groups were associated with nasal discomfort.
Conclusion
Self-administered intranasal ROX-828 was well tolerated. While the primary endpoint was not met, the results provide preliminary evidence that ROX-828 improves migraine pain.
Introduction
Routes of administration that avoid the gastrointestinal tract and that have high bioavailability may offer significant advantages to oral migraine medications. Nausea and vomiting during a migraine can be particularly severe and incapacitating, and can be among the most problematic symptoms experienced by migraineurs (1,2). Ninety percent of migraineurs have experienced nausea during migraine episodes as determined by patient survey (3). For the majority of migraine occurrences, a significant percentage are associated with nausea (52%) and vomiting (22%) (3). Migraine-related nausea can cause patients to delay or avoid taking oral medication, resulting in decreased therapeutic efficacy (3). The gastric stasis in migraineurs (4) can also affect drug absorption after oral medications (5,6) and result in compromised therapeutic efficacy.
Intranasal formulations of migraine medications, including triptans (5-HT1 receptor agonists), are currently available for use when oral treatment is not desirable (7). Although triptans are widely used for migraine, non-triptan intranasal drugs that provide rapid relief of migraine symptoms are needed when triptans should be avoided (e.g. patients with vascular disease, uncontrolled hypertension or hemiplegic migraine) or are ineffective. For example, in patients who experience allodynia, non-triptan options are used when additional pain medication is necessary (8).
Intranasal ketorolac (ketorolac tromethamine, SPRIX®) is a non-opioid, mixed COX1/2-inhibitor non-steroidal anti-inflammatory drug (NSAID) approved in adults for short-term (up to five days) management of moderate-to-moderately-severe pain that requires analgesia at the opioid level. The pharmacokinetics of intranasal ketorolac are similar to those following intramuscular ketorolac administration (9). Intravenous ketorolac has shown promising efficacy in the acute treatment of migraine in a small comparative study against intranasal sumatriptan (10), but no clinical studies have been conducted with intranasal ketorolac in migraineurs. The intranasal formulation is well tolerated. The most frequent adverse events in clinical trials were transient episodes of nasal burning or stinging, the majority of which were rated mild in severity (11–14). Comparison of intranasal formulations of ketorolac with and without lidocaine indicated that the addition of low concentrations of lidocaine (5% or 6%, but not 4%) significantly decreased the time to Tmax without affecting the Cmax, and in addition improved local tolerability (data on file, Luitpold Pharmaceuticals). The objectives of the present study were to evaluate the efficacy, safety and tolerability of a single dose of intranasal ketorolac tromethamine (SPRIX®) containing 6% lidocaine (ROX-828) in patients with migraine.
Methods
Patients were enrolled in a phase 2, multicenter, double-blind, placebo-controlled study (Protocol ROX-2007-01; registration number for the EU Clinical Trials Register: 2007-001431-58) evaluating the efficacy and safety of ROX-828 in patients with a history of recurrent migraine. Enrollment was from September 2007 to February 2008 at 16 centers in Germany (10 sites) and Finland (six sites). The study protocol and the informed consent form were approved by the institutional review boards of each participating center (German sites: Ethik-Kommmission der Bayerischen Landesärztekammer; Finnish sites: Helsinki and Uusimaa Hospital District/Ethics Committee for Obstetrics and Gynecology, Otology and Ophthalmology, Neurology and Neurosurgery). The trial was conducted in accordance with the ethical standards of all responsible institutional and national committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2000. All enrolled patients provided informed consent.
Men and women 18–65 years old were eligible if they had a history of migraine with or without aura according to International Headache Society (IHS) criteria (15), onset of migraine history prior to age 50 years and between two and eight moderate or severe migraine headaches per month. Use of migraine prophylactic drugs was permitted if used at stable doses for at least 12 weeks prior to study entry. Patients were excluded if they had a medical or psychiatric condition, or circumstance which, in the opinion of the investigator, would adversely affect their participation in the trial. Other exclusion criteria were continuous use of aspirin or NSAIDs, and use of another NSAID for the migraine attack for which study drug was used.
During the screening visit, patients underwent medical history and clinical evaluations. Patients meeting entry criteria were randomized 1:1 to the ROX-828 or IN placebo groups using a blocked randomization schedule within each investigative site. The ROX-828 and placebo nasal solutions were provided in disposable, multidose, metered-spray devices that were identical in appearance. All doses were administered as one spray containing 100 µL into each nostril (i.e. one spray into the left nostril and one spray into the right nostril). Each spray of ROX 828 delivered 15.75 mg of ketorolac tromethamine, for a total dose 31.5 mg. Both ROX-828 and the placebo control also contained 6% lidocaine, 0.02% sodium ethylene diamine tetraacetic acid (EDTA) and water.
Patients were instructed to use study medication within four hours of a new migraine attack that was rated ≥ moderate in pain intensity. Patients were to use study medication as the first treatment and not to use rescue medication until at least two hours after taking study medication. Patients recorded the time and date of onset and characteristics of the attack (headache severity, aura, nausea, vomiting, photophobia/phonophobia, clinical disability). Once an attack was treated, patients recorded responses over 48 hours using diary cards. Patients had a follow-up visit within seven days of treatment for physical examination, hematology and serum chemistry evaluations and questioning regarding adverse events and concomitant medication use.
Efficacy analyses were performed on the intent-to-treat (ITT) population, defined as patients who took study medication except those who did not use study medication as the first treatment, or who did not have a time of dosing. The primary endpoint of the study was the proportion of patients who became pain-free (i.e. a grade 0 on the (IHS) scale where 0 = no pain, 1 = mild pain, 2 = moderate pain and 3 = severe pain) two hours after dosing.
Secondary endpoints included presence or absence of migraine-associated symptoms (nausea, vomiting, photophobia and phonophobia), meaningful pain relief (defined as a reduction of a moderate or severe headache to mild or none), freedom from pain (defined as a reduction in pain to none), global impression of efficacy (measured two hours after taking study medication using a seven-point scale where 1 = very much better, 2 = much better, 3 = a little better, 4 = no change, 5 = a little worse, 6 = much worse and 7 = very much worse), time to rescue medication use (recorded on the subject diary card) and clinical disability (rated by the patient using a four-point scale where 0 = none, 1 = mild, 2 = moderate and 3 = severe). Times to meaningful pain relief, freedom from pain and use of rescue medication were determined using Kaplan-Meier time-to-event analyses.
Safety analyses were performed for all subjects who self-treated with study medication (safety population). Safety assessments included reported and observed adverse events, and results of physical examinations, vital sign measurements, hematology, blood chemistry and urinalysis.
Statistics
A sample size of 65 provided 90% power based on a two-sided test at the 5% level of significance. All continuous variables were summarized using descriptive statistics including number, mean, standard deviation (SD), median, maximum and minimum. All categorical variables were summarized using frequency counts and percentages.
The primary variable, the proportion of subjects pain-free two hours post-dosing, was compared between the active and placebo groups using an asymptotic Pearson’s Chi-square test, based on a two-sided 5% level of significance using the ITT population. Descriptive statistics were provided for secondary variables and the Pearson’s Chi-square test compared proportions of subjects between treatment groups. Ninety-five percent confidence intervals for the differences between groups in freedom from pain and pain relief were calculated. The 95% confidence intervals have not been corrected for multiplicity and should be viewed as descriptive, as statistical analysis of secondary endpoints was considered exploratory. Clinical disability was compared between the placebo and active treatment group using the asymptotic Pearson’s Chi-square test (without continuity adjustment). Time-to-event analyses using Kaplan-Meier methods were compared with the log-rank test.
Results
Screening of 176 patients resulted in randomization of 173 patients to the ROX-828 (N = 87) and placebo (N = 86) groups. The disposition of subjects is summarized in Figure 1. The number of patients with migraine who self-treated with study medication was 142 (safety population), and 140 patients were evaluable for efficacy analysis: 68 in the ROX-828 group and 72 in the placebo group (ITT population). The majority of patients who used study medication completed the study (136/142, 96%).
Patient disposition.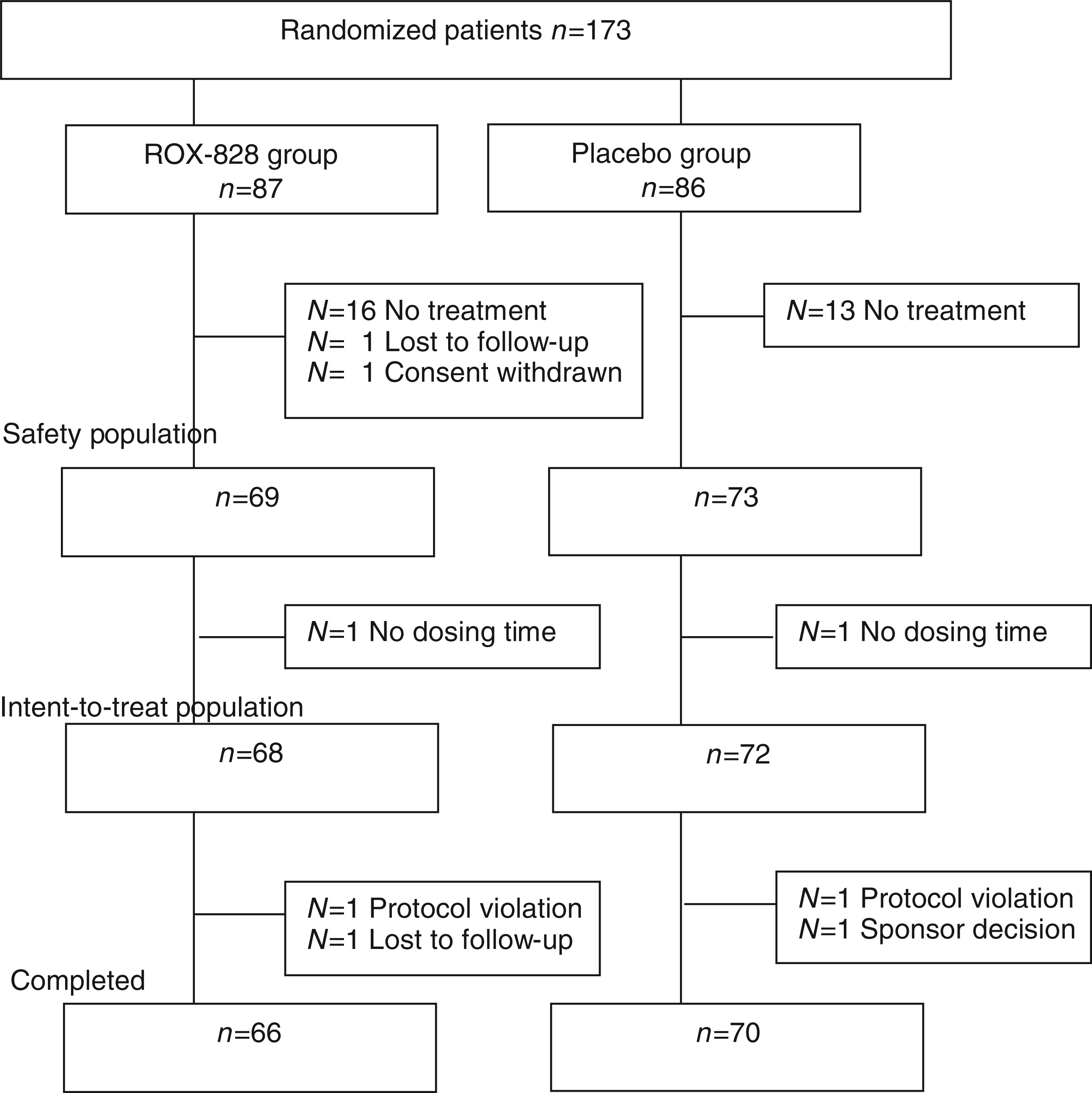
Demographics and migraine characteristics of patients (intent-to-treat population).

SD: standard deviation.
Efficacy
The proportions of patients with freedom from pain (IHS scale grade 0) and meaningful pain relief (IHS scale grade 0 or 1) from 30 minutes to 48 hours after intranasal dosing with ROX-828 or placebo are shown in Figures 2A and B. Statistically significantly more subjects achieved freedom from pain with ROX-828 treatment than with placebo at 1.5, 3, 4, 24 and 48 hours post-dosing (Figure 2A). Confidence intervals for the difference between groups at these time points did not include zero, consistent with the treatments being different. Although statistical significance at two hours (primary endpoint) was not reached, the proportion of patients free of pain was greater in the ROX-828 group compared with placebo: 18% (12 patients) versus 10% (seven patients), p = 0.17. The proportion of subjects with meaningful pain relief was larger in the ROX-828 than placebo group at all time points, and the differences were statistically significant at all time points except 0.5 and 24 hours post-dosing (Figure 2B). Confidence intervals for the difference between groups at all time points except 0.5 and 24 hours also did not include zero and are consistent with the treatments being different. At two hours post-dosing, 51.5% of ROX-828 treated and 31.9% of placebo-treated subjects presented with pain relief.
(A) Proportion of patients with freedom from pain (International Headache Society grade 0) and (B) meaningful Pain Relief (IHS grade 0 or 1) versus time after dosing in the ROX-828 and placebo groups. p values and 95% confidence intervals for differences between ROX-828 and placebo are shown in the side bar. *p < 0.05, **p < 0.01. CI: confidence interval.
A Kaplan-Meier plot for time to meaningful pain relief (as reported in the subject’s diary) is shown in Figure 3A. Overall, time to meaningful pain relief was shorter in the ROX-828 group as compared to the placebo group. The log-rank test revealed a statistically significant difference between the meaningful pain relief curves of the two groups (p = 0.008). Median times to pain relief were 3.6 hours for ROX-828 and 11.3 hours for placebo. The time to freedom from pain also occurred more rapidly after dosing with ROX-828 than with placebo, with median times of 7.7 and 18.9 hours in the ROX-828 and placebo groups, respectively.
Kaplan-Meier distribution analysis of (A) time to meaningful pain relief and (B) time to rescue medication use following dosing.
Kaplan-Meier analyses for time to use of rescue medication are shown in Figure 3B. Overall, time to rescue medication was shorter in the placebo group as compared to the ROX-828 group and there was a statistically significant difference between the use of rescue medication curves of the two treatments (p = 0.005). The median time to rescue medication was 17.7 hours in the ROX-828 group and 3.8 hours in the placebo group. The most commonly used (used by at least 4% of patients) types of rescue medication were similar in the two groups, and overall were triptans (62%), followed by anilides (14%), propionic acid derivatives (8%) and salicylic acid derivatives (4%).
The percentage of patients with nausea, vomiting, photophobia and phonophobia after dosing with ROX-828 or placebo is shown in Figure 4. Patients in the ROX-828 group had numerically fewer migraine-related symptoms at all time points for all symptoms. Statistically significant differences between the ROX828 and placebo groups occurred at three hours and later for nausea and vomiting and four hours and later for photophobia.
Percentage of patients with common migraine symptoms of nausea, vomiting, photophobia and phonophobia from 30 minutes to 48 hours after dosing with ROX-828 or placebo. Significant difference between groups *p < 0.05, **p < 0.01, ***p < 0.001.
Compared with placebo-treated subjects, subjects treated with ROX-828 showed a statistically significant improvement in the severity of clinical disability from 1.5 hours (p = 0.02) through 48 hours (p = 0.02) post-dosing. Clinical disability at dosing and at two hours post-dosing is shown in Figure 5. At dosing, clinical disability was comparable between treatment groups. At two hours post-dosing, 24% of ROX-828-treated and 10% of placebo-treated subjects reported no clinical disability (p value for treatment differences p = 0.009). From dosing until two hours post-dosing, the proportion of subjects in the ROX-828 group reporting no clinical disability increased while the proportion of subjects reporting severe disability decreased. In the placebo group the proportion of subjects with no clinical disability remained rather constant, and the proportion with severe disability increased. The proportion of subjects with mild disability remained stable in both groups during the two hours. After two hours post-dosing, the use of rescue medication was allowed, making data less interpretable.
Clinical disability at dosing (time 0) and two hours post-dosing in the (A) ROX-828 and (B) placebo groups. Proportions were similar at time 0 in the two groups. At two hours, the proportion of subjects in the ROX-828 group reporting no clinical disability increased while the proportion of subjects reporting severe disability decreased. In the placebo group, the proportion of subjects with no clinical disability changed only slightly, and the proportion with severe disability increased. The proportion of subjects with mild disability remained stable in both groups during the two hours. The proportion of subjects with moderate disability decreased to a similar degree in both groups during the two hours. The p value for differences in treatment between ROX-828 and placebo at two hours is 0.009.
Patients’ global impression of efficacy at two hours post-treatment is shown in Figure 6. Improved efficacy (very much better and much better on the seven-point scale) was reported by 40% of ROX-828 - and 18% of placebo-treated patients, and the difference between treatments was statistically significant (p = 0.008, Chi-square; p < 0.0001, rank-sum).
Patients’ global impression of study medication efficacy at two hours post-dosing. Improved efficacy was defined as patients reporting much better or very much better efficacy on the seven-point scale. The effect was significant between the treatment groups (p = 0.008 Chi-square; p < 0.0001 rank-sum).
Safety
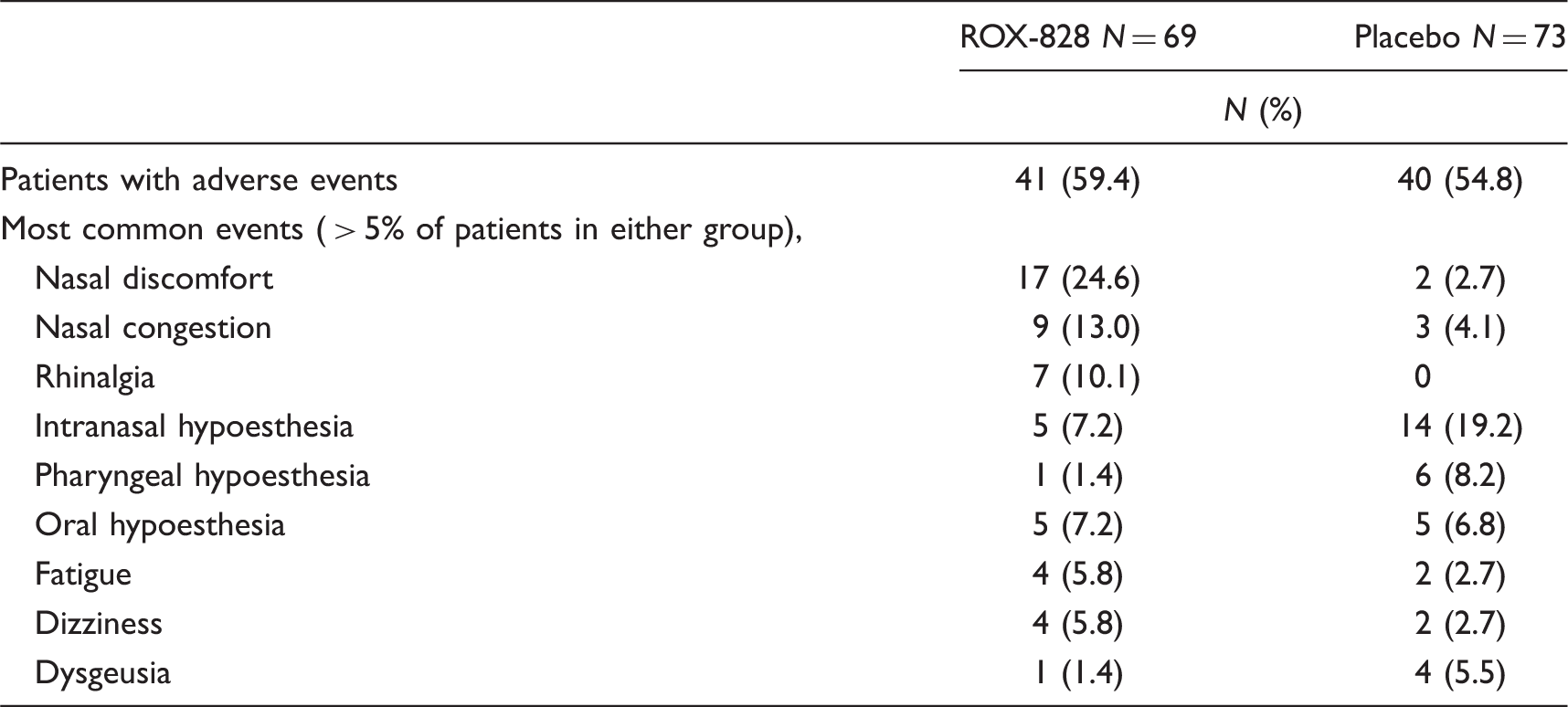
Treatment emergent adverse events occurring within the 48-hour observation period in the ROX-828 or placebo groups (safety population).
Discussion
The results of this double-blind, placebo-controlled study indicate that intranasal ROX-828 (ketorolac tromethamine, SPRIX® containing 6% of lidocaine) is effective in the treatment of acute migraine. Although several studies have shown that intravenous ketorolac tromethamine is effective in reducing headache pain (10,16,17), this is the first study to assess the effect of intranasal ketorolac tromethamine on migraine pain. The onset of efficacy of intranasal ketorolac in treating acute migraine associated with moderate-to-severe headache pain was very rapid, and the differences in response rates between the ROX-828 and placebo groups at 1.5 hours were comparable to those observed in placebo-controlled trials with triptan therapy at two hours (18,19).
Although the primary variable, freedom from pain at two hours post-dosing, did not achieve statistical significance, substantially more ROX-828-treated patients achieved a pain-free status two hours post-dosing compared to the placebo group. In general, the times to freedom from pain and meaningful pain relief were shorter with ROX-828 than placebo treatment, beginning at one hour and persisting over 48 hours. Although the study was not powered to determine significance of secondary endpoints, confidence intervals for the differences in freedom from pain and pain relief between groups at the majority of time points did not include zero, consistent with the treatments being different. The 95% confidence intervals have not been corrected for multiplicity and should be viewed as descriptive, as all secondary variables were considered exploratory. Effective and realistic adjustments for multiplicity are based on the complete set of comparisons that were made. The complete set of comparisons was not defined prospectively, and thus any multiplicity correction at this time would be post hoc.
Subjects participating in this study had headache characteristics reflective of a typical population of migraineurs (20). Headaches were associated with moderate disability and associated symptoms of nausea/vomiting, photophobia and phonophobia. Symptoms other than headache pain contribute significantly to disability caused by acute migraine (1). Both disability and health-related quality of life worsen with increasing migraine symptom severity (21). Significant improvements in the ROX-828 group versus placebo were seen in patients’ global impression of efficacy at two hours, in clinical disability beginning at 1.5 hours and throughout the study, and in migraine-associated symptoms, including nausea and vomiting beginning at three hours.
The use of rescue medication was allowed after two hours post-dosing, thus differences between the placebo and ROX-828 groups in improvements in related symptom data were more difficult to demonstrate, but results trended toward improvements in the ROX-828 group for most symptoms. The mean time for use of rescue medication in the placebo group was between three and four hours, and indeed, the proportion of patients with improvements in associated symptoms increased in the placebo group after three hours. However, there were consistent trends or significant differences in the ROX-828 group compared to placebo for the majority of time and endpoints even with rescue medication use in the placebo group. Patients in the ROX-828 group had significantly longer times to use of first rescue medication (mean 17 hours) after dosing compared to placebo (mean 3.8 hours).
Both ROX-828 and the placebo control contained lidocaine. Several studies have shown that lidocaine may provide relief in acute migraine attacks, (22–24) although there are conflicting data (25) and large, controlled studies are lacking (26). While we cannot rule out that the presence of lidocaine had an effect on pain relief, the ROX-828 group showed consistently better efficacy that was significantly different from that of the placebo group.
ROX-828 treatment was well tolerated and most adverse events were mild or moderate in severity. The most common adverse event was transient nasal discomfort that was not associated with abnormalities of the anterior nares. Nasal discomfort is a frequently reported event with nasally administered medications from a variety of different classes (7,27). While migraine patients may be particularly sensitive to stimuli such as this, in previous studies of intranasal ketorolac tromethamine, mild nasal irritation was of short duration and was not associated with any changes in nasal mucosa (11–14). Nasal discomfort was observed in the ROX-828 group despite the presence of lidocaine. Hypoesthesias attributable to the presence of lidocaine occurred in both the ROX-828 and placebo groups, and intranasal and pharyngeal hypoesthesia occurred at a greater incidence in the placebo group.
Current acute treatments for migraine management include analgesics, including oral NSAIDs and migraine-specific abortive treatments such as oral and non-oral triptan formulations. New treatment options are still needed when oral medications and/or triptan therapy are not preferred or are contraindicated. Parenteral ketorolac is widely used to treat moderate-to-severe pain. Parenteral ketorolac is effective for treating migraine (10), and is used as a first-line option for managing migraine in the emergency department (28,29), particularly for patients with allodynia (16) or for whom triptans are not appropriate. A recent study showed that 50% of patients with migraine seen in the emergency department had contraindications that prevented them from receiving triptans (29). Thus, an intranasal ketorolac tromethamine formulation may be useful in any setting when triptans fail or are not appropriate, and in emergency departments to avoid use of parenteral ketorolac.
Conclusion
This is the first study to assess a single dose of ROX-828 (the intranasal formulation of ketorolac tromethamine, SPRIX®, containing 6% of lidocaine) in reducing migraine pain. While statistical significance for the primary endpoint of freedom from pain at two hours was not reached, the data provide sufficient preliminary evidence that self-administration of intranasal ROX-828 can relieve migraine pain and these results warrant further investigation. Intranasal ROX-828 was well tolerated and nasal discomfort was the most common adverse event. Gastrointestinal tract absorption is often significantly delayed during the acute phase of a migraine attack, and nausea and vomiting can be prominent features of migraine attacks. ROX-828 avoids the gastrointestinal tract and has good bioavailability that may be advantageous in the acute management of migraine.
Footnotes
Funding
Funding for the study, including medical writing assistance, was provided by Roxro Pharma, a subsidiary of Luitpold Pharmaceuticals (Shirley, NY, USA). Medical writing and editing support was provided by Patrice Ferriola.
Acknowledgements
We thank Drs. Diego Provvedini and Alberto Tajana of Recordati Industria Chimica e Farmaceutica for critical review of the manuscript.
Declaration of competing interests and financial disclosure
V. Pfaffenrath, E. Fenzl, and M. Färkkila participated as clinical investigators for the study. D. Bregman is an employee of Luitpold Pharmaceuticals.
