Abstract
Intranasal corticosteroids (INCs) act predominantly locally and are considered to exert minimal systemic effects. On reviewing the international data collected in the World Health Organization's global pharmacovigilance programme an unexpected cluster was found of 38 case reports of migraine in suspected connection with INCs. These reports came from five countries (May 2007) and concerned six different drugs. In all reports the INC was the sole suspect drug. In nine cases re-exposure to the drug had taken place, leading to the recurrence of the event in eight of these patients. However, INCs are mainly used for rhinitis, and there is a known connection between rhinitis and migraine. Although representing only 0.6% of the total of case reports, international pharmacovigilance data suggest that the use of INCs may cause or trigger migraine or migraine-like headache. Further study is needed to determine if the reported association is true or not and, if so, what the possible mechanism is.
Introduction
Intranasal corticosteroids (INCs) act predominantly locally and are considered to exert minimal systemic effects (1–3). Nevertheless, in rare cases symptoms of hypercorticism have occurred, including menstrual irregularities, acneiform lesions, cataract and cushingoid features, mainly when the recommended doses were exceeded or in individuals who were particularly sensitive or predisposed by virtue of recent systemic steroid therapy (4–7). In a previous study we have reported on the unexpected connection between the administration of INCs for rhinitis and the development of a variety of neuropsychiatric disorders (NPDs) (8). Whereas most of these NPDs are established adverse reactions to systemic corticosteroids, this is not the case for migraine or unilateral headache (9). We reviewed an unexpected cluster of case reports of migraine in occurring in suspected connection with the use of INCs, as stored into Vigibase, the international database of the World Health Organization (WHO) Uppsala Monitoring Centre (UMC).
Materials and methods
The UMC maintains the international pharmacovigilance programme of the WHO, in collaboration with 83 national pharmacovigilance centres around the world (10, 11). A primary aim of this programme is the early detection after the release of medicines of possible new adverse reactions and interactions. Yearly the UMC receives about 250 000 case reports of patients with suspected adverse drug reactions from the national centres that are stored in Vigibase, the central database of the UMC. The case reports are anonymous and heterogeneous and vary as regards source, documentation and relationship likelihood, and are stored in an electronic E2B compatible format, using the WHOART adverse reactions terminology and the WHO Drug Dictionary (12, 13). The WHO-UMC database (Vigibase) currently contains > 3.7 million case reports.
Study design
Case series.
Data selection
All case reports of single-ingredient INC products with reported nasal route of administration and WHOART preferred terms ‘Migraine’ and ‘Migraine aggravated’ were retrieved from Vigibase (May 2007). Duplicate case reports or reports with a miscoded route of administration were excluded. We reviewed case reports with respect to suspected drugs, nature of the events, presence of confounders, time relationship, prescribed dose and outcome.
Potential confounders
Age, sex, concomitant disease (allergic rhinitis, migraine), drug–drug interactions and comedication (known to cause migraine) disorders were considered as potential confounders.
Concomitant disease was identified by the relevant WHOART terms (e.g. migraine aggravated) or the use of concomitant medication for the treatment of headache or migraine. Drugs that are established to enhance INC effect include anti-infective drugs (macrolides, fluoroquinolones, antimycotics, protease inhibitors), calcium-channel blockers (diltiazem), gastrointestinal drug (aprepitant) and sex hormones (14).
Results
Vigibase contains a total of 6268 case reports concerning INCs (from 1981 to May 2007), including 36 reports of migraine and two of migraine-aggravated (WHOART/Medical Dictionary for Regulatory Activities terms) in association with INCs, together 0.6% of the total, coming from five different countries (Table 1). Characteristics of the case reports are shown in Table 2. In all reports the INC was the sole suspected drug and in 24 it was the only drug taken. The most frequently reported INC was fluticasone (n = 15) and beclomethasone (n = 14) (Table 2). Women represented 75% (n = 27) of case reports with reported gender information (n = 36). The mean age was 43.1 years (n = 24:
Reporting countries

Case reports’ characteristics
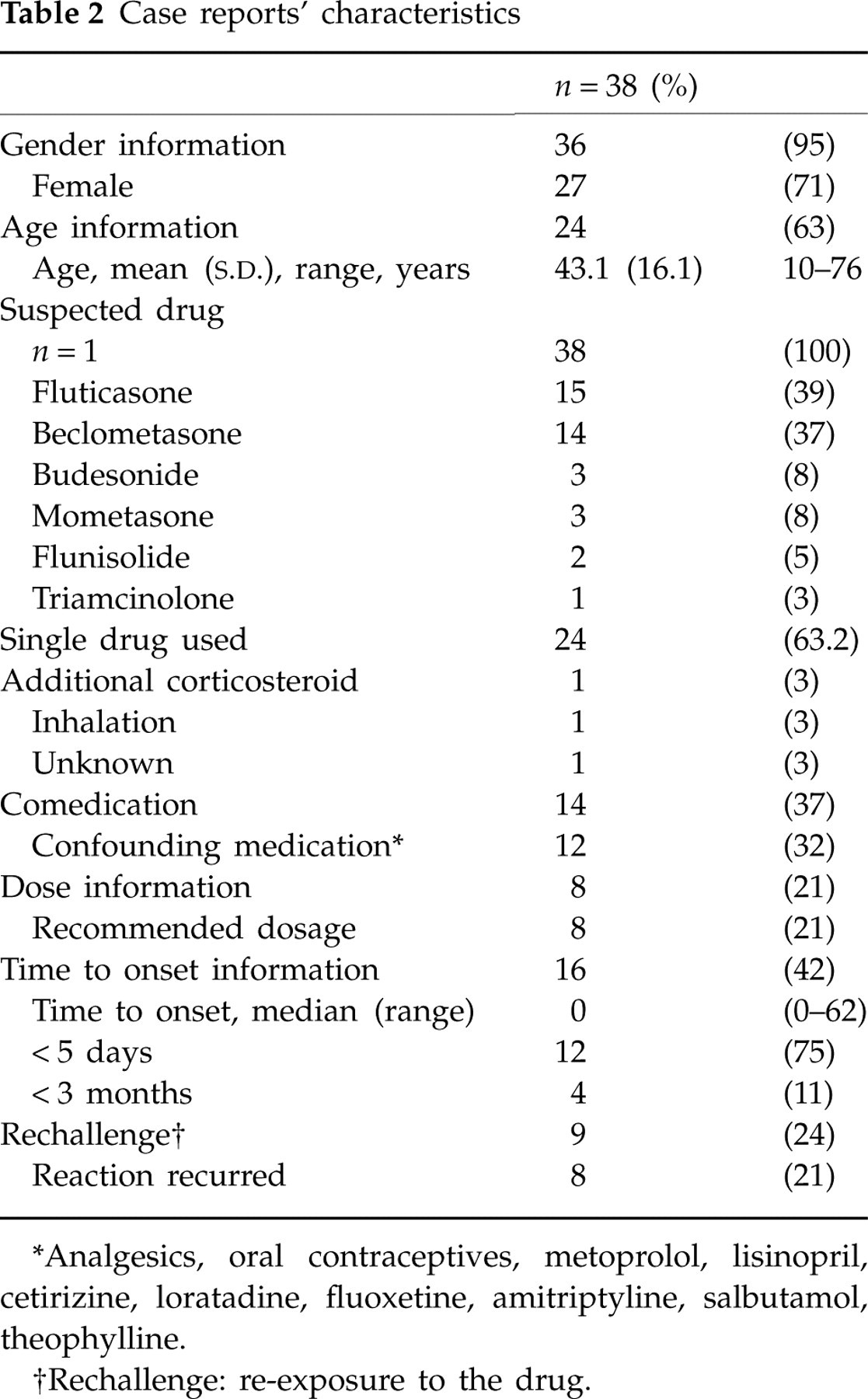
∗Analgesics, oral contraceptives, metoprolol, lisinopril, cetirizine, loratadine, fluoxetine, amitriptyline, salbutamol, theophylline.
†Rechallenge: re-exposure to the drug.
In 24 reports there was follow-up information given. In eight patients it was recorded that re-exposure to the INC had led to a relapse of migraine, including one of the reports of migraine aggravated. In 14 further reports the patient had recovered or improved. In two other reports, on the other hand, a ‘negative dechallenge’ was recorded. In only two cases had a causality estimation been attached by the reporting National Centre: ‘possible’ in one and ‘probable’ in the other.
In none of the case reports with recorded dose information did the daily dose exceed the maximum daily recommended dose range (n = 8). Migraine developed early in the course of INC treatment (within the first 4 days) in 12 of the 16 reports where the time to onset was recorded. In four other reports the data showed that migraine had occurred within the first 2 months of INC use. In 12 of 14 reports with listed comedication, a drug known to cause a headache/migraine and/or a drug indicated for the treatment of a headache/migraine had been taken. One of these patients had been using an ethinylestradiol/gestoden combination, which can potentiate the INC as well as trigger an attack of migraine. Six patients had co-reported adverse events that may be of interest: two had simultaneously rhinitis, two had sinusitis, two sinus pain and one epistaxis.
Discussion
Vigibase contains > 6268 reports of INCs, illustrating the huge world-wide use of these drugs. The 38 reports concerning migraine represent only a tiny subset of 0.6%. The reports in Vigibase are short in detail; usually the connection between drugs and events is uncertain, while a clinical appraisal and relationship-likelihood estimation of the cases is often difficult. Although there was a predominance of reports from the USA (28 cases), similar observations were made in four other countries.
It is noteworthy that in all 38 reports in this review the INCs had been the sole suspected drug, and an additional drug used for the treatment of headache/migraine and/or causing a migraine (paracetamol, acetylsalicylic acid, carbasalate, ethynilestradiol/gestoden, loratadine, cetirizine, lisinopril, amytriptyline, metoprolol, salbutamol, theophylline) was listed in only 12 cases. The distribution of the various INCs is likely to represent exposure figures in the first place.
Headache is commonly observed in clinical trials and mentioned as adverse reaction in the product information of all approved INCs, but migraine is not. However, in clinical trials with intranasal fluticasone, mometasone and triamcinolone the occurrence of headache was found to be comparable to that observed with placebo (9, 15–21). Women complained about migraine more frequently than did men in our study. This may reflect an overall higher prevalence of migraine in women compared with men (22).
As has been shown by Ku and co-workers, there is a strong connection between allergic rhinitis and the occurrence of migraine (23). According to this study > 34% of patients with allergic rhinitis experienced migraine headaches, compared with only 4% of people without allergic rhinitis. Another study by Aamodt et al. found migraine and non-migrainous headache to be approximately 1.5 times more likely among patients with current asthma, hey fever and chronic bronchitis (24). Neither study addressed a possible additional link with the treatment of rhinitis, however. Since allergic rhinitis is the major indication for INCs, all reports are potentially confounded by indication. In addition, selection may have taken place, since INCs may (erroneously) have been prescribed to patients with genuine migraine, which was misdiagnosed as rhinitis-induced headache.
One of several mechanisms that have been proposed to act in the development of migraine in patients with comorbid atopic disorders is involvement of the immune system with histamine release playing an important role. Elevated plasma IgE, histamine, decreased lymphocyte phagocytotic function and increased plasma tumour necrosis factor-alpha were found in subjects with migraine. It has been hypothesized that stress relief enhances immune activity and triggers a burst of circulating vasoactive compounds that function as mediators of inflammation and potential precipitators of a migraine attack in vulnerable subjects (25).
According to the review article by Trangsrud and colleagues, INCs commonly cause some form of irritation of the nasal mucosa (9). Nasal dryness, burning, stinging and sneezing together with headache and epistaxis occur in 5–10% of patients, regardless of the INC product used. It is noteworthy that there are altogether 895 reports of rhinitis or rhinitis ulcerative and 117 reports of sinusitis in suspected connection with various INCs in Vigibase. If INCs do play a role in the development of aggravation of unilateral ‘migraine-like’ headache, it may be more likely to be secondary to a localized INC-related process, i.e. in the nose or in the sinuses, rather than the induction of genuine migraine. The co-reported terms sinusitis, sinus pain, rhinitis and epistaxis together with migraine in six of the 38 reports may be indicative of such an underlying mechanism. Alternatively, intranasally administered corticosteroids might reach the central nervous system through olfactory neural and perineural transportation (26, 27).
There is a possible link between platelet function and migraine, and corticosteroids may have a prothrombotic action (28, 29). The systemic levels of corticosteroids after intranasal administration are low, however, and unlikely to lead to significant effects on the clotting and fibrinolytic cascades. A few case reports have been described of cerebral venous sinus thrombosis in connection with oral corticosteroid use, presenting as migraine-like headache (30). However, the occurrence of transient migraine is most unlikely to be to have been linked with cerebral thrombosis.
The relatively small number of case reports regarding INCs containing the term migraine may underestimate the real number of such reactions, due to a general trend in underreporting of adverse drug reactions as well as the coding of the case reports. A coding term is not the same as a clinical diagnosis and may not reflect established diagnostic criteria. The WHOART preferred term migraine also covers the included terms migraine aggravated, migraine-like headache, Horton's headache and cluster headache, although these may not be identical entities. In a study by Schreiber et al., the majority (80%) of patients with a history of self-reported or physician-diagnosed sinus headache had been diagnosed as having migraine based on the current International Headache Society's criteria (31). In light of the comparatively large number of reports of INCs in Vigibase with the term headache (n = 425), it is possible that a proportion of these cases may refer to an additional number of patients with migraine-like headache. Furthermore, there are in Vigibase seven cases of benign intracranial hypertension reported in connection with INCs, and headache could also have occurred as a sign of this condition.
Although there is a known connection between allergic rhinitis and migraine, the reports in Vigibase suggest that, in addition, INCs might cause or worsen migraine or migraine-like headache. Perhaps an INC-related inflammatory process of the mucosa in the nose or paranasal sinuses may in turn lead to (unilateral) headache. In order to collect more information regarding the possible but ill-understood connection between INCs and migraine-like headache, healthcare practitioners are requested to report similar observations to the national pharmacovigilance programmes in their countries. In addition, further studies are needed to determine whether the reported association between INCs and migraine or unilateral headache is real or not and, if so, what the possible mechanism might be.
