Abstract
Background
Questionnaires administered to orthostatic hypotension (OH) patients reveal frequent occurrence of coat-hanger ache (CHA), but laboratory-based precipitation of CHA during head-up tilt (HUT) has not been investigated. This study compared the frequency and clinical aspects of CHA in the same group of OH patients during daily activities versus during HUT.
Methods
Retrospective IRB-approved review of prospectively collected data on 22 dysautonomic patients. Heart rate response to deep breathing, Valsalva manoeuver, HUT and thermoregulatory sweat test evaluated cardiovagal, adrenergic and sudomotor functions. Occurrence and clinical features of CHA during daily activities and during HUT were recorded. Data were analysed with descriptive statistics.
Results
All patients demonstrated severe adrenergic (OH), cardiovagal and sudomotor impairment. Of 22 patients, 13 (59%) reported CHA within 3–5 minutes of standing or after 10 minutes to 2 hours of sitting that was relieved within 5–20 minutes of recumbency. During HUT, 4 of 13 (18%) patients developed CHA. Clinical features varied.
Conclusions
CHA was reported by 59% of OH patients during daily activities and by only 18% during HUT. The clinical characteristics of CHA episodes displayed inter-individual and intra-individual variability. These findings militate against direct association between OH and CHA and suggest a complex pathophysiology.
Introduction
Orthostatic headache, a hallmark of intracranial hypotension, is defined as a headache that worsens within 15 minutes after sitting or standing and improves within 15 minutes after recumbency (1). It can be caused by pathological processes other than intracranial hypotension such as colloid cyst of the III ventricle, skull base tumour, Arnold–Chiari malformation, cerebellar haemorrhage, cervical spine metastasis, filum myxopapillary ependymoma, and ocular hypertension (2–8). The unfolding spectrum of orthostatic headache also includes dysautonomic patients with postural tachycardia syndrome (9), baroreflex failure (10), autonomic dysreflexia (11) or orthostatic hypotension (OH) (12).
OH may affect 18% of individuals aged >65 years causing syncope, injury and even death (13). Orthostatic head and neck discomfort is a useful but poorly recognized symptom of OH. In patient surveys, Robertson and colleagues observed orthostatic discomfort localized to the occiput, nape of the neck, and shoulders (coat-hanger ache) (CHA) in 92% of 25 dysautonomic patients (12) and Bleasdale-Barr and Mathias found CHA in 93% of patients with pure autonomic failure (PAF) and in 51% of patients with multiple system atrophy (MSA). CHA was attributed to OH-caused hypoperfusion of tonically active neck muscles (14). In contrast, two laboratory-based studies reported CHA in only 11.4% (15) and 29% (16) of patients during HUT. To our knowledge, no one has yet compared self-reported CHA with laboratory-based HUT-induced CHA.
The objectives were to document the severity and diagnoses of autonomic dysfunction and to prospectively assess the frequency and clinical characteristics of CHA in 22 consecutive dysautonomic patients with OH during daily activities (by history) versus during HUT (in the laboratory) in the same group.
Methods
OH was defined as sustained reduction of systolic blood pressure of at least 20 mmHg or diastolic blood pressure of 10 mmHg within 3 minutes of standing or HUT to at least 60° (17). PAF was defined as primary autonomic failure syndrome characterized by symptomatic OH, bladder and sexual dysfunction without any sensory, pyramidal, extrapyramidal or cerebellar dysfunction during 5 years of follow-up (18). MSA was defined as a primary autonomic failure syndrome characterized by urogenital and cardiovascular dysfunction with motor disorder, incorporating parkinsonism, cerebellar or pyramidal features (19).
Institutional review board approval was obtained for this study. Twenty-two prospectively evaluated patients were reviewed retrospectively: PAF, 6; MSA, 5; Parkinson’s disease, 3; autonomic neuropathies, 6; baroreflex failure, 2. Mean age was 62.4 years (range, 47–76 years). Detailed history, somatic and autonomic neurological examination, and blood tests were performed before laboratory autonomic evaluation. Complete blood cell count, erythrocyte sedimentation rate, serum electrolytes, vitamin B12 levels, serology for syphilis, haemoglobin A1c, thyroid function tests, morning and evening cortisol levels, serum protein immunoelectrophoresis, and paraneoplastic profile were obtained for all patients. All patients had electrocardiogram and neuroimaging.
Informed consent was obtained before performing standardized autonomic function tests to document the diagnosis and to assess severity and distribution of adrenergic, cardiovagal and sudomotor dysfunctions. Blood pressure response to the Valsalva manoeuver (VM) and HUT were employed to assess sympathetic adrenergic function. The heart rate response to deep breathing (HRDB) and Valsalva ratio (VR) were used to assess cardiovagal function. Sympathetic cholinergic function was assessed with the thermoregulatory sweat test (TST) (20).
Blood pressure was recorded continuously and non-invasively by Ohmeda Finapres 2300, a photoplethysmographic device. An appropriately-sized finger cuff was used, and the left hand was supported in a sling to keep the hand at heart level. Heart rate was recorded continuously from a precordial electrocardiograph (lead II) using a Hewlett-Packard HR monitor. For TST, oral and skin temperatures were monitored continuously. The data were collected using Biopac Student Lab Pro.
Procedures
The patients were free from neck ache and headache at the time of the laboratory evaluation. They were off all medications for 5 days except for three patients with diabetes, who continued taking diabetes medications. Patients were instructed to avoid alcohol, caffeine and nicotine overnight and to arrive at the laboratory after a light breakfast. The tests were performed in a quiet room at 22°–23°C.
For HRDB, patients were instructed to breathe deeply and evenly, 5 seconds in and 5 seconds out, without holding breath during the cycle. They took eight breaths at the rate of six breaths per minute. The difference between the maximum and minimum HR during each respiratory cycle was calculated. HRDB was the mean of differences during the six best consecutive breathing cycles (20).
For the VM, the patient, in a semi-recumbent position, was asked to start forced expiration at the end of normal inspiration and maintain expiratory strain of 40 mmHg for 15 seconds. Care was taken to ensure that pressure rose sharply at the onset, was sustained at 40 mmHg for duration of the manoeuver, and fell abruptly at the termination of strain. At least 3 minutes of recovery time was allowed after each VM. VR, an index of cardiovagal activity, was calculated from the ratio of maximal to minimal HR. The presence or absence of late phase II and systolic blood pressure (SBP) overshoot during 30 seconds after termination of strain were recorded. BP and HR responses were obtained in triplicate, and the largest responses were accepted (21).
The physician performed the HUT test. The patient was strapped to the table across the knees, thighs and chest. BP and HR were recorded from 3 minutes before to 3 minutes after the test, and the table was tilted to 90° for 10 minutes, if tolerated. Patients were asked to report symptoms as they occurred (22).
In the TST, the patient, unclothed and recumbent, was exposed to controlled heat (45°–50°C) and humidity (35–40%) in a sweat chamber. An iodine and starch combination was employed as a colour indicator. The test was continued until generalized sweating or rise in oral temperature of >1°C above baseline was obtained (20).
All patients were queried via questionnaire about neck injury and headache before onset of illness. They were asked if they had experienced CHA or orthostatic headache during daily activities since the onset of illness. During HUT, patients were asked to report occurrence of all symptoms. They were specifically asked about neck ache and headache at 1, 3, 5, 7 and 10 minutes of HUT. Occurrence of orthostatic CHA or headache during daily activities or during HUT prompted questions about the duration of upright posture required to precipitate it and about improvement following recumbency. The patients were asked to grade pain on a visual analogue scale of 1 to 10, one being minimally perceptible and 10 being extremely severe. They were also asked about the location and character of ache and precipitating and relieving factors.
Statistical analysis
Because of the possibility that abnormalities of autonomic function might be a consequence of aging in our study group, we employed age-adjusted criteria to delineate abnormal from normal, using normal ranges from studies with methods similar to ours (23,24). The data were tabulated and analysed by descriptive statistics.
Results
No patient had CHA as the presenting complaint. The presenting complaints of patients were orthostatic intolerance of varying severity ranging from light-headedness to fainting (17), impotence (3), weight loss (1) and dry mouth (1). The mean duration of symptoms was 6.77 ± 3.84 years, exceeding 2 years in all patients. Patients with PAF had symptoms for at least 5 years. All routine laboratory tests were normal in patients with PAF. Three patients had elevated haemoglobin Alc levels and a known history of diabetes mellitus. One patient had Sjogren’s syndrome with elevated antibodies to Ro (SS-A) and La (SS-B) autoantigens. One patient had received total body radiation for Hodgkin’s disease.
Individual patient data for autonomic tests and coat-hanger ache.

HRDB: heart rate response to deep breathing; VR: Valsalva ratio; VM II phase: Valsalva manoeuver phase 2; VM IV phase SBP: Valsalva manoeuver phase 4 systolic blood pressure; HUT: head-up tilt; ↑: increase; ↓: decrease; TST: thermoregulatory sweat test; PCHA: postural coat-hanger ache; HUT CHA: head-up tilt-induced coat-hanger ache.
Results of autonomic laboratory evaluation in 22 patients.
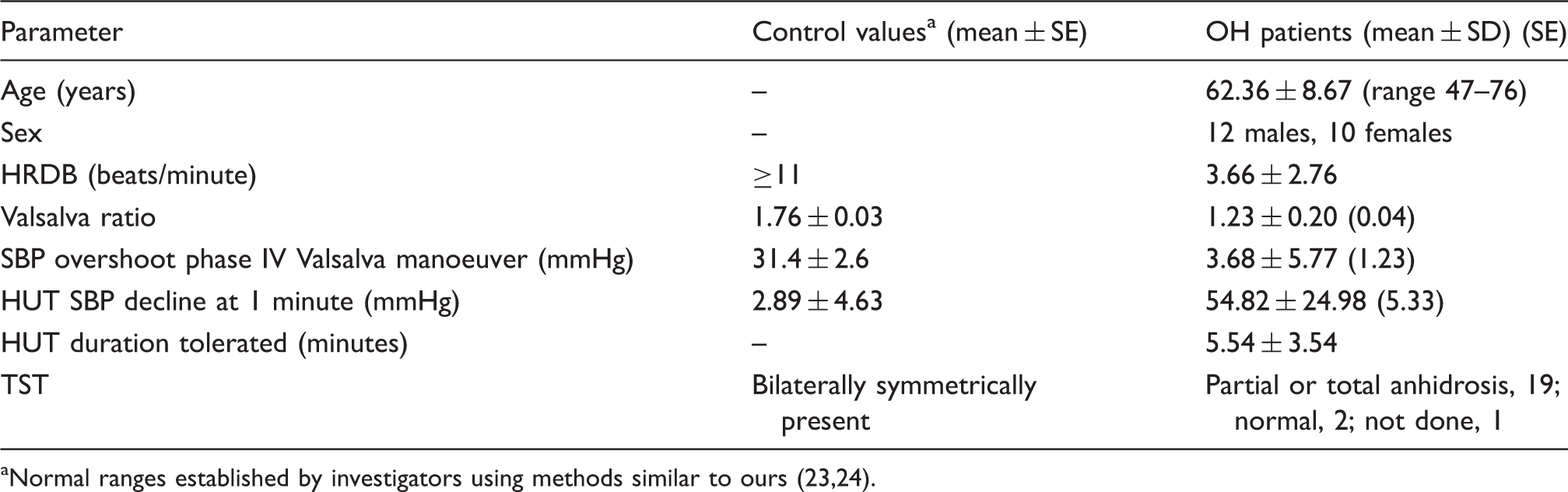
Normal ranges established by investigators using methods similar to ours (23,24).
Blood pressure response to HUT was abnormal in all patients. The range of HUT duration tolerated varied from 1 to 10 minutes, with a mean of 5.54 ± 3.54 minutes. All subjects tolerated HUT up to 1 minute. Thereafter, the number of subjects decreased with increasing duration of tilt. Consequently, for statistical comparisons, mean data are presented for 1-minute HUT and duration of tilt tolerated. The test had to be discontinued in less than 5 minutes in 13 patients, and only seven patients tolerated 10 minutes of HUT.
The decline in SBP within the first minute of HUT exceeded 40 mmHg in 14 patients, indicating severe OH (12). Four patients were asymptomatic. Symptoms accompanying OH in the other 18 patients, in decreasing order of frequency, were light-headedness, visual disturbances, presyncope, weak legs, heavy chest, speech disturbance, jerking of arms, and back pain. The results of HUT and VM revealed severe adrenergic involvement. The TST was done in 21 patients. One patient each with Parkinson’s disease and baroreflex failure displayed normal sweating. The other 19 patients demonstrated anhidrosis of varying patterns and severity.
Two patients had past history of motor vehicle accidents without persisting neck symptoms, one had suffered concussion after sustaining a fall from a horse, one had a cycling accident, and one had undergone cervical discectomy. Six patients had intermittent occurrence of migraine without aura. Three of six gave a history of CHA. Examination did not reveal tenderness in the sub-occipital, cervical paraspinal or trapezii regions. Neurological examination demonstrated C5 radiculopathy in one patient but no evidence of cervical myelopathy.
Of the 22 patients, 13 (59%) reported CHA occurrence during daily activities. Patients with CHA had a mean duration of illness of 7.9 ± 3.8 years, and those without CHA had a mean duration of illness of 4.9 ± 3.2 years. The majority of these patients experienced CHA after prolonged sitting (10 minutes to 2 hours), and a few developed CHA on standing (3 to 5 minutes) or when walking. The discomfort was described as pressure, dull ache or tightness involving posterolateral neck and suprascapular regions in a coat-hanger pattern. Sometimes only neck, interscapular region or shoulders were affected. In one patient, the neck was affected unilaterally. Two of the 13 patients complained of ‘numbness of head’ in addition to CHA. The frequency of CHA ranged from a few times a day to twice per month. It was more easily precipitated in the morning, when medication wore off, or when the patient was taking a shower or dressing. The severity ranged from 3 to 10 on a visual analogue scale and varied in the same individual on different occasions. When severe, the patient had to cease physical activity. Attaining supine position provided relief in the majority of patients in 5 to 20 minutes, but others took up to 2 hours to improve. One PAF patient had CHA when evaluated 11 years earlier, but denied current CHA.
Of the 22 patients, nine did not develop CHA during daily activities or during HUT. Of the 13 patients who developed CHA during daily activities, four developed neck or head discomfort during HUT. Two patients reported CHA within 2 minutes of HUT, one with tightness of neck and shoulders and the other with neck pain. Both displayed a maximum decline in SBP of 80–90 mmHg. Two patients described head discomfort, one complaining of tightness of the forehead after 9 minutes of HUT and the other noting pressure in the head and constriction of the throat within 90 seconds of HUT. Both demonstrated a maximum decline of SBP of ∼100 mmHg. One patient, who initially complained of CHA during HUT, reported loss of this symptom during the presyncopal phase. The other three reported improvement upon attaining a supine position. Four of the 22 patients were asymptomatic during HUT: one with Parkinson’s disease, one with PAF, and two with MSA. These four patients had a decline in SBP of less than 50 mmHg (mean, 32 mmHg) and tolerated HUT duration of 8–10 minutes.
Discussion
Several studies have reported an association between CHA and OH based on the co-occurrence of CHA and OH (12,14). Furthermore, CHA is induced by factors that exacerbate OH such as morning (reduced blood volume due to nocturnal diuresis), food (splanchnic vasodilation) and warm environment (cutaneous vasodilation). Cariga and colleagues (26) have observed a positive correlation between the degree of fall in systolic and diastolic blood pressure and adjusted score for neck pain intensity. Some authors (14) state that quality of pain reported by patients (e.g. cramp-like, gripping) favours an ischemic origin and attribute CHA to ischemia of neck muscles. Recently, Humm and colleagues measured velocity recovery cycle (VRC) of muscle action potentials by direct stimulation of the trapezius muscle of eight selected MSA patients with CHA (27). The muscle VRC was the same in patients and normal subjects when supine. During HUT, VRC was unchanged in normal subjects, but the MSA patients showed a progressive decrease (44%) in early supernormality (depolarization) and an increase (17%) in relative refractory period (27). These studies imply that OH is the aetiology of CHA and that ischemia of neck muscles is its only pathophysiology.
In the current study, 13 of 22 consecutive dysautonomic patients with OH provided a history of orthostatic CHA during activities of daily living. During HUT, only four of 13 developed neck or head discomfort, two reporting CHA and two reporting head discomfort characterized by bifrontal pressure/tightness. The frequency of occurrence of CHA was variable between patients and within the same individual. Time to onset of CHA after sitting or standing also varied. Severity of CHA varied considerably within the same individual from being minimal on one occasion to extremely severe on another. Association with other symptoms of orthostatic intolerance and duration required for improvement after recumbency were also variable. These findings suggest that CHA aetiopathogenesis may be more complex than was previously thought.
CHA has been attributed to neck muscle ischemia. Ischemia should be global above the heart level, affecting cerebral cortex, brainstem, cervical spinal cord, dorsal root ganglia, brachial plexus, dural sinuses, and the musculoskeletal system. There is, however, differential vulnerability of organs to ischemia based on their capacity to autoregulate their blood flow and for their cellular structures to survive. For example, patients with OH show an expansion of cerebrovascular autoregulation such that cerebral perfusion may remain relatively constant when the patient is supine and in response to standing (28). Therefore, if ischemia is the expected primary cause of CHA, then numerous other structures besides muscles could contribute to CHA.
Variability and inconsistency of CHA may be caused by differential contribution from multiple sources of pain, influence of ischemia and autonomic dysfunction on the central processing of pain, and altered central perception of pain. Pain referred to posterolateral neck and shoulder regions (CHA) has been elicited in humans by stimulation of infratentorial arteries, the occipital sinus, and duramater of the floor of the posterior fossa (29). Noxious stimulation of the interspinous muscles at C3-4, C4-5 and C5-6 may also produce pain in a similar distribution. Infratentorial structures have dual innervation – trigeminal and upper cervical – and convergence of this input at the trigeminocervical nucleus facilitates the referral of pain to the frontal region (30). Nociceptive structures in the cervical region include joints, periosteum, ligaments of the cervical spine, paraspinal cervical muscles, cervical nerve roots and vertebral arteries. OH may activate nociceptive input from any or all of these structures through various mechanisms, including reduction in muscle tone (a known component of OH and syncope) (17,31), causing neck flexion and non-physiological stretching of cervical musculoskeletal structures. OH may cause epidural hypotension. Upright posture may accentuate this phenomenon by overdrainage from the lumbar epidural veins into the inferior vena cava. This reduction in spinal epidural pressure can alter the gradient between epidural space and cerebrospinal fluid, leading to absorption of cerebrospinal fluid from the subarachnoid to the epidural space and vein, with partial collapse of the spinal duramater and stimulation of nociceptors (32). Ischemia of the dural and pial blood vessels may alter brain ion homeostasis and cause efflux of excitatory amino acids to promote nociception (33).
The autonomic nervous system, both parasympathetic and sympathetic components, exerts powerful modulatory effects on nociception. Trigeminal and vagal afferents overlap in spinal segments C1-C2. Vagal nerve stimulation induces nuclear Fos immunolabelling in the dorsal vagal nerve nucleus and paratrigeminal nucleus. Vagal afferent stimulation has an intensity-dependent modulatory effect on pain. High-intensity stimulation inhibits nociception by co-activating descending serotonergic and non-adrenergic systems, and low-intensity stimulation of cervical vagal afferents facilitates nociception (34). Our patients had moderate to severe loss of vagal function that may have altered headache threshold. Of interest also is the effect of the sympathetic component. Bilateral removal of superior cervical ganglion causes a rise in calcitonin gene-related peptide in the rat trigeminovascular system (35), a peptide with an established role in migraine pain. Although we did not test pupillary sympathetic function, loss of facial sweating was evident in several patients and sympathetic failure caused OH.
Perception and recall of pain may also be affected by OH. Abrogast et al. observed that a third of their patients with OH were completely asymptomatic during HUT (15). Four of 22 patients in the present study were asymptomatic, including one who had prior history of CHA. The author has seen patients with OH who experience knee buckling and gradually faint without complaining of any symptom during HUT (31). One patient in the present study felt CHA during HUT that disappeared during presyncope. Low et al. observed impaired cognition in almost half of the patients in the OH group with mean age of 63.6 years (16). Poda and colleagues demonstrated an acute worsening of global cognition including immediate and delayed recall during HUT (36).
In summary, CHA was frequent during daily activities (59% of patients) and infrequent during HUT (18% of patients). Clinical characteristics of CHA displayed inter-individual and intra-individual variability. These findings favour a multifactorial contribution to CHA. In addition to OH, a set of mechanisms may be operational in an individual patient at a particular time to produce a variable end result. Awareness of the symptom should indicate the need for orthostatic blood pressure measurements before proceeding with work-up of other causes of neck ache.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The author declares that there is no conflict of interest.
