Abstract
Background: Vascular dysfunction may be involved in migraine pathophysiology and contribute to the increased risk of ischemic stroke in migraine, particularly in women with migraine with aura (MA). However, data on endothelial function in MA are controversial. Here, we investigated whether systemic endothelial function and arterial stiffness are altered in women with MA, using a novel peripheral arterial tonometry device for the first time.
Methods: Twenty-nine female MA patients without comorbidities and 30 healthy women were included, and carotid intima-media thickness was assessed by a standardized procedure. Endothelial function was assessed using peripheral arterial tonometry. Reactive hyperaemic response of digital pulse amplitude was measured following 5 minutes of forearm occlusion of the brachial artery. Arterial stiffness was assessed by fingertip tonometry derived and heart-rate-adjusted augmentation index.
Results: No differences were found in peripheral arterial tonometry ratio (2.3 ± 0.6 vs 2.2 ± 0.8; p = 0.58) and left carotid intima-media thickness (in µm: 484 ± 119 vs 508 ± 60; p = 0.37). Women with MA had higher heart-rate-averaged augmentation index [median (interquartile range, IQR) of 5 (IQR 0.5 to 18) vs –5 (IQR −16.8 to 8.3), p = 0.005] and heart-rate-adjusted augmentation index [1 (IQR −6 to 12.5) vs −8 (IQR −20.3 to 2.5), p = 0.008] than healthy controls.
Conclusion: Peripheral endothelial function is not impaired in women with MA, but they have greater arterial stiffness. This may contribute to the increased stroke risk in women with MA.
Keywords
Introduction
Migraine is a common neurological disorder affecting approximately 10% of the working population (1). Observational studies have consistently reported an independent association between increased risk of ischemic stroke and migraine with highest risk in women with migraine with aura (MA) (2–4). Moreover, MA is the only primary headache type associated with brain infarcts in a recent population-based MRI study (5). However, the mechanisms underlying the relationship between ischemic stroke risk and MA are uncertain. Endothelial dysfunction, increased aortic pulse wave velocity and a procoagulatory and inflammatory state have been proposed as possible mechanisms to close this gap (6,7), but studies are rare. A few case-control studies that have investigated peripheral endothelial function with flow-mediated dilatation (FMD) showed conflicting results. FMD is a non-invasive standard method to assess endothelial function by measuring the relative change of brachial artery diameter after post-occlusive reactive hyperaemia (8). In migraine some studies found decreased FMD in the interictal period (6,9,10), whereas others reported no differences in FMD compared with age-matched healthy individuals (11,12). Furthermore, emerging evidence suggest that functional properties of large arteries are altered in migraine patients (6,13–15). Thus, arterial stiffness indicated by increased aortic augmentation index (AI) (13,15) and increased aortic and brachial artery pulse wave velocity (PWV) was higher in migraine patients, especially in MA (13,14). Arterial stiffness is an established, independent predictor of future risk of cardiovascular events and stroke in healthy participants (16,17). The purpose of this study was to investigate systemic endothelial function and AI as a correlate for arterial stiffness in women with MA using a novel non-invasive peripheral finger plethysmograph (EndoPAT). We hypothesized that AI as a surrogate measure for arterial stiffness is increased in women with MA.
Methods
Study population
Women with MA were consecutively recruited from the outpatient headache clinic of the Charité University Mitte, Berlin between October 2010 and September 2011. Twenty-nine women with MA aged ≤ 50 years who fulfilled the diagnostic criteria of MA according to the International Headache Society criteria (2nd edition) and with frequency of MA of at least one attack per month were included in this study (18). All patients had a detailed clinical evaluation and a semi-structured interview about history of headache by an experienced neurologist.
Thirty healthy female participants were recruited from volunteers among the staff of the Charité University hospital and served as control group. Healthy volunteers were matched with patients by age and blood pressure. Healthy volunteers were free of any overt cardiovascular or systemic disease and were of similar age, body composition, and main clinical characteristics, including lipid status, smoking habits and use of oral contraceptives. One woman with MA and two healthy volunteers were not included into the study after screening at admission as a result of exclusion criteria.
Exclusion criteria were severe infectious disease, history of cardiovascular disease (stroke, coronary heart disease, myocardial infarction), body mass index (BMI) < 18 or > 25 kg/m2, arterial hypertension (systolic blood pressure > 140 mmHg, diastolic blood pressure > 90 mmHg), diabetes mellitus, pregnancy, drug abuse and medication with statins, anticoagulants, aspirin, clopidogrel or intake of triptans within the previous 24 hours.
The study was approved by the ethics committee of Charité-Universitätsmedizin Berlin and participants gave their written informed consent to participate. The study has been registered in clinicaltrials.org (NCT01388699).
Vascular endothelial function
Quantitative determination of endothelial function was assessed by application of a non-invasive finger plethysmograph (EndoPAT2000; Itamar Medical, Caesarea, Israel). Assessment of endothelial function was based on the measurement of peripheral arterial tonometry (PAT) of the index finger during reactive hyperaemia of the forearm vascular bed (Figure 1) as previously described (19,20). The reactive hyperaemic PAT response has been shown to significantly depend on nitric oxide synthesis (21). In summary, all EndoPAT measurements were performed after an overnight fasting and under standardized conditions in the supine position after a resting period of at least 15 minutes in a quiet room maintained at room temperature. Pneumatic probes were placed on both index fingers. Baseline measurement was carried out for 5 minutes. Complete arterial occlusion for 5 minutes was induced by external compression of the upper arm using an inflatable cuff with at least 30 mmHg above systolic blood pressure followed by 5 minutes of post-occlusion measurements. Pulse amplitude measurements were continuously recorded, digitized and the PAT ratio was calculated by computerized algorithm of the EndoPAT2000 software system using the post- and pre-occlusion values. Values were normalized to the measurements from the non-experimental arm, which serves as a control for non-endothelial dependent systemic effects and fluctuations due to influences of the autonomic nervous system that might induce changes in peripheral arterial tone. EndoPAT measurements were not performed under blinded conditions. However, a strength of the method is that results are analysed with as computerized, automated algorithm, and therefore measurements are reported as being independent of operator-dependent errors and subjective bias (22).
Representative peripheral arterial tonometry (PAT) recordings of reactive hyperaemic response after 5 minutes of transient occlusion of the brachial artery from the experimental arm and PAT recordings from the control arm.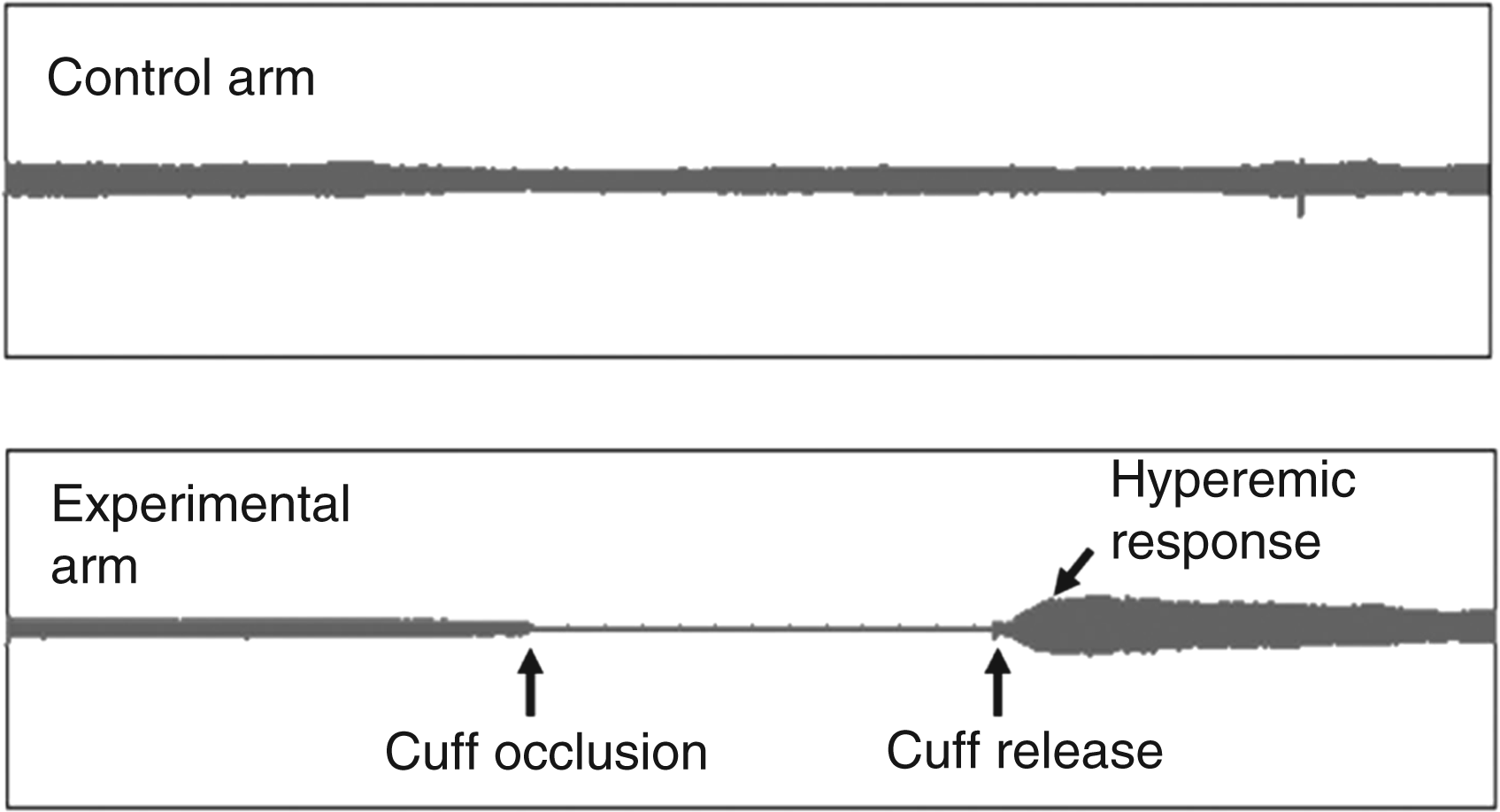
Arterial stiffness
Arterial stiffness was assessed by the finger tonometry derived AI, which is calculated automatically by analyzing the waveform of the PAT signal and averaged from all beat-to-beat signals of the finger arterial pulse-wave amplitude during the baseline period as previously described (23). AI is calculated as the difference between the second (P2) and first systolic peak pressure (P1), expressed as a percentage of the central pulse pressure (PP): AI (%) = [(P2 – P1)/PP] × 100, as shown in detail in Figure 2.
Representative recordings of pulse waves with normal Augmentation Index (AI = −14%) and increased Augmentation Index (AI = 21%). Augmentation index (AI) is the difference between the first (P1) and the second (P2) peaks of the arterial waveform. Values are expressed a percentage of the central pulse pressure (PP): AI (%) = [(P2 – P1)/PP] × 100.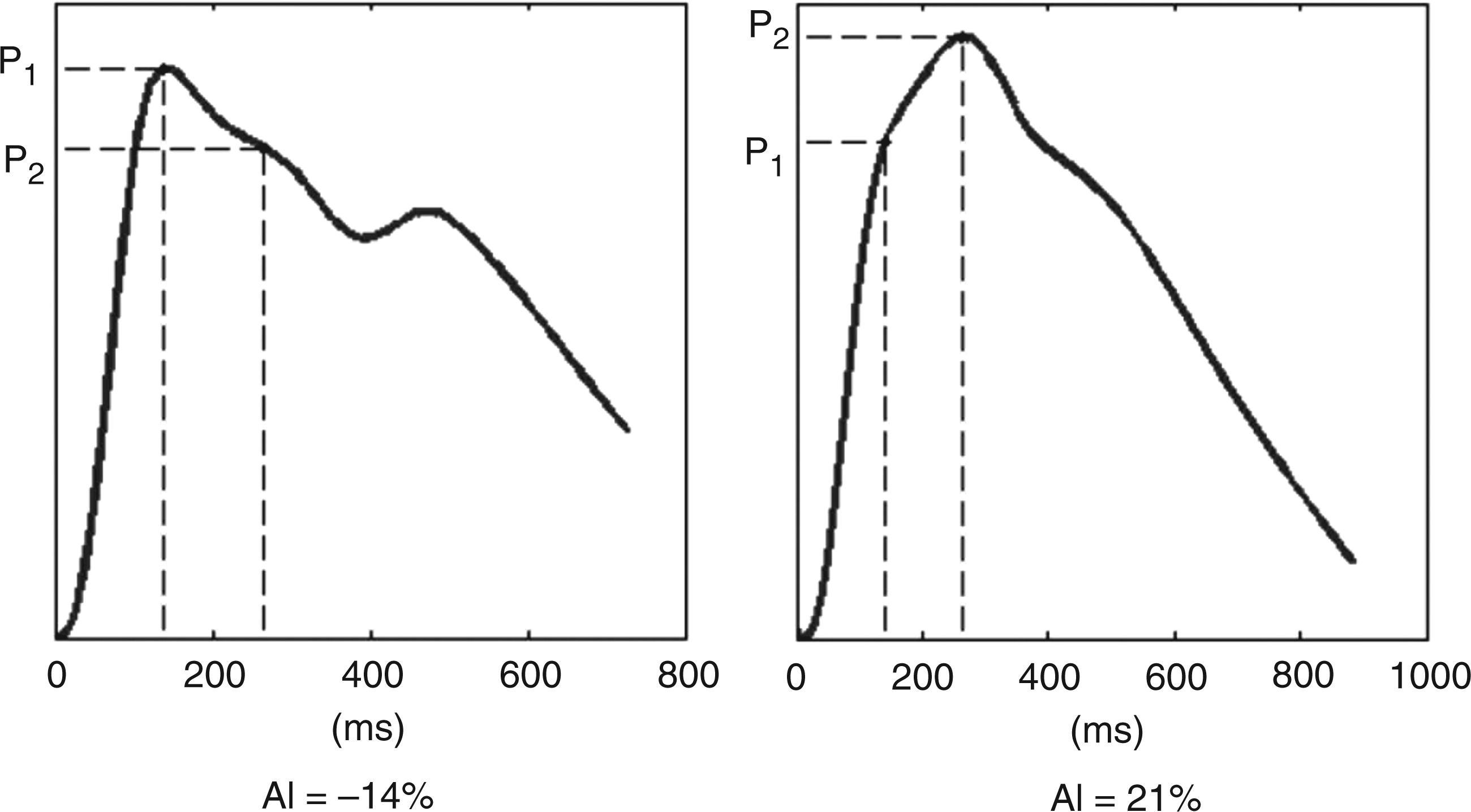
In addition, because AI is influenced by changes in heart rate, all values of AI were corrected to 75 beats per minute (AI@75 bpm). In summary, large values of AI correspond to a higher stiffness or less compliance of the arterial system. The EndoPAT method correlates with other validated methods of AI quantification as previously shown (24).
Carotid intima-media thickness
Duplex sonography of the right and left common carotid artery was performed in all participants. All recordings were obtained with the patient resting in a supine position, with the head turned to the contralateral side. IMT was assessed according to the Mannheim Intima Media Thickness Consensus (25). IMT of the far wall was measured at 2 cm proximal to the carotid bulb using a standardized real-time measurement method (radiofrequency-based quality IMT, Esaote Mylab25Gold) as previously described (26,27).
IMT was defined as the distance between the characteristic echoes of the lumen-intima interface and the media-adventitia interface. Mean values of a minimum of three IMT measurements and vessel diameter were calculated for both sides.
Statistical analysis
Sample size estimation was based on a previous study that investigated differences in endothelial-dependent flow-mediated dilatation between migraine and healthy controls (10). Data are expressed as mean ± SD or median and interquartile range (IQR) for continuous variables and as frequencies and proportions for categorical variables. Variances were not statistically significantly different between groups. Continuous data from the two groups were compared using the Mann-Whitney U-test or unpaired t-test depending on normality of distribution. Categorical variables were compared using the Pearson χ-test, for correlation the Spearman rank correlation was used. All tests were two-tailed, and statistical significance was determined at an alpha level of 0.05. Statistical analyses were performed with PASW software (version 18, SPSS, Chicago).
Results
Demographic, clinical and laboratory variables in 29 women with aura and 30 healthy women
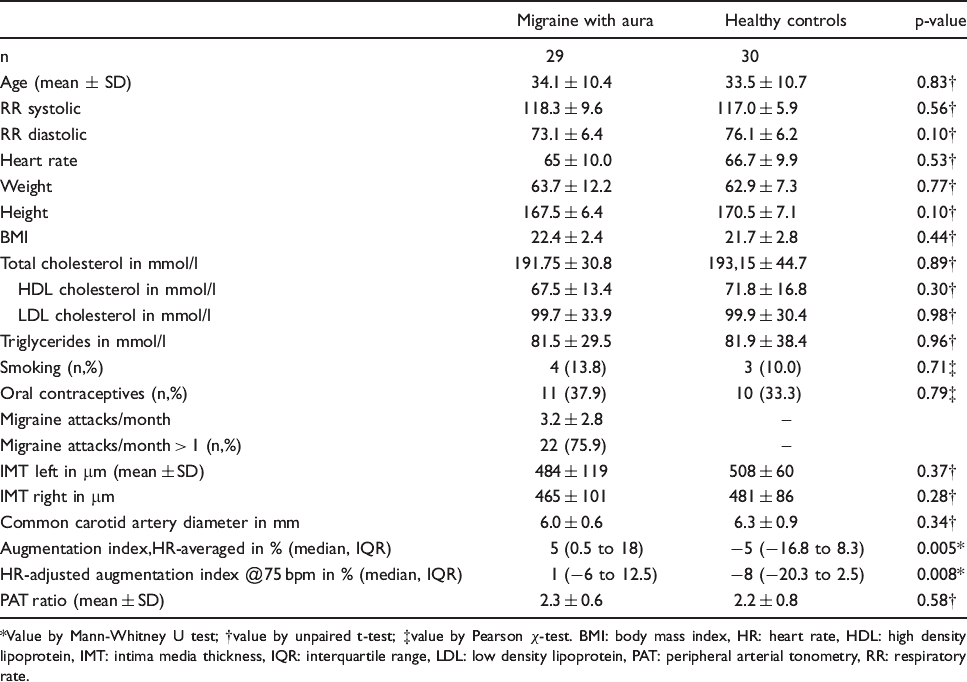
Value by Mann-Whitney U test; †value by unpaired t-test; ‡value by Pearson χ-test. BMI: body mass index, HR: heart rate, HDL: high density lipoprotein, IMT: intima media thickness, IQR: interquartile range, LDL: low density lipoprotein, PAT: peripheral arterial tonometry, RR: respiratory rate.
All participants were Caucasian. Four of the MA patients (13.8%) received prophylactic treatment (two with beta-blocker, one with lamotrigine, one with topiramate). Frequency of migraine attacks per months was 3.2 ± 2.8 with 4.4 ± 3.5 days of migraine per month. Twenty-two patients (75.9%) had more than one migraine attack per month.
No significant differences were found for blood pressure values, height, weight, heart rate, BMI, triglycerides, cholesterol levels, IMT thickness of the right and left common carotid artery (CCA), diameter of right CCA, and PAT ratio between the two groups, as shown in Table 1. Women with MA had higher heart-rate-averaged AI [median (interquartile range) of 5 (IQR 0.5–18) vs −5 (IQR −16.8–8.3), p = 0.005] and heart-rate-adjusted AI [1 (IQR −6 to 12.5) vs −8 (IQR −20.3 to 2.5), p = 0.008] as shown in Figure 3.
Box-and-whisker diagram (sample minimum, 5th percentile, lower quartile, median, upper quartile, 95th percentile, sample maximum) of augmentation index (A), heart-rate-adjusted AI at 75 beats per minute (B) and peripheral arterial tonometry (PAT) index (C) of 29 women with migraine with aura (MA) and 30 female healthy controls. P-values for group comparisons were (A) p = 0.005, (B) p = 0.008, and (C) p = 0.58, respectively.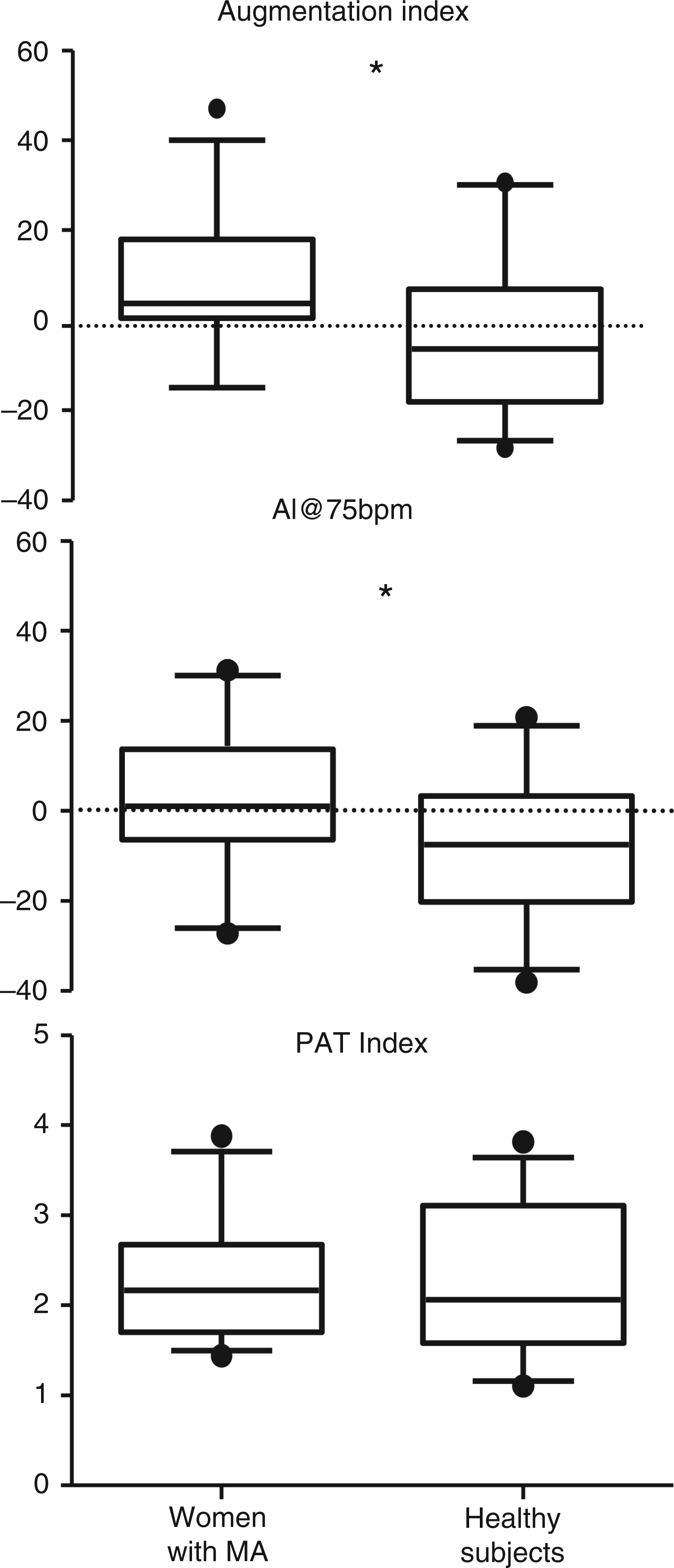
A direct correlation with age was found for AI (r = 0.46, p < 0.001) and heart-rate-adjusted AI (r = 0.44, p < 0.001)
Discussion
This study has the following major findings: 1) AI as a marker for arterial stiffness is increased in female MA patients without overt cardiovascular risk factors compared with matched healthy women; and 2) no differences were found for systemic endothelial function using digital reactive hyperaemic peripheral arterial tonometry.
Our findings on endothelial function are in line with a recent study investigating brachial FMD in migraine patients that particularly focussed on excluding possible confounders such as by strict inclusion criteria, application of CIMT assessment, and measurement of lipid levels (12). Perko et al. found that systemic endothelial function is not impaired in patients with MA and migraine without aura without comorbidities. In our study, we used similarly strict criteria (e.g. assessment of CIMT for detection of subclinical atherosclerosis) to avoid possible changes in PAT hyperaemic response caused by confounding factors (12). In contrast to our findings, a limited number of previous case-control studies reported decreased systemic endothelial function measured with FMD in migraine patients during the interictal period suggesting that endothelial dysfunction might be the missing link between migraine and increased cardiovascular risk (6,9,10). However, other case-control studies could not detect any differences (11,12,28). These conflicting results might be explained by differences in age, gender distribution, method of analysis, and adjustment for possible confounders. For example, Vanmolkot et al. reported a higher BMI in migraineurs that could have possibly biased their results (6). Furthermore, several studies included migraine with (MA) and without aura and, as highlighted by Perko et al., CIMT was rarely measured in available studies (12). Similar to FMD, CIMT is considered to be an early marker for atherosclerosis and used as a surrogate marker of subclinical atherosclerosis in observational studies. Thus, exclusion of differences in CIMT that rule out early atherosclerosis could help to further adjust for possible confounders.
Compared with studies on endothelial function, reports on arterial stiffness measured with AI and PWV in migraine patients are more consistent. Four studies have investigated AI and PWV in migraine patients so far (6,13–15). Ikeda et al. could show that in 22 MA and 89 MO patients brachial PWV was increased in migraineurs without cardiovascular risk factors (14).
In line with our findings, two studies reported higher heart-rate-adjusted AI indicative of increased aortic stiffness in migraine using a tonometry device that derives AI indices from radial artery pulse wave form analysis (29). AI and PWV have been validated as markers for arterial stiffness and are widely used in observational studies (30). Increased arterial stiffness is an established independent predictor of overall cardiovascular risk and ischemic stroke in the general population (16,17). Independent association of migraine and increased event rate has been described for ischemic stroke (4,31) and cardiovascular diseases (32,33). A recent systematic review reported an increased risk of ischemic stroke that was apparent only in MA patients and highest in women (4). Interestingly, higher values of aortic PWV and AI were found in MA patients than in migraine patients without aura (6,13). Moreover, Schillaci et al. reported that in multivariate analysis, female gender was independently associated with increased aortic AI together with migraine, age, and mean arterial pressure (13). Thus, this might point towards a contribution of arterial stiffness to the increased risk of ischemic stroke and cardiovascular risk in women with MA.
Pronounced changes in arterial stiffness result in increased pulse pressure. However, in our study no significant changes were found in systemic pulse pressure. Thus, the significant differences in AI in our study might be more subtle. Moreover, underlying mechanisms are unclear, but structural changes in the arterial wall in migraine patients might have a role, because, for example, Tzourio et al. have shown that the activity of the responsible elastin degradation enzyme is elevated in migraine (34).
The fingertip EndoPAT device used in our study is a novel, non-invasive technique to assess endothelial function and PAT-derived AI (35). The advantages of the EndoPAT technique are that it is easy and quick to learn, significantly less susceptible to faults and less operator-dependent than FMD. Thus, it could provide a method for assessing endothelial function that may be suitable for large-scale population studies.
Two studies showed that the PAT hyperaemic response is positively correlated with brachial FMD healthy participants (36) and in 89 participants with chest pain (r = 0.55, p < 0.001) (20). Furthermore, Bonetti and coworkers reported a significant correlation between PAT hyperaemic response and cerebral blood flow response to intracoronary administered acetylcholine, which is considered as the ‘gold standard’ for testing endothelial function (37). Recently, we demonstrated that endothelial dysfunction indicated by low PAT hyperaemic response is present in patients with acute ischemic stroke to a similar degree to that in cardiac heart failure patients (38). Others have demonstrated decreased PAT ratio in other diseases that go along with endothelial dysfunction (23,39).
Our study has the following strengths and limitations. Diagnosis of MA was made by an expert neurologist with a clinical semi-structured interview that is probably superior to self-administered questionnaire used in previous studies (15). Furthermore, to the best of our knowledge, ours is the first study on endothelial function and arterial stiffness in migraine using PAT hyperaemic response.
There are certain limitations that should be acknowledged. Firstly, AI is not a direct measure of arterial stiffness. It has been suggested that arterial stiffness and augmented pressure from wave reflections significantly contribute to the digital volume pulse inflection point; however, AI is influenced by several other factors, such as gender differences in vascular geometry, peripheral resistance, height, and pulse wave velocity. Moreover, although several studies could demonstrate the correlation between PAT-derived AI and other validated methods of AI and PWV quantification (19), the correspondence between PAT-derived AI and arterial stiffness needs further investigation. However, the AI method (pulse wave contour analysis from peripheral artery tonometry) has emerged as a novel non-invasive method to estimate aortic pulse pressure changes that carries meaningful clinical information on outcome and vascular risk in various patient populations (40–42). Given the small sample size in our study, our findings and its generalizability have to be interpreted with caution, and further evidence from larger observational studies is needed.
We did not find evidence for systemic endothelial dysfunction in MA patients. However, recurrent vascular inflammation of cranial arteries has been suggested to contribute to migraine pathophysiology (43). Thus, endothelial dysfunction might be primarily located in the cerebral vasculature. Perko and coworkers reported that endothelial function in the cerebral circulation probably differs from that in the systemic circulation in migraine patients (44). Furthermore, they found a lower cerebrovascular reactivity to
In conclusion, based on available results from this and preceding studies, there is no evidence for a systemic endothelial dysfunction in MA in the interictal period. In line with our findings, existing studies consistently show that arterial stiffness is altered in migraineurs and particularly in women with MA. This strongly suggests that a systemic vascular involvement is present in MA that may contribute to the increased risk of ischemic stroke found in epidemiological studies.
Footnotes
Acknowledgements
This study was funded by the Federal Ministry of Education and Research (BMBF) through the Grant Centre for Stroke Research Berlin (01 EO 0801) and the German Research Foundation (DFG) through the Grant NeuroCure (SFB-TR43). ME receives funding from Volkswagen Foundation (Lichtenberg program) and the European Stroke Network.
Conflict of interests
None declared.
