Abstract
Introduction
According to the second edition of the International Classification of Headache Disorders (ICHD-II) and the subsequently revised ICHD-II criteria, chronic migraine (CM) is characterized by headache on at least 15 days per month, of which at least 8 headache days per month meet the criteria for migraine without aura or respond to migraine-specific treatment (CM-R, A1.5.1) (1,2). It is believed that medication overuse contributes to its development and blocks attempts at prevention (3). Approximately 1.4% to 2.2% of the general population suffers from CM (4–8). Patients with CM present a clinical treatment challenge. This population is associated with significant disability, psychological distress, reduced health-related quality of life, and considerable healthcare cost (9–11). Until now, only topiramate and local injections of botulinum toxin have been shown to be effective in placebo-controlled randomized trials for CM prophylaxis (12–19). However, the high cost of botulinum toxin and considerable side effects of topiramate preclude their wide use for CM.
Acupuncture is one of the most commonly researched and widely accepted complementary and alternative medicine therapies in the treatment of migraine (20–23). A Cochrane systemic review concluded that there is no evidence for an effect of true acupuncture over sham interventions, although this is difficult to interpret, as the exact point location could be of limited importance. Available studies suggest that acupuncture is at least as effective as, or possibly more effective than, prophylactic drug treatment, and has fewer adverse effects (AEs). Acupuncture should be considered a treatment option for patients willing to undergo this treatment (23). However, most of these studies were conducted in patients with episodic migraine. There have not been any rigorous trials for the evaluation of the efficacy and safety of acupuncture treatment versus proven effective drugs on CM.
In an attempt to investigate the efficacy and tolerability of acupuncture compared with topiramate treatment in patients with CM, we conducted a prospective, randomized clinical study under conditions similar to routine care. In addition, we evaluated the subgroup of patients with medication overuse in both treatment groups.
Methods
Study design
The present study was developed at the Department of Neurology of Kuang-Tien General Hospital. The protocol and supporting documents (information sheet, consent form, and protocol) were approved by the institutional ethics committee in June 2008. The recruitment of patients was done from August 2008 to October 2010, and the trial was completed in January 2011. Patients were recruited via the outpatient department. They were scheduled by the research assistants and evaluated by an experienced neurologist. This study consisted of two periods: prospective baseline period before enrolment (week -4 to week 0) and treatment period (week 0 to week 12) after enrolment.
Inclusion and exclusion criteria were checked against data from the baseline period. The inclusion criteria were: an age of 18–65 years; a diagnosis based on the published guidelines of the Task Force of the International Headache Society Clinical Trials Subcommittee for controlled trials of prophylactic treatment of CM in adults criteria A–C (referring to the clinical characteristics of headache) but not criterion D of CM-R (the criteria for medication overuse headache, MOH-R, can also be met) during the 3 months before trial entry; and an established migraine history for at least 1 year (24). Participants with MOH-R must be stratified accordingly (25).
Patients were excluded if any of the following were present: 1) the headache was experienced for ≥15 days per month or there was no response to triptans or ergots on at least 8 days during the prospective baseline period (a 4-week preliminary phase); 2) headaches other than CM (such as another primary chronic headache or any secondary headache except medication overuse headache (25)); 3) migraine prophylaxis agents had been used in the past 3 months, such as β-blockers, anti-depressants, calcium channel blockers, anti-epileptic agents, cycle-modulating hormonal drugs, or vessel dilatation agents; 4) migraine onset after the age of 50, or over 60 years of age at onset of CM; 5) history of hepatic disorder, nephrolithiasis or other severe illness; 6) cognitive impairment interfering with the participant's ability to follow instructions and describe symptoms; 7) previous fearful experience of acupuncture or receiving acupuncture in the past 3 months; 8) bleeding diathesis or using anticoagulation agents; 9) pregnancy or nursing; or 10) severe depression (Beck Depression Inventory-II (BDI-II) score ≥30 at baseline day 1). If the patients fulfilled the criteria and gave written informed consent, they were enrolled in the study.
After enrolment, the patients were randomized into two treatment arms: 1) acupuncture group: acupuncture administered in 24 sessions over 12 weeks; and 2) topiramate group: a 4-week titration, initiated at 25 mg/day and increased by 25 mg/day weekly to a maximum of 100 mg/day (or to the maximal tolerated dose) followed by an 8-week maintenance period. At the end of the baseline screening phase, randomization was done provided the participants met the quantitative inclusion and exclusion criteria as per their reported diary data. The randomization was done according to computer-generated randomly allocated treatment codes, and data were kept by a person not involved in the care or evaluation of the patients or in the data analysis. Patients were allowed to take acute headache medication prescribed by our neurologists. We required that the acute headache medication be the same throughout the baseline period and for the duration of treatment period, and this was documented daily throughout the course of the study. All patients were scheduled so as to avoid any overlap during which they could share clinical information and experiences with each other.
Clinical assessments
All participants began the study by completing a set of outcome measures: the Migraine Disability Assessment (MIDAS), Short-Form 36 (SF-36), BDI-II and Hospital Anxiety and Depression Scale (HADS). All four questionnaires were administered on day 1 of the baseline period and repeated at the end of the treatment phase. To ensure consistency, the evaluating physician was the same person on each occasion for each patient, and was blinded to the type of treatment. Patients had to keep a headache diary from the baseline period (diary 1) and treatment period (diaries 2–4). Each diary covered 4 weeks. Daily calendar entries were designed to assess the frequency, location, duration, peak severity (1, mild; 2, moderate; 3, severe headache pain), presence of aura, menstruation, concomitant symptoms, the number of days with acute headache medication intake (including amount and frequency of analgesic drugs) for each migraine attack and study medication received.
Treatments
Acupuncture treatment
Acupuncture consisted of 24 sessions of 30 min, administered over 12 weeks (two sessions per week). Each patient had fixed and classic acupuncture points, bilateral Cuanzhu (BL-2), Fengchi (GB-20), Taiyang (EX-HN-5) and midline Yintang (Ex-HN-3) (Figure 1) in their 24 sessions without modification for the specific symptoms of the patient. All of the selected acupoints were in the distribution of trigeminal and cervical dermatomes related to the trigeminal sensory pathway. We placed patients in the sitting position and acupuncture needles were tapped into place through sterile plastic guide tubes. At each point, the needle was manipulated by twirling with lifting-thrusting slowly until the participant felt Location of the acupoints used (all bilateral except Ex-HN-3, which is located at the head midline) and their relation to the dermatomes. Dermatomes are listed with the following abbreviations: V1, ophthalmic branch of the trigeminal nerve; V2, maxillary branch of the trigeminal nerve; C2, dermatome of the second cervical nerve. Standards for reporting interventions in controlled trials of acupuncture (STRICTA)

Topiramate treatment
Patients assigned to the topiramate group also underwent treatment for 12 consecutive weeks. The treatment phase consisted of a 4-week titration period and an 8-week maintenance period. During the titration period, participants were given topiramate 25 mg/day at bedtime for 1 week, followed by weekly increases of 25 mg up to either 100 mg/day of topiramate or to the maximal tolerated dose. Starting in week 2, topiramate was given daily in equally divided twice daily doses. During the maintenance period, a stable topiramate dose of at least 50 mg/day was required. All participants were required to follow the same titration schedule. The mean final topiramate maintenance dose was 84.0 mg/day.
Primary efficacy parameters
The monthly number of moderate/severe headache days (a day with headache pain that lasted ≥ 4 hours with a peak severity of moderate or severe intensity, or any severity or duration if the participant took and responded to a triptan or ergot) from the second to the fourth diaries compared with the first diary (baseline period).
Secondary efficacy parameters
There were eight secondary efficacy parameters, as follows: 1) monthly number of headache days (any intensity) from the second to the fourth diaries compared with the first diary (baseline period); 2) change of MIDAS scores from baseline to endpoint; 3) change of HADS scores from baseline to endpoint; 4) change of SF-36 scores from baseline to endpoint; 5) change of BDI-II scores from baseline to endpoint; 6) reduction from baseline in the number of days with acute headache medications, from the second to the fourth diaries compared with the first diary (baseline period); 7) ≥50% reduction in monthly moderate/severe headache days from the second to the fourth diaries compared with the first diary (baseline period); 8) ≥50% reduction in monthly headache days from the second to the fourth diaries compared with the first diary (baseline period).
Safety assessments
Medical and headache histories were recorded, physical and neurological examinations were performed, and clinical laboratory parameters (hematology and chemistry evaluations) were assessed at screening and the final visit (week 12). Women of childbearing potential had urine pregnancy tests. After enrolment, participants in the topiramate group were required to return for a doctor visit once a week during the first month of the titration period, and then return to monitor any AEs of the drug at the end of a month during the following 2 months of treatment period (six visits in total). Participants in the acupuncture group were seen twice a week for acupuncture treatment and to monitor AEs in each session for the whole three months (24 visits in total). We recorded AEs such as nausea, dizziness, paresthesia, fatigue, dyspepsia, somnolence, and nausea along with ecchymosis, local paresthesia or bleeding for all enrolled patients.
Statistical analysis
Data analysis was performed separately for all study participants and for a subgroup of participants who were overusing acute headache medication. Medication overuse was defined as intake of simple analgesics (e.g. aspirin, acetaminophen, or non-steroid anti-inflammation drugs) on more than 15 days per month or the intake of a combination of analgesics, opioids, ergots, or triptans on more than 10 days per month (25). A last-observation-carried-forward approach was used to impute missing data with the intent-to-treat analysis principle. All outcome variables were examined with the assumption of normality using the Kolmogorov-Smirnov test, and the assumption was held for all outcome variables. Thus, independent two-sample t-tests were performed to compare the changes in mean monthly number of moderate/severe headache days and other efficacy assessments between the two groups. Paired t-tests were performed for changes in all efficacy evaluations between month 3 data and baseline data within each treatment group. The proportions of participants with ≥50% reduction of moderate/severe headache days and participants with ≥50% reduction of headache days were compared between the two groups with the chi-square test or Fisher's exact test when appropriate. All hypothesis testing was two-tailed and the level of significance was set at 0.05. All statistical analyses were performed using SPSS Version 15.0 for Windows (SPSS Inc., Chicago, IL, USA)
Results
Enrolment of patients and baseline characteristics
A total of 66 patients who were eligible and agreed to participate in our study were randomly allocated to either the acupuncture or topiramate treatment group. Of the 66 patients, one patient in the acupuncture group dropped out owing to an inability to take time off work, and three patients in the topiramate group did not finish the study owing to intolerance of side effects. The dropout rate was low for both topiramate and acupuncture groups. We imputed baseline values for the missing data of the four patients who did not complete the study (setting the differences from baseline to zero). Figure 2 illustrates patient enrolment and random allocation of the patients to the study groups. The baseline characteristics of the two groups were similar in the intention-to-treat population (Table 2). Most patients met the medication overuse headache criteria at baseline (Table 2). The subgroup of patients who were overusing acute headache medication (n = 49, 74.2%) consisted of 24 patients receiving acupuncture and 25 receiving topiramate treatment. There were also no significant differences in demographics and baseline characteristics between the acupuncture-treated and topiramate treated patients for this subgroup (Table 2).
Flow chart of process and disposition of patients. Baseline demographics and characteristics of the two groups Values are n, mean (SD), or percent. MIDAS, Migraine Disability Assessment; BDI-II, Beck Depression Inventory-II; SF-36, Short-Form 36; HADS, Hospital Anxiety and Depression Scale; SD, standard deviation.

Outcome of treatment
Primary efficacy parameters: numbers of moderate/severe headache days
Mean changes from baseline of the two groups
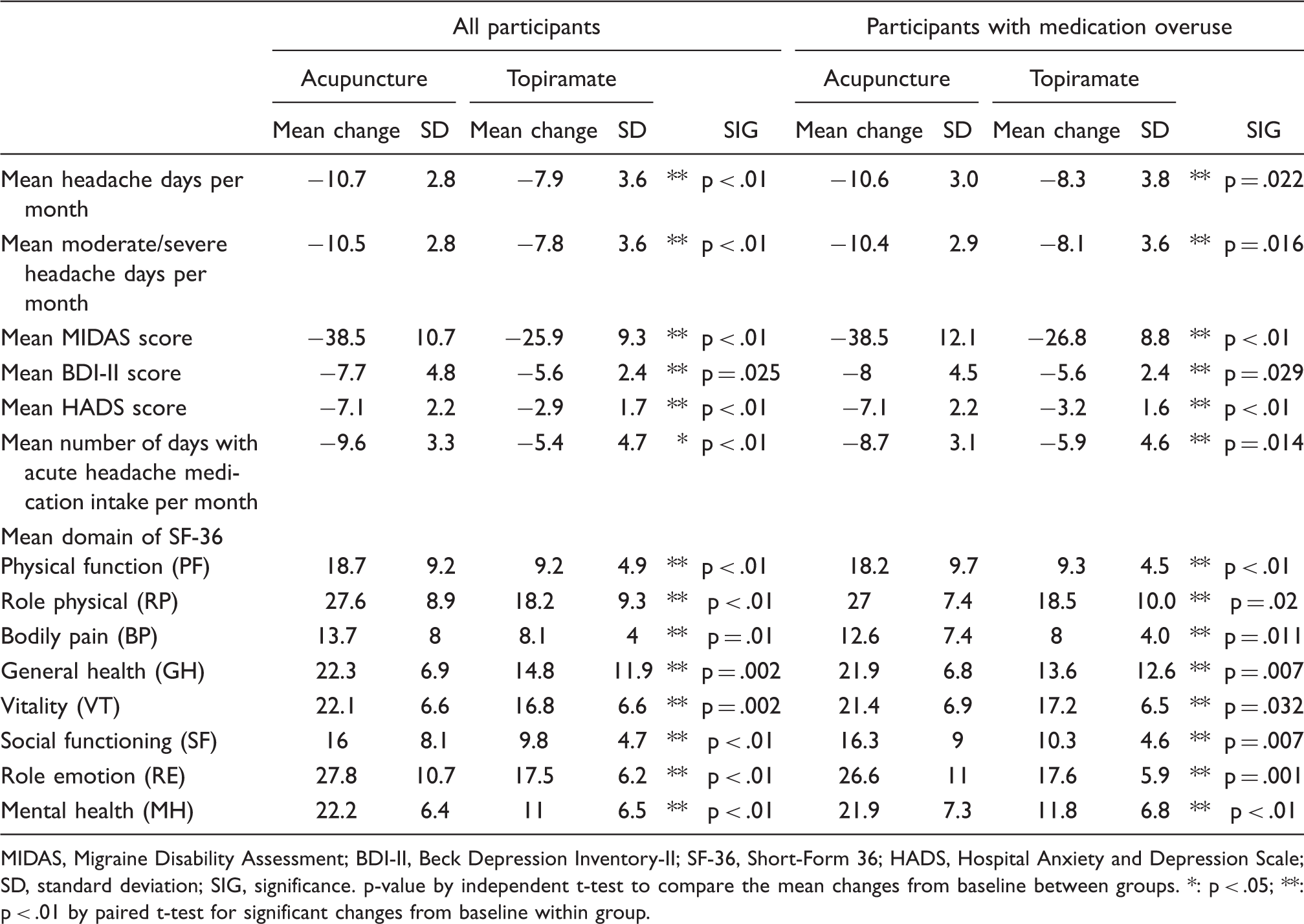
MIDAS, Migraine Disability Assessment; BDI-II, Beck Depression Inventory-II; SF-36, Short-Form 36; HADS, Hospital Anxiety and Depression Scale; SD, standard deviation; SIG, significance. p-value by independent t-test to compare the mean changes from baseline between groups. *: p < .05; **: p < .01 by paired t-test for significant changes from baseline within group.
Acute headache medications taken
There was a significant reduction in the number of days with acute headache medication intake in both groups, and a significantly greater reduction was observed in the acupuncture group compared with the topiramate group among all participants (−9.6 ± 3.3 acupuncture vs −5.4 ± 4.7 topiramate; p < 0.01) and in the participants who were overusing acute headache medication (−8.7 ± 3.1 acupuncture vs −5.9 ± 4.6 topiramate; p
50% responder analyses
A significantly greater percentage of the participants in acupuncture group than in topiramate group had at least a 50% decrease from baseline in the number of moderate/severe headache days (acupuncture 75.8% and 79.2% vs topiramate 30.3% and 32.0% for all participants and the medication overuse subgroup, respectively; all p < .01). In addition, a greater percentage of the participants in acupuncture group than in topiramate group had at least a 50% decrease from baseline in the number of headache days (acupuncture 63.6% and 70.8% vs topiramate 15.2% and 20.0% for all participants and the medication overuse subgroup, respectively; all p < .01) (Figure 3).
Percentages of participants having at least a 50% decrease from baseline in the number of moderate/severe headache days. A significantly greater percentage of the acupuncture group than topiramate group had at least a 50% decrease from baseline in both measures by Fisher's exact test for all participants and the subgroup of patients who were overusing acute medication (all p < .01).
Headache impact on disability, psychological distress and health-related quality of life
Statistically significant and clinically meaningful differences for acupuncture versus topiramate were observed in the mean change from baseline to endpoint in MIDAS score for all participants and the subgroup of patients who were overusing acute headache medication (p < .01) (Table 3). Acupuncture treatment showed a significantly greater improvement when compared to topiramate treatment in HADS (p < .01), BDI-II (p = .025) and overall health-related quality of life (SF36) (p < .05), as measured by changes from baseline for all participants (Table 3). The same pattern was also observed for the subgroup who were overusing acute headache medication (p < .01 for MIDAS and HADS, p = .029 for BDI-II and p < .05 for overall health-related quality of life (SF36) (Table 3). All efficacy evaluations showed significant improvement from baseline for each group (all p < .05), and the acupuncture group demonstrated a significantly better improvement for all of the assessments when compared with the topiramate group among all participants as well as for the subgroup of patients who were overusing acute headache medication (all p < .05).
Adverse side effects
No serious AEs or deaths were noted. In the acupuncture group, side effects were reported by 6% of the patients. Most AEs were related to the local insertion of the needles, such as local pain after the session, ecchymosis, and local paresthesia during the session. Acupuncture was well tolerated by the patients, and no one discontinued prematurely because of needle-related side effects. One patient in the acupuncture group discontinued treatment because of inability to take time off work. In the topiramate group, AEs were reported by 66% of the patients. The most frequently noted adverse effects were paresthesia (n = 16, 48.4%), difficulty with memory (n = 12, 36.3%), dyspepsia (n = 12, 36.3%), fatigue (n = 8, 24.2%), dizziness (n = 7, 21.2%), somnolence (n = 6, 18.1%), and nausea (n = 5, 12.1%). Most of the AEs were mild and self-limiting, but three (9%) of the patients withdrew from treatment because of intolerable adverse effects.
Discussion
The present study is one of the most rigorous trials of the efficacy of acupuncture treatment versus proven effective available drugs on CM. The strengths of our prospective study include interventions, assessments, and outcome measurements as recommended in the guidelines for trials on CM (24). A clearly described procedure for acupuncture treatment following the STRICTA (standards for reporting interventions in controlled trials of acupuncture) guidelines makes our study reproducible (26), and our study demonstrated the efficacy in the prophylactic treatment of CM in both acupuncture and topiramate groups. Acupuncture, when compared with topiramate, proved to be more effective in reducing the monthly number of moderate/severe headache days, monthly number of headache days, headache disability, psychological distress, and overall health-related quality of life at month 3 from the baseline. In addition to the results of the primary efficacy endpoint, a significantly greater percentage of acupuncture-treated than topiramate treated patients had at least a 50% decrease from baseline in the number of headache days and number of moderate/severe headache days. Acupuncture versus topiramate treatment also resulted in highly significant improvements from baseline in disability, psychological distress, and overall health-related quality of life. These findings indicate that the benefits of the acupuncture treatment were clinically meaningful to the patients. It can thus be concluded that acupuncture treatment has a superior efficacy and less side effects when compared with topiramate treatment for patients with CM.
Safety is an important consideration in the management of chronic conditions such as CM, and fewer treatment-related AEs were reported among patients who received acupuncture (6%) than among those treated with topiramate (66%). Three (9%) of the topiramate patients discontinued the study owing to AEs. In contrast, no patients in the acupuncture group discontinued the study owing to AEs, a result that is consistent with the known safety and tolerability profiles of acupuncture (27–30). The low drop-out rates may be due to three factors: 1) Our hospital is the largest in the region and is ranked as the highest in terms of quality. We build good relationships with our patients, resulting in low drop-out rates. 2) In topiramate group, AEs were reported by 66% of the patients; however, most of the AEs were mild and self-limiting so most did not lead to further drop-outs. In acupuncture group, minor side effects were reported by 6% of the patients and none discontinued prematurely because of needle-related side effects. 3) The treatment sessions were relatively short.
With CM being a disabling and rather difficult condition to treat, alternative therapeutic options such as acupuncture should be explored. A recent Cochrane systemic review concluded that acupuncture is at least as effective as, or possibly more effective than, prophylactic drug treatment, and has fewer adverse effects. However, there is no evidence for an effect of true acupuncture over sham interventions. Acupuncture should be considered a treatment option for patients willing to undergo this treatment (23). Indeed, sham acupuncture may still induce a wide range of peripheral, segmental and central physiological responses to an unpredictable degree (31–35). The possible reasons that previous trials could not detect the difference between true and sham acupuncture was that they assume there was no treatment effect for sham acupuncture in estimating the effect size. True acupuncture is widely used in Asia and topiramate treatment is the usual therapy for CM. Our findings of the treatment effects of true acupuncture versus topiramate provide useful information in clinical practice.
Currently, the US Food and Drug Administration has approved onabotulinumtoxinA, based on PREEMPT (Phase III REsearch Evaluating Migraine Prophylaxis Therapy with Botulinum Toxin Type A) trials for the prophylactic treatment of headaches in adults with CM (17–19). However, the high cost precludes its routine use for CM. Other preventive treatments, such as propranolol, valproic acid, flunarizine and topiramate for episodic migraines, are widely used for CM management, but none of these drugs has been approved for CM prophylaxis (36–38). Topiramate, a relatively new anticonvulsant, has been shown to be effective in placebo-controlled randomized trials for CM (13,14,16). We compared our results with those of the other two CM preventive studies; one is the largest trial for topiramate prevention for CM and the other is pooled data of PREEMPT studies (13,19). The mean number of headache days decreased by 10.7 days per 4 weeks for acupuncture group and by 7.9 days per 4 weeks for topiramate group in our study, and this difference was significant. The 7.9 days decrease in the mean number of headache days for topiramate group in our study is comparable to that for topiramate group reported by Silberstein et al. (13) (6.4 days per 4 weeks), and to that for botulinum toxin group reported by the pooled data of PREMMPT studies (19) (8.4 days per 4 weeks). In our study, the mean dosage of topiramate was 84.0 mg/day, which is consistent with previous studies where low dose topiramate (
This trial has another important finding. It is well known that for preventive agents to be successful, medication overuse must first be controlled (detoxification) (3). It is widespread practice to withdraw patients from medication overuse on an out-patient or in-patient basis with adjunctive treatment. This was clearly not done in the present study because we observed an estimated reduction in the number of monthly moderate/severe headache days and number of headache days for the acupuncture group for all participants and for participants who were overusing acute headache medication during the 28-day baseline period. Similar effects were observed for the topiramate group. This indicates that overusing medication did not have an impact on treatment effect, and that withdrawing patients with CM from medication overuse may not be necessary prior to the initiation of acupuncture or topiramate treatment. These findings agree with previously published data observed on topiramate for CM and PREMMPT studies (12,16–19). From another point of view, the improvement across multiple efficacy endpoints among the acupuncture-treated patients compared with topiramate-treated patients, with an accompanying significant difference in the mean number of days on which acute headache medication was taken, may be that acupuncture made it easier for the patients to withdraw the acute headache medication and that the improvement resulted from detoxification and not from the acupuncture itself.
Traditional acupuncture has its own diagnostic system for headaches, in that treatment should be individualized (acupoints vary from person to person or from treatment to treatment). However, the absence of standardized approaches to acupuncture treatments obstructs data reproducibility across the discipline. With this in mind, we used fixed and classic acupuncture points (standardized acupuncture) in all patients during every therapeutic session. This makes our study reproducible and as scientifically rigorous as possible. However, this procedure could have limited the real effect of individualized acupuncture. Further studies are needed to confirm whether real differences can be appreciated between individualized and standardized acupuncture.
Some might ask how we chose the acupoints and how they work for CM. It is assumed that a variable combination of peripheral effects, spinal and supraspinal mechanisms, and cortical, psychological or 'placebo’ mechanisms contribute to the clinical effects of acupuncture (41). Current theories suggest that migraine is a neurovascular disorder involving cortical spreading depression, neurogenic inflammation, and vasodilation (42). Sensitization and facilitation of pain transmission in central trigeminal sensory pathways may have a particularly important role in the development of CM (43). A recent study suggests that acupuncture may have anti-inflammatory action via release of neuropeptides from nerve endings, including calcitonin gene-related peptide (CGRP), an important mediator of neurogenic inflammation and a potent vasodilator during migraine attack (41,44). We selected acupoints in the distribution of the trigeminal and cervical dermatomes because we postulated that an interaction between trigeminal and cervical nociceptive inputs to the trigeminocervical complex via acupuncture may inhibit trigeminal-vascular activation and thus may inhibit migraine attack. It would be of great interest to explore the effects of these acupoints on the trigeminal-sensory pathway to provide insight into the mechanism of these acupoints, although it is beyond the scope of this article. It is also important to understand what roles the peripheral as well as the central mechanisms have in CM patients after acupuncture treatment in future studies.
There were several limitations to our study that deserve consideration. The most relevant is the use of an active comparator without sham controls, which cannot rule out the possibility of a placebo effect. A high placebo response has been observed in migraine prophylaxis studies, which may also contribute to the between-group difference observed in our study (45). Parenteral procedures such as acupuncture treatment consistently report higher placebo response rates than those seen in trials using oral medication (46,47). The placebo response by placebo injection in the PREEMPT trials was very high, and this mode of intervention is very similar to acupuncture. We were aware of and capable of using sham acupuncture, but we decided not to because the validity of sham acupuncture and the best way of inducing its placebo effect are still open questions (31,48,49). In addition, the aim of this study was to evaluate the effectiveness of acupuncture in comparison with the optimal treatment for CM in current clinical practice. Furthermore, if both treatments are possibly effective, it is easier to encourage patients to join the study and there are fewer ethical problems in adopting an active treatment arm for patients who are looking for a treatment for their discomfort. Therefore, an active group instead of sham control was used in this study, and topiramate, one of the most effective drugs in CM prophylaxis, was chosen as a comparison. Another limitation is that acupuncture group had more study visits (24 visits in total) compared with the topiramate group (6 visits in total). In addition, longer contact time with a close interaction between patient and acupuncturist certainly also contributed to the observed effect. Although the researchers were advised not to make additional contact with the study participants if possible, a greater placebo effect could have occurred in acupuncture group, in which patients unavoidably received more attention and hands-on treatment. Other limitations of our study are the study design, having a small number of patients, the mono-centric study, and the unblinded study combined with the use of subjective outcomes only. Consequently, the present findings must be interpreted with caution.
Conclusions
Despite the limitations, this randomized, controlled study indicates that acupuncture treatment is superior to topiramate treatment for patients with CM across multiple clinically relevant variables, including number of moderate/severe headache days per month, headache days per month, headache disability, and several patient-reported quality of life measures as well as psychological distress. These significant differences still existed when we focused on those patients who were overusing acute headache medication. Acupuncture should be considered as a treatment option for CM patients willing to undergo this prophylactic treatment, even for those patients with medication overuse. Future clinical trials should focus on the long-term effects of acupuncture on CM.
Footnotes
Acknowledgements
This study was registered with the National Institutes of Health, ClinicalTrials.gov identifier NCT01096420.
Funding
This work was supported by a grant from Hung Kuang University and Kuang Tien General Hospital.
