Abstract
Background: The hypothalamus has been discussed as a pivotal structure for both cluster headache (CH) and aggressiveness, but little is known about the extent of self-reported aggressiveness in patients with CH.
Patients and methods: Twenty-six patients with chronic, 25 with active episodic and 22 with episodic CH outside the active period were examined interictally with a validated questionnaire quantifying factors of aggression and compared with 24 migraine patients and 31 headache-free volunteers.
Results: The ANOVA was significant for the subscale ‘self-aggression/depression’ (F4, 123 = 5.771, p < 0.001) with significant differences between chronic and episodic CH and healthy volunteers. No significant changes were found for other subscales and the sum scale (F4, 123 < 1.421, p > 0.230). Especially in the clinically most affected group of patients (chronic CH and active episodic CH), high levels of “self-aggression/depression” correlate with higher prevalence of depressive symptoms and higher impairment measured on an emotional and functional level.
Discussion: Self-aggressive and depressive cognitions with highest scores in chronic CH seem to be reactive as they correlate with depressive symptoms and impairment. They should be considered as an important therapeutic target since they impair the patient’s life significantly.
Introduction
Cluster headache (CH) is a severe and disabling condition characterized by excruciating unilateral pain attacks located mainly in the first trigeminal division lasting between 15 and 180 minutes (1). Attacks are accompanied by striking ipsilateral signs of parasympathetic activation and sympathetic hypoactivity (2) and lead to significant impairment (3,4). As many as 90% of patients report restlessness, with visible motor symptoms such as pacing around, rocking or agitation (5). Patients often report further behavioural peculiarities ranging from complex stereotyped actions to seeking seclusion and often even refuse to see their family members. Self-inflicted injuries (such as banging their head or knocking their fists against a wall), and other self-aggressive respectively self-mutilating behaviour have also been reported (5–8). The rate of self-inflicted traumatic head injuries is reported to be higher in patients with CH than in migraine, especially for brawls (9). However, increased aggressiveness towards others has never been described as a typical finding and is mentioned only anecdotally (10–12).
Aggressiveness is a complex phenomenon and can be a correlate of other underlying disorders such as depression and pain (13,14). As previous studies have reported significant psychiatric symptoms in patients with CH (4,15–17), higher levels of aggression – if present – could be explained by this concomitant spectrum of disease. From a pathophysiological perspective, there is good evidence for the involvement of the ventro- and dorsomedial as well as the posterior hypothalamus in the pathophysiology of aggression (18). Interventional data from animal experiments (19) and stereotactic neurosurgery of the posterior hypothalamic region in humans (20) have shown striking effects on aggressive behaviour. However, aggression cannot be reduced to the hypothalamic system alone. Extensive changes in neurotransmitter release have been described in an intricate network of areas such as the orbitofrontal and the anterior cingulate cortex (top-down control) and limbic structures, mainly the amygdala (21).
It is intriguing that neuroimaging studies have found morphological and functional changes in the posterior hypothalamus in patients with CH (22,23) as well, suggesting a role in the pathophysiology of CH. This is supported by the efficacy of deep brain stimulation of the posterior hypothalamus in medically intractable CH (24–26).
It is not known whether patients with CH have higher levels of aggression compared to other primary headaches and healthy volunteers. In addition, it is unknown whether these changes are confined to acute attacks as a facet of intermittent hypothalamic activation or active periods (as a transient state). Alternatively, they could be constantly present as personality traits.
The aim of the study was therefore to answer the following questions in a prospective and controlled multicentre study:
Do patients with episodic CH in the active period, episodic CH outside the active period, or chronic CH show higher degrees of interictal aggression and its submodalities compared to patients with migraine and healthy volunteers? Is there any relationship between aggression scores and depressive symptoms? Is there any relationship between aggression scores and headache-specific emotional and functional impairment?
Patients and methods
Patients
Clinical details of patients included and results. Results on depressive symptoms have been published as part of a previous study (17)

CCH: chronic cluster headache; ECHa: episodic cluster headache in the active phase; ECHi: episodic cluster headache outside the active period; MIG: migraine; Control: healthy controls; SD: standard deviation; FAF: Questionnaire for Measuring Factors of Aggression. One subject each in the CCH and ECHa group did not complete the FAF questionnaire.
Study design
All participants completed a face-to-face standardized interview to screen for psychiatric disorders (axis I), which is used widely in Germany (Mini-DIPS) (27). Responses to screening questions indicative for depressive syndrome were explored. Afterwards, subjects were asked to complete a questionnaire assessing modalities of aggression (FAF; see below for further information) and return it in a sealed envelope to minimize under-reporting as a modification of the sealed envelope approach (28). To ensure that data was collected interictally and to avoid loss of data patients were requested to complete all questionnaires in the clinic.
Questionnaire for Measuring Factors of Aggression (FAF)
This questionnaire reliably assesses various modalities of aggressive behaviour with 77 items. Subjects affirmed or negated FAF items in a dichotomous fashion (‘Occasionally I lose my temper and become angry’: yes or no) (29). The FAF evaluates five dimensions of aggressive behaviour: 1 spontaneous aggression (thoughts about aggression against others); 2 reactive aggression (degree of socially accepted self-assertion); 3 irritability (tolerance to frustration); 4 self-aggression/depression (dissatisfaction with own personality, self-reproach); and 5 inhibition of aggression (ability to regulate aggressive thoughts). A sum score can be calculated for the first three scales. The FAF shares some items with the Freiburg Personality Inventory and is partly based on the American Buss Durkee Hostility Inventory (30). A simple translation of the latter would not have adequately addressed cultural peculiarities for a German-speaking population.
German FAF norms were established in 630 subjects (males and females ranging from 15 to 75 years of age). The construct was validated in juvenile and adult delinquents who showed significantly increased scores for the scales 1 (spontaneous aggression), 3 (irritability), 4 (self-aggression/depression) and the sum score compared to the sample drawn from the general population. The scales do not correlate with the general intelligence quotient measured by the Wechsler Adult Intelligence Scale. The internal consistency (Cronbach’s α) differs between 0.61 and 0.79 for the subscales and reaches 0.85 for the sum score. The split-half reliability (Spearman–Brown) reaches 0.857.
Adult delinquents (n = 77) showed significant differences in the irritability and self-aggression scales and in the sum of aggression scales in comparison to controls. The difference in self-aggression was attributed to the consequences of the imprisonment. Hampel and Selg (29) also report that the spontaneous aggression scale reached higher scores in psychiatric patients than in psychosomatic patients, lower scores in reactive aggression for both groups and higher scores for both patient groups in the irritability and the self-aggression scales. For the inhibition scale higher scores were found in psychiatric patients, whereas psychosomatic patients scored lower.
Because of this sensitivity to different aspects of aggressive behaviour, the questionnaire has been used widely in German-speaking populations in various clinical and non-clinical contexts. It was used to measure the relationship between aggression and allergic disorders (31,32), rheumatoid arthritis (33) and patients with hand dermatitis (34). Additionally, the FAF was used to correlate behaviour with specific genotypes in psychiatric patients (35–39) and in personality diagnostics in Austrian (40) and Swiss psychiatric patients (41).
Headache Disability Inventory (HDI)
All participants completed the German adaptation of the 25-item Henry Ford Hospital Headache Disability Inventory (HDI) (42,43) with a ‘total’ score and two subscores (EMOTION with 13 items for the influence of headache on mood) and FUNCTION (12 items to detect restrictions in activities of daily living). Detailed results can be found elsewhere (4).
Statistics
Data were analysed with SPSS 18 for Windows (SPSS; Chicago, IL, USA). The scores of the six subscales and the sum scale were entered separately in univariate analyses of variance (ANOVAs) with the between-factor ‘group’. If variance homogeneity was violated, effects were verified by means of non-parametric Kruskal–Wallis H-test. Post hoc tests were used to further investigate significant main effects. Bonferroni correction was used to correct for multiple testing (10 comparisons required puncorrected < 0.005 to achieve global p < 0.05). When necessary for detailed understanding of results non-corrected least significant differences are reported (i.e. puncorrected < 0.05).
Further analyses tested if revealed that a linear model would fit best to detect a polynomial contrast despite non-significant main effects (such as a linear increase or decrease in a measure from the chronic CH patients over other patient groups to healthy controls). We thus applied a polynomial linear contrast weighting groups (2 1 0 –1 –2) in the expected order (CCH, ECHa, ECHi, MIG, HC). As we had hypotheses about the order of groups (highest values in the CCH group, lowest values in the HC group), we applied one-tailed testing for post hoc comparisons.
To investigate any association of the self-aggression/depression scale with depressive symptoms we tested whether subjects reporting previous depressive symptoms had increased scores on this scale by means of an ANOVA.
Bivariate correlations were calculated to examine a potential relation between the degree of impairment and the extent of FAF4 scores for self-aggression/depression (Bonferroni-corrected). Pairwise comparisons between the presence of depressive symptoms in the past and total scores on the HDI were calculated by means of Mann–Whitney U-tests (non-parametric tests were used because variables were unevenly distributed).
Results
Clinical data for the headache patients and the controls are shown in Table 1. FAF data were incomplete and thus regarded as missing for one chronic and one CH patient in the active episode. Results for FAF total score are given in Table 1, for all subscores in Figure 1.
Mean FAF subscores given for the different groups (cluster headache, migraine, healthy volunteers). Whiskers represent standard error of the mean. Results of Bonferroni corrected post hoc t-tests are given as follows: ***< 0.001, **< 0.01, +< 0.10 (one-tailed). FAF: Questionnaire for Measuring Factors of Aggression; FAF 1: spontaneous aggression (thoughts about aggression against others); FAF 2: reactive aggression (degree of socially accepted self-assertion); FAF 3: irritability (tolerance to frustration); FAF 4: self-aggression/depression (dissatisfaction with own personality, self-reproach); FAF 5: inhibition of aggression (ability to regulate aggressive thoughts). CCH: chronic cluster headache; ECHa: episodic cluster headache in the active phase; ECHi: episodic cluster headache outside the active period; MIG: migraine; HC: healthy controls.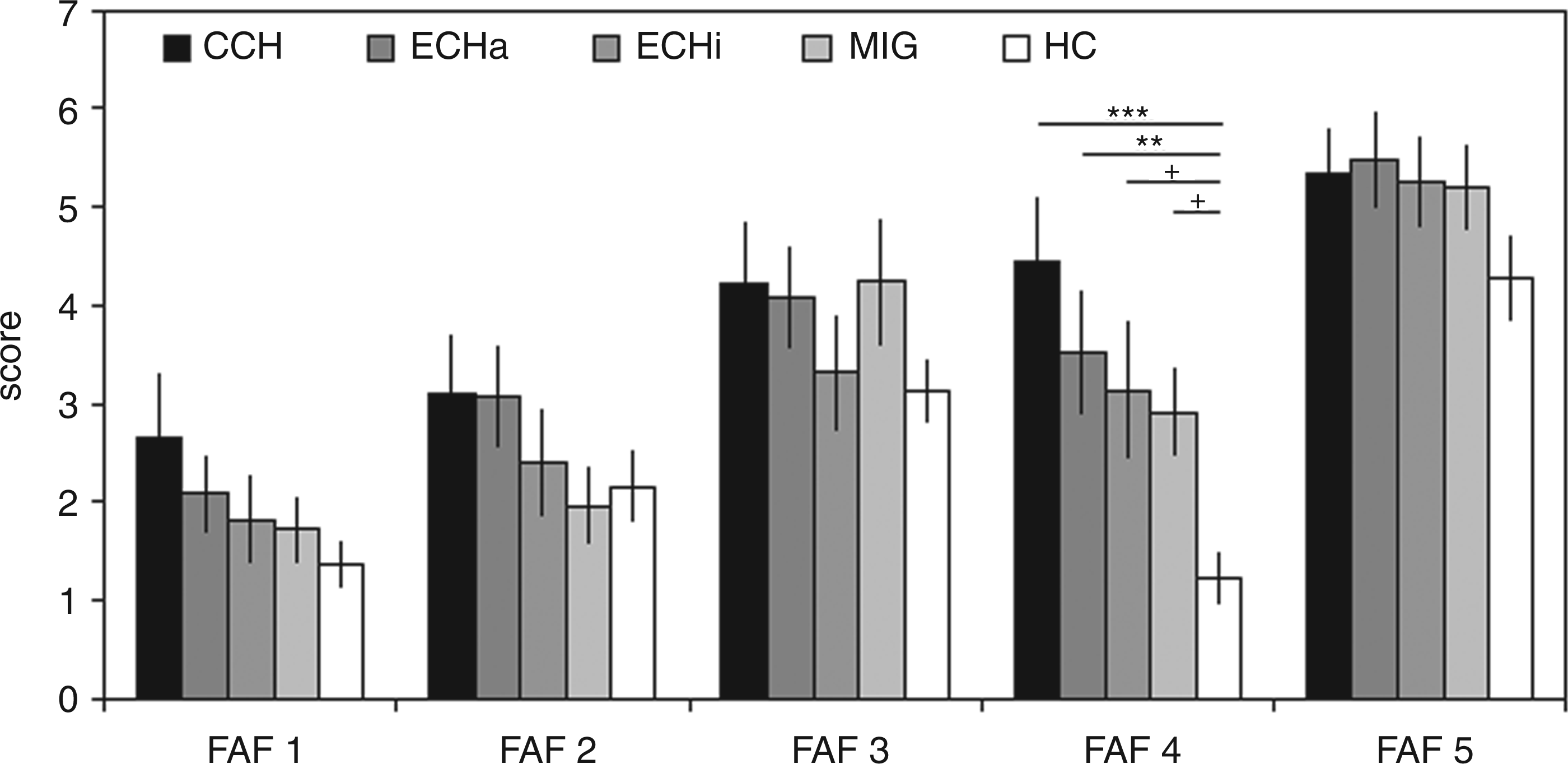
FAF scores
The ANOVA proved to be significant for the self-aggression/depression subscale (F4, 123 = 5.771, p < 0.001), but not for the other subscales and the sum scale, respectively (F4, 123 < 1.421, p > 0.230). Due to violation of variance homogeneity for some scales, the Kruskal–Wallis H-test was used, the results of which matched those from the ANOVA. Bonferroni-corrected post hoc t-tests revealed that for self-aggression the HC group displayed significantly lower values compared to CCH (p < 0.001) and ECHa (p = 0.007), and marginally lower values compared to the ECHi (p = 0.052) and MIG groups (p = 0.099). No significant differences between the headache patient groups could be found. At an uncorrected level, CCH patients also had significantly higher values than the ECHi and MIG groups (see Figure 1).
A significant linear trend indicating decreasing levels of self-aggression/depression from CCH to HC was found (F1, 123 = 20.997, p < 0.001, Figure 1). Although main effects for spontaneous and reactive aggression and for the sum scale did not reach significance, linear trends were significant for these three scales (F1, 123 > 4.246, p < 0.041). At an uncorrected level, post hoc t-tests reached significant differences (see Figure 1). Namely, for spontaneous aggression CCH had higher values than the HC (p = 0.012), for reactive aggression patients with CCH and ECHa had higher values than the MIG group (p < 0.05), and for the sum scale aggression CCH had higher values than the HC group (p = 0.018).
Relationship with depressive symptoms
A 2 × 5 ANOVA was performed with the factors ‘depressive symptoms’ (depressed in the past vs. not depressed in the past) and ‘group’ (active and inactive episodic and chronic CH, migraine and healthy controls) for each of the five FAF subscores and the sum score to explore a significant main effect of ‘depressive symptoms’. For the dependent variable self-aggression/depression the analysis revealed a significant main effect for ‘depressive symptoms’ (F1, 113 = 17.257, p < 0.001) and group (F4, 113 = 4.201, p = 0.003) while the interaction term proved to be non-significant (F1, 113 = 0.346, p = 0.846). That is, all patient groups and the HC group displayed a pattern of higher scores in self-aggression/depression when reporting previous depressive symptoms. The group effect replicates the general finding mentioned above. No significant main effects for ‘depressive symptoms’ were found in the other FAF subscales and the sum scale (Figure 2).
Mean FAF subscores in all participants subject to depressive symptoms in the past. Occurrence of depressive symptoms for subscale FAF4 is indicated in the small additional figure in a dichotomous fashion (depressive symptoms present in the past or not) to illustrate comparable effects across groups. Whiskers represent standard error of the mean. FAF: Questionnaire for Measuring Factors of Aggression; FAF 1: spontaneous aggression (thoughts about aggression against others); FAF 2: reactive aggression (degree of socially accepted self-assertion); FAF 3: irritability (tolerance to frustration); FAF 4: self-aggression/depression (dissatisfaction with own personality, self-reproach); FAF 5: inhibition of aggression (ability to regulate aggressive thoughts). CCH: chronic cluster headache; ECHa: episodic cluster headache in the active phase; ECHi: episodic cluster headache outside the active period; MIG: migraine; HC: healthy controls.
Relationship with impairment
Correlation of FAF 4 scores (self-aggression/depression) with impairment scores in the German adaption of the Headache Disability Inventory. Pearson’s correlation coefficient is reported with p-values in parentheses. Results that remain statistically significant (p < 0.05) after Bonferroni’s correction are printed in bold

CCH: chronic cluster headache; ECHa: episodic cluster headache in the active phase; ECHi: episodic cluster headache outside the active period; MIG: migraine; Control: healthy controls; FAF: Questionnaire for Measuring Factors of Aggression; HDI: Headache Disability Inventory.
Relationship of depressive symptoms and impairment
HDI scores depending on depressive symptoms in the investigated groups. Statistics refer to results of non-parametric Mann–Whitney U-tests, as distribution of subjects retrospectively reporting depression within the groups was uneven. For some controls, HDI scores were not available. Significant results (p < 0.05) are printed in bold

CCH: chronic cluster headache; ECHa: episodic cluster headache in the active phase; ECHi: episodic cluster headache outside the active period; MIG: migraine; Control: healthy controls; FAF: Questionnaire for Measuring Factors of Aggression; HDI: Headache Disability Inventory.
Discussion
The ANOVA proved to be significant for the self-aggression/depression subscale but not for the other subscales and the sum scale. Post hoc tests revealed significantly increased self-aggression scores for patients with chronic CH and episodic CH in the active period compared to healthy volunteers, but not among the headache groups. Participants who reported being depressed in the past showed an increase in the FAF self-aggression/depression subscore. Additionally, the degree of self-aggression/depression was associated with headache-specific impairment, especially in patients with chronic and active episodic CH. This, â history of depression coincided with greater impairment.
Self-aggressive cognition, depressive symptoms and impairment
The ‘self-aggression/depression’ domain, as measured in the FAF, represents feelings of dissatisfaction with one’s own personality, tendencies of self-reproach and depressive symptoms. Self-aggression in terms of physical aggression directed against oneself leading to self-mutilation or self-inflicted injuries are not covered. In a previous study (4) in the same sample, the lifetime prevalence of depressive symptoms was found to be highest in patients with chronic CH, followed by those with episodic CH and migraine and was shown lowest in healthy controls. Impairment scores showed a similar distribution, with highest scores among patients with active CH, lower scores in the ECHi and MIG groups and lowest scores in healthy controls. We could show that patients with active CH and depressive symptoms in the past suffered from significantly higher impairment (as confirmed with the total score of the HDI) than patients with inactive CH, migraine or healthy controls. These results are in line with studies showing elevated incidence of depression in CH patients (15,17). Increased disability was associated with increased depression scores (17).
Our findings corroborate these data as participants reporting depressive symptoms in the past also displayed increased scores in the self-aggression/depression domain only. In line with these observations, a positive relationship was found between levels of self-aggression and scores on the German adaptation of the HDI (total, emotion and function). Headache patients with a history of depressive symptoms showed higher scores of self-reproach and dissatisfaction than those without symptoms of affective distress in the past; these were most pronounced for patients in the CCH and the ECHa groups. These results support the notion that severely affected patients with CH suffer from a significant and complex psychosocial impairment, as shown in other studies (see (4) for further review). The factors ‘self-aggression’, ‘depressive symptoms’ and ‘impairment’ correlate with each other and it is tempting to propose a common pathophysiological ground.
Higher levels of self-aggression/depression could at least be partly explained by higher pain densities in patients with CH. However, it seems noteworthy that patients with inactive ECH, who have a significantly lower current pain density than migraine patients, had higher levels of self-aggression/depression than migraine patients. This would argue against pure pain-related effects.
It is conceivable that the observed psychiatric comorbidity could be one reason for increased aggression (14), as found in other studies which focused mainly on delinquents, patients with personality disorders and patients with suicidal tendencies (35–41). Interestingly, in our study population patients with CCH frequently reported suicidal tendencies in the past. However, we could only find increased scores of internalizing aggressive behaviour that is more associated with depressive symptoms/self-reproach, whereas increased externalizing aggressive behaviour could not be found. Therefore, any causal attribution would be speculative and is far beyond the scope of our study.
Role of the hypothalamus in CH and aggression
Several lines of evidence point to the hypothalamus as a crucial pathophysiological structure in patients with CHs. Neuroimaging (22,23) and studies on hormonal regulation suggest hypothalamic involvement (44,45) and consequently deep brain stimulation of the posterior hypothalamus was successfully established in refractory CH (25,26,46–49). Interestingly, the posteromedial hypothalamus had also been a target for stereotactic lesions not only in intractable pain (50) but also in aggressive behaviour (20). Accidental intraoperative stimulation of the adjacent ‘triangle of Sano’ (region between the posterior hypothalamic area medially and the medial subthalamic nucleus laterally) led to an outburst of previously unknown aggressive behaviour in a patient with Parkinson’s disease (51). In animal experiments, various parts of the hypothalamus were shown to be involved in different facets of aggression (18,19,52,53). Accordingly, in humans aggressive behaviour can be ameliorated by stimulation of the posterior hypothalamus (54,55).
Although these studies suggest a potential dual role of the posterior hypothalamus in both aggression and CH, our results cannot support such a hypothesis.
Aggression in migraine
In the present study, no significant differences were found for factors of aggression in migraine patients compared with CH patients and healthy controls apart from self-aggression/depression. Self-aggression in migraine patients compared to healthy controls showed a trend towards higher scores. Likewise, the positive correlation between self-aggression and impairment reached significance on an uncorrected level for the total score and the emotion subscale on the HDI. However, a history of depressive symptoms had no influence on the level of self-aggression.
Limited data is available for aggression in migraine. Although not statistically significant, the low level of inhibition of aggression in migraine patients was comparable to that of CH patients. This is supported by studies in adults, adolescents and children with migraine, which have shown a reduced ability to repress aggression and anger (56–58). Irritability was highest on the day before an attack (46%) and still present in 21% of migraine patients interictally (59).
Our findings of increased self-aggression in migraine patients are generally in line with the changes observed for patients with active CH, however they were less pronounced. Increased levels of depression coincide with increased self-aggression. Depression has been extensively reported in migraine (60), but there is only limited data comparing the prevalence of depression in different primary headaches. As reported elsewhere (4), in our study population a history of depressive symptoms could be found most frequently in patients with CCH, while those with active and inactive episodic CH reported depressive symptoms as frequently as those with migraine. Gesztelyi and co-workers found that migraine patients had even higher median scores in Beck’s Depression Inventory (median: 8) than patients with CH (median: 5) and healthy volunteers (median: 2) with highest disability in the CH group followed by the migraine group. However, as a limitation group sizes differed strongly (61). Although a history of depressive symptoms was not associated with higher impairment in our study, quality of life in patients with migraine was lower than in healthy controls and higher than in patients with active CH in another study (62).
Limitations
Because of the nature of our study, aggression was only examined interictally. Therefore, we cannot rule out that ictal changes within an acute attack were missed. However, it did not seem feasible to ask the patients with active CH to complete a questionnaire during an attack. In future studies, interviewing partners of patients should also be considered and eventually searching for signs of aggression on a physical level, in contrast to the cognitive approach with specific questionnaires in this study.
Conclusion
Our findings did not corroborate significantly increased levels of aggression other than self-aggression in the context of self-reproach as a correlate of depression in patients with active CH. In line with previous studies, self-aggression correlated with emotional and functional measures of impairment with highest disability in the clinically most affected groups with active CH. Likewise, we could substantiate a close relationship between reporting of depressive symptoms and headache-specific impairment. As our study was not designed to detect a causal interrelationship between these three dimensions, further studies will have to address this issue.
As we could not corroborate any significant effect beyond self-aggression, our data do not support a dual role of the hypothalamus in the generation of both aggression and CH. As a clinical consequence, self-aggression in the context of depression should be clinically monitored and therapeutically targeted.
Footnotes
Acknowledgements
This study was conducted by members of the DMKG (German Migraine and Headache Society) network of young researchers.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
RL, TD, YPH, AL and EL: no conflict of interest. KH: has received honoraria from Gruenenthal. CG: has received research grants and honoraria from the Roux-Program of the University of Halle-Wittenberg, MSD, Berlin Chemie, Medtronic, Allergan and Böhringer Ingelheim. TPJ: has received grants and honoraria from MSD.
