Abstract
Background: The occurrence of hemifacial spasm (HFS) during an episode of migraine has been seldom reported. Here we describe three new cases presenting with HFS in association with migraine attacks.
Case results: Three patients (one woman and two men, aged 31–36 years) developed HFS in close temporal relationship with migraine headaches. All of them started having the muscle spasms after pain onset. Two of them had electromyographic evidence of facial nerve damage, and continued having HFS once the pain abated.
Conclusions: Migraine attacks may be associated with HFS. The appearance of HFS could be related to migraine activity. A mechanism of central hyperexcitability in connection with nociceptive inputs on the trigeminal nucleus caudalis and/or a dilation of vessels compressing the facial nerve at the root exit zone could lead to the development of HFS in predisposed patients. ‘Migraine-triggered hemifacial spasm’ could possibly be regarded as a complication of migraine.
Introduction
Hemifacial spasm (HFS) is characterized by involuntary contractions of the facial muscles innervated by the facial nerve. Most cases are caused by vascular compression of the nerve at the root exit zone. The appearance of HFS during an episode of migraine has already been acknowledged, but it has been reported only very rarely (1,2). Here we describe the clinical features of three new patients presenting with HFS in close temporal relationship with migraine headaches. We consider the possibility that the development of this movement disorder could be related to migraine activity.
Case reports
Magnetic resonance imaging (MRI) of the brain, a magnetic resonance angiography (MRA) and an electroencephalogram (EEG) were normal. An electromyogram (EMG) and a blink reflex were performed between attacks, and showed no signs of facial nerve damage. After initiating treatment with topiramate 100 mg per day, the patient experienced a significant decrease in both the frequency and intensity of the headaches, which were no longer associated with muscle spasms.
A head computerized tomography (CT) scan was normal and a lumbar puncture revealed no xanthochromia. The headache abated spontaneously and the spasms improved in the emergency room, but some facial twitching remained. An EEG was performed, and showed no significant abnormalities. Twenty-four hours after onset, an EMG demonstrated bursts of muscle activity in the right orbicularis oculi and the right orbicularis oris consistent with HFS (Figure 1). The blink reflex showed an increase in the R1 latency on the right side compared with the healthy side (14.2 ms vs. 12.4 ms), reflecting minor damage of the right facial nerve. An MRA study revealed a vascular loop of the anteroinferior cerebellar artery penetrating into the right internal auditory canal, just close to the affected nerve (Figure 2). The patient was eventually discharged, and 2 weeks later he had no headache but continued to have mild HFS.
EMG demonstrating bursts of muscle activity in the right orbicularis oculi and the right orbicularis oris consistent with hemifacial spasm. Magnetic resonance angiography, three-dimensional time-of-fligh, maximum intensity projection reconstruction, axial view at the level of the cerebellopontine angle. A vascular loop of the right anteroinferior cerebellar artery can be seen inside the right internal auditory canal (arrow).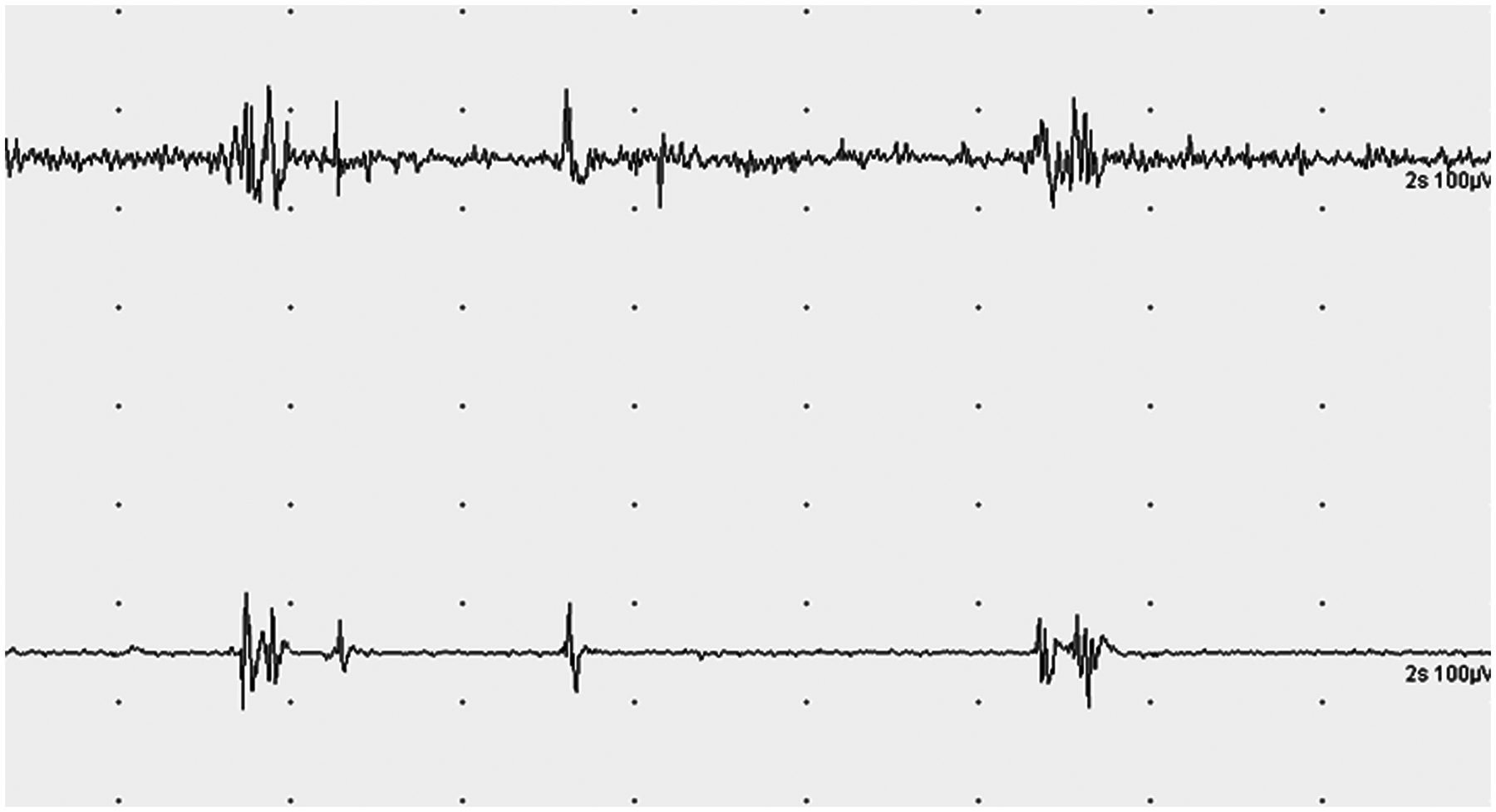
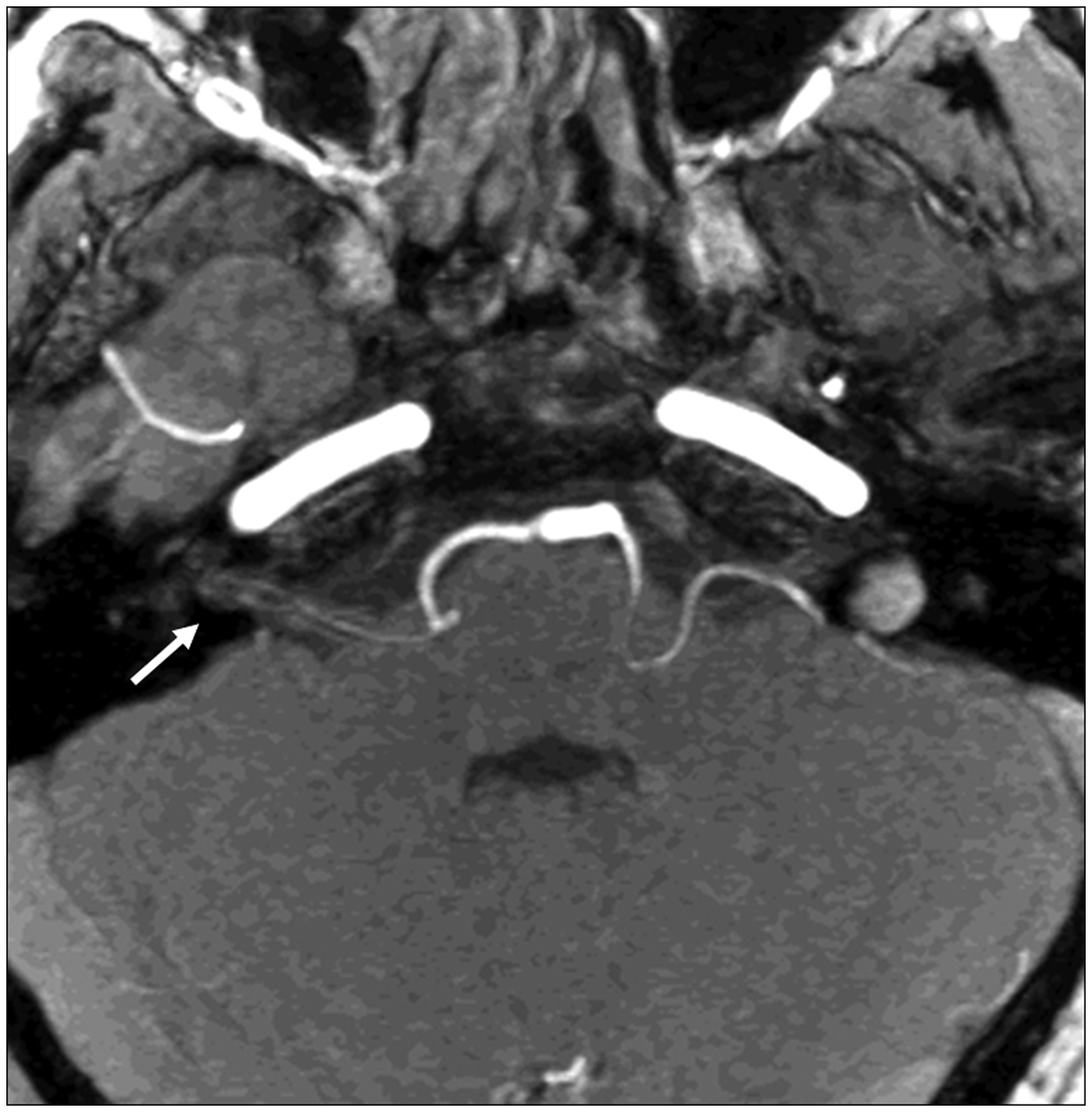
An initial head CT was normal. The pain subsided in 4 hours, but the facial muscles remained asymmetrical. Twenty-four hours later, an EMG showed persistent muscle activity in the left orbicularis oculi. The blink reflex exacerbated such activity, with superposition of a clonic component. In addition, the R1 latency was prolonged on the affected side. High-resolution MRI identified a vessel in contact with the left facial nerve at the cerebellopontine angle (Figure 3). At discharge, the patient showed minor facial weakness. Despite treatment with beta-blockers, he had daily headaches through the following month. A tonic contraction of the left facial muscles was almost constant, and clonic jerks were added each time he had a headache.
Three-dimensional fast imaging using steady-state acquisition (3D-FIESTA) magnetic resonance imaging, minimum intensity projection, axial view at the level of the cerebellopontine angle. A blood vessel (long arrow) is seen close to the left facial nerve (short arrow).
Discussion
Main features of all cases of migraine-triggered hemifacial spasm described to date
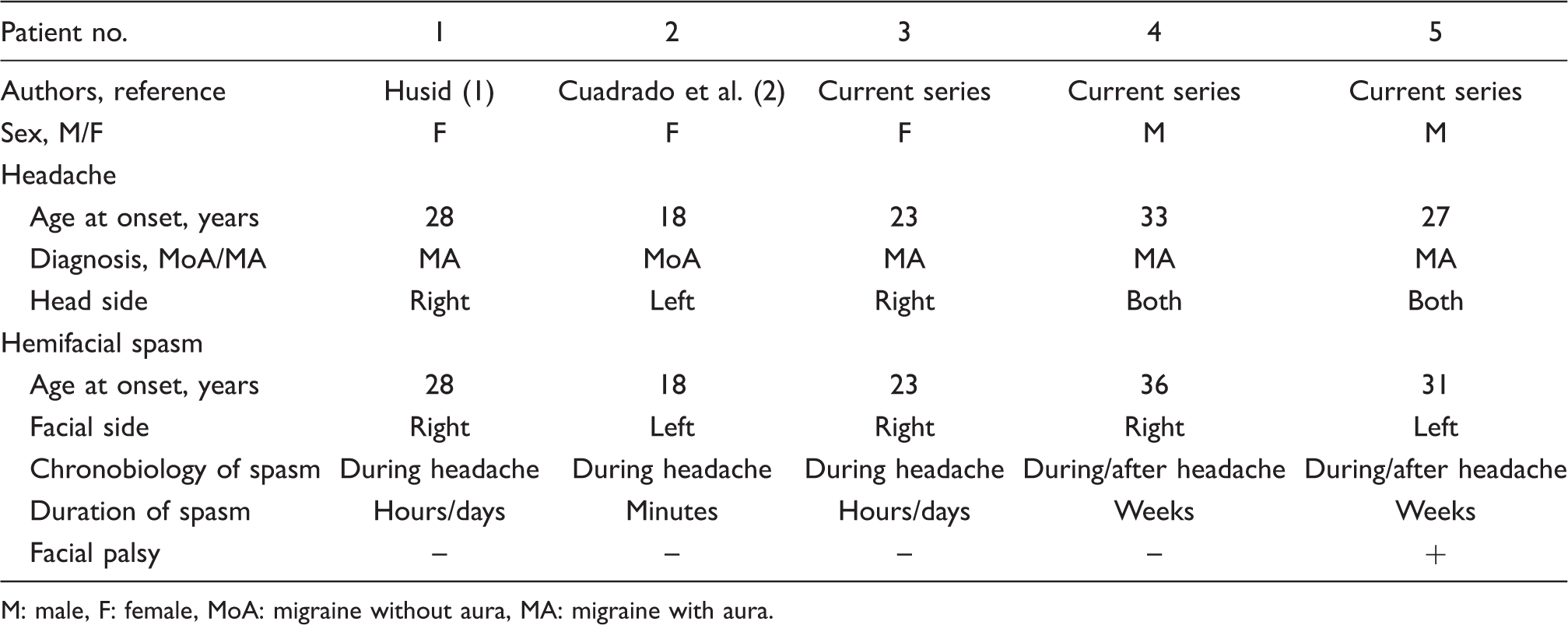
M: male, F: female, MoA: migraine without aura, MA: migraine with aura.
It is unlikely that the two phenomena are simply coincidental because there has been a clear temporal relation of events in each patient. In all the reported cases, the HFS started once the headache reached its maximum intensity. Hence, Cuadrado et al. considered that migraine could be a trigger for HFS, rather than the latter being an aura with positive signs. They suggested that a potentiation of certain trigeminofacial reflexes might trigger the appearance of facial spasms during migraine attacks. Indeed, their patient also had facial spasms when either nociceptive or electrical stimuli were applied within the V-1 region (2). Similar observations were made in our third patient. He had both pain and ipsilateral clonic movements of the face upon compression of the left supraorbital or trochlear areas. In addition, his HFS was exacerbated by the blink reflex test. As formerly proposed, nociceptive impulses converging on the trigeminal nucleus caudalis could result in temporal and spatial summation, thus leading to increased activation of the trigeminofacial connections within the brainstem (2,3).
Although the pathophysiology of migraine is still obscure, a hyperexcitable brain state has been postulated (4,5). Migraine may share a state of neural hyperexcitability with HFS. Kobata et al. described the co-occurrence of HFS with both trigeminal and glossopharyngeal neuralgia, and postulated the existence of a combined hyperactive dysfunction syndrome of the cranial nerves (6). Interestingly, Alonso-Navarro et al. described a 52-year-old woman with migraine and unrelated HFS who experienced significant improvement of HFS after the introduction of topiramate for the treatment of migraine. They speculated on the possibility that topiramate could reduce the ephaptic transmission of the facial nerve (7). Gross reported the successful treatment of HFS with pizotifen in a 43-year-old woman with migraine and suggested that antagonizing 5HT1 receptors might inhibit the nucleus of the seventh cranial nerve (8). Lastly, Husid, who first described the association between migraine and HSF (1), speculated on the possibility of central neuronal hyperexcitability underlying both disorders. Another possible pathophysiological link for this author was the migraine-induced trigeminovascular dilation of vertebrobasilar vessels compressing the facial nerve (1).
Proposed diagnostic criteria for migraine-triggered hemifacial spasm

In conclusion, a coincidental occurrence of migraine and HFS seems unlikely because both phenomena were closely coupled in all the reported patients. Within a migraine attack, a mechanism of central hyperexcitability in connection with nociceptive inputs on the trigeminal nucleus caudalis and/or a dilation of vessels compressing the facial nerve at the root exit zone could lead to the development of HFS in predisposed patients. In some patients both phenomena would resolve together, whereas others would have facial nerve damage perpetuating the spasm. The latter would have a worse prognosis for complete recovery. These three new cases contribute to the idea that ‘migraine-triggered hemifacial spasm’ might be eventually included among the complications of migraine.
