Abstract
Background: It is not known whether smoking by mothers during pregnancy is associated with headache in their offspring.
Methods: Two prospective cohorts of 869 children aged 10–11 years from Ribeirão Preto (RP) and 805 children aged 7–9 years from São Luís (SL) were studied. Data on maternal smoking were collected at birth. Primary headache was defined as a reporting of ≥2 episodes of headache in the past 2 weeks, without any associated organic symptoms.
Results: Prevalence of headache was 28.1% in RP and 13.1% in SL as reported by the mothers and 17.5% in RP and 29.4% in SL as reported by the children. Agreement between mothers’ report and children’s self-report of primary headache in the child was poor. After adjustment, children whose mothers smoked ≥10 cigarettes per day during pregnancy presented higher prevalence of primary headache than their counterparts in both cohorts, as reported by the mother and in RP as reported by the children.
Conclusions: Maternal smoking during pregnancy was associated with headache in 7- to 11-year-olds. With one exception, the consistency of the results, despite poor agreement between maternal and children reports of headache, indicates that maternal smoking during pregnancy may contribute to headaches in their children.
Introduction
Primary headache (migraine and tension-type headache) is an important clinical problem that may start at school age and frequently compromises learning and socialization, leading to specialized medical care (1–4). It is also a good predictor of headache in adolescents (1).
Prevalence of primary headache among schoolchildren varies from 16% in Finland (5) to 49% in Istanbul (2), possibly because of differences in diagnostic criteria and the difficulties that young children may have in explaining their symptoms (2,6). Prevalence rates tend to increase with age and are slightly higher for girls (2).
The etiology of primary headache is still under discussion (2,7,8). Early somatic and psychiatric disorders have been associated with primary headache in preschool children (7). Having a family history of headache, anxiety or psychological problems, low socioeconomic status and exposure to passive smoking at home have also been independently associated with headache (2,9–11). However, variables included in most studies explain about a third of the total variance of headache (8).
Clinical and population studies have shown an association between current tobacco smoking and headache (9,12,13) but there is still doubt that smoking causes headache because most studies were cross-sectional. Tobacco smoking may cause headache as a result of alterations in nitric oxide levels in the brain, decreased monoamine oxidase activity and vascular changes induced by anoxia due to carbon monoxide (12).
If current smoking is associated with primary headache, it is also plausible that smoking of mothers during pregnancy may lead to headache in their offspring. In a recent cross-sectional study, retrospectively recalled prenatal exposure to tobacco was associated with increased prevalence of chronic daily headache in childhood (14).
We are not aware that another group has assessed such a hypothesis in school children using a longitudinal design. Thus data from two birth cohort studies in Brazil were used to investigate whether maternal smoking during pregnancy is associated with primary headache in school-aged children.
Methods
Samples
Data from two cohorts were used; one from Ribeirão Preto (RP) of children born in 1994 and the other from São Luís (SL) born in 1997/98. RP is a wealthy city and SL is located in one of the poorest regions of the country.
The RP cohort consisted of 2,911 individuals born over a period of 4 months (April to August 1994) at ten hospitals in the city, representing 99% of all live births during this period (15). Fewer than 5% of mothers did not answer the questionnaire owing to refusal or early hospital discharge. The SL cohort consisted of 2,541 newborns enrolled in a systematic sample in which one out of each seven births at ten hospitals in the city were selected from March 1997 to February 1998 (16). Losses due to refusal or inability to trace the mother occurred in 5.8% of cases. The sample represented 96.3% of all city births (16).
Samples from the two cohorts were followed up in primary school (17). We stratified five birth weight groups for follow-up: very low birth weight (VLBW, <1500 g), low birth weight (LBW, 1500–2499 g), insufficient birth weight (2500 g–2999 g), normal birth weight (3000–4249 g) and high birth weight (HBW, ≥4250 g, children whose birth weight was at least two standard deviations above the population mean). Children within the weight ranges with the smallest number of newborns (VLBW, LBW, HBW) were over-sampled. In the two cities, children from the samples were searched for in schools rolls. Mothers or caregivers of children in the VLBW, LBW or HBW groups and a fraction of one out of three in the insufficient and normal birth weight groups were invited to participate in the study by telephone or by post. In SL, after excluding multiple births (n = 50), stillbirths (n = 48) and deaths occurring in the first year of life (n = 65), 926 children were eligible for follow-up. We were able to trace 673 children from the original sample, comprising 5 VLBW, 76 LBW, 19 HBW and 573 normal and insufficient birth weight, which is a follow-up rate of 72.6%. In RP, after excluding multiple births (n = 65) and deaths occurring in the first year of life (n = 47), 1,150 children were eligible for follow-up. The follow-up rate was 68.7% and included 790 school-aged children (24 VLBW, 145 LBW, 28 HBW and 593 normal and insufficient birth weight). Missing data on headache (n = 27 in RP and n = 18 in SL when considering maternal report and n = 33 in RP and n = 57 in SL when child report was used), and girls who had already reached menarche (n = 21 in RP) were excluded because menses are associated with headaches (18).
We needed a sample size of 698 to detect, at a 5% level with a power of 80%, a 10% difference in the prevalence of headache among exposed and non-exposed groups (comparing mothers who smoked with those who did not smoke during pregnancy), assuming the prevalence of headache at about 25%.
Data collection and instruments for investigation
Anthropometric infant data and information about pregnancy, delivery, and postpartum period were obtained at birth. Information about headache was obtained between 2004 and 2005 in RP when the children were 10 to 11 years old, and between 2005 and 2006 in SL, when the children were seven to nine years of age. A questionnaire addressed to the person responsible for each child (usually the mother) was used. A research assistant explained carefully the questions to participating mothers and children. The completed questionnaires were reviewed for accuracy.
Variables
The dependent variable was primary headache, dichotomized into present or absent. Primary headache was defined as ≥2 episodes of headache in the past 2 weeks, without any associated organic symptoms, regardless of duration, intensity or location of the episode, as endorsed by the mother. Headache in the past week may have included acute episodes that could have been associated with viral diseases. This criterion was established following the International Classification of Headache Disorders guidelines (19). The children were also independently asked the same question.
The independent variable was maternal smoking during pregnancy (zero, 1–9 and ≥10 cigarettes per day). The continuous covariate factors in the analysis were maternal age (years), age of child (months), birth weight (grams), body mass index (kg/m2) and gestational age at birth based on the date of the last normal menstrual period (weeks). The categorical covariates were marital status (married, cohabiting, single), maternal full time education in years (0 to 4, 5 to 8, 9 to 11, ≥12), paternal smoking during child’s pregnancy (yes, no) and sex of the child.
Data on variables collected at school age, exposure to second-hand smoke at home, high blood pressure and mental health problems were also used in the analysis. Exposure to second-hand smoke at home was assessed with the question: ‘Has anyone who lives in the child’s house smoked at home in the last 12 months?’ Possible mental health problems were assessed using the Strengths and Difficulties Questionnaire (SDQ) (20), as completed by the caregiver. Total score was classified as normal (0 to 13), borderline (14 to 16) or positive (≥17) (20). The questionnaire has been adapted and validated for Brazilian children (21).
Blood pressure was measured three times with a 15-minute interval between measurements using an OMRON 712C digital sphygmomanometer. The same person took the measurements with the child resting and seated, having his/her arms on the same line with the heart, using recommended dimensions for blood pressure cuff bladders (22). The average of the last two readings was used. High blood pressure was considered when both systolic and diastolic blood pressures were ≥ 95th percentile for age, sex and height (22).
Statistical analyses
The characteristics according to participation in the follow-up study were compared. Agreement using kappa statistics was assessed between source of reporting of primary headaches, child or mother (23).
The Poisson regression method was used because when the prevalence of an event is higher than 10%, the use of logistic regression to estimate the odds ratio leads to an overestimate of the risk (24). The models were stratified according to city. The prevalence rate ratio (PRR) and its 95% confidence interval (95% CI) for primary headache were calculated by the robust method to avoid overestimation of the standard errors (24). Models were adjusted for variables collected at birth (maternal age, age of child, birth weight, body mass index, gestational age, marital status and schooling, paternal smoking during child’s pregnancy and sex of child) and at school age (blood pressure, mental health problems and exposure to passive smoking at home of the child). Models using primary headaches as reported by the children were also fitted. Because results could differ by sex, interactions between sex of child and maternal smoking during pregnancy on primary headache were tested.
Owing to the complex sampling design, because children of low birth weight (<2500 g) and high birth weight (≥4250 g) were overrepresented, the prevalence estimates and their standard errors were weighted using the svy set of commands in STATA. The weighting took into account the different probabilities of selection of each group of birth weight and preterm birth. Standard errors were corrected taking into account previous stratification of the sample by birth weight. Analyses were carried out using the statistical package STATA (version 10).
The Research Ethics Committees of the Universities in RP and SL approved the study. The legal guardians for the children gave written informed consent.
Results
Initial sample, eligible to follow-up, number and percentage followed up in the 1994 Ribeirão Preto birth cohort and 1997/98 São Luís birth cohort

p value calculated by the chi-squared test.
The prevalence of primary headache as reported by the mothers was higher in RP (28.1%) than in SL (13.1%) (p < 0.001), whereas the prevalence of primary headache as reported by the child was higher in SL (29.4%) than in RP (17.5%) (p < 0.001). Agreement based on kappa statistics between children’s and mothers’ reporting of primary headache in the child was poor, 0.21 for RP and 0.11 for SL. Agreement did not differ by age in each city. Mean age of children was 10.6 years in RP and 8.1 years in SL.
Poisson regression analysis of the risk factors for primary headache among school-aged children as reported by the mother, 1994 Ribeirão Preto birth cohort

PRR: prevalence rate ratio estimated by Poisson regression model, CI: confidence interval. Estimates are weighted and took into account the complex sampling design. Some variables do not total 790 because of missing values (n = 27 on headache and n = 21 girls who already had menarche). The model was adjusted for all variables in the table.
Poisson regression analysis of the risk factors for primary headache among school children as reported by their mothers, 1997/98 São Luís birth cohort

PRR: prevalence rate ratio estimated by Poisson regression model, CI: confidence interval. Estimates are weighted and took into account the complex sampling design. Some variables do not total 673 because of missing values (n = 18 on headache). The model was adjusted for all variables in the table.
Interactions between the effect of sex of the child and maternal smoking during pregnancy on primary headaches were not significant in either city (p = 0.998 for RP and p = 0.962 for SL).
Poisson regression analysis of the risk factors for self-report primary headache among school children, 1994 Ribeirão Preto birth cohort

PRR: prevalence rate ratio estimated by Poisson regression model, CI: confidence interval. Estimates are weighted and took into account the complex sampling design. Some variables do not total 790 because of missing values (n = 33 on headache and n = 21 girls who already had menarche). The model was adjusted for all variables in the table.
Poisson regression analysis of the risk factors for self-report primary headache among school children, 1997/98 São Luís birth cohort
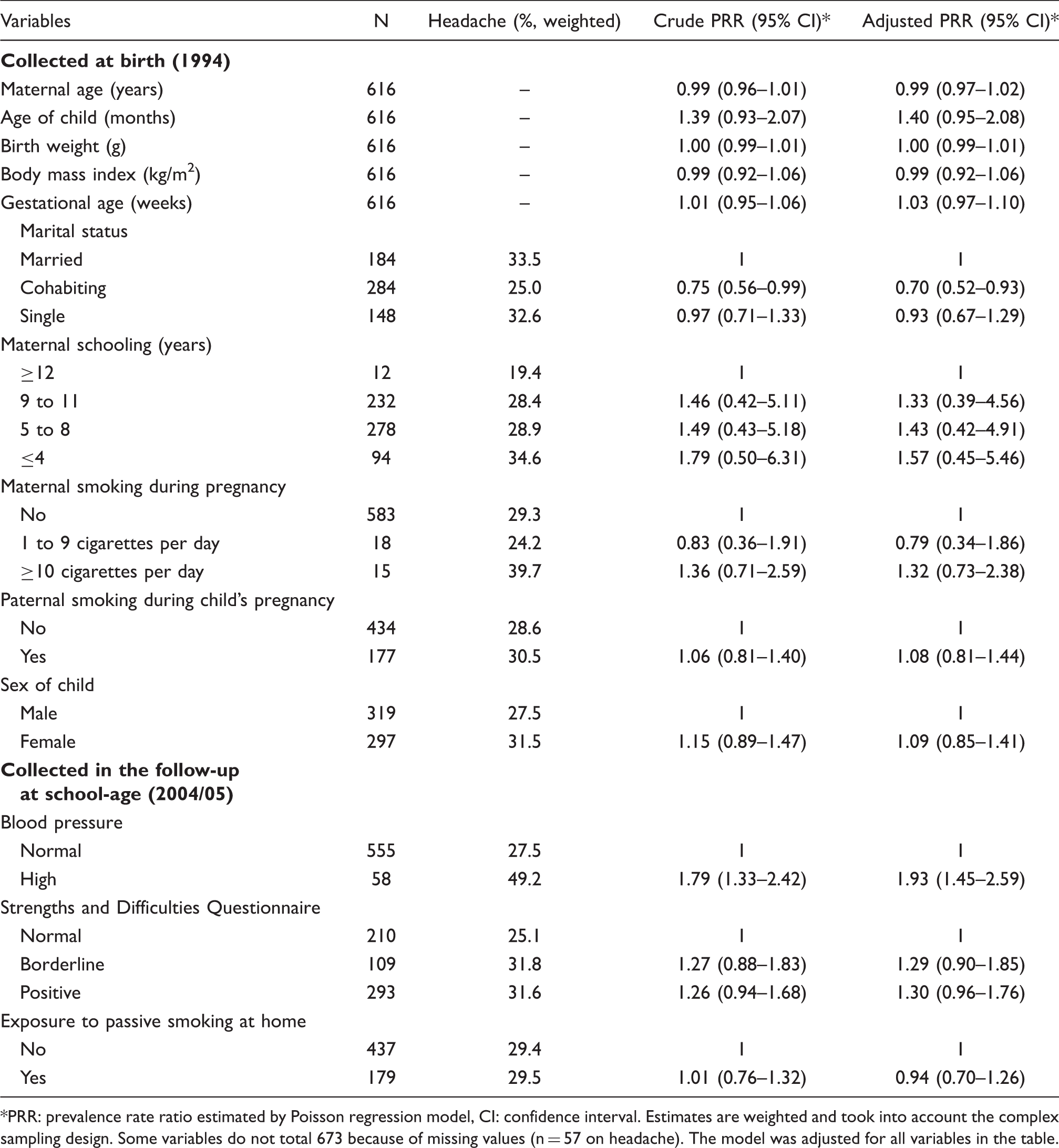
PRR: prevalence rate ratio estimated by Poisson regression model, CI: confidence interval. Estimates are weighted and took into account the complex sampling design. Some variables do not total 673 because of missing values (n = 57 on headache). The model was adjusted for all variables in the table.
Discussion
The main finding of this study is that maternal smoking, ≥10 cigarettes per day during pregnancy, was associated with primary headache among children aged 7 to 11 years old, as reported by their mothers. The association persisted even after adjustment for confounders, including possible mental health problems in the child, socioeconomic status and exposure to passive smoking at home. The association was consistent in the two birth cohort studies, despite the contrasting level of economic development between RP and SL and the age difference of the children in the two cohorts. The association was independent of psychological problems detected with the SDQ test. When we considered headache as reported by the children the association was significant only in the most developed city (RP), where children were older.
The major strengths of this study is that it was population based, used a longitudinal design, the results were replicated in two cities separately, we adjusted for the most important potential confounders and the response rate was satisfactory (68.7% in RP and 72.7% in SL). The longitudinal design ensured a more accurate assessment of prenatal maternal smoking.
There are some weaknesses in the present study. We did not collect information on active smoking of the children, but the prevalence of smoking in children under the age of 11 years is low in Brazil. In Pelotas, prevalence of smoking in the last 30 days was only 1.2% among 10–13 years old in 1999/2000 (25). We did not obtain information on medication. We also did not have information on maternal headache. Given that headache may have a genetic component, offspring of mothers who have headaches may be more likely to suffer from headache (26). However, it is also possible that children who listen to their mothers complain of headaches may be more ready to acknowledge suffering from headaches.
Another limitation is that the participation rates were lower for mothers who smoked ≥10 cigarettes per day during pregnancy, were less than 20 years of age and single or married in RP, for male babies in SL, and for mothers of low and high full time education in both cities. This could have increased the likelihood of selection bias.
The children were also asked the same question on headaches as their mother and the level of agreement based on kappa statistics was poor (23). Most studies have assessed headaches in childhood using parental reports on the assumption that information provided by younger children is less reliable (2,11). Few studies have assessed concordance between parental and children’s reports on headache. Some have found that children’s answers are reliable (6,27) whereas in others rates based on children’s self-report were higher than those based on parental assessment (28,29). There is no convincing evidence that children’s answers are less reliable than their parents’ (27). However, even when children’s reports were used, the association between maternal smoking during pregnancy and primary headache was highly significant in RP, in which the children were older than those from SL. It is possible that older children understand better the meaning of headache in a questionnaire. This might explain why the association based on children’s self-report between maternal smoking during pregnancy and primary headache in schoolchildren was not significant in SL. Lack of conceptual understanding of headache in small children may have caused a degree of misclassification, which may have been responsible for the results in SL as the number of mothers who smoked in pregnancy in SL was low (only 15 mothers smoked ≥10 cigarettes per day during pregnancy).
The only other study that tested the hypothesis in this study also concluded that prenatal exposure to tobacco was associated with increased prevalence rate of chronic daily headache in childhood (14). However, in that study the design was cross-sectional, no adjustment for current passive smoking was carried out and information about maternal smoking during pregnancy was assessed retrospectively, when children were 5–12 years old.
Exposure to second-hand smoke at home was not associated with headache in children in either cohort, possibly because exposure is much lower than exposure to maternal smoking in the uterus. A possible explanation for our finding is that Brazilian children by ages 7–11 spend most of their time in school and playing with friends outside the home. Thus, they may be spared some of the negative effects of maternal smoking during the day.
An unexpected result was that mothers with fewer years of full time education were less likely to report headache in their children in SL, but not in RP. In SL mothers with 12 or more years of full time education reported headache in their child more frequently than mothers in any other education group. Recall bias may explain this finding; for example, mothers with more education may be more aware of health problems in their children or may have a lower threshold for recognizing a health issue in their child. However, even if the explanation of this finding were due to recall bias, the main results of our study cannot be explained by recall bias, as we adjusted for years of full time education of the mother.
We note that psychological problems, as measured by the SDQ, were also positively associated with headache in the children in the two cities. The association between psychological problems and headaches has been reported by others (8,10,11). Because maternal smoking in pregnancy has been related to behavioral and emotional disorders in the offspring (30,31), this could have acted as a confounder. However, it is worth noting that adjustment for psychological problems did not change the estimates of the association between maternal smoking in pregnancy and child’s headache.
Smoking is typically considered a risk factor for headache (9,10,12), possibly as a result of the pharmacological effects of tobacco smoke (12). There are several possible mechanisms to explain the effect of maternal smoking in pregnancy on childhood headache. Prenatal nicotine exposure influences neuronal development, including effects on several neurotransmitter systems (32). Nicotine interferes with catecholamine and brainstem autonomic nucleus development during the prenatal period of rodents (equivalent to first and second trimester of humans) and alters the neocortex, hippocampus, and cerebellum during the early postnatal period (third trimester of humans) (33). It has also been reported that exposure to nicotine during pregnancy has detrimental effects on cholinergic modulation of rat brain development (34). In addition to the changes in pain processing, smoking can induce structural changes in other systems that may predispose individuals to painful conditions (35). It is also plausible that maternal smoking during pregnancy may induce structural modifications in the fetal brain or alter angiogenesis (36), causing headache in childhood (37). Inhibition of brain monoamine oxidase during fetal brain development, secondary to maternal cigarette smoking, which may lead to brain morphological and functional changes, could also be implicated (38).
Conclusions
Maternal smoking during pregnancy was associated with primary headache among 7- to 11-year-old school children in two prospective, population-based cohorts in a middle-income country. Regardless of location the association was consistent based on maternal report and self-report by 10- to 11-year-olds, but not self-report by 7- to 9-year-olds after adjustment for confounders, including children’s mental health problems, socioeconomic status and exposure to passive smoking at home. Poor agreement between maternal and children reports of headache suggests that reliance on parental report only for assessment of children headaches may be misleading. Smoking by mothers in pregnancy may be a contributor to headaches in their children.
Footnotes
Funding
This work was supported by CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico), Brazilian National Research Council [grant number 523474/96-2 and 520664/98-1], and FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo) São Paulo Research Foundation [grant number 93/0525-0, 97/09517-1 and 00/0908-7].
Conflicts of interest
The authors declare that there is no conflict of interest.
